Abstract
Alpha-glucosidase inhibitors have been available for clinical use for about 20 years. They have shown reasonably good efficacy comparable to other oral blood glucose lowering drugs and in some parts of the world are the most commonly prescribed oral diabetes medication, especially in Asian countries. Unlike as has been observed with some other blood glucose lowering agents, however, no adverse signals of potential cardiovascular harm have emerged in relation to their use. On the contrary, significant beneficial cardiovascular outcome results have been observed in the post-hoc analyses of randomised placebo-controlled trials with the alpha-glucosidase inhibitor acarbose. Targeting mainly postprandial hyperglycaemia, alpha-glucosidase inhibitors favourably affect several cardiovascular risk factors, such as obesity, hypertension and high glycaemic variability with little to no risk for hypoglycaemia. Furthermore, acarbose favourably affects endothelial dysfunction and carotid intima media thickening in humans and, in animal models, improves cardiac interstitial fibrosis and hypertrophy of cardiomyocytes. The ultimate determination of the cardiovascular effects of alpha-glucosidase inhibitors in terms of clinical outcomes awaits the results of ongoing long-term, randomised, placebo-controlled trials.
Keywords
Introduction
The golden paradigm to reduce cardiovascular (CV) complications in patients with diabetes mellitus remains a multifactorial approach based on therapeutic lifestyle management, targeting hypertension, dyslipidaemia, hyperglycaemia and hypercoagulability. 1 In recent years, however, evidence has emerged that intensification of blood pressure and glucose control therapies yield no clear CV benefits and, in some cases, may also cause CV harm,2–7 especially in patients with pre-existing CV disease.2–7 Looking more specifically at various glucose-lowering drugs, it was found that treatment with some of them such as rosiglitazone, 8 most sulfonylureas and even insulin might be associated with a higher risk of CV complications, at least when compared with metformin treatment.9–11 For example, a large nationwide epidemiological study in Denmark has recently reiterated these concerns about a potentially increased mortality and CV risk associated with some sulphonylureas. 12
This paper attempts to evaluate the case of alpha-glucosidase inhibitors, i.e. acarbose, voglibose and miglitol, which have been available as oral anti-diabetic drugs since the early 1990s.13,14 Indeed, in some Asian countries with large diabetic populations, such as China, this class of drugs (especially acarbose) has become the most commonly prescribed glucose-lowering medication. 15 Given the penetrant uptake of the alpha- glucosidase inhibitors and the availability of generic formulations in some cases, the issue of their CV affects is of practical importance.
Mode of action, dosing, efficacy
Alpha-glucosidases in the brush-border of the small intestine are competitively inhibited by the pseudo-carbohydrates acarbose, voglibose or miglitol. This inhibition delays digestion of complex carbohydrates in the upper small bowel and subsequently retards absorption of glucose and ‘blunts’ postprandial hyperglycaemia (Figure 1).13,14,16,17 Intact acarbose is poorly absorbed and is excreted in the faeces, mostly intact but with up to 30% undergoing metabolism predominantly via fermentation by colonic microbiota.13,14,16 Similarly, voglibose is slowly and poorly absorbed and rapidly excreted in stools, 14 with no metabolites identified to date. 18 In contrast, miglitol is fully absorbed in the gut and cleared unchanged by the kidneys. 19
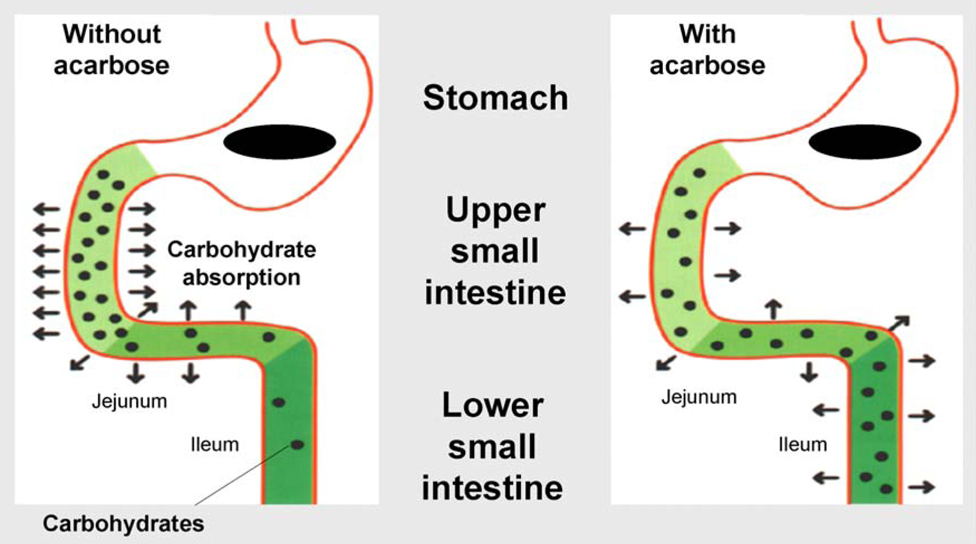
Acarbose acts non-systemically to delay carbohydrate absorption.
