Abstract
Biodegradable magnesium composites are attractive materials for bone implant applications. This study compares synthetic (HAS) and natural (HAN) hydroxyapatite reinforcements in Mg1Ag composites regarding mechanical and corrosion performance. Microstructural analysis showed that reinforcements were distributed along grain boundaries as secondary phases. The addition of 10 wt-% HAS increased the hardness by 23.6%. Higher compressive strength was achieved with 5-10 wt-% HAN, reaching 187 MPa (65% improvement over Mg1Ag), while 5 wt-% HAS improved strength by ∼ 9% and enhancing ductility. Corrosion results indicated 15 wt-% HAS and HAN composites exhibited the most anodic Eocp (+36 mV and +31 mV vs. Mg1Ag). Potentiodynamic analysis identified 15 wt-% HAS as the most corrosion resistant (Ecorr: − 1210 mV, icorr : 127 μA), whereas HAN composites showed higher icorr (>304 μA) than the Mg1Ag. Finally, MTT assays confirmed all composites are cytocompatible with Saos-2 cells, showing no significant cytotoxic effects.
Keywords
Introduction
Magnesium (Mg)-based biomaterials have attracted considerable attention for biomedical applications due to their unique combination of biodegradability, biocompatibility, and suitable mechanical properties.1–8 Mg alloys naturally degrade in physiological environments, thereby eliminating the need for secondary surgical procedures to remove implants, unlike permanent materials such as titanium and stainless steel.2,4 This characteristic makes Mg-based materials particularly attractive for temporary biomedical devices, including bone plates, screws, and vascular stents. 3 Moreover, Mg is an essential mineral in the human body, playing a vital role in bone metabolism and enzyme activation.9,10 Unlike traditional metal implants that can cause toxicity or chronic inflammation, Mg alloys typically degrade into harmless byproducts. 1 Regarding the mechanical properties of Mg, its elastic modulus (41–45 GPa) 11 is closer to that of natural bone (∼10–30 GPa) 12 compared to traditional implant materials, such as titanium (∼110 GPa) 13 or stainless steel (∼200 GPa). 14 This lower rigidity reduces stress shielding, a common problem where stiffer implants cause surrounding bone to weaken due to a lack of mechanical loading. 15 Despite their advantages, Mg alloys face significant challenges, most notably rapid degradation; they corrode quickly in physiological environments, which can lead to premature implant failure. A second limitation is their mechanical strength, which is lower than that of titanium for high-load applications. To address these issues, researchers are developing surface coatings, utilising alloying elements such as Zn, Ca, and rare-earth metals, and applying bioactive coatings to fine-tune degradation rates and enhance mechanical properties.16–19
The addition of bioactive materials, such as hydroxyapatite (HA) derived from fish bones, can improve the osteogenic properties of Mg composites while potentially influencing their corrosion behaviour. For instance, studies have shown that the presence of HA can facilitate the formation of a protective layer that slows down the corrosion rate of Mg alloys by forming corrosion products like Mg(OH)2, which can act as a barrier against further degradation.20–22 This is particularly relevant in the context of Mg–Zn composites, where the incorporation of HA has been linked to an enhanced corrosion resistance due to the formation of a stable surface layer.20,22 For example, the addition of 10 wt-% of HA to a Mg–Zn/Al2O3 composite increased the compressive strength to ∼126 MPa and the Young's modulus of ∼35 GPa. In addition, the Ecorr shifted towards the anodic direction and icorr decreased after the addition of 15 wt-% of HA into the composite. 23 In another study, a Mg–6.5 wt-% Zn composite reinforced with 10 wt-% HA was fabricated using mechanical milling, and its corrosion performance was compared to pure Mg and a Mg–6.5 wt-% Zn composite. 24 The results revealed that the Ecorr of the composite reinforced with HA was −1.46 V, which is more positive than the other sample in Hank's solution. This improved corrosion performance was attributed to the formation of fibrous globular apatite on the surface of the composite. The influence of different quantities of HA on the micro-structure and properties of a Mg/3Al matrix was also examined, and it was reported that the addition of HA enhanced the mechanical properties. 25 The highest hardness (∼60 HV) and compressive strength (180 MPa) were obtained with the addition of 9 wt-% HA. Furthermore, the corrosion rate of the Mg/3Al composite decreased with the content of HA. Recently, it has been reported that the pure Mg with 5 wt-% of HA produced by an ultrasonic-assisted rheo-squeeze casting method exhibited a compression strength of ∼245 MPa and a hardness of ∼69 HV. 26
Natural hydroxyapatite (HAN) has become of significant interest to researchers due to its low-cost and superior biocompatibility compared to synthetic hydroxyapatite (HAS).27–31 However, the chemical composition of HAN plays a major role in determining its biological performance. 32 It has been reported that the introduction of secondary phases, such as those derived from fish bones, can improve mechanical properties while simultaneously contributing to corrosion resistance through the formation of a more complex micro-structure capable of withstand corrosive environments.33,34 Recent studies revealed that Mg–Zn–HAN (fish bone-derived) composites developed a micro-lamellar grain structure after 20 h of planetary ball milling. 35 Furthermore, composites containing 1 wt-% HAN exhibited a lower weight loss compared to those with 2 wt-% HAN in simulated body fluid (SBF). Vandana et al. 36 demonstrated that the Mg–HAN (fish bone-derived) produced by friction stir welding exhibited a strength 65 MPa lower than pure Mg. They also found that both the Ecorr and icorr of the Mg–HAN composite were lower than pure Mg in a 3.5 wt-% NaCl. The addition of varying quantities of HA slightly influences the hardness, according to the study conducted on Mg–HAN derived from cow bone. 37 It was reported that incorporating a minimum of 3 wt-% HAN and a maximum of 9.0 wt-% HAN into pure Mg resulted in hardness values of 50.56 and 49.70 HV, respectively. Additionally, the corrosion rate was found to be optimised with the addition of 5 wt-% HAN compared to other samples in the SBF.
Mechanical alloying is a solid-state powder processing technique that involves the repeated welding, fracturing, and re-welding of powder particles in a high-energy ball mill. 38 This process allows for the production of homogeneous materials with fine micro-structures and the capacity to incorporate alloying elements that are typically difficult to combine through conventional melting methods. The objective of this research is to compare the mechanical and corrosion performance of varying quantities of nano-sized HAS and HAN derived from Argyrosomus regius fish incorporated into a Mg–Ag (1 wt-%) biodegradable composite. In our earlier work, we demonstrated the fabrication of HAN powder from A. regius fish at a low cost. 30 Therefore, in the present work, 5, 10, and 15 wt-% of each reinforcement type were selected to produce Mg–Ag (1 wt-%)/HA biodegradable composite via mechanical mixing. The micro-structural alterations, thermal behaviours, mechanical properties, and corrosion performance of these composites were evaluated and are presented in this study.
Materials and methods
Material preparation
The nano-sized HAS powder (20–50 nm), silver powder (0.7–1.3 µm, 99.9%), and pure magnesium powder (325 mesh, 99.8% (assay)) were purchased from Alfa Aesar, respectively. While the HAN powder (average particle size ∼19.1 μm) employed in this study was calcined fish head bone source of the species A. regius. The chemical composition and preparation of the calcined fish head powder (HAN) was given in detail in the literature. 30 To prepare the composite given in Table 1, the powder mixture was prepared via mechanical mixing (model Spex 8000D) at room temperature. The process was carried out in a 420 stainless steel vial (inner diameter: 37.5 mm, height: 57 mm) using hardened steel balls. The grinding media consisted of two Ø12 mm and four Ø6 mm balls, totalling approximately 17.80 g, to achieve a ball-to-powder weight ratio of 10:1. To minimise oxidation, the powders and balls were loaded into the vial inside a glove box under a protective argon atmosphere. The mixing was performed for 5 min at 1060 cycles min−1; no process control agent was used due to the short duration and the mixing nature of the process.
The chemical composition of the composite (wt-%).
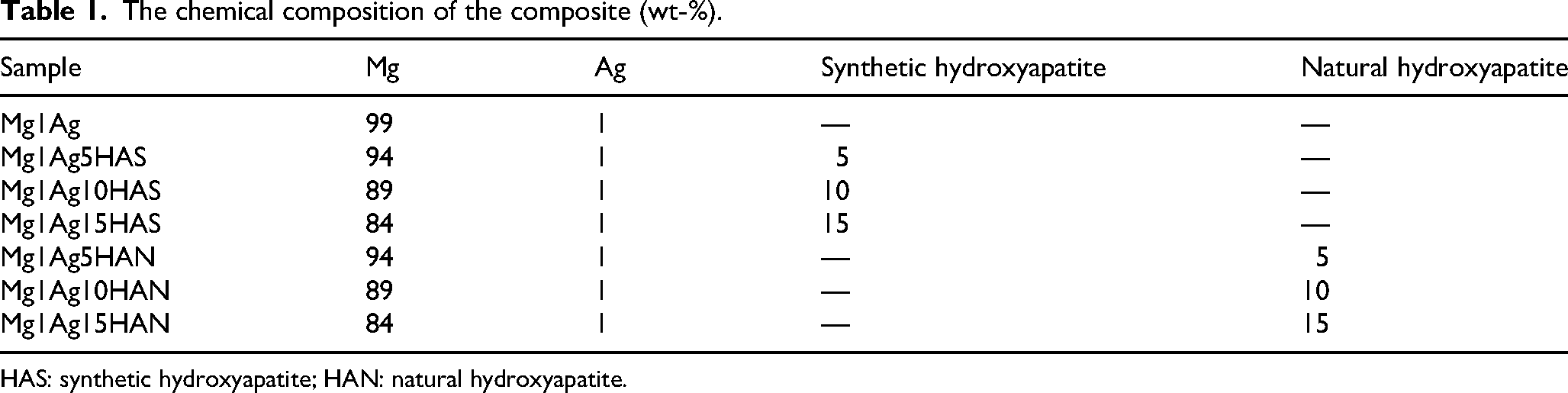
HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
Sintering was carried out in a horizontal tube furnace (Protherm) under a flowing argon atmosphere (100 mL min−1). The green compacts were placed in an Al2O3 ceramic crucible and heated at a constant rate of 5 °C min−1 up to 550 °C, held at this temperature for 3 h, and then furnace-cooled to room temperature under Ar. Before sintering, powder mixtures were uniaxially cold-pressed using a hydraulic press at 150 bar into cylindrical pellets of 10 mm diameter and 20 mm height. The peak temperature (550 °C) was selected to promote diffusion-controlled necking and densification while remaining below the melting point of Mg (650 °C), thereby avoiding melting-related shape loss, segregation, and micro-structural coarsening.
Material characterisation
The micro-structures of the samples were imaged using field-emission scanning electron microscopy (FESEM) and semi-quantitative analysis was performed using energy-dispersive spectroscopy (EDS) using Hitachi Regulus 8230. To find the crystal phase peaks a scan between 5° and 90° at a scan rate of 5° min−1 was performed using X-ray diffraction (XRD) model Panalytical Empyrean. Thermogravimetric and differential thermal analyses were carried out using a Perkin Elmer Diamond brand thermogravimetric/differential thermal analyser. It was used to determine the phase transitions from room temperature to 600 °C at a heating rate of 10 °C min−1. The weights of the samples were determined using a Precisa 125 A brand precision balance with a precision of ±0.0001 g. The apparent density after sintering was determined using the Archimedes principle; the theoretical density was determined according to the law of mixing and these were used in the calculation of relative density: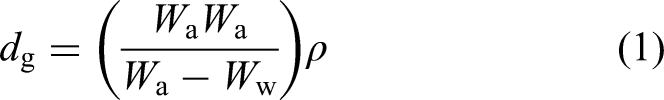

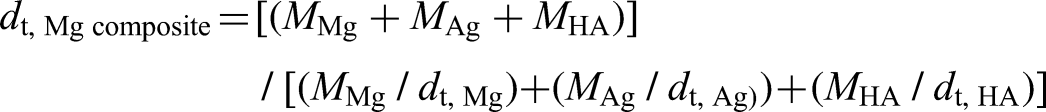
The theoretical density of the Mg composite (dt, Mg composite) is calculated using the masses and weight fractions of Mg, Ag, and HA, along with their respective theoretical densities (dt, Mg = 1.74 g cm−3, dt, Ag = 10.49 g cm−3, dt, HA = 3.05 g cm−3). For fish bone-derived (biological) HA, a reference theoretical density of 3.05 g cm−3 was used, which is commonly reported for biological/nanoscale HA and widely adopted for relative density calculations. 39
Mechanical test
Before hardness measurement, the composites (Ø12 mm × 20 mm) were cold-moulded and subsequently ground using 1200 grid SiC. The hardness of the composite was measured using a micro-hardness tester Future-Tech FM-800. In this regard, a force of 100 gf with a dwell time of 15 s was applied to the surface of the samples. The compressive test of the sample was carried out using a Universal Test Machine model Shimadzu AG-IS. According the ASTM-E9, the prepared sample test was achieved at a crosshead speed of 1 mm·min−1. Mechanical properties, including compressive strength, elastic modulus, and strain, were determined from the stress-strain curves.
Corrosion test
The electrochemical test was accomplished in SBF solution 40 at 35 °C in corrosion cells. The counter electrode and reference electrode were graphite rod and Ag/AgCl reference electrode (saturated KCl), respectively. The composites (Ø12 mm × 20 mm) were prepared by wiring with a conductive wire and mounted on cold epoxy resin. Then, the samples were ground with 1200 SiC adhesive paper using tap water. Then, all the samples were cleaned using distilled water and left to dry at room temperature. The corrosion test was executed as following: after immersion of samples in the corrosion solution, all the electrochemical tests were started after 15 min. The open circuit potential (OCP), potentiodynamic, and electrochemical impedance spectroscopy (EIS) tests were performed on the composite. The OCP test recorded the corrosion potential (Eocp) alteration for 1 h and the results are presented as Eocp versus time. The passivation behaviour, corrosion potential (Ecorr), and corrosion current density (icorr) were found using the potentiodynamic test. For this, a range of −2000 to 2000 mV at a scan rate of 1 mV·s−1 was chosen as potentiodynamic parameters. The last applied corrosion technique, EIS, was implemented to evaluate the impedance resistance of the composite. The following parameters were set to draw the Nyquist plots: an amplitude of 10 mV with a frequency range of 105 to 10−2 Hz. Furthermore, the electrical equivalent circuit was deduced by fitting the Nyquist curves. The tests were replicated at least three times.
