Abstract
The insulin receptor (IR) and low-density lipoprotein receptor (LDLR) maintain glucose and lipid metabolism, respectively. Diabetes is associated with increased blood glucose, dyslipidaemia and increased risk of atherosclerosis. We hypothesise that interactions between IR and LDLR play a role in the atherosclerotic process in subjects with diabetes. Therefore, in this work we studied potential interactions between these two receptors. Our data show an intracellular and surface membrane-bound co-association of IR and LDLR. The co-association makes LDLR functionally poor in clearing extra-cellular LDL particles. A short 10 min exposure of cells to insulin disrupts the association between the two receptors and generates LDLR with higher LDL clearing activity without any change in protein expression. This co-association of LDLR with IR and their dissociation by insulin may be an important part of the regulatory mechanism of the normal physiological receptor function in a biological system. Modulation of receptor co-association is potentially a therapeutic target to reduce cardiovascular risk, and further studies are needed to investigate this possibility.
Introduction
The low-density lipoprotein receptor (LDLR), a type-I transmembrane glycoprotein, is involved in the control of blood cholesterol1–3 by clearing cholesterol-laden low-density lipoprotein (LDL) particles,2,4 whereas the insulin receptor (IR) maintains blood glucose homeostasis.5–7 Diabetes is associated with dyslipidaemia, although the exact mechanisms are not entirely clear.8–10 Furthermore, diabetes is characterised by abnormalities in IR, either through absolute deficiency of insulin (type 1 diabetes) or secondary to transcriptional / translational / post-translational abnormality of the receptor protein itself (type 2 diabetes). Insulin is known to regulate multiple biological activities,11–15 resulting in enhanced glucose transport and maintenance of adequate blood glucose levels.16–19 It is also known that insulin increases LDLR expression,20–23 but it is unknown whether the LDL-clearing activity of LDLR is dependent on the interaction of insulin with IR.
Impairment in IR function results in abnormal glucose metabolism, which manifests clinically as diabetes or impaired glucose tolerance, conditions commonly associated with obesity and insulin-resistant states.24–26 Insulin resistance is also associated with LDL accumulation in blood vessels as a result of poor clearance by functionally impaired LDLR.19,27 This in turn predisposes to atherosclerosis and cardiovascular disease, the main cause of mortality in patients with diabetes. Previous reports have shown that the presence of insulin increases LDLR activity in HepG2 cells. 21 Insulin treatment has also been shown to increase the expression of cell-surface LDLR, resulting in a reduction in LDL cholesterol levels. 19 However, there are no clear mechanistic pathways linking LDLR activity to insulin. Therefore, in this work we explore the potential interdependency between LDLR and IR through a series of experiments to address the following questions: (a) Is insulin a key driver of LDLR activity? (b) If IR and LDLR are interdependent, how is LDLR activity controlled by IR? (c) If LDLR is dependent on insulin for functional existence, what are the consequences of inactive IR on LDLR function?
Materials and methods
Cell culture
HepG2 cells were obtained from NCCS, Ganeshkind, Pune, India, and maintained as described elsewhere. 23 Peripheral blood monocytes were isolated from blood samples of healthy controls and subjects with diabetes following the guidelines of the Institute Ethics Committee.
The following procedures are described only briefly. The full details can be found in the Supplementary information.
Electron microscopy
Electron microscopy was performed using the standard protocol in use at the Analytical Instruments Facility (AIF) at AIIMS, New Delhi, India. Antibodies conjugated with colloidal gold of various sizes were used to label receptor proteins. Images were captured using a Philips CM-10 transmission electron microscope at different magnifications.
Immunoprecipitation and western blotting
Receptor proteins were immunoprecipitated from unlabelled and 35S-labelled cell lysates. Co-immunoprecipitation of receptor proteins was verified on immunoblots. 28 The effects of insulin on co-immunoprecipitates were observed with 35S-labelled cell lysates.
Sub-fractionation of endoplasmic reticulum, Golgi and plasma membrane
Endoplasmic reticulum, Golgi and plasma membrane were isolated by ultracentrifugation. 29 Co-existence of LDLR and IR was examined by checking the cross-reactivity of two receptor proteins in these organelles.
LDL receptor activity
Functional activity of LDL receptor(s) was determined by measuring the rate of uptake of Dil-LDL particles (Dil: 1,1′-Dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate) by cells in culture. Dil-LDL was prepared and quantified using procedures described by Havel et al. 30 and Stephan and Yurachek. 31
Expression of receptor proteins and transcriptional regulation
Expression of receptor proteins was determined by immunoblotting, and real-time PCR was used to measure LDLR transcription. More details are provided in the online Supplementary material.