Adaptive changes in the lower smaller intestine, which may take several weeks to occur following initiation of therapy with alpha-glucosidase inhibitors, ensure full carbohydrate absorption, mitigate initial gastrointestinal side effects, such as abdominal distension, flatulence or diarrhoea, and also seem to be associated with a higher endogenous stimulation of glucagon-like peptide-1 (GLP-1).16,17 In the context of this adaptation and the interval gastrointestinal side effects, it is advisable to pursue a dosing concept of ‘start low, go slow’. The average effective doses are 100 mg tid for acarbose, 0.2 mg tid for voglibose and 50 mg tid for miglitol.13,14
In terms of glucose-lowering efficacy, acarbose is the most extensively evaluated agent. A Cochrane systematic review (Figure 2) 20 confirmed a placebo-subtracted decrease of HbA1c with acarbose therapy of 0.8% (95% confidence intervals (CI) 0.64–0.90), a 2.3 mmol/l reduction in postprandial glucose (95% CI 1.92–2.73), and a 1.1 mmol/l reduction in fasting glucose (95% CI 0.83–1.35). Comparable efficacy seems to be apparent from studies with voglibose and miglitol.18,19
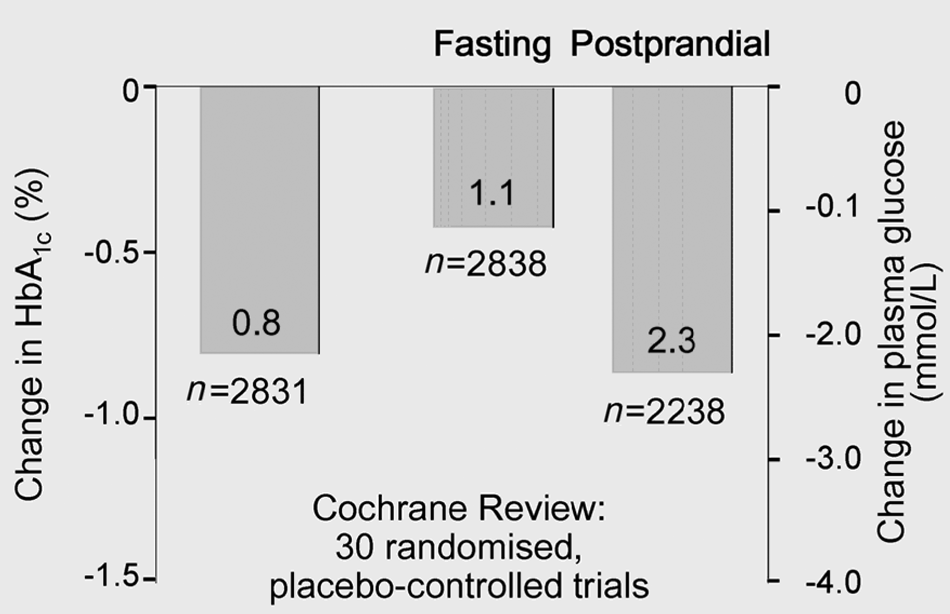
Acarbose is an effective monotherapy for type 2 diabetes. 20
Head to head comparisons have been performed against metformin, most sulphonylureas and meglitinides and the dipeptidyl-peptidase-4(DPP4) inhibitors.21–23 In summary, the blood glucose-lowering potency as assessed by HbA1c measurements has been largely found comparable to those drugs, with an edge in favour of metformin in some studies. Of course, by their mode of action, the predominant blood-glucose lowering of alpha-glucosidase inhibitors occurs postprandially.13,14,19–23
Epidemiological and pathogenetic links of postprandial hyperglycaemia to CV disease
This postprandial aspect of action as seen with alpha- glucosidase inhibitors seems to be of importance. Compelling evidence has been accumulated over the last 20 years that elevated postprandial glucose values, even in the high non-diabetic impaired glucose tolerance (IGT) range, contribute to an approximately three-fold increase in the risk of developing coronary heart disease or other major adverse CV event. 24 Controversy, however, exists whether elevated fasting plasma glucose and post-load glucose contribute differently to all-cause mortality or CV outcomes, respectively. A meta-analysis of Coutinho, et al. has suggested that both parameters contribute more or less equally, 25 in contrast with publications, for example, from the DECODE, 26 DECODA 27 or Funagata 28 studies that seem to support a unique role of postprandial hyperglycaemia contributing to CV risk. The ongoing prospective AusDiab