In vitro cytotoxicity assessment of Mg composites using Saos-2 cells
The osteosarcoma cell line (Saos-2) was used to evaluate the cytotoxicity of Mg composites. Saos-2 cells were cultured in Dulbecco's modified Eagle's medium (DMEM, Gibco, Germany) supplemented with 10% foetal bovine serum (Gibco), 1% penicillin–streptomycin (Gibco), and 1% Glutamax (Gibco), under a humidified atmosphere containing 5% CO2 at 37 °C. Cytotoxicity analyses were performed by both indirect and direct contact methods. For the indirect contact method: Mg composites were incubated in 10 mL DMEM at 37 °C for 14 days to obtain extracts. At the end of the incubation periods (1, 3, 5, 7, and 14 days), the supernatants were collected, centrifuged at 500 g for 10 min, and stored at 4 °C until cytotoxicity testing. For control groups, DMEM was used as the negative control. Saos-2 cells were seeded at a density of 5 × 103 cells per well into 96-well culture plates and incubated for 24 h to allow cell attachment. Then, the extracts were added to the cells and incubated for 24, 48, and 72 h, after which MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was performed. For the direct contact method: equal-sized Mg composites were seeded with 5 × 104 cells and incubated for 72 h. At the end of each designated time point, the cells were detached from the composite surfaces, transferred to 96-well culture plates, and subjected to MTT assay following an additional 24 h incubation. For the MTT assay: in both methods, 10 µL of MTT solution was added to each well, and the plates were incubated at 37 °C for 2–4 h. Subsequently, 100 µL dimethyl sulfoxide was added to dissolve the formazan crystals, and the absorbance was measured at 570 nm using a micro-plate reader (Biotek, USA). 41
Results and discussion
Density analysis of the composites
The reduction in density of reinforced samples is commonly attributed to particle agglomeration, the formation of interfacial voids, gas entrapment, and poor wettability between the Mg matrix and ceramic particles. In this study, the density of the composites was evaluated by comparing theoretical and measured values (Figure 1). As expected, the theoretical density exhibited a linear increase with a higher reinforcement content, reflecting the higher intrinsic density of HA compared to the magnesium matrix. However, the experimental results revealed a distinct trend; while densification improved up to 10 wt-% reinforcement, a significant decline in both apparent and relative density was observed at 15 wt-%. This reduction is most pronounced in the Mg1Ag15HAN sample, suggesting that HAN may be more prone to processing challenges at higher concentrations.
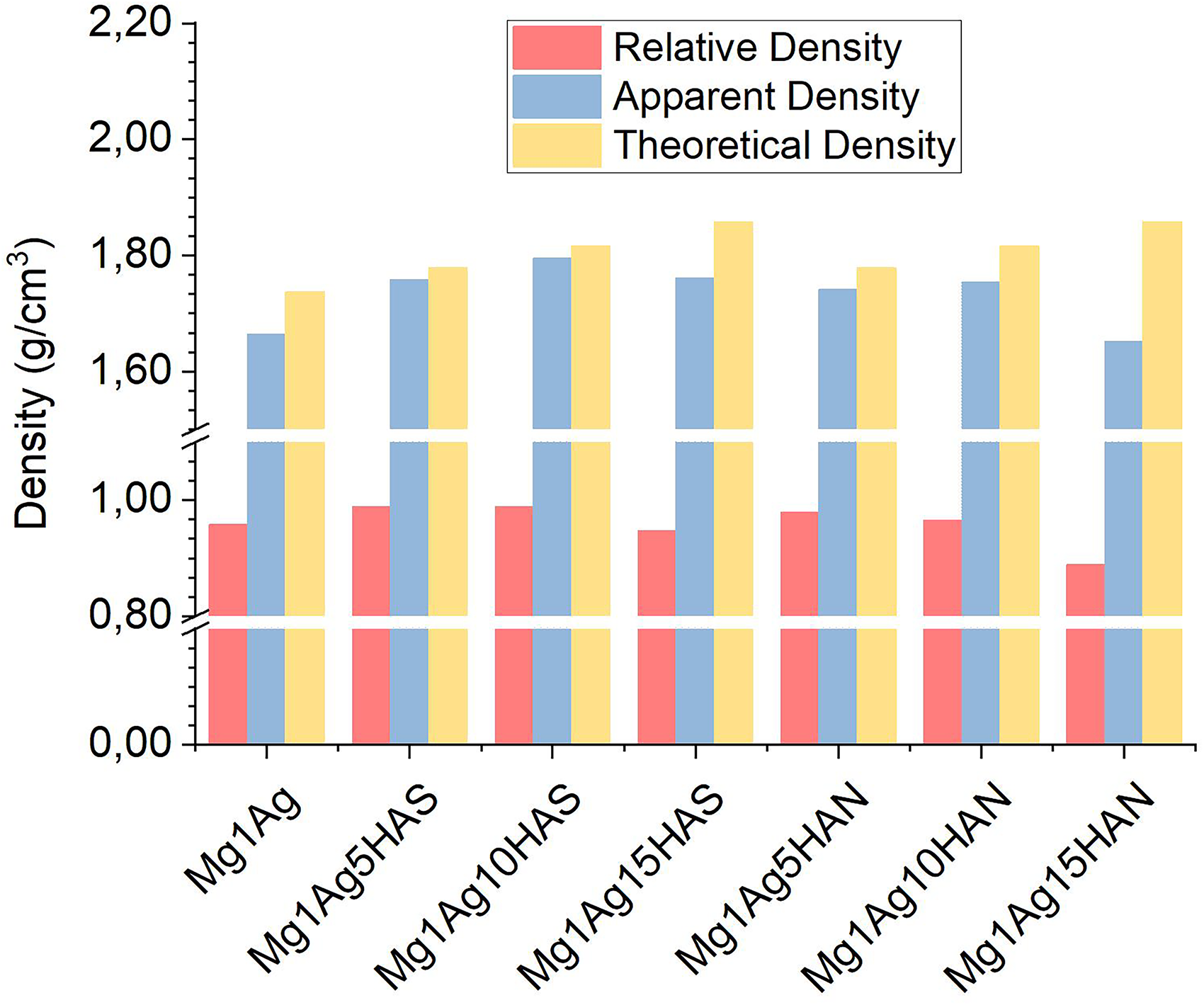
Relative, apparent and theoretical density of Mg1Ag, Mg1Ag–HAS and Mg1Ag–HAN composites. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The increasing ratios of both HAS and HAN reinforcements tend to decrease density, with Mg1Ag15HAS and Mg1Ag15HAN exhibiting the lowest relative densities. This reduction is most pronounced in the Mg1Ag15HAN sample, suggesting that HAN may be more prone to processing challenges at higher concentrations. The divergence between theoretical and measured values at high loading levels is primarily attributed to particle agglomeration, interfacial void formation, and gas entrapment during the powder metallurgy process. It was reported that the theoretical densities of pure Mg and MHA (Mg + 5 wt-% HA) were calculated as 1.73 and 1.79 g·cm−3, respectively. However, the experimentally measured densities after rheo-squeeze casting with ultrasonic assistance were found to be 1.73 and 1.71 g·cm−3, respectively. The slight discrepancies between theoretical and experimental densities are likely associated with particle agglomeration and the presence of residual porosity formed during processing. 26 On the other hand, the addition of HA at small quantities such as 0.5, 1.0, and 1.5 wt-% into the Mg–Zn–Ti alloy increased experimental density. 42
Micro-structure analysis of the composites
The FESEM analysis highlights the effects of natural and synthetic hydroxyapatite additions on the microstructural evolution of Mg1Ag composite following mechanical mixing and sintering (Figure 2). The unreinforced Mg1Ag sample exhibits a coarse-grained structure with moderate porosity (Figure 2(a)). The addition of 5 wt-% natural hydroxyapatite from fish bone (HAN) in the Mg1Ag5HAN sample leads to secondary phase in structure (Figure 2(b)). However, milling-induced striations are still visible, suggesting incomplete recrystallisation and the presence of residual deformation from the mechanical mixture process. Moreover, the porosity slightly increases compared to the unreinforced alloy, likely due to the HAN particles partially hindering densification during sintering. In contrast, the addition of 5 wt-% HAS (Mg1Ag5HAS) results in a much cleaner and more homogeneous micro-structure (Figure 2(c)). The grains are finer and more equiaxed, with no visible milling traces, and porosity is significantly reduced. These observations suggest that HAS enhances diffusion and sintering behaviour more effectively than HAN, possibly due to better chemical purity, uniformity, or particle morphology. At higher reinforcement levels (15 wt-%), the differences between HAN and HAS become even more pronounced. The Mg1Ag15HAN sample shows a dense distribution of the secondary phase particles with clear agglomerations and increased porosity (Figure 2(d)). These voids were also observed in the fabricated Mg–12.5 wt-% HA–10 wt-% MgO nano-composite. 31 These defects are typical when large amounts of natural particles, potentially containing impurities or irregularities, are introduced into the matrix, leading to incomplete particle dispersion and stress concentration zones. On the other hand, the Mg1Ag15HAS sample exhibits a more uniform distribution of the secondary phases with fewer and smaller agglomerates (Figure 2(e)). Although porosity is still higher compared to the 5 wt-% addition, HAS demonstrates better compatibility with the matrix even at higher concentrations, resulting in a more stable and continuous micro-structure.

FESEM image of (a) Mg1Ag (b) Mg1Ag5HAN (c) Mg1Ag5HAS (d) Mg1Ag15HAN (e) Mg1Ag15HAS. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The EDS results demonstrate that the composite maintains a homogeneous magnesium-rich matrix with slight oxygen enrichment and minimal silver content, aligning with the nominal Mg1Ag composition. The good dispersion of elements, particularly the lack of large agglomerates of silver, suggests effective mechanical mixing and homogeneous sintering of the material (Figure 3). The presence of oxygen suggests the formation of a native oxide layer (likely MgO) on the surface, which is common for magnesium alloys exposed to air. Silver is detected at a very low concentration of 0.08 wt-%, indicating that silver is either finely dispersed within the matrix or present at concentrations below the detection limit in some areas. Concerning the Mg1Ag15HAS composite, the EDS confirm the successful incorporation of HAS at a 15 wt-% reinforcement level (Figure 4). Magnesium remains the dominant element (94.24 wt-%), reflecting the matrix phase. A moderate content of oxygen (4.60 wt-%) is detected, which can be attributed to both a thin native MgO layer and the oxygen from the HA structure itself. The detection of calcium (0.07 wt-%) and phosphorus (0.01 wt-%) in the scanned region shows the presence of HAS nano-particles dispersed within the matrix. Given the nanoscale size and high distribution uniformity, these elements are not expected to appear in large localised concentrations, which is consistent with the fine, widespread signals observed in the elemental map. The uniform distribution and the absence of large HAS clusters suggest that mechanical mixing and sintering processes were effective in embedding the nano-particles into the magnesium matrix without severe particle aggregation. In contrast, the Mg1Ag15HAN composite, reinforced with natural fish bone-derived HA, exhibited higher oxygen levels (17.88 wt-%) and slightly increased Ca (1.03 wt-%) and P (0.31 wt-%) signals (Figure 5). This suggests that the HAN particles, which tend to be larger and less pure than synthetic ones, introduced more oxygen and localised reinforcement zones within the matrix. The elemental mapping showed a somewhat more heterogeneous distribution of Ca and P compared to the HAS composite. This variability likely arises from the inherent compositional and morphological differences of biogenic HA, such as residual organic matter and irregular particle shapes.
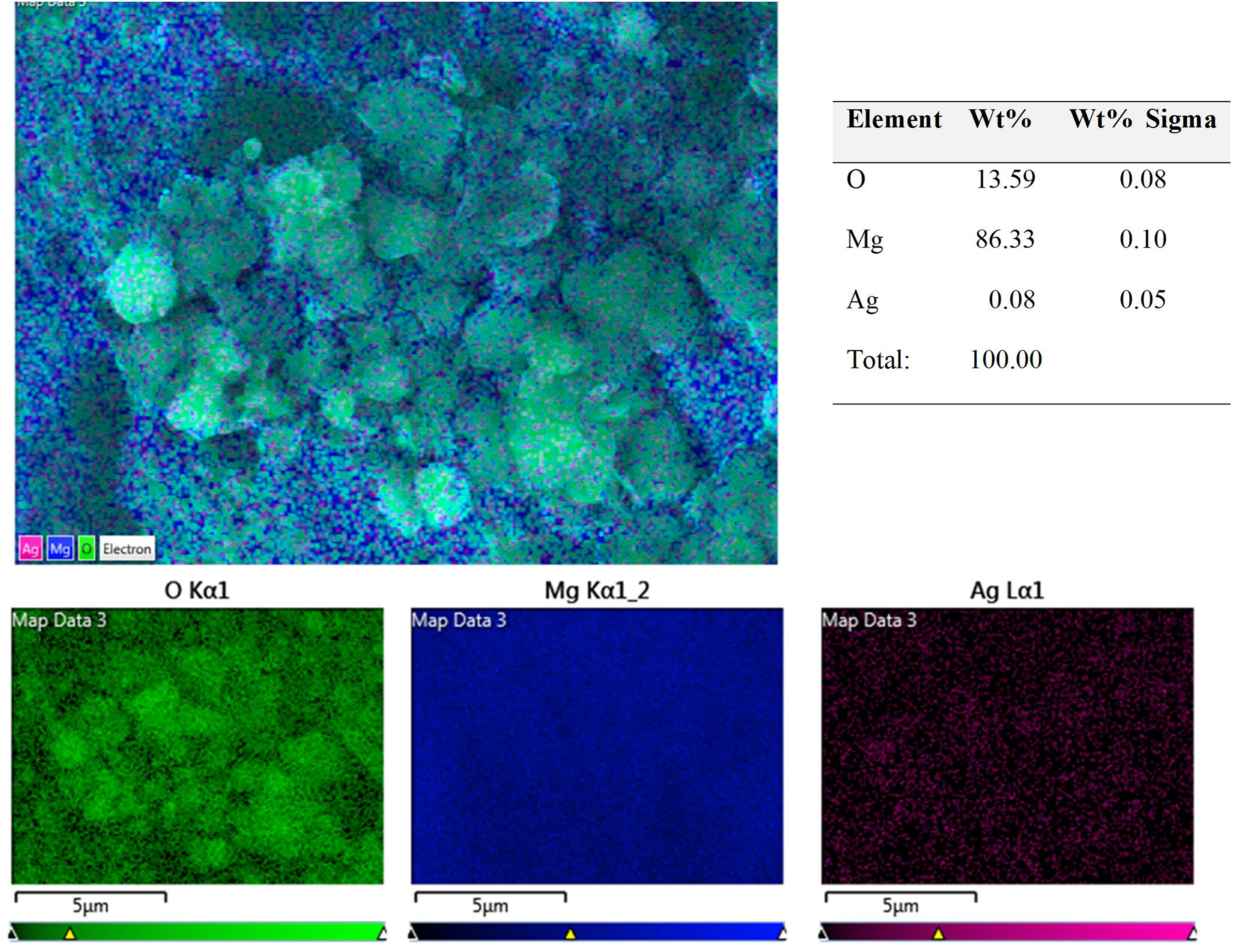
EDS mapping of Mg1Ag sample showing the Mg, O, and Ag dispersion in the composite.

EDS mapping of Mg1Ag15HAS sample showing the Mg, O, Ag, Ca, and P dispersion in the composite. HAS: synthetic hydroxyapatite.

EDS mapping of Mg1Ag15HAN sample showing the Mg, O, Ag, Ca, and P dispersion in the composite. HAN: natural hydroxyapatite.