Knock down of LDLR expression
LDLR-siRNA sc-35802 (Santa Cruz Biotech, CA, USA) was used to knock down LDLR expression in cultured cells. Transfection of siRNA to cultured HepG2 cells was carried out according to the manufacturer’s protocol using transfection reagent (sc-29528) provided by the same company. The cells were then lysed as before and western blot analysis was performed using anti-LDLR antibody as described above. More details are provided in the online Supplementary material.
Results
LDLR and IR remain in complex
LDLR and IR were immunoprecipitated from 35S-methionine-labelled HepG2 cells and run on SDS-PAGE in two batches (Figure 1A and 1B). One was treated with 0.1% SDS and 5% beta-mercaptoethanol (β-ME) (Figure 1A) and the other with 1% SDS and 5% β-ME (Figure 1B). Since native PAGE did not allow the protein precipitates to move an adequate distance, 0.1% SDS was used to provide mobility to the immunoprecipitated proteins. It was noticed that at 0.1% SDS the immunoprecipitates of both receptors appeared together at the same higher molecular mass (Figure 1A, lanes 1 and 2), whereas with 1% SDS, 32 the respective bands for LDLR and IR moved to their appropriate molecular masses (Figure 1B, lanes 1 and 2). Appearance of LDLR and IR in one band at a region of higher molecular weight suggested the probability of association of the two receptors together. On the other hand, the immunoprecipitates treated with 1% SDS (Figure 1B, lanes 1 and 2) separated the bands to their respective molecular masses. It was also apparent from panel B that both the receptor proteins were immunoprecipitated together by either antibody, as the immunoprecipitates by antibodies of each kind showed bands for both the receptors. Since 5% β-ME was present in all the lanes (panel A and B) and only the higher concentration of SDS gel separated the individual receptors, it was concluded that the two proteins were bound by weak non-covalent interaction(s) and not by covalent bonding.
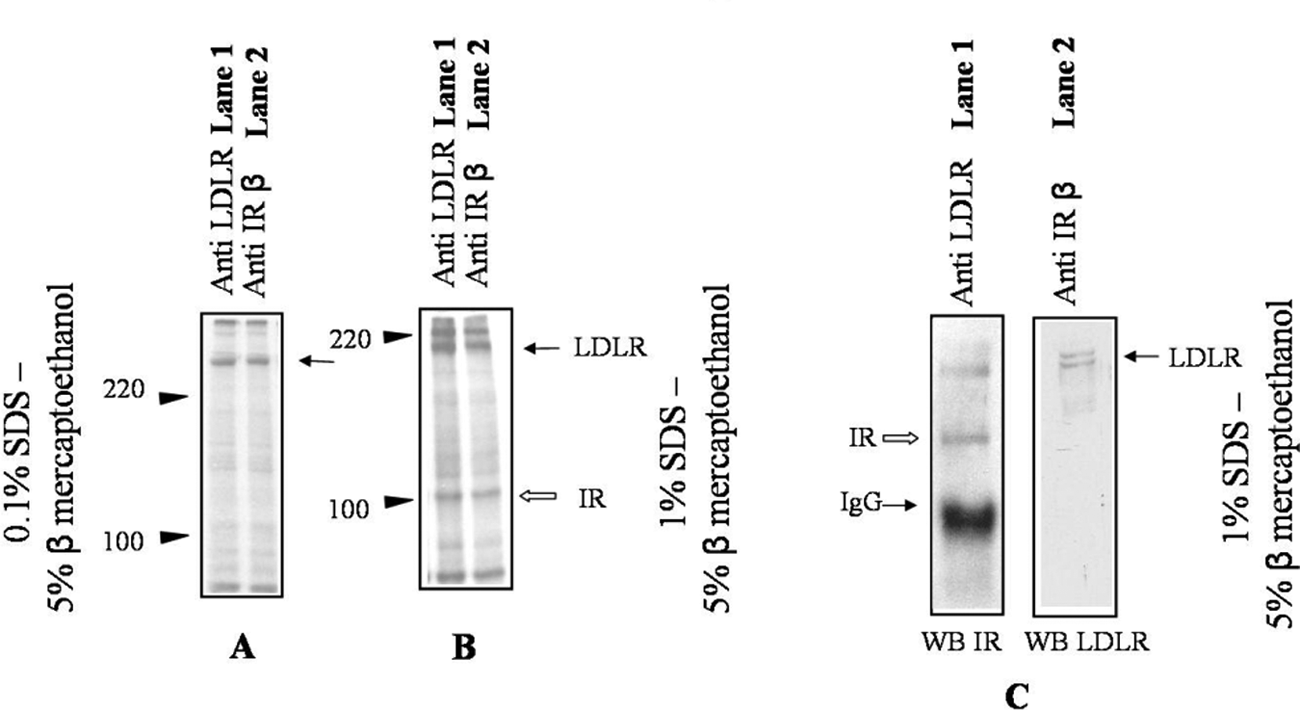
Detergent dissociates component receptor proteins from the low-density lipoprotein receptor–insulin receptor (LDLR–IR) complex.