Study, a cohort study of approximately 10,000 people across Australia with baseline glucose tolerance testing, has indicated a graded association between baseline glycaemic indices and subsequent CV clinical events and mortality over approximately 5-years of study follow-up, with similar model performance using impaired fasting glucose or IGT classifications.29,30 The observational CAPRI study in overtly diabetic patients diet-treated or on oral glucose-lowering therapy has described an additional relationship between the maximal postprandial blood glucose peak and carotid intima-media thickness above and beyond the association observed with HbA1c. 31 Furthermore, the Diabetes Intervention Study 32 and the San Luigi Gonzaga Study 33 investigators have reported specific associations between postprandial glucose concentrations and risk for subsequent CV events, such as myocardial infarction, in patients with type 2 diabetes.
A serious limitation of these epidemiological studies is that most often postprandial hyperglycaemia was not measured, but rather glycaemia after an oral glucose load with 75 g glucose was evaluated. It is noteworthy, therefore, that although the 2-h glucose values after an oral glucose load are closely correlated with the 2-h values after a standardised meal, the correlation with postprandial blood glucose experienced in the ambulatory setting are much more variable. 34 Continuous glucose monitoring with sensors (CGMS) is probably a necessary prerequisite for studies trying to elucidate the specific impact of postprandial glycaemic parameters on CV outcomes, especially when it comes to glycaemic variability. 24 This approach has been applied in a number of pathophysiological studies. 24
As discussed recently, acute increases of blood glucose concentrations and fluctuating glucose levels with high peaks may induce haemodynamic alterations and are associated with endothelial dysfunction and signs of oxidative stress,24,35 even in non-diabetic patients. The seminal work of Monnier et al. has shown that using CGMS in patients with type 2 diabetes, the urinary excretion of 8-isoprostanes as a sign of oxidative stress is closely correlated with the post-meal glucose levels and even more so with the maximal glucose excursions, in contrast to no such correlation with the HbA1c value or the mean glucose concentration. 36 Ceriello et al. have used a model of generating glucose fluctuations by glucose infusion both in diabetic and non- diabetic subjects. 35 They found a very close association of the glucose peaks with a decrease of the flow-mediated dilation of the forearm and an increase of nitrotyrosine, which reflects oxidative stress at the mitochondrial level.