The micro-structural analysis of the Mg1Ag15HAN composite, fabricated via powder metallurgy, reveals a heterogeneous distribution of elements and reinforcement phases within the magnesium matrix (Figure 6). High-magnification SEM imaging of the matrix, as shown in (Figure 6(a)), clearly depicts the presence of the secondary phase particles; the corresponding EDS spectrum 13 confirms these as HAN through the distinct signals of calcium (Ca) and phosphorus (P). The successful incorporation of the alloying element is evidenced in Figure 6(b), where spectrum 19 provides definitive proof of silver (Ag) presence with a significant weight percentage detected in the localised region. Furthermore, micrograph (Figure 6(c)) illustrates the characteristic localisation of the 15 wt-% calcined fish bone-derived micro-HA at the grain boundaries. The elemental signature in spectrum 8 validates the presence of Ca and P at these grain boundaries, suggesting that the HAN particles act as physical barriers that pin the grain boundaries during the consolidation process, a common phenomenon in powder metallurgy-prepared magnesium–ceramic composites.22,43
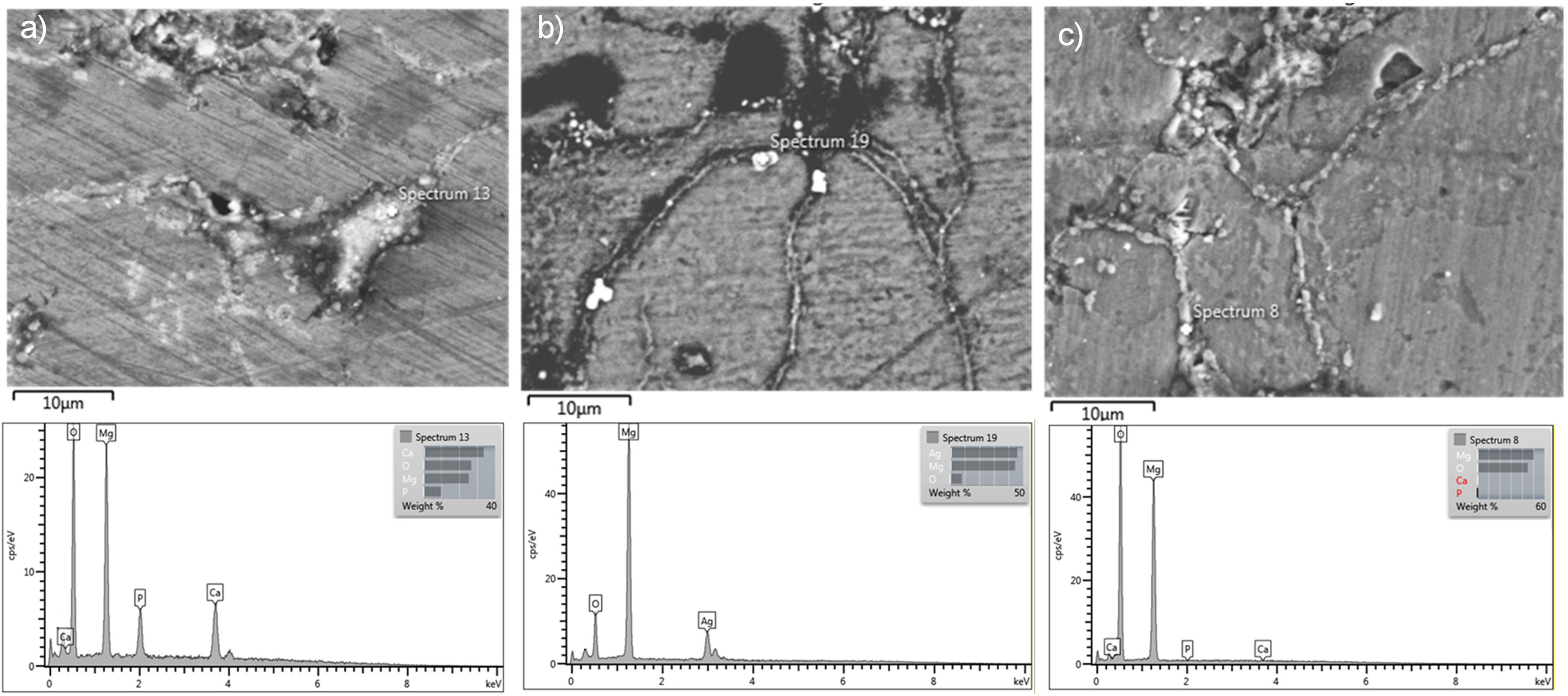
Micro-structural and elemental characterisation of the Mg1Ag15HAN composite: (a) distribution of the secondary hydroxyapatite (HAN) phase within the magnesium matrix; (b) verification of the silver (Ag) alloying element; and (c) localisation of micro-hydroxyapatite particles at the grain boundaries. HAN: natural hydroxyapatite.
The XRD patterns of the Mg1Ag composites reinforced with varying amounts of HAS and fish bone-derived HAN reveal key differences in phase formation and crystallographic characteristics (Figure 7). For all the samples, the dominant peaks correspond to Mg (32°, 34°, and 36°), confirming that magnesium remains the primary phase after mechanical mixing and sintering. These peaks correspond to the (100), (002), and (101) planes, respectively. Peaks associated with the Ag (38.1° and 44.3°) and Ag0.03Mg0.97 inter-metallic phase (near of Mg peaks) were also detected, indicating that silver successfully diffused and formed inter-metallic compound within the Mg matrix. In the HAS-reinforced samples, distinct peaks corresponding to HA (29.7° and 32.2°) are observed, becoming more prominent with increasing HA content. The HAS's nanoscale nature seems to enhance its crystallinity and visibility in the XRD patterns. Furthermore, MgO peaks (36.9°, 42.9°, 62.3°) are detected, suggesting partial surface oxidation during processing, though they remain relatively minor. Notably, the HAS samples show sharper and more defined HA peaks, indicating better crystalline reinforcement phases. In the HAN-reinforced samples, HA peaks are also present but appear broader and less intense compared to the HAS samples. This suggests that the HAN, obtained from fish bone, possesses lower crystallinity and more structural imperfections, likely due to its biogenic origin and calcination process. Interestingly, the HAN samples also show minor peaks of Mg3P2 (27.3° and 47.5°) and a magnesium phosphide phase, especially noticeable in Mg1Ag10HAN. This phase formation is attributed to the reaction of phosphorus present in the HAN with magnesium during sintering, which is less evident in the HAS composites.
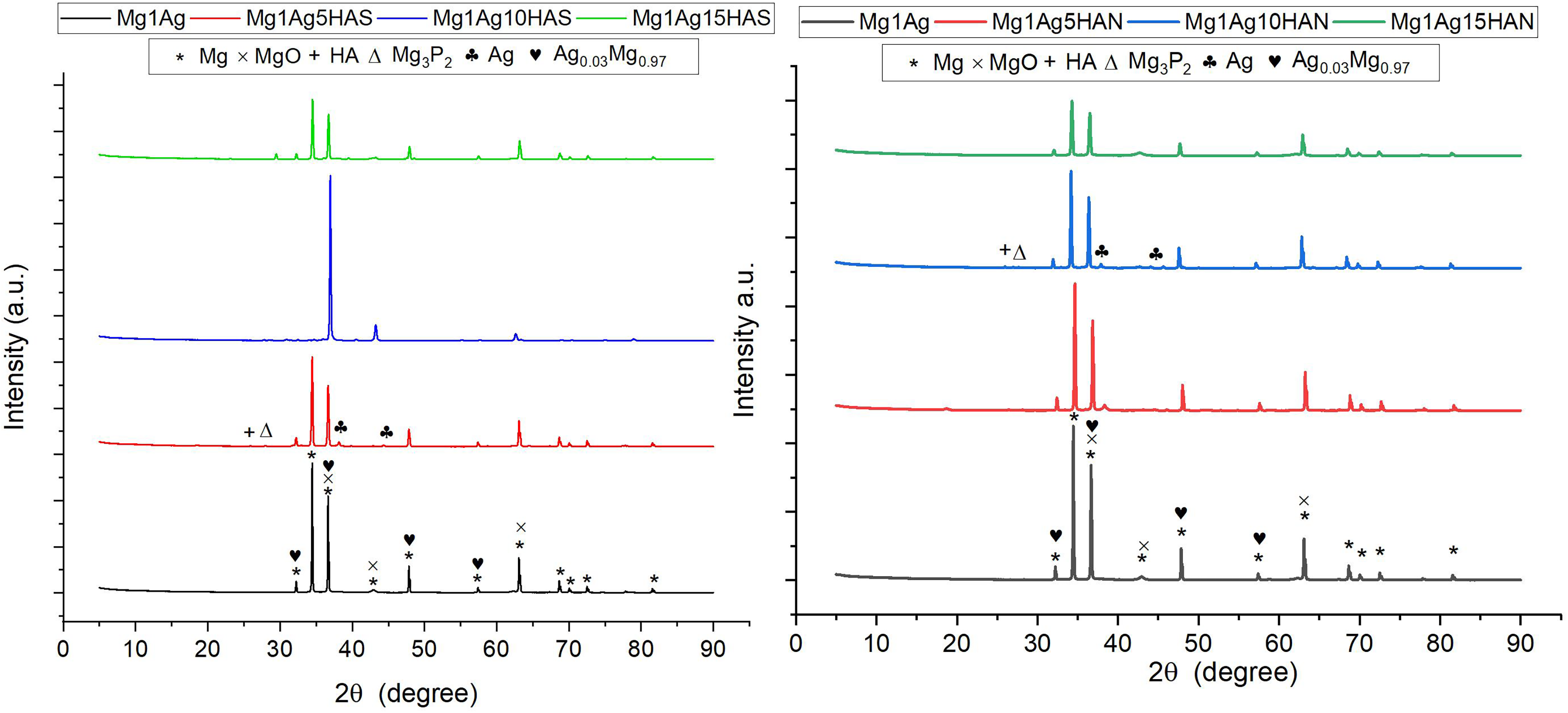
XRD spectra of Mg1Ag, Mg1Ag–HAS and Mg1Ag–HAN composites. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
According to the Differential Thermal Analysis (DTA) results (Figure 8(a)), it was observed that the HAS contribution caused a significant decrease in the spinning temperature and phase transition temperatures of the alloy and the amount of energy absorbed during these transitions. This effect was observed clearly, especially in the samples containing 15% HAS and HAN. In the DTA analyses, while the sample containing 5% HAS and HAN showed higher temperature and enthalpy changes, it was determined that thermal stability increased and phase transition temperatures decreased as the HAS and HAN ratio increased. Thermogravimetric analyses revealed that high HAS and HAN content balances the mass change rate by increasing the oxidation resistance of the alloy (Figure 8(b)). For example, 15% HAS-doped Mg1Ag alloy exhibited the least mass change, showing higher oxidation resistance. This situation shows that HAS additive enhances thermal stability and provides a more stable structure for Mg1Ag alloy at high temperatures. These findings show that the thermal behaviour of Mg1Ag alloys to be used in biomedical implant materials can be optimised according to the HAS and HAN additive ratio. The thermal stability and corrosion resistance of the Mg–Ag alloy increases with increasing HAS and HAN ratio, indicating the potential to improve the performance of the material in applications such as biomedical and dental implants. Therefore, the thermal and chemical stability effects of HAS and HAN addition on Mg–Ag alloys provide important information for the development of alloys to be used in biomaterials and other industrial applications.
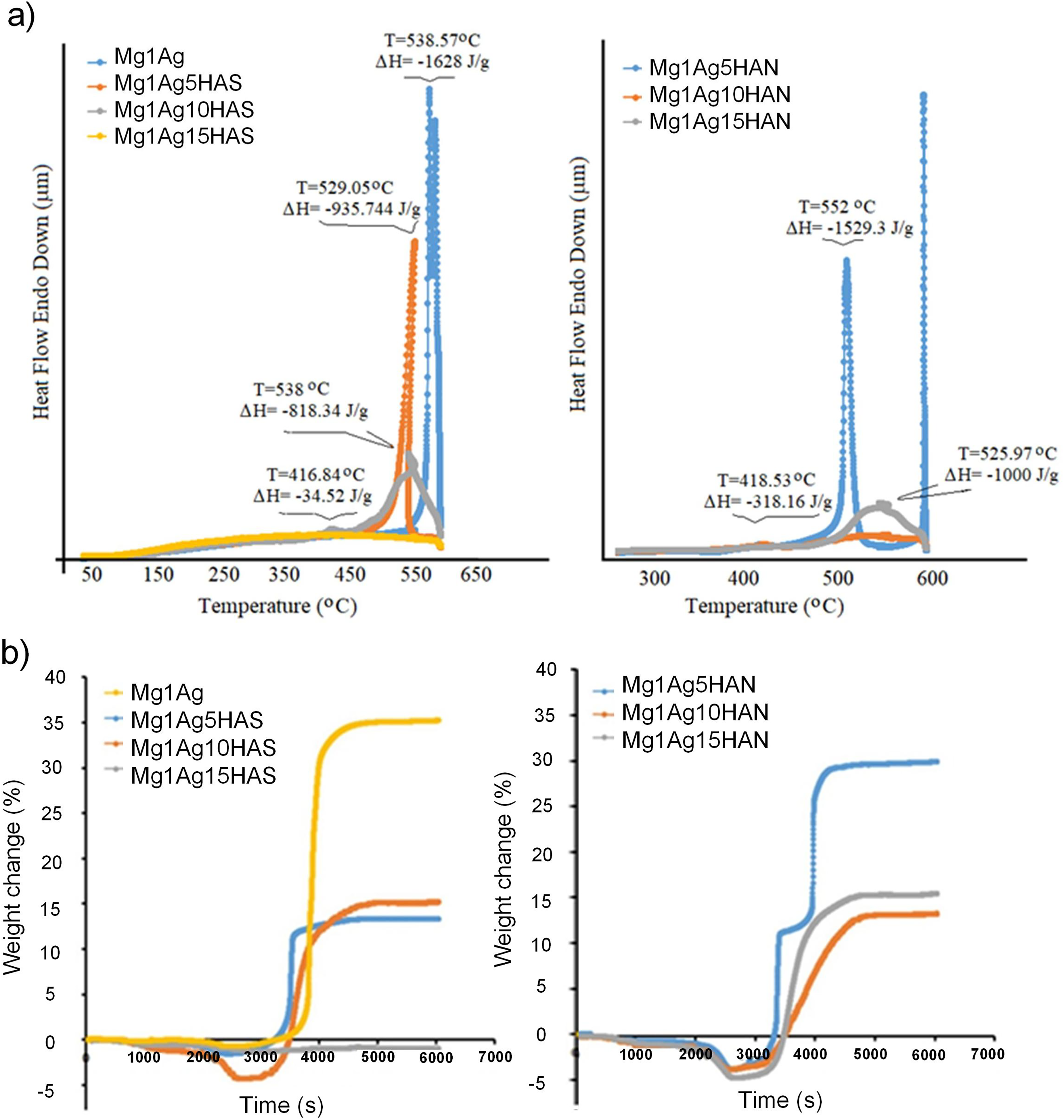
DTA (a) and TG (b) graphs of Mg1Ag, Mg1Ag–HAS, and Mg1Ag–HAN composites. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
Mechanical analysis of the composites
The effect of HA addition on the hardness of mechanical mixed Mg–Ag composites was investigated by incorporating two types of HA: HAS and natural fish bone-derived HA (HAN) (Figure 9). The baseline Mg1Ag alloy exhibited a hardness of 55 HV. Upon the addition of HAS, a significant enhancement in hardness was observed, particularly at 10 wt-% HAS, where the hardness reached a maximum value of 68 HV. This improvement can be attributed to the homogeneous dispersion and strong interfacial bonding between the HAS particles and the Mg–Ag matrix, which effectively hindered dislocation motion during deformation. Moreover, this composite was slightly 8 HV higher than Mg–3Al–9% HA composite. 25 However, at 15 wt-% HAS, a slight reduction in hardness to 61 HV was observed, likely due to particle agglomeration at higher reinforcement contents. In contrast, the addition of HAN consistently led to a decrease in hardness compared to the unreinforced alloy, with the lowest value recorded at 47 HV for the Mg1Ag10HAN sample. The inferior performance of HAN-reinforced composites may be related to the presence of irregular particle morphology, and weaker particle–matrix interactions. According to the Hall–Petch relationship, 42 the reduction in grain size significantly contributes to the observed increase in hardness, particularly in the Mg1Ag10HAS sample, which reached a peak of 68 HV. This hardness was ∼8 HV higher compared to Mg/3Al base reinforced with nano-sized 9 wt-% of HA. 25 The HA particles effectively act as obstacles to grain boundary migration (Figure 2) during sintering. However, at 15 wt-% reinforcement, the refinement effect is likely countered by particle agglomeration and increased porosity, as evidenced by the subsequent drop in both relative density and hardness.
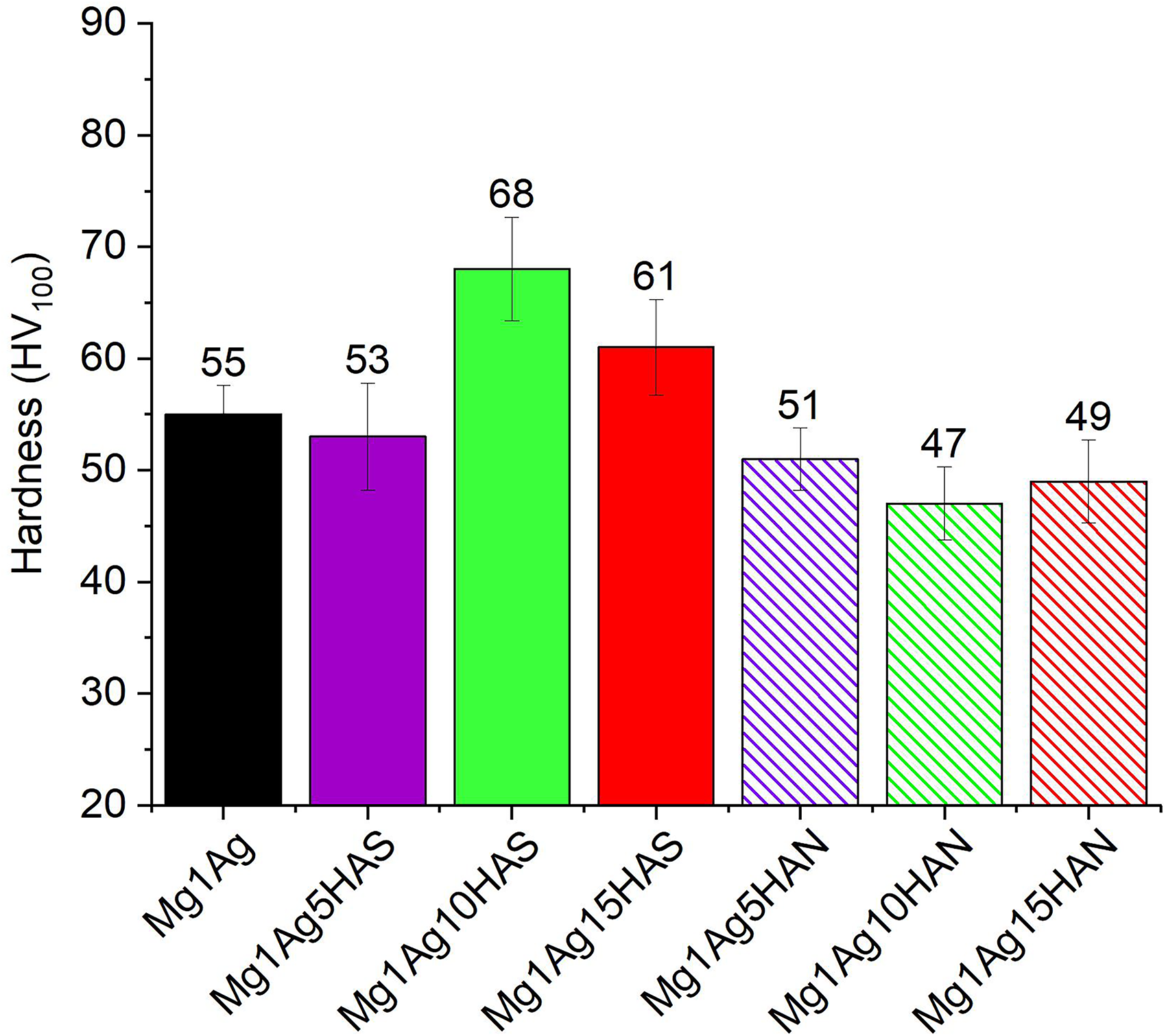
The micro-hardness values of Mg1Ag, Mg1Ag–HAS, and Mg1Ag–HAN composites. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The compressive mechanical behaviour of Mg–Ag composites reinforced with HAS and HAN was systematically investigated, and the results are presented in Figures 10 and 11. For the Mg1Ag base alloy, a moderate compressive strength was observed, reaching around 121 MPa with notable ductility (Figure 10(a)). The incorporation of 5 wt-% HAS (Mg1Ag5HAS) led to a significant enhancement in compressive strength, peaking at approximately 134 MPa (Figure 11), alongside a substantial increase in strain capacity (over 30%), indicating improved energy absorption before failure. This enhancement is primarily attributed to the distribution of inter-metallic phases at the grain boundaries, which impeded dislocation movement, thereby improving both strength and ductility. 42 However, increasing the HAS content to 10 wt-% (Mg1Ag10HAS) and 15 wt-% (Mg1Ag15HAS) resulted in a decline in both compressive strength and ductility, attributed to particle agglomeration and stress concentration points promoting early failure. 44 The post-compression images reveal severe cracking and fragmentation in samples with higher HAS content (Figure 10(a)). The compressive strength of the Mg–1Ag–10HAS composite was found to be 66 MPa lower than that of the Mg–3Al matrix reinforced with 9 wt-% nano-HA, a discrepancy likely stemming from the varying reinforcement efficiency of HA within the Mg–Al system. 25 Similarly, the Mg–1Ag–15HAS sample exhibited a strength of 19 MPa below the value reported for the Mg–Zn composite doped with 15 wt-% HA. 23 In contrast, composites reinforced with fish bone-derived HAN exhibited different behaviour. While the Mg1Ag5HAN and Mg1Ag10HAN samples showed improved compressive strength compared to the base alloy, higher additions of HAN (Mg1Ag15HAN) caused a noticeable reduction in compressive performance (Figure 10(b)). Mg1Ag5HAN and Mg1Ag10HAN demonstrated the highest strength among HAN composites (187 MPa), yet suffered from brittle fracture at lower strains (10.2% and 7.29%, respectively) (Figure 11). It has also been reported in the literature that reducing the HA content from 27.5 to 12.5 wt-% leads to an increase in compressive failure strain from 4.2% to 11.5%. 31 The brittle nature of HAN composites was visually confirmed by sharp, catastrophic cracks and complete disintegration of some samples after testing, as shown in Figure 10(b). This behaviour is likely due to the intrinsic structural imperfections within the HAN, weakening the bonding with the Mg matrix.
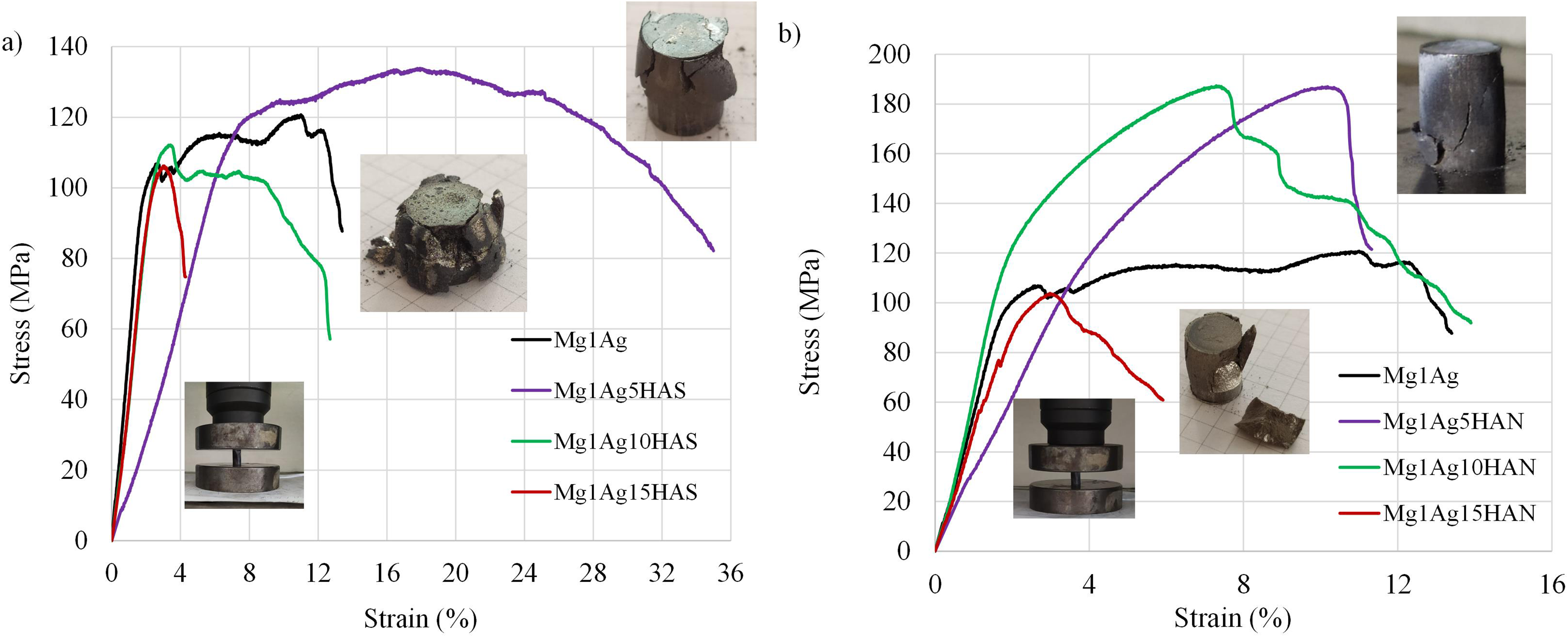
The compression test curves of Mg1Ag, Mg1Ag–HAS (a) and Mg1Ag–HAN (b) composites. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
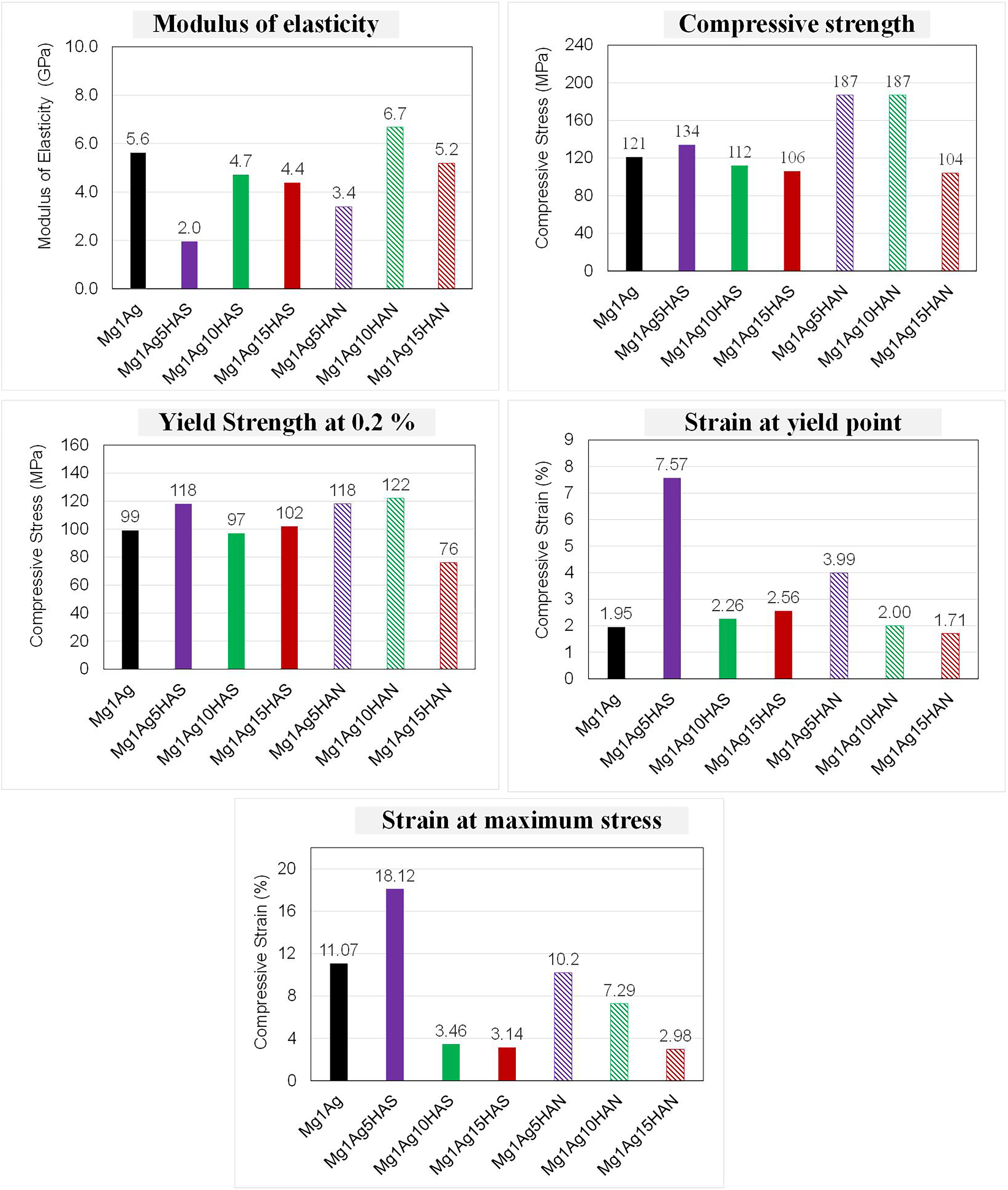
Mechanical properties of Mg-based composites reinforced with 1 wt-% Ag and varying contents (5%, 10%, 15%) of synthetic hydroxyapatite (HAS) or natural hydroxyapatite derived from fish bone (HAN): (a) modulus of elasticity, (b) compressive strength, (c) yield strength at 0.2% strain, (d) strain at yield point, and (e) strain at maximum stress. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The deeper analysis of the tensile curves showed that the modulus of elasticity was highest for the Mg1Ag10HAN composite (6.7 GPa), indicating a notable improvement in stiffness compared to the base Mg1Ag alloy (5.6 GPa), while the lowest modulus was observed in Mg1Ag5HAS (2.0 GPa), suggesting limited reinforcement effectiveness of HAS at lower content (Figure 11). Compressive strength followed a similar trend, peaking at 187 MPa for both Mg1Ag5HAN and Mg1Ag10HAN, representing a substantial increase over the unreinforced Mg1Ag (121 MPa). A similar behaviour was observed for Mg–HA; it increased from ∼140 MPa (3% HA) to ∼175 MPa (9% HA). 25 However, the strength dropped to 104 MPa in Mg1Ag15HAN, indicating that excessive HAN content may lead to particle agglomeration or interfacial weakening. Yield strength at 0.2% strain also improved with HAN addition, reaching 122 MPa in Mg1Ag10HAN, whereas the lowest value was found in Mg1Ag15HAN (76 MPa), further confirming the negative impact of overloading HAN (Figure 11). In terms of ductility, HAS-reinforced samples showed superior performance. Mg1Ag5HAS demonstrated the highest strain at both yield point (7.57%) and maximum stress (18.12%), highlighting its ability to sustain plastic deformation. Conversely, HAN additions, particularly at higher levels, significantly reduced ductility, with Mg1Ag15HAN exhibiting the lowest strain values (1.71% at yield and 2.98% at maximum stress).
Corrosion performance of the composites
The corrosion performance of HAS- and HAN-doped magnesium alloys was evaluated using electrochemical techniques. At first glance, the unreinforced Mg1Ag alloy exhibited the most negative and stable potential, indicating the highest susceptibility to corrosion. The addition of HAS notably improved the corrosion resistance of the composites; the Eocp of HAS incorporated into the Mg composites were slightly anodic compared to Mg1Ag (Figure 12). The Mg1Ag15HAS sample showed significantly more anodic potentials, reflecting a reduction in corrosion activity due to the protective effect of the ceramic phase. Notably, the Mg1Ag10HAS sample initially exhibited a potential similar to the uncoated alloy, but gradually shifted towards a more positive value over time, suggesting the formation of a stable and protective surface layer, likely consisting of HA-derived compounds or corrosion products that reduced ion exchange with the electrolyte. The Eocp of Mg1Ag15HA, Mg1Ag10HA, Mg1Ag5HA, and Mg1Ag after 1 h of immersion in the corrosive solution was −1462, −1469, −1470, and −1498 mV, respectively.
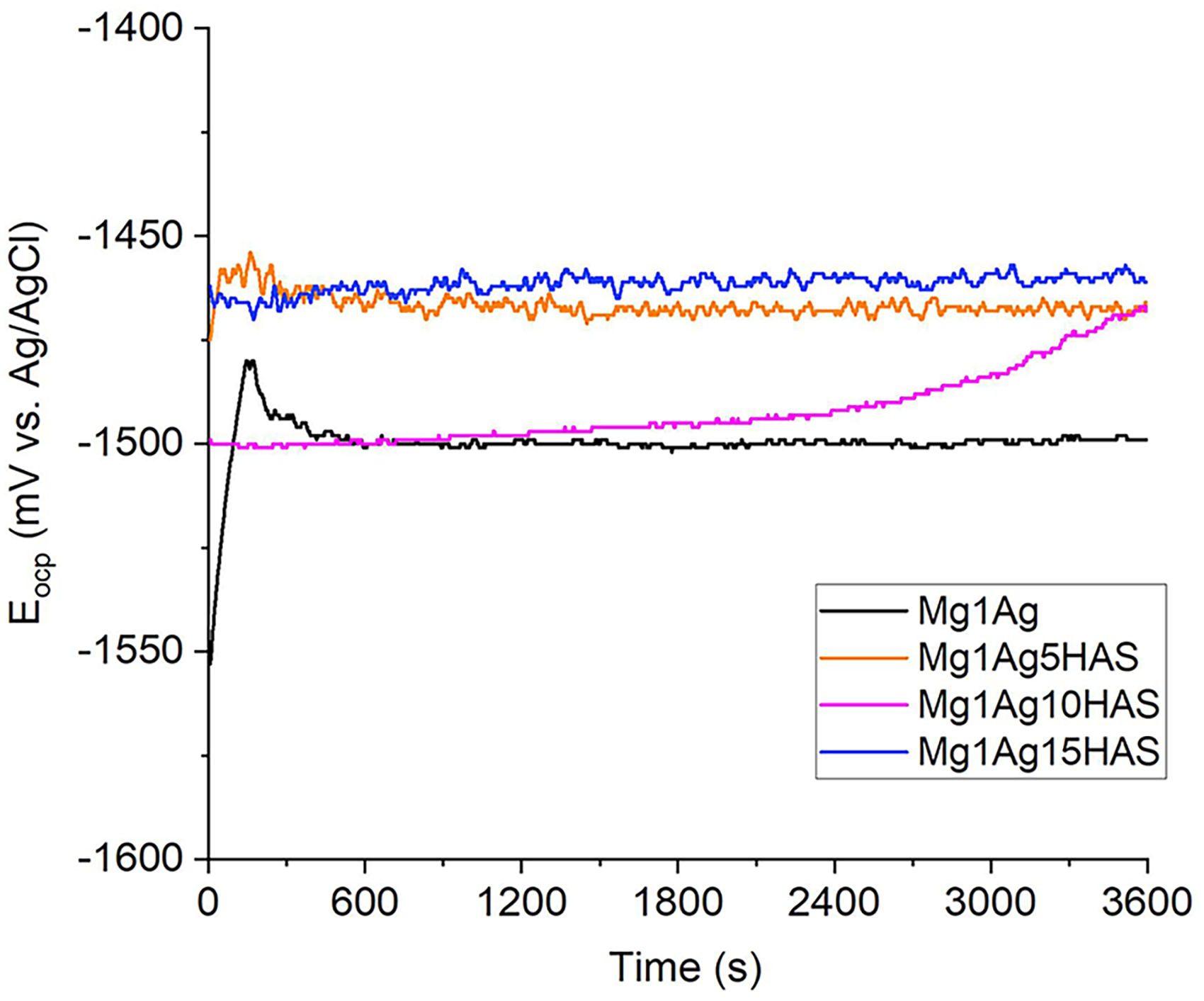
The Eocp behaviour of Mg1Ag–HAS composite. HAS: synthetic hydroxyapatite.
The surface morphology of composites reveals significant differences in corrosion product formation based on the reinforcement type (Figure 13). For the Mg1Ag binary alloy (Figure 13(a)), the SEM micrographs show localised degradation with small pits, and the corresponding EDS spectrum (spectrum 4) indicates a surface dominated by magnesium (Mg) and oxygen (O), with Mg reaching approximately 80% by weight. This suggests the formation of a primary magnesium hydroxide Mg(OH)2 layer. In contrast, the incorporation of HA into Mg1Ag15HAS (Figure 13(b)) and Mg1Ag15HAN (Figure 13(c)) promotes a more bioactive surface response. Specifically, the Mg1Ag15HAS sample, containing nano-sized HAS, exhibits a denser deposition of corrosion products with clear peaks for calcium (Ca) and phosphorus (P) in the EDS analysis (spectrum 5), accompanied by a decrease in the metallic Mg weight percentage to 50%. Similarly, the Mg1Ag15HAN sample, reinforced with micro-HA derived from calcined fish bone, displays an apatite-like layer formation with distinct Ca and P signals (spectrum 6), maintaining a Mg weight percentage of 60%. The presence of these mineral peaks, alongside chlorine (Cl) from the SBF solution, indicates that the addition of both HAS and HAN facilitates the nucleation of a protective calcium phosphate layer, which is essential for biocompatibility and controlled degradation in physiological environments.
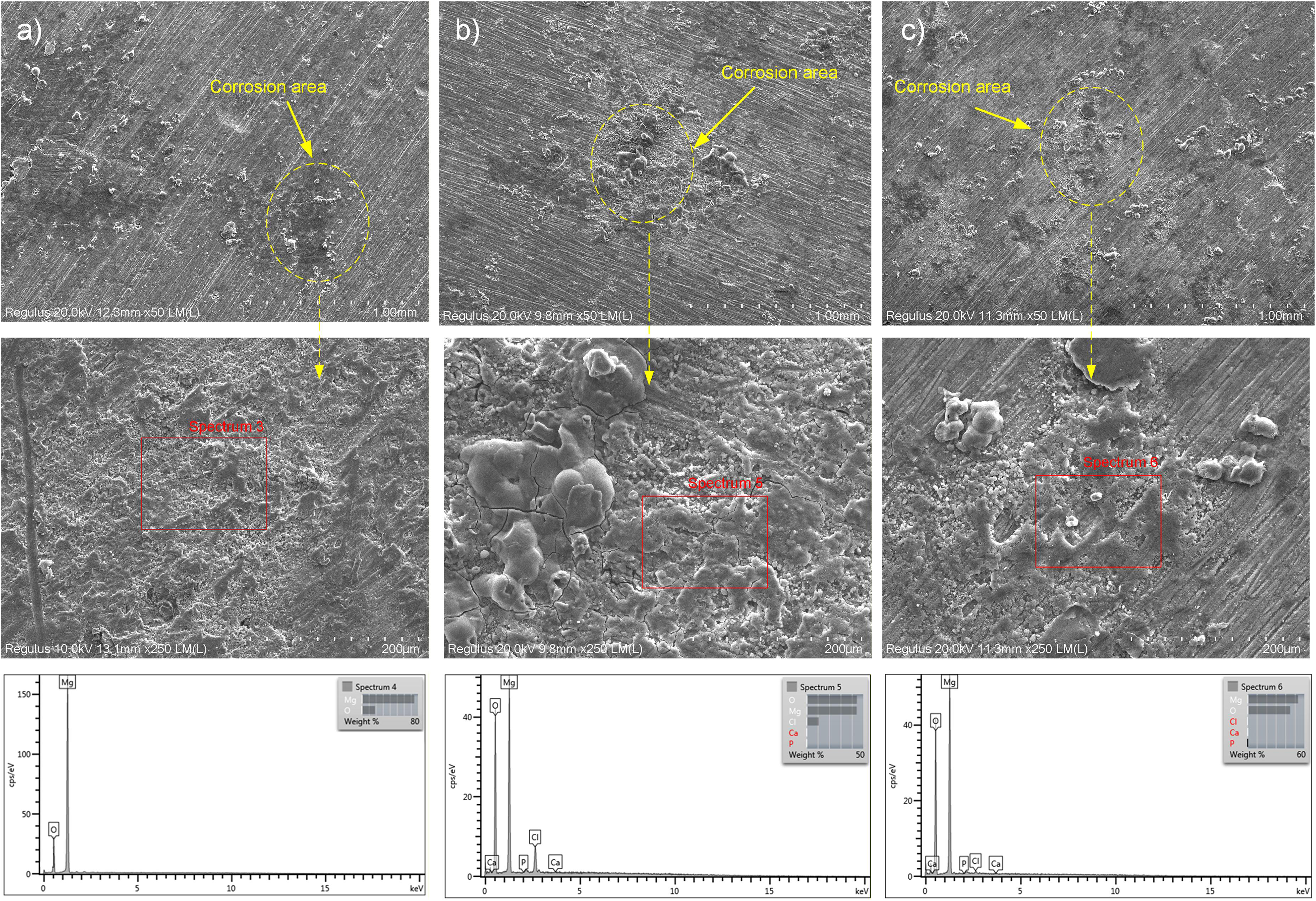
SEM/EDS analysis of (a) Mg1Ag, (b) Mg1Ag15HAS, and (c) Mg1Ag15HAN after SBF immersion for 1 h. Top row (x50): surface overview of corrosion sites. Middle row (x250): high-magnification morphological details of corrosion site. Bottom row: EDS spectra highlighting the semi quantitative analysis. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite; SBF: simulated body fluid.
Regarding the effect of HAN, it was observed that addition of all levels of HAN improved the corrosion such as HAS (Figure 14). Among them, Mg1Ag15HAN showed the most anodic and stable potential (−1467 mV), indicating a superior ability to passivate and resist corrosive attack, likely due to the formation of a protective surface barrier containing calcium phosphate compounds. The Mg1Ag5HAN and Mg1Ag10HAN samples also showed improved stability with slightly more negative but steady Eocp values compared to Mg1Ag, suggesting the continued protective role of the HAN reinforcement. The improvement in corrosion behaviour is attributed to the ceramic phase acting as a barrier to electrolyte penetration and promoting passivation. The Eocp of Mg1Ag5HAN, Mg1Ag10HAN, and Mg1Ag15HAN after one hour of immersion in the corrosive solution was −1475, −1469, and −1467 mV, respectively.
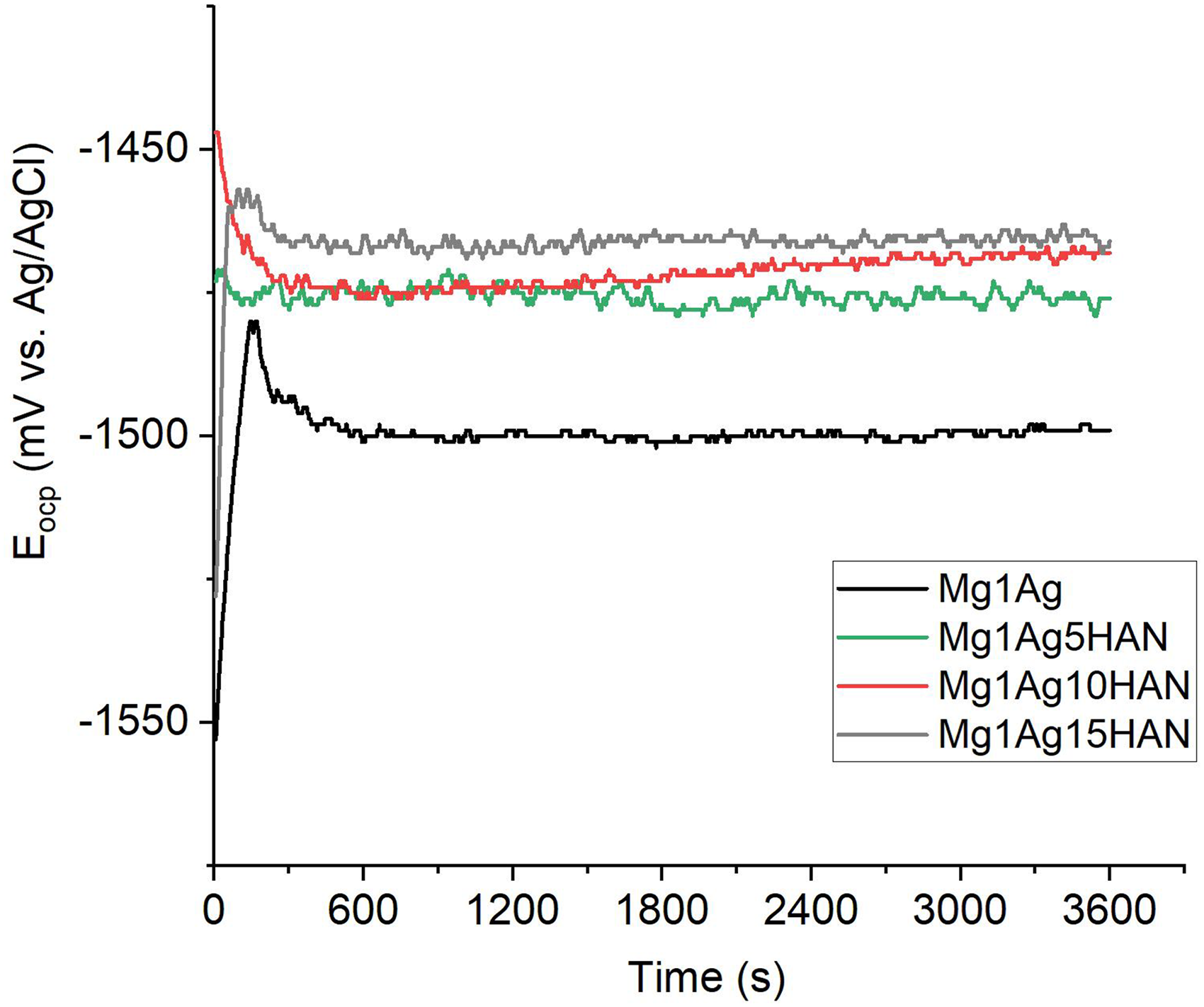
The Eocp behaviour of Mg1Ag–HAN composite.
The potentiodynamic curves of the HAS composite groups are shown in Figure 15. While a minor addition of HAS accelerated the corrosion rate relative to the base Mg1Ag composite, the corrosion potential (Ecorr) for Mg1Ag5HAS was recorded at −1350 mV, a value comparable to that of the Mg1Ag alloy. In contrast, for an equivalent reinforcement fraction, the corrosion potential of Mg–Zn-based composites has been reported to be significantly more negative. 45 Among the composites, Mg1Ag15HAS displayed the most positive corrosion potential of −1210 mV and the lowest corrosion current density (icorr), indicating its superior electrochemical stability and passivation behaviour. The sample Mg1Ag10HAS alloy exhibited the most negative corrosion potential (Ecorr) and Mg1Ag5HAS showed the high corrosion rate indicating their higher susceptibility to corrosion (Table 2). The Ecorr of Mg1Ag15HAS was 390 mV more anodic than the Mg–Zn/HAP (15%) composite. 22 Similarly, the Mg1Ag10HAS was 80 mV more noble than the Mg–Zn/HA (10%) composite. 24 Both Mg1Ag5HAS and Mg1Ag10HAS showed intermediate improvements compared to the unreinforced alloy, with gradual anodic shifts and reduced current densities, confirming that corrosion resistance improves proportionally with the HAS content.
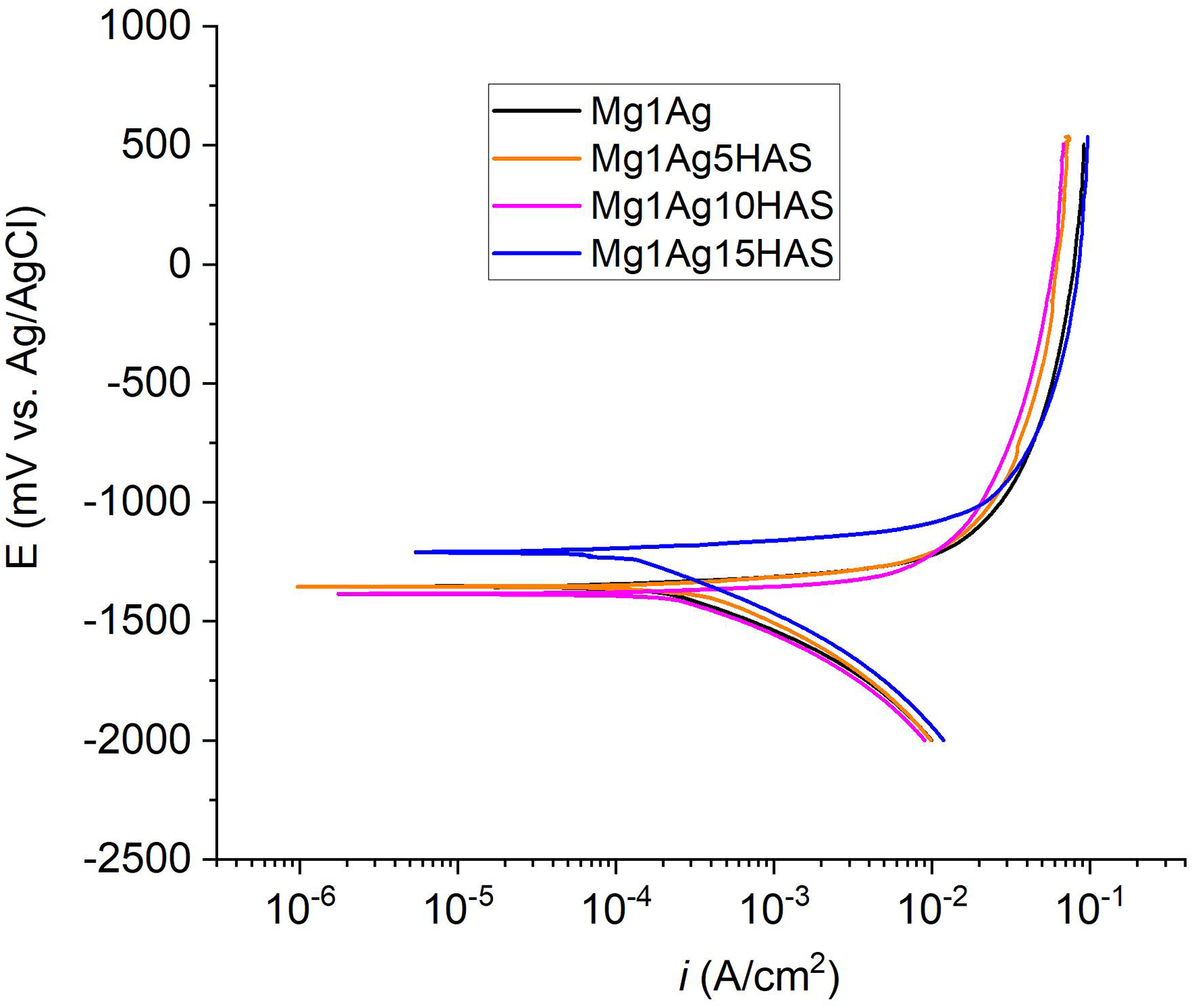
Potentiodynamic polarisation curves of Mg1Ag and Mg1Ag–xHAS (x = 5, 10, 15 wt-%) composites. HAS: synthetic hydroxyapatite.
The Ecorr and icorr of Mg1Ag and Mg1Ag–xHAS (x = 5, 10, 15 wt-%) composites obtained with Tafel methods from Figure 15.
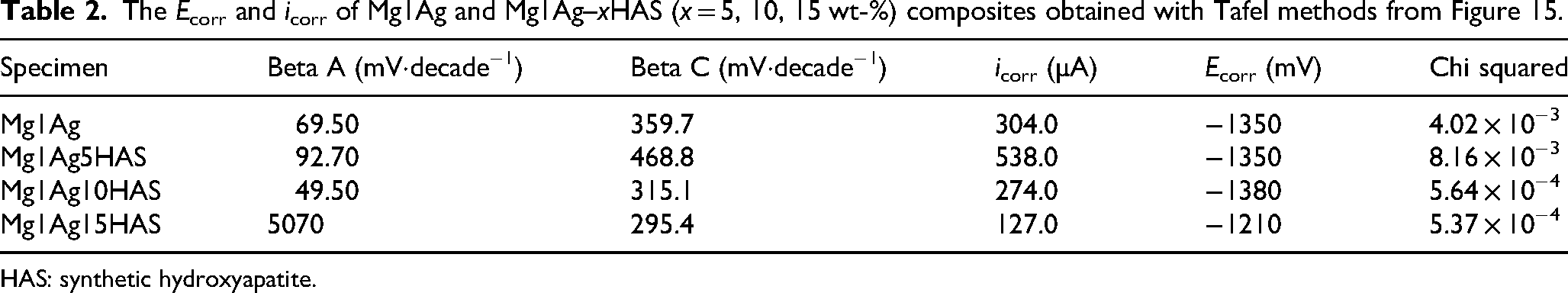
HAS: synthetic hydroxyapatite.
Regarding the potentiodynamic curves of Mg composite with HAN, a typical anodic dissolution curve was observed for all the sample as shown in Figure 16. The base alloy Mg1Ag exhibits a relatively low corrosion current density, indicating a lower corrosion rate (Table 3). It can be deduced that the addition of HA into the Mg1Ag for HAS or HAN makes the composite more cathodic. This tendency was also observed by 5% addition of HA into the pure Mg. 46 The impact of Ca concentration was also demonstrated on Mg–xCa alloys by the polarisation curves, which revealed that cathodic reaction rates rise as the Ca content increases. 47 This acceleration is primarily attributed to the increased volume fraction of the secondary phase. In contrast, all HAN-reinforced composites showed slightly higher corrosion current densities, suggesting a decrease in corrosion resistance. However, the Mg15HAN showed a more noble corrosion potential; two passivation regions, one located between −1050 and −1000 mV and the second one between −495 and −490 mV. The Ecorr of Mg15HAN was −1330 mV, it was slightly more anodic than the other composites, and the corrosion density of pure Mg was 37 µA lower compared to the composites with HAN. These results suggest that the incorporation of HAN improves the electrochemical stability of Mg1Ag, likely by acting as a barrier to aggressive chloride ions and promoting the formation of a more stable passive layer on the surface. However, at higher HAN content (15 wt-%), the protective effect may begin to diminish due to possible agglomeration or micro-structural defects. Although the addition of 5 and 10 wt-% HA shifted the Ecorr towards more negative values and increased the corrosion rate, a higher loading of HA (15 wt-%) shifted the Ecorr towards more positive values while further accelerating the corrosion rate. Similarly, it has been reported that the gradual addition of Ca from 1.34 up to 16 wt-% results in greater anodic dissolution and a higher corrosion rate. 47
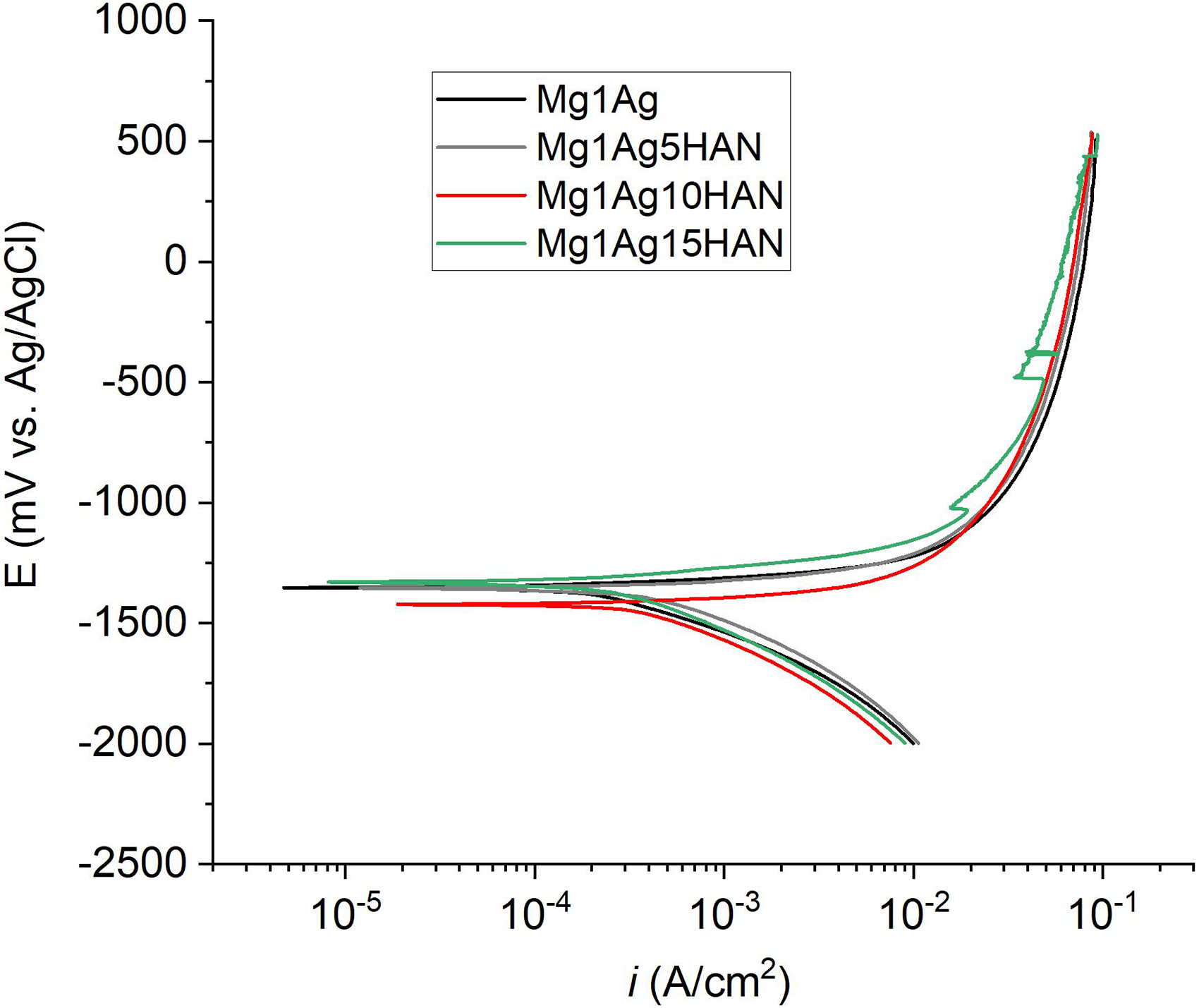
Potentiodynamic polarisation curves of Mg1Ag and Mg1Ag–xHAN (x = 5, 10, 15 wt-%) composites.
The Ecorr and icorr of Mg1Ag and Mg1Ag–xHAN (x = 5, 10, 15 wt-%) composites obtained with Tafel methods from Figure 16.
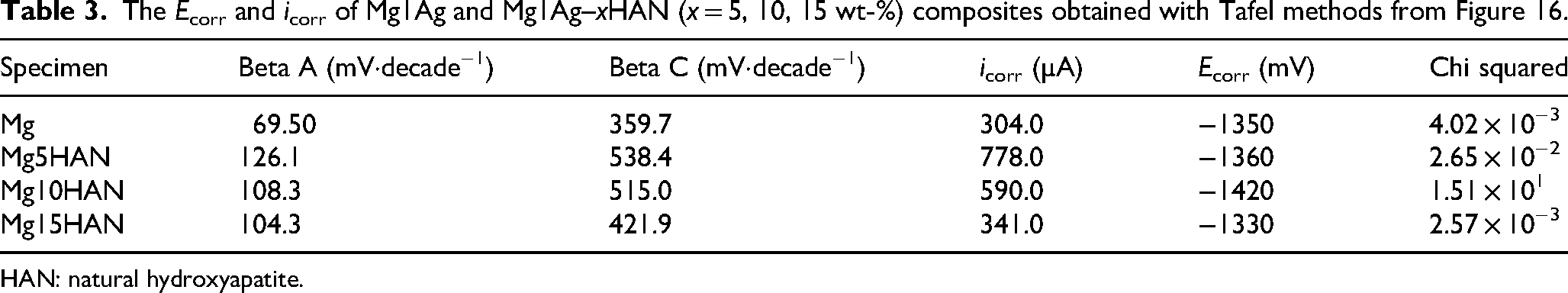
HAN: natural hydroxyapatite.