This receptor complex was subsequently confirmed when the immunoprecipitate of one receptor protein was blotted for the other receptor protein (Figure 1C). The immunoprecipitate of LDLR showed the band for IR when probed with anti-IR antibody on the western blot (Figure 1C, lane 1). The same was also true in the immunoprecipitate of IR, as a band for LDLR was obtained on the western blot by anti-LDLR antibody (Figure 1C, lane 2). In both cases the gel was run with 1% SDS and 5% β-ME. The IR was immunoprecipitated in all above cases with anti- IRβ antibody. Similar results were also obtained when anti-IRα chain antibody was used (Supplementary information, Figure S1, S2).
The inability to separate the two receptors at 0.1% SDS was not due to any non-specific binding between the two proteins. This is supported by the lack of such interaction when an unrelated plasma membrane receptor (TNF-R1) was immunoblotted along with the LDLR and IR from HepG2 cell lysate by their respective antibodies (see Supplementary material; Figure S1 and S2).
LDLR and IR remain in co-association
The co-aggregation of the two receptors was also verified by double immunogold electron microscopy. IR was linked to a gold particle of 30 nm and LDLR to one of 15 nm in size. The co-aggregation of gold particles of two different sizes provided further evidence for the association of two receptor proteins in cell cytoplasm as well as on the plasma membrane (Figure 2a and 2b). On the plasma membrane the existence of such an association was clearly visible within and around the invaginations, as captured by the electron microscope (Figure 2b). The relative labelling index (RLI) for receptor-linked gold was calculated according to Griffiths et al. 33 Plasma membranes were found to be preferentially labelled by both the receptors. The RLI indicated the ratio of the number of receptors found on the plasma membrane of a cell to the total number of receptors found within the cell. The RLI was close to 1.0 for both receptors (RLILDLR: 0.98; RLIIR: 1.02). The two receptors showed about 10–30% co-association at random zones on plasma membrane (in and outside of clathrin coat). Inter-association with less than 50 nm inter-gold distance was only considered to mark co-associated receptors.
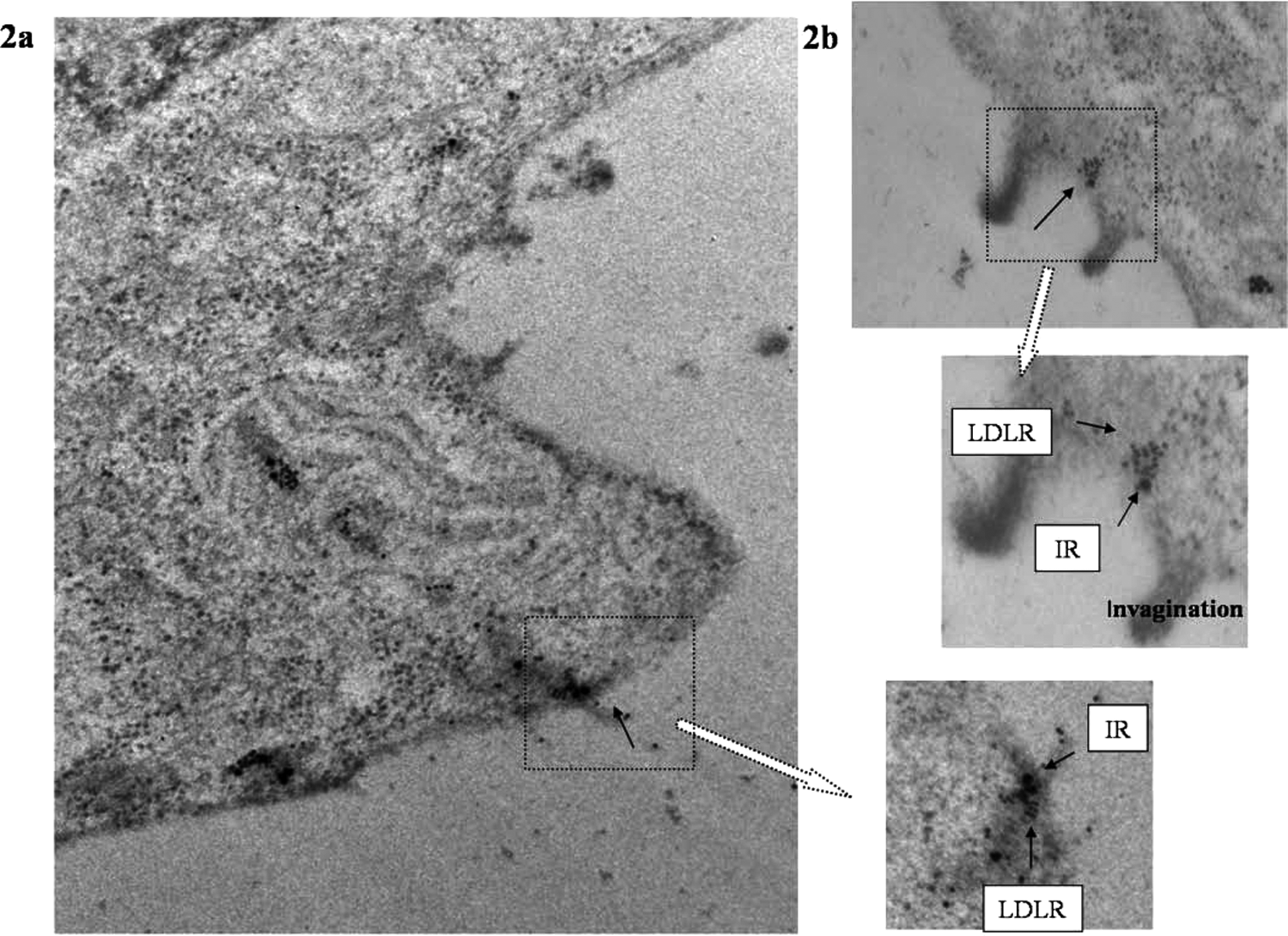
Co-association of low-density lipoprotein receptor (LDLR) and insulin receptor (IR) by electron microscopy.
Evidence of co-association in sub-cellular organelles
Golgi complex, endoplasmic reticulum and plasma membrane fractions were purified from goat liver by ultracentrifugation. 34 The immunoprecipitates of LDLR from the organelles were blotted with anti-IR antibody (Figure 3A). The gel was run on 1% SDS and 5% β-ME. The blot showed that IR was present in the immunoprecipitate of LDLR, indicating that IR was immunoprecipitated along with LDLR. The presence of IR in the immunoprecipitate of LDLR from each of the three compartments further supported their existence in complexes. Repeat experiments with human term placental tissue also showed similar results (Supplementary information, Figure S3).
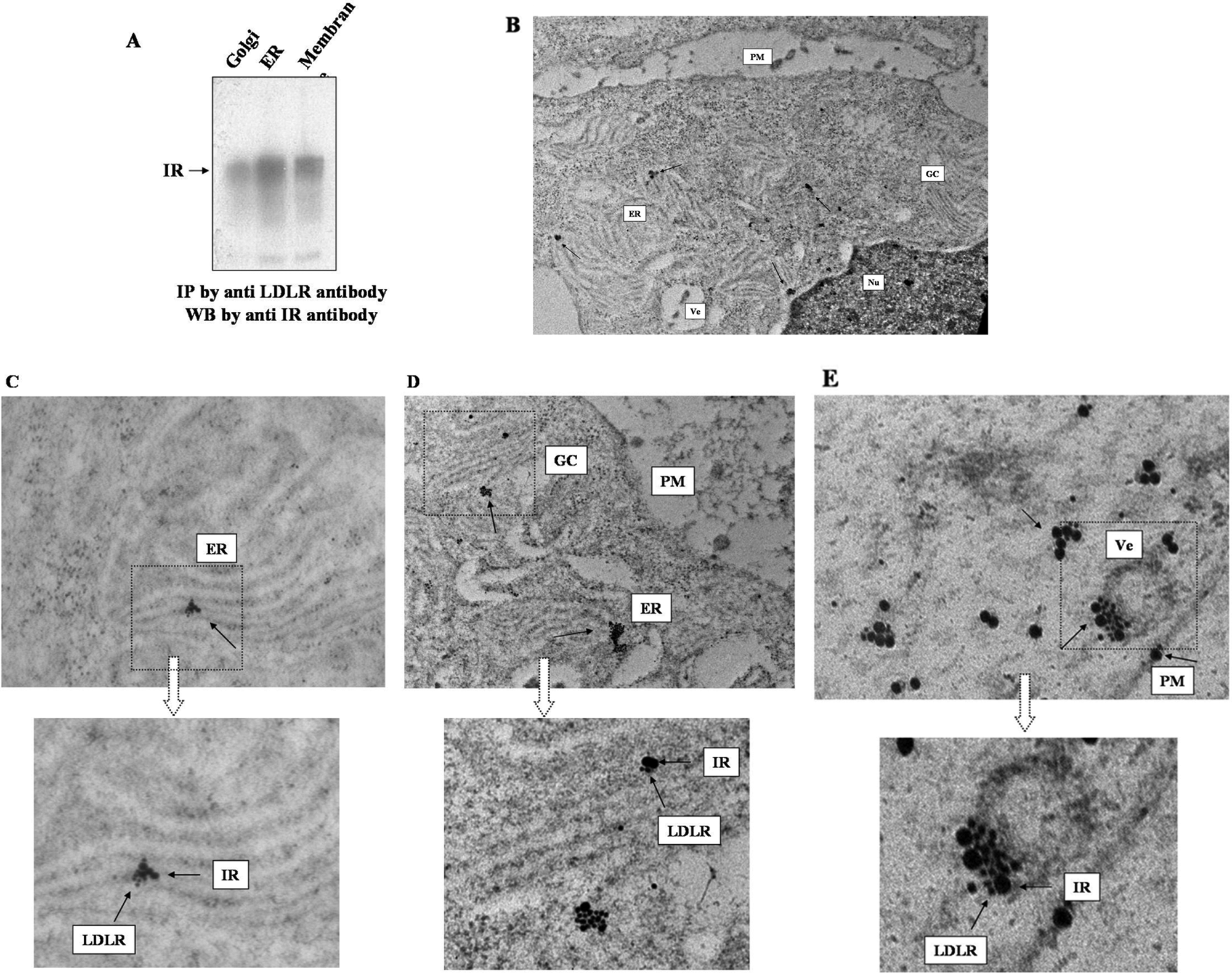
Co-association of low-density lipoprotein receptor (LDLR) and insulin receptor (IR) in sub-cellular organelles.
Electron microscopy (Figure 3, panels B, C, D and E) also showed the association of the two receptors in Golgi complex, endoplasmic reticulum, vesicle, plasma membrane and cytoplasm.
Dissociation of LDLR and IR from their co-association by insulin
We further investigated LDLR and IR co-association upon stimulation with insulin. HepG2 cells were labelled with 35S-Methionine and incubated with 15 µg/ml of insulin for 10 min. The concentration of insulin (15 µg/ml) and the incubation time (10 min) were chosen after validation experiments with various insulin concentrations at different time points (data not shown). LDLR and IR were subsequently immunoprecipitated from HepG2 cell lysate by anti-LDLR and anti-IRβ antibodies. SDS-PAGE was run at 0.1% SDS with 5% β-ME. The receptor bands were found at the region of their specific molecular weight (MW). In contrast to previous experiments, no co-association was observed at any higher MW region. This showed that insulin separates the two receptors, thus allowing for individual immunoprecipitation by respective antibodies (Figure 4).

Dissociation of low-density lipoprotein receptor (LDLR) and insulin receptor (IR) from their co-association by insulin.
Co-relation between LDLR expression and LDL uptake by insulin
Although there is a general consensus that increased receptor protein expression can increase functional activity, role of insulin on LDLR is an exception. Incubation of HepG2 cells (~2 × 105 cells/incubation) with 15 µg/ml insulin for 10 min did not show any increase in transcription (mRNA) or LDLR protein synthesis (Figure 5A and D; B: positive control for real-time PCR). However, incubation with Dil-LDL for 10 min in presence of 15 µg/ml of insulin showed a significant difference (threefold increase) in the functional activity of the LDLR (Figure 5C). Uptake of Dil-LDL by HepG2 cells during 10 min incubation with 15 µg/ml of insulin was found to be significantly higher than the uptake found in the absence of insulin (Figure 5C). This showed that increased uptake of Dil-LDL by insulin at 10 min was not dependent on gene expression of the LDLR protein (Figure 5A, D)), but was related to insulin-induced separation of LDLR from the co-association with IR (Figure 4). Insulin did also increase LDLR expression at 5 h, but only showed a marginal effect on the functional activity of LDLR (see Supplementary information Figure S4 for stimulatory effect of insulin on LDLR).