Finally, Ceriello et al. went on to in vitro studies using human endothelial cells and exposing them to various glucose patterns, i.e. normal or constantly high glucose for 21 d vs. oscillating high and normal glucose for 21 d vs. oscillating high and low or permanently high glucose for 14 d followed by normal glucose by 7 d. 37 They consistently showed the highest up-regulation of pathways related to apoptosis; at the gene transcription level, the highest generation of oxidative stress markers and DNA damage; and the highest reactive oxygen species accumulation with oscillating glucose concentrations. These alterations could not be restored to normal despite normalisation of glucose levels, suggesting thus a metabolic memory of human endothelial cells. 37
So, in all, impressive evidence has been accumulated for the case of postprandial hyperglycaemia and fluctuating glucose concentrations with peaks as a pathophysiological link to disordered vascular reactions of large blood vessels and as a risk predictor for CV complications longer term.
Intervening on postprandial hyperglycaemia: Acute and longer-term vascular effects
Measuring endothelial function at the myocardial level with positron emission tomography (PET) technology in insulin-requiring diabetic patients and targeting postprandial hyperglycaemia with various kinds of insulin regimens, a clear proof of principle could be obtained. 38 When postprandial hyperglycaemia was abolished, myocardial blood flow was restored to normal, with paradoxical vasoconstriction with more marked postprandial hyperglycaemia evident. 38 Similarly, using acarbose to target postprandial hyperglycaemia in non-insulin requiring diabetic subjects, forearm flow-mediated dilation improved significantly in two separate studies.39,40 This was in contrast to a 3-month study of nateglinide treatment to assess postprandial hyperglycaemia and its association with myocardial blood flow measured with PET at baseline and follow-up. 41 Withholding the drug on the day of PET evaluation, no change of myocardial flow parameters could be demonstrated. On the other hand, there was also rather little effect on postprandial hyperglycaemia. 41 So, not all drug interventions of postprandial hyperglycaemia are equally effective at the vascular level and some drugs, such as acarbose, may show a unique effectiveness. This was also supported by animal studies in which exposure to acarbose was associated both with prevention of cardiac interstitial fibrosis and cardiomyocyte hypertrophy in a model of intermittent hypoxia. 42
The landmark Study to Prevent Non-Insulin-Dependent Diabetes Mellitus (STOP-NIDDM) Trial evaluated the efficacy of 3 years of treatment with acarbose vs. placebo in preventing the transition to overt diabetes among 1429 patients with IGT. 43 Targeting postprandial hyperglycaemia with acarbose was not only found to be associated with a 36% reduction of new onset diabetes, but also with a highly significant reduction of myocardial infarction (1 vs. 12, p = 0.0226) and any CV event (15 vs. 32, p = 0.0326) as assessed by a post hoc analysis of secondary outcomes (Figure 3). 