The EIS is a strong technique to evaluate the kinetic variables of the corrosion mechanism. Figure 17 shows the Nyquist plots with equivalent electrical circuit (ECC) fitting plots. At first glance, all the composite samples displayed three regions with well-defined loops: the high-frequency capacitive loop was attributed to exchange ion between metal surface and oxide layer, the medium-frequency capacitive loop correspond to the mass transfer/diffusion, and the low-frequency inductive loop assigned to the surface relaxation. The Nyquist plot reveals that the corrosion resistance of the Mg1Ag alloy significantly improves with the addition of nano-sized HAS particles. Among the tested samples, Mg1Ag15HAS exhibits the largest semi-circle diameter, indicating the highest charge transfer resistance and, therefore, the best corrosion protection in SBF solution (Figure 17(a)). This trend suggests that increasing the HAS content enhances the barrier properties of the surface, likely by forming a more stable and protective layer that limits the ingress of aggressive chloride ions. Based on the Nyquist plot, the charge transfer resistance (Rct), which reflects the material's resistance to corrosion, increased with the addition of HAS. The estimated Rct values were approximately 160 Ω for Mg1Ag, 100 Ω for Mg1Ag5HAS, 130 Ω for Mg1Ag10HAS, and 250 Ω for Mg1Ag15HAS. The presence of silver further contributes to corrosion resistance, possibly by promoting a more uniform passive film and reducing localised attack by acting as a noble metal inclusion, slowing anodic reactions. Mg1Ag5HAS and Mg1Ag10HAS show improvement, but the protective effect is limited, possibly due to incomplete surface coverage or insufficient formation of a barrier layer. On the other hand, the Nyquist spectra for Mg1Ag alloys reinforced with calcined fish-bone HAN show a clear, composition-dependent evolution of charge-transfer resistance (Rct) in the SBF medium (Figure 17(b)). The Mg1Ag composite exhibits a moderate semi-circle Rct (160 Ω), whereas the addition of 5 wt-% HAN actually slightly diminishes Rct (140 Ω), suggesting that low HAN loadings may interrupt the continuity of the protective Mg–OH/PO4 layer. At 10 wt-% HAN, Rct nearly returns to the base alloy value (155 Ω), indicating partial restoration of barrier function. Strikingly, 15 wt-% HAN delivers the largest semi-circle by far Rct (300 Ω), demonstrating that high HAN content forms a robust, ion-impermeable network that synergises with Ag to stabilise the passive film and inhibit Cl−.
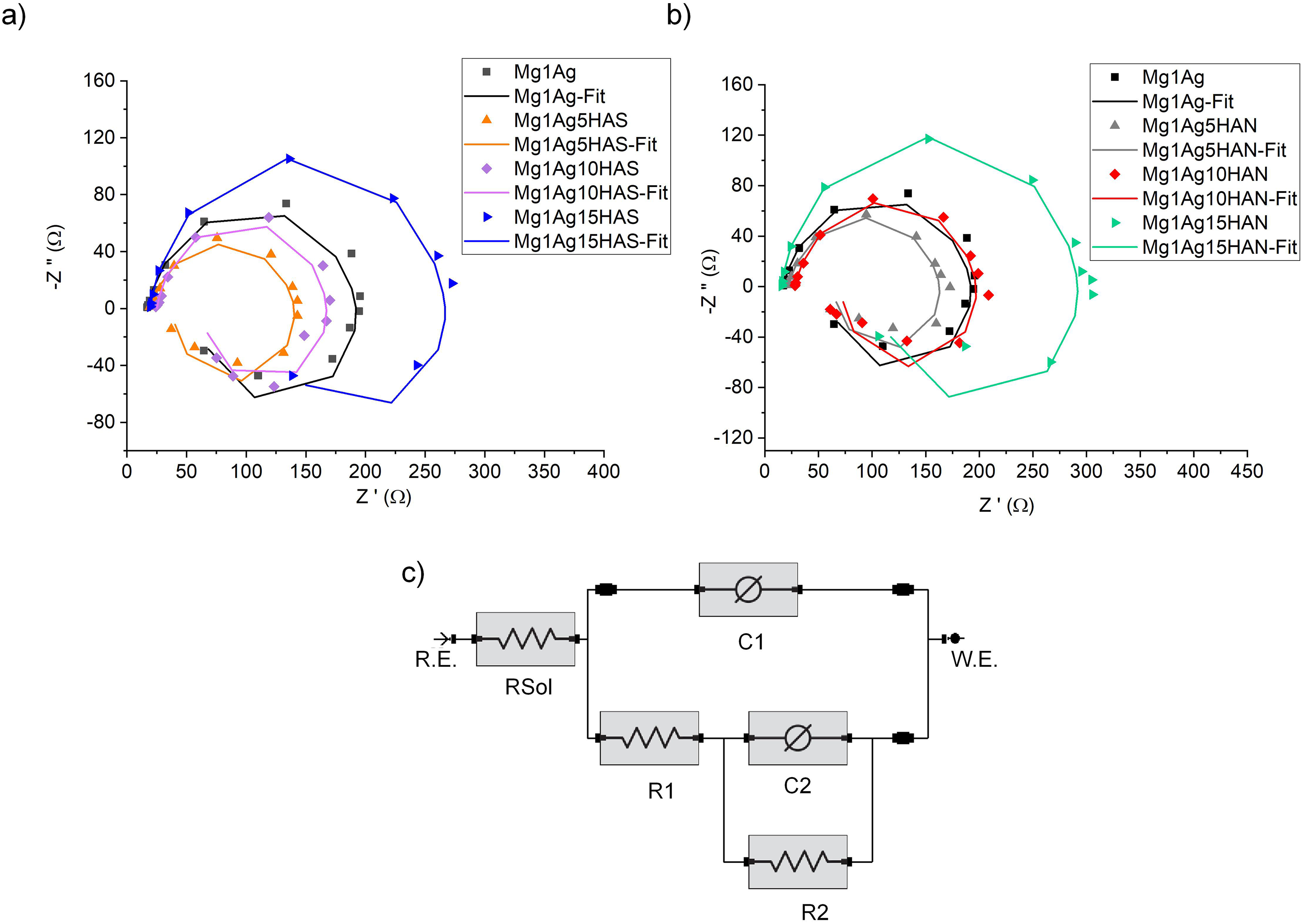
Nyquist plots of (a) Mg1Ag–xHAS (x = 5, 10, 15 wt-%), (b) Mg1Ag–xHAN (x = 5, 10, 15 wt-%), and (c) the equivalent electrical circuit (ECC) model. HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The ECC models were used to interpret the findings of impedance and are shown in Figure 17(c) and the data extracted was given in Tables 4 and 5. The equivalent circuit model comprises two constant phase elements (CPEs) and three resistive elements: Rsol (solution resistance), R1 (resistance at the solution/oxide interface), and R2 (resistance at the oxide/metal interface). The C1 and C2 elements represent CPEs, used instead of ideal capacitors to model real electrochemical interfaces. CPEs account for surface heterogeneities such as roughness, porosity, and compositional non-uniformity, which affect the charge distribution and capacitive response. According Table 4, the base Mg1Ag alloy exhibits a relatively high R2 value (83.76 Ω), indicating a moderate level of resistance at the oxide/metal interface, and a similar R1 value (19.79 Ω), reflecting initial film formation resistance. When 5% HAS is introduced (Mg1Ag5HAS), R2 significantly decreases to 37.91 Ω, suggesting a weakened or more porous oxide layer interface. This could be attributed to non-uniform dispersion of HAS particles or disruption of passive film integrity, reducing corrosion resistance. At 10% HAS (Mg1Ag10HAS), R2 nearly recovers to the base alloy value (83.81 Ω), while R1 remains modest (16.51 Ω), implying improved oxide layer stability and interface protection – likely due to better integration and distribution of HAS nano-particles within the matrix. Interestingly, the composite with 15% HAS (Mg1Ag15HAS) exhibits the highest R2 (102.64 Ω), indicating enhanced resistance at the metal/oxide interface. This suggests that a higher concentration of HAS may contribute to the formation of a more stable and protective barrier layer. However, the accompanying drop in C2 (from 1.08 × 10−5 to 5.17 × 10−6 S·s a ) for this sample indicates decreased interfacial capacitance, consistent with a denser or less reactive oxide film. The constancy of the exponent n (close to 1.0) across all the samples for C2 implies near-ideal capacitive behaviour at the oxide/metal interface. The behaviour of C1 and R1 (solution/oxide interface) across the samples shows less consistent trends, though generally, C1 values decrease with increasing HAS, suggesting a more stable and less reactive outer oxide layer.
Electrical equivalence of Mg1Ag and Mg1Ag–HAS composites.
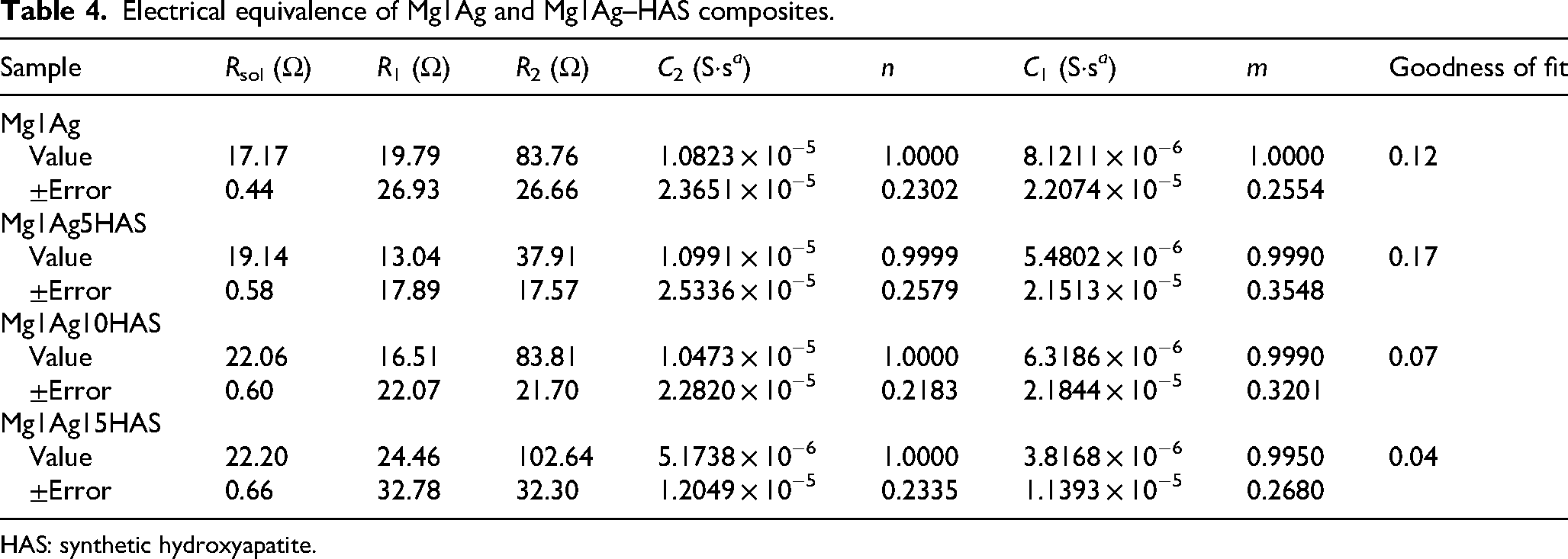
HAS: synthetic hydroxyapatite.
Electrical equivalence of Mg1Ag and Mg1Ag–HAN composites.
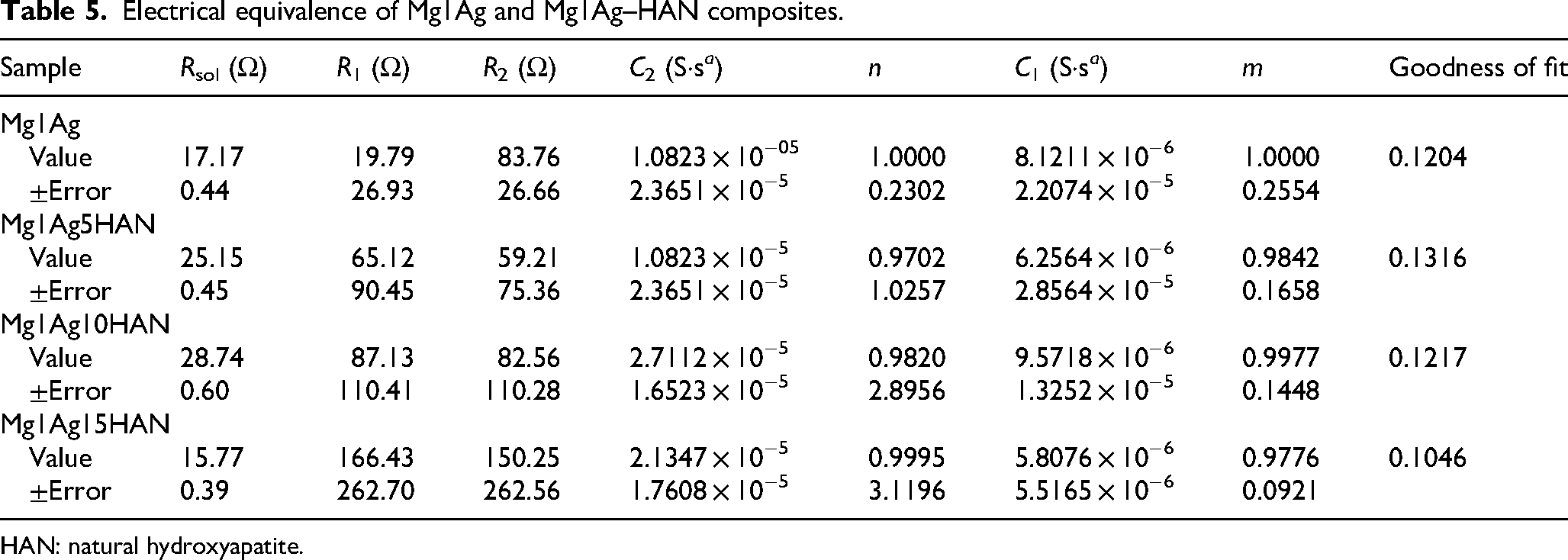
HAN: natural hydroxyapatite.
Concerning the addition of HAN into the Mg1Ag composite indicates that both the solution/oxide interface resistance (R1) and the oxide/metal interface resistance (R2) increase substantially as the HAN content increases, particularly in the Mg1Ag15HAN sample, which exhibits the highest R1 (166.43 Ω) and R2 (150.25 Ω) values (Table 5). This suggests that HAN promotes the formation of a more protective and stable oxide layer, effectively impeding corrosion processes. The base alloy Mg1Ag exhibits relatively low C1 and C2 values, with ideal capacitive behaviour (m and n = 1), indicating a thin and uniform passive film with a limited charge storage capacity. Upon the addition of 5% HAN, C1 slightly decreases while C2 remains nearly unchanged, and minor deviations in m and n suggest increased surface heterogeneity due to particle incorporation. In the Mg1Ag10HAN sample, both C1 and C2 increase, particularly C2, reflecting enhanced interfacial capacitance likely due to a thicker or more complex oxide layer. The high m and n values indicate the formation of a more homogeneous and stable passive film. The Mg1Ag15HAN composition exhibits the highest degree of capacitive behaviour at the oxide/metal interface (C2), along with nearly ideal m and n values (0.9776 and 0.9995), suggesting a highly uniform and protective oxide layer.