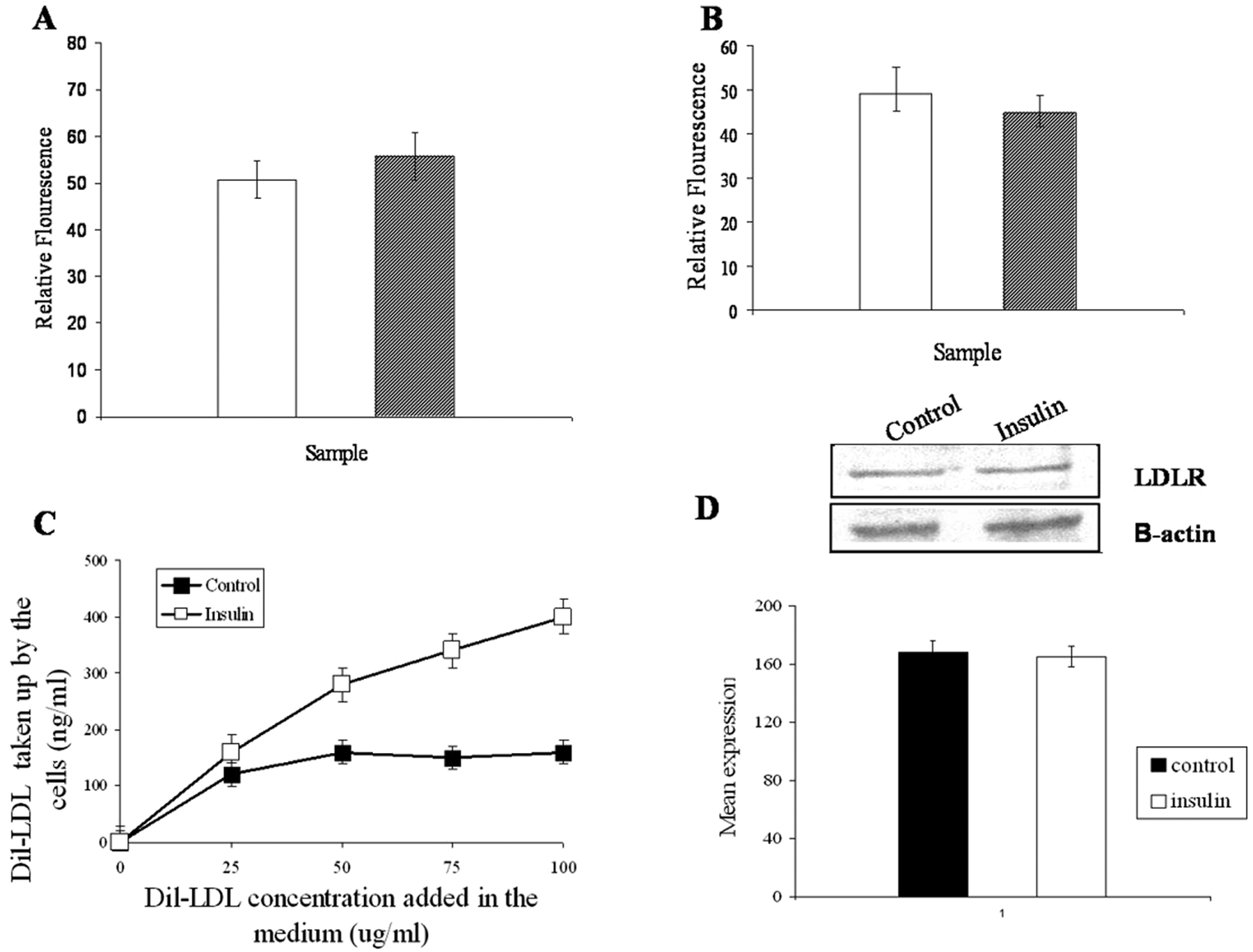
Low-density lipoprotein receptor (LDLR) expression and low-density lipoprotein (LDL) uptake by insulin.
Co-immunoprecipitation of LDLR-IR complex after knocking down mRNALDLR by siRNA treatment
Cells were treated with commercially available (Santa Cruz Biotech, CA, USA) LDLR-specific siRNA. Almost 55% inhibition of receptor expression was found at 10 nM concentration of siRNA. No further inhibition of receptor expression was observed, even after a threefold increase of siRNA concentration (Figure 6 panel A). The inter-variation of the integrated density value (IDV) in three experimental repeats is also shown. These concentrations of siRNA had no effect on cell viability. Therefore, 10 nM siRNA-treated cells were used, and LDLR/IR were immunoprecipitated by treating cell lysates with anti-LDLR/anti-IR antibodies, respectively. These immunoprecipitates were then cross-checked on immunoblot by anti-LDLR and anti-IRβ antibodies (Figure 6, panel B). In all cases, SDS-PAGE was run at 1% SDS and 5% β-ME, with siRNA-untreated cells used as controls. Immunoprecipitates of LDLR from control cells contained both LDLR and IR proteins, whereas bands were faint for LDLR and IR from siRNA-treated cells (Figure 6B1 and B2). A bright LDLR band was also found in the immunoprecipitates of IR-β from control cells but, only a trace of LDLR protein was detected in the immunoprecipitates of IR-β from siRNA-treated cells (Figure 6B3). The trace of either LDLR or IR band in siRNA-treated cells was only expected from 45% expressed LDLR and LDLR-associated IR because siRNA could only knock down 55% of total LDLR expression.
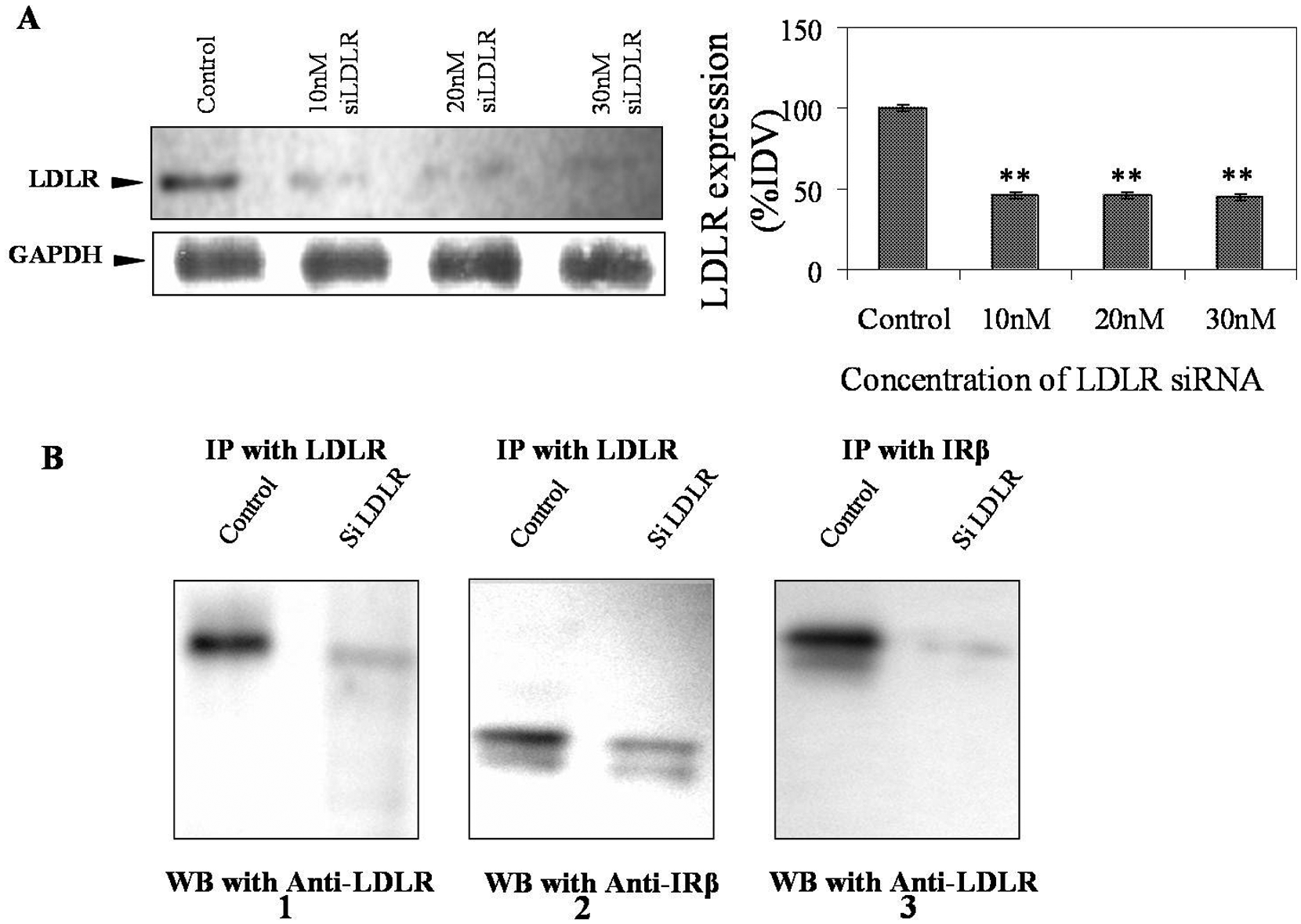
Co-immunoprecipitation of low-density lipoprotein receptor (LDLR) and insulin receptor (IR) from LDLR-specific siRNA-treated cells.
Discussion
The LDL receptor is one of the primary candidates involved in the regulation of plasma cholesterol level. 2 LDLR achieves its functional activity under strict regulation of the SREBP2-mediated feedback mechanism.35–37 Insulin has been found to stimulate cellular expression of LDLR as well as its functional activity. 23 Insulin deficiency or compromised activity by an impaired IR in conditions such as diabetes mellitus has been reported to modulate plasma LDL levels.8–10,38 Here, we report an intracellular co-association of IR and LDLR, and co-localisation on the plasma membrane. This association has been found to be a key modulator of insulin-mediated LDLR activation.
Our data suggest that the LDLR–IR complex becomes dissociated by a detergent such as SDS and remains undisturbed by reducing compounds such as β-ME, providing evidence that the binding force of the complex is non-covalent. Our data also show that at an extremely low detergent concentration (0.1% SDS) these two receptors immunoprecipitate together, whereas a higher detergent concentration (1% SDS) 32 disrupts their association (Figure 1). Studies with respect to other plasma membrane receptors (TNFR1) (Supplementary information Figure S1, S2) have shown that the association described above is specific. The existence of co-localisation of the two receptors in membrane invaginations (e.g. coated pits on plasma membrane) provides information of the existence of a co-localised precursor before endocytosis of LDL in biological cells (Figure 2b). The intracellular co-existence of the two receptors in sub-cellular compartments has also been evidenced by immunoprecipitation from organelles as viewed by electron microscopy (Figure 3). We have reported in our study that the two receptors were separated from their cohesion by insulin in a short time interval (Figure 4).
Although it is generally accepted that a highly expressed receptor could be also functionally more competent, the evidence obtained in the present study represents a deviation from such a belief. Even though only 10 min of incubation of HepG2 cells with 15 µg/ml insulin did not show any stimulation of LDLR expression (transcriptional or translational), the uptake rate of LDL (using fluorescent labelled Dil-LDL) by these LDLRs has been found to be significantly increased at 10 min with the same insulin concentration (Figure 5). This indicates direct modulation of receptor function by insulin rather than altered gene expression. The insulin-mediated generation of free LDLRs from the LDLR–IR co-localised complex is expected to internalise the extra-cellular LDL particles at a faster rate into the cells. Although there has been no excess receptor protein synthesis in 10 min by insulin, the availability of more free LDL receptors through the insulin-mediated disruption of the LDLR–IR complex could account for the significant difference in uptake rate of LDL by LDLR as observed in our study (Figure 5). Since insulin is an extra-cellular messenger5–7 and cannot enter the cell interior, such LDLR protein activation by insulin through the disruption of the LDLR–IR co-localised complex can occur only on the plasma membrane of the cell.
Knock down of LDLR by LDLR-specific siRNA also showed a reduced amount of immunoprecipitation of IR in association with LDLR (Figure 6). Although IR was not knocked down by IR-specific siRNA, the result of the converse effect of IR inactivation was verified in monocyte cells of subjects with diabetic ketoacidosis. An in vitro study 39 with cultured cells has shown that acetoacetate (which is produced in individuals with diabetic ketoacidosis) lowered cellular levels of IR and IR-mRNA by around 25% at 6 h. Also, confocal microscopy in similar studies in our laboratory has shown that the receptor complex of IR and LDLR was practically absent in the overnight sample of type-1 diabetic subject who had severe ketoacidosis (Figure S5, Supplementary material). The predominance of red colour showed the evidence of existence of only single (free) LDLRs.
The association of IR and LDLR may thus provide a mechanism for accelerated atherosclerosis in subjects with excess LDLR–IR co-associated forms. The association between the two receptors is reversible and can be modulated by insulin; when insulin is withdrawn the two receptors re-associate. 40 Lack of insulin, or insulin resistance in diabetic subjects, is expected to prevent the generation of free, functionally active receptors, hence LDLRs will remain unable to clear LDL, thereby increasing atherosclerotic risk. Since insulin is an extra-cellular messenger5–7 and cannot enter the cell interior, the functional activation of LDLR protein by insulin through the disruption of the LDLR–IR co-association can occur only on the plasma membrane of the cell. Although this study shows for the first time an association of the two receptors LDLR and IR, future work is needed to understand in more detail the factors that modulate dissociation and hence function of these receptors.
Footnotes
Acknowledgements
We are greatly thankful to Dr TK Das, Dr TC Nag for access to electron microscope. We are also thankful to Dr P Chattopadhyay and Mr Sanjay Kumar for providing the real-time PCR facility.
Funding
This work was supported with funds from the Department of Science and Technology (DST) (Project no: SR/SO/BB-44/2005, http://www.dst.gov.in/), Government of India, and GR was supported by a Senior Research Fellowship from the Council of Scientific and Industrial Research (CSIR), Government of India (Fellowship no: 10-2(5)/2003, ![]() ). The sponsors had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
). The sponsors had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest statement
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