44 In addition, new onset hypertension was significantly reduced with acarbose. 44 In a subgroup of patients in the STOP-NIDDM trial in whom carotid intima-media-thickness was assessed, reduced thickening was observed under acarbose treatment over time. 45 Consistent with these findings, another post hoc meta-analysis of seven randomised trials with acarbose in 2180 patients with type 2 diabetes and a follow-up of 1 year or longer (MeRia Study), 46 likewise revealed a significant decrease of myocardial infarction (9 vs. 19, p = 0.012) and any CV event ( 76 vs. 88, p = 0.006). Similar to the observations in STOP-NIDDM, a significant reduction of blood pressure was also observed in the acarbose-treated patients in the MeRia Study, 46 a phenomenon seen in another randomised controlled trial over 6 months in comparison with glibenclamide. 47
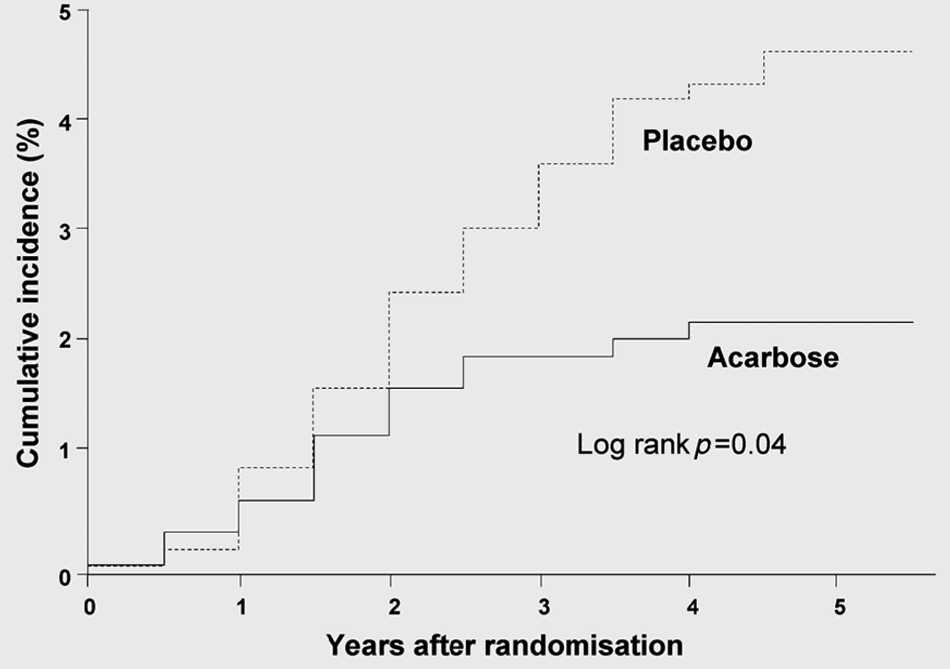
STOP-NIDDM trial: Lower incidence of cardiovascular events in acarbose treated patients.
Voglibose has also been evaluated in a randomised trial in terms of diabetes prevention in 1780 IGT subjects. A close to 50% reduction (multivariate analysis, hazard ratio, 0.512, CI 0.360–0.727, p = 0.0002) was found after 1 year, but no CV outcome data have been reported so far. 48
Other randomised clinical trials seeking to specifically target postprandial hyperglycaemia have been less successful in improving CV outcomes, such as the Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research (NAVIGATOR) Trial 49 or the Hyperglycemia and Its Effect After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus (HEART2D) Trial. 50 In NAVIGATOR, the previously mentioned short-acting insulin secretagogue nateglinide compared with placebo was assessed in 9306 subjects with IGT and elevated risk for CV disease over 6 years. Nateglinide had no effect on incident diabetes risk nor CV risk and, when participants underwent oral glucose tolerance testing without the study drug on the day of testing, there was no decrease in 2-h glucose levels. 49 Similarly, the HEART2D trial assessed prandial vs. basal insulin treatment in 1115 patients with type 2 diabetes following acute myocardial infarction 50 and failed to achieve the per-protocol intended postprandial decrease of blood glucose and was an essentially negative trial in terms of improving CV outcomes. So, while these trials were negative with regard to the principal efficacy assessments, the failure within each trial to achieve the targeted postprandial glucose goals maintains the viability of the concept of potential CV efficacy with targeted postprandial glucose modulation and support ongoing assessments of the alpha-glucosidase inhibitors or other drugs similarly effective at modifying this parameter. Although intriguing, the data to date with regard to the CV safety and efficacy of existing alpha-glucosidase inhibitors are not definitive due to the extremely few CV events to analyse across the databases available.
Definitive evaluation of the CV effects of treatment with acarbose is underway in the ongoing Acarbose Cardiovascular Evaluation (ACE) Trial. The trial comprises 7500 patients in China with IGT and who are at least 3 months out from a prior myocardial infarction randomised to acarbose treatment vs. placebo. The trial is designed with co-primary endpoints of (a) prevention of diabetes and (b) major adverse CV events. 51
Recent clinical studies with alpha-glucosidase inhibitors
As already mentioned, acarbose remains the most widely studied drug of the class. It can be administered as mono-therapy or in combination with any other blood glucose-lowering drug, including insulin, as has been recently demonstrated again in a large-scale, observational study enrolling some 15,000 patients in China, Taiwan, the Middle-East, Morocco, Poland, Indonesia, Pakistan and the Philippines and with a follow-up of up to 3 years. 52 It seems to offer specific advantages in terms of attenuating postprandial hyperglycaemia, glucose variability, hypoglycaemia and weight gain. 52 A notable weight decrease was seen with acarbose over time in all treatment groups (mono-therapy, combination with metformin or sulfonylureas or insulin) despite a 1% lower HbA1c level. Mild gastrointestinal side effects, however, were apparent in a small minority of patients, which subsided with increasing duration of follow-up. In another study of patients with metformin-treated type 2 diabetes with continuous glucose monitoring used, randomised treatment with acarbose vs. glibenclamide, a comparable decrease of HbA1c was observed between the groups, but a much smaller intra-day and inter-day glucose variability was seen with acarbose. This effect was mainly due to a flattening of postprandial hyperglycaemia. 53 Several episodes of hypoglycaemia occurred with glibenclamide during the 12-week evaluation period, in contrast to none with acarbose. Body weight decreased with acarbose by 1.4 kg and showed a tendency to increase with glibenclamide. 53
In terms of preventing weight gain, acarbose was also highly effective in a 20-week placebo-controlled, randomised trial in patients who were newly started on insulin therapy. In this study, acarbose was superior in decreasing HbA1c and postprandial insulin levels. 54 Finally, in a head to head comparison with the DPP4 inhibitor vildagliptin, acarbose was similarly effective in blood glucose lowering, yet superior in decreasing body weight. 22
So in summary, clinical studies evaluating acarbose indicate a good glycaemic efficacy across all stages and all treatment options of type 2 diabetes. Acarbose seems to influence the CV risk factor profile positively, in particular by virtue of its excellent effect on weight control without the risk of hypoglycaemia. Hypoglycaemia in turn has been found to be a significant predictor of CV mortality in several of the recent intervention trials in type 2 diabetes.2–4 In addition, the emerging CV risk constellations of postprandial hyperglycaemia and high glucose variability are clearly attenuated by acarbose.
Perspectives
Twenty years after introduction of alpha-glucosidase inhibitors into the therapy of type 2 diabetes, still new discoveries can be made. In view of the recent issue of potential CV harm caused by some blood glucose-lowering agents, it is reassuring that no such signals have emerged in the context of the use of alpha-glucosidase inhibitors. On the contrary, significant beneficial CV outcome effects have been observed in the STOP-NIDDM Trial43–45 and in a post-hoc analysis of randomised placebo-controlled trials with acarbose. 46 Furthermore, CV risk factors such as obesity, hypertension, postprandial hyperglycaemia, high glucose variability seem to be influenced favourably. In contrast to other treatment options of blood glucose lowering, there is no risk of hypoglycaemia, which has turned out to also be a risk predictor of CV mortality in its own right. The ultimate proof for a significant CV benefit of acarbose is hoped to come from the ongoing ACE Trial. 51 Since many of the ongoing randomised, long-term trials looking into CV outcomes in relation to the use of DPP4 inhibitors or the GLP-1 analogues, as discussed elsewhere in this issue by Jose and Inzucchi which also mainly target postprandial hyperglycaemia, it will remain interesting how these trials will compare with the ACE Trial.
Footnotes
Funding
This activity has been supported by an educational grant of the Association for the Support of International Scientific Communication in Diabetology e.V., Munich, Germany.
Conflicts of interest
Conflicts of interest for Professor Standl:
Task force: First Joint EASD/ESC Guidelines; DSMBs: PROActive, Navigator, ACE Trials; Investigator: studies initiated by Oxford Trial Unit, Bayer Schering, Menarini, Novartis, NovoNordisk, Sanofi-Aventis, Servier, MSD/Merck; Lecturing honoraria and consultation fees: Astra Zenica, Bayer, BMS, GSK, Johnson & Johnson, Lilly, Menarini, Merck-Serono, MSD/Merck, Novartis, NovoNordisk, Rambaxi, Roche, Sanofi-Aventis, Servier, Takeda.
Conflicts of interest for Professor Schnell:
Lecturing honoraria and consultation fees: Bayer Schering Pharma.