Biocompatibility of the composites
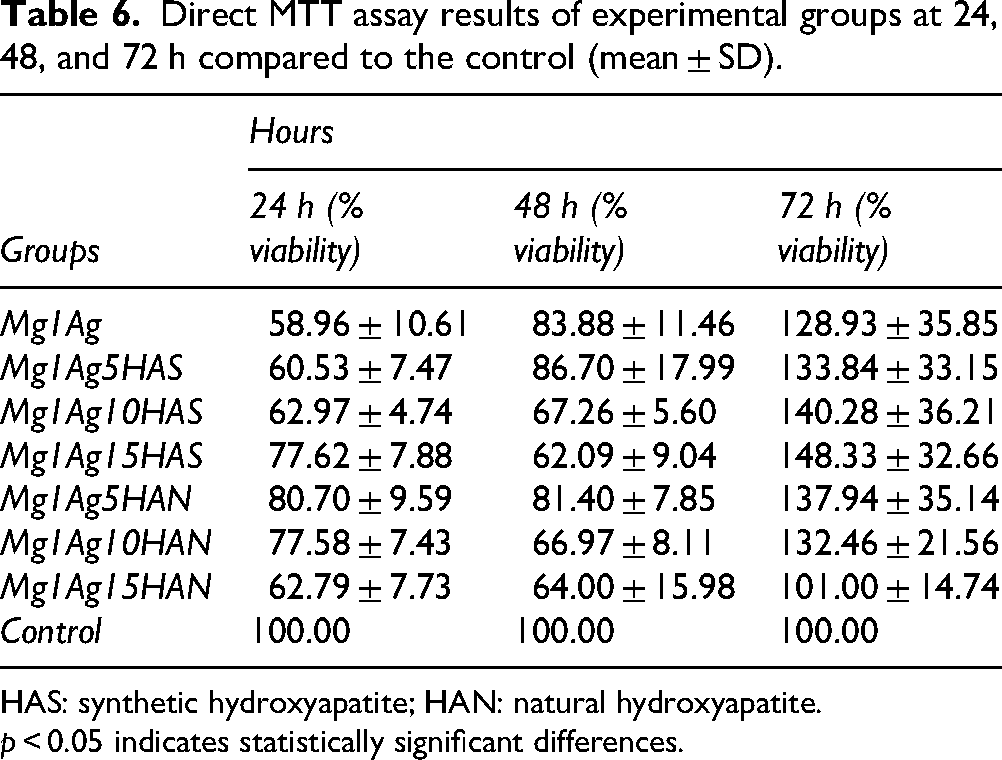
According to the direct MTT assay results (Figure 18 and Table 6), a significant reduction in cell viability was observed in all experimental groups at 24 h compared to the control group (58.96–80.70%). At 24 h, the viability values of most Mg–Ag–HA groups were slightly lower than the control, suggesting a mild initial cytotoxic effect likely due to rapid Mg2+ and Ag+ ion release immediately after cell contact. Notably, the Mg1Ag10HAN (77.58%) and Mg1Ag5HAN (80.70%) groups exhibited relatively higher viability rates when compared with the other samples. At 48 h, a heterogeneous distribution of viability values was detected: while the Mg1Ag (83.88%), Mg1Ag5HAS (86.70%), and Mg1Ag5HAN (81.40%) groups reached levels close to the control, marked decreases were recorded in the Mg1Ag15HAN, Mg1Ag10HAN, and Mg1Ag15HAS containing groups (62–67%). However, by 72 h, a proliferative response surpassing the control group was evident in all experimental groups, with viability rates ranging between 128.93% and 148.33%. This increase was particularly pronounced in the Mg1Ag15HAS, Mg1Ag10HAS, Mg1Ag5HAN, and Mg1Ag5HAS groups, which demonstrated approximately 1.4–1.5-fold higher viability compared to the control. This trend reflects cell adaptation to the ionic environment and the gradual reduction of initial burst ion release. Statistical analysis revealed significant differences between early and later time points (p < 0.001), with the 72 h results showing no significant difference among the composite groups, confirming comparable biocompatibility. Collectively, these findings suggest that although Mg and HAS/HAN-containing structures initially reduced cell viability, they subsequently exerted a proliferative effect over time, significantly enhancing cell viability at later stages.
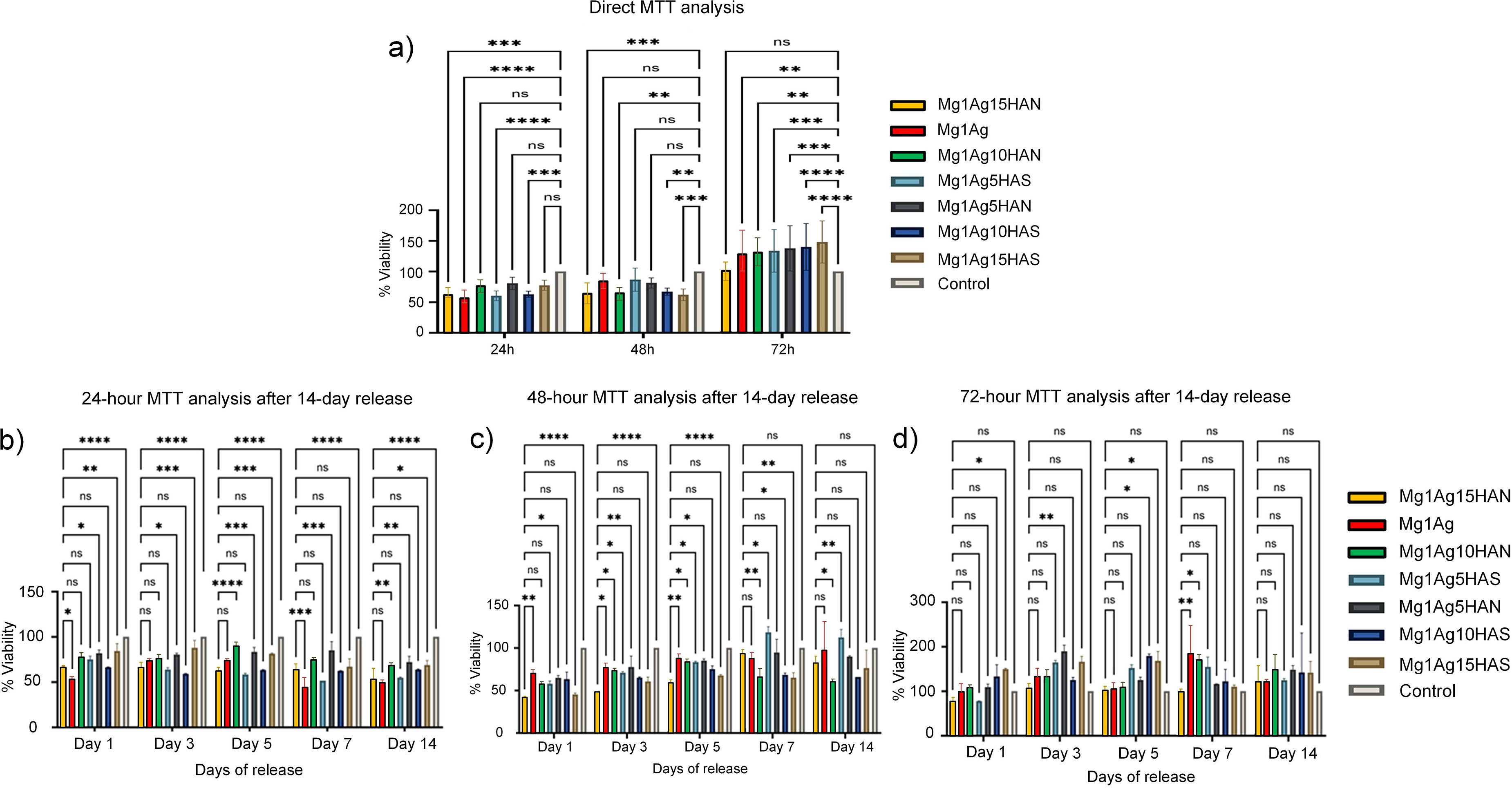
(a) Direct MTT assay (24, 48, 72 h), (b) indirect MTT assay (24 h), (c) indirect MTT assay (48 h), and (d) indirect MTT assay (72 h) results showing cell viability (%) of experimental groups.
Direct MTT assay results of experimental groups at 24, 48, and 72 h compared to the control (mean ± SD).
HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
p < 0.05 indicates statistically significant differences.
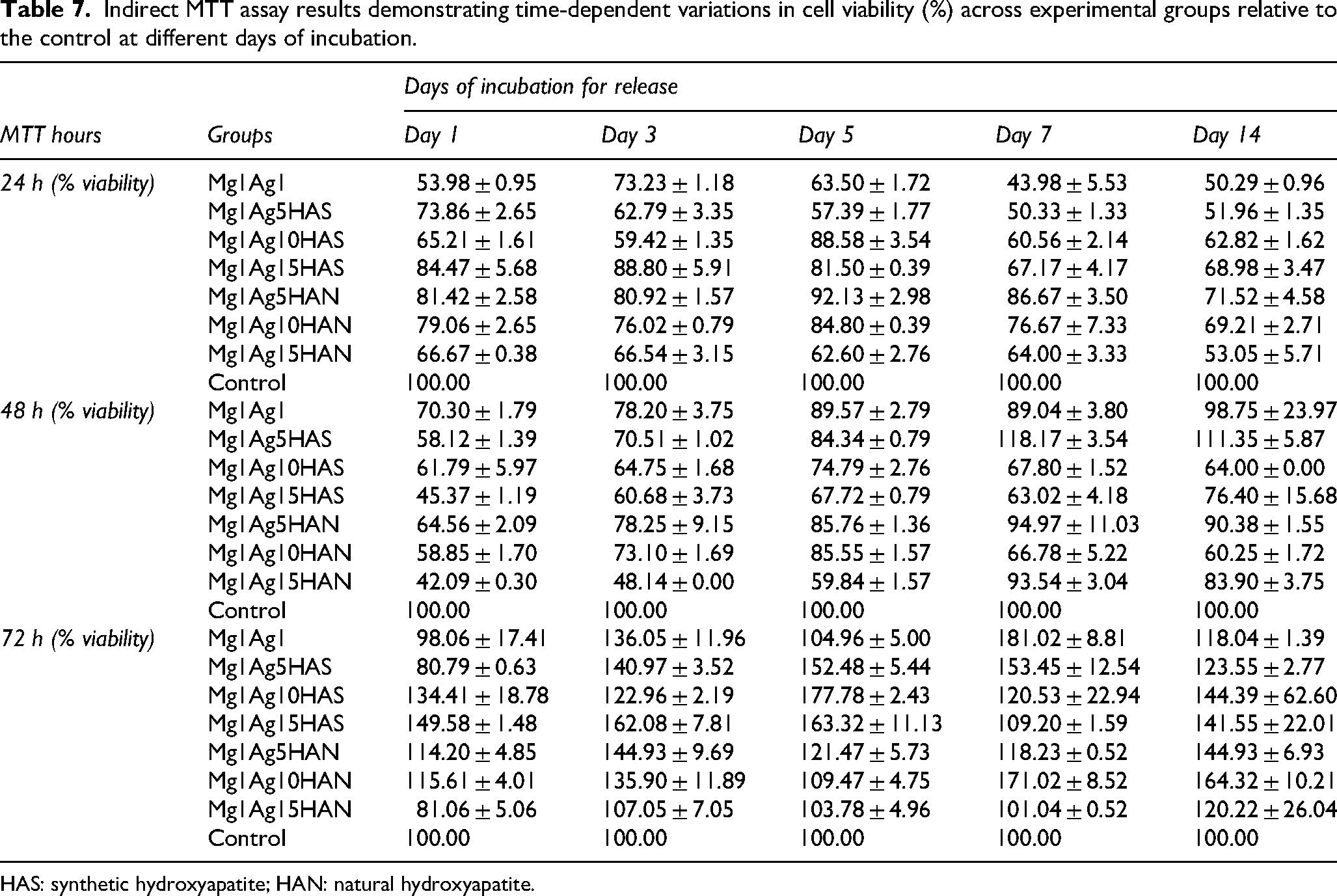
The results from the 24-h MTT assay after 14-day release show clear trends in cell viability in response to the Mg–Ag–HA composite extracts (Figure 18(b) and Table 7). Indirect MTT assays revealed that cell viability exhibited time-dependent variations across experimental groups when compared to the control. At 24 h, all groups showed a reduction in viability relative to the control (100%), with values ranging from 53.98% (Mg1Ag1) to 84.47% (Mg1Ag15HAS) for day 1. In this period, Mg1Ag5HAN (81.42%), Mg1Ag10HAN (79.06%), and Mg1Ag15HAS (84.47%) maintained relatively higher viability compared to other groups. All experimental groups exhibit cell viabilities above 50%, indicating no severe cytotoxic effects. HAS-containing samples and HAN showed higher cell viabilities than pure Mg1Ag. This indicates both forms of HA improve cell compatibility. The cytotoxicity appears slightly higher (lower viability) at day 1 for Mg1Ag alone, likely due to rapid ion release at early stages. Over time (days 3–7), the viability improves, indicating stabilisation of the ionic environment. For day 3, cell viability increases slightly in several groups (Mg1Ag1 from 54% → 73%, Mg1Ag115HAS from 84.47 → 88.80). At day 5, most groups maintain moderate viability (50–80%), indicating cell adaptation. Mg1Ag5HAN shows the highest values (∼92%), while Mg1Ag1 and Mg1Ag5HAS remain lower (∼57–63%). At day 7, the Mg1Ag1 drops sharply (∼44%), indicating continued Mg degradation and local alkalinisation may be stressing cell while HA groups maintain higher viability (∼60–86%), showing HA's protective effect. By day 14, most groups show lower viability than the control, suggesting long-term tolerability. A further decline is seen in Mg1Ag1 (∼50%). HA-containing groups show moderate to good viability (∼52–72%), particularly Mg1Ag5HAN (71.5%). This indicates long-term stability of HA-containing composites compared to pure Mg–Ag.
Indirect MTT assay results demonstrating time-dependent variations in cell viability (%) across experimental groups relative to the control at different days of incubation.
HAS: synthetic hydroxyapatite; HAN: natural hydroxyapatite.
The cytotoxicity of the Mg–Ag–HA composites was evaluated using the 48-h MTT assay on Saos-2 cells after exposure to the 14-day extracts obtained from indirect contact experiments. As shown in Figure 18(c) and Table 7, all the samples exhibited cell viability values above 50%, indicating non-cytotoxic behaviour according to ISO 10993-5. At 7th day (48 h), viability is slightly higher than at 24 h for Mg1Ag1 as ∼70%. HA groups are around 58–65%, still showing mild cytotoxicity. On the other hand, Mg1Ag15HAN had the lowest (42%), indicating sensitivity to higher HA concentrations. At days 3 and 5, cell viability increased in most groups (e.g. Mg1Ag5HAS ∼70.5%, Mg1Ag5HAN ∼78%) and viability reaches its peak for many groups (Mg1Ag1 ∼90%, Mg1Ag5HAS ∼84.3%), respectively. The day 7 indicates a transition from cytotoxic to biocompatible environment where an improvement in viability for HA groups is observed (Mg1Ag5HAS ∼118%, Mg1Ag5HAN ∼94%).
On day 14, it was observed a high sustained viability for HA (e.g. Mg1Ag15HAS ∼114%, Mg1Ag5HAS ∼111%). In contrast, Mg1Ag1 remains high (≈98%) suggesting that after the initial burst, degradation stabilises.
MTT assay results (72-h incubation after 14-day extract release) demonstrated that all Mg–Ag–HA composites exhibited good cytocompatibility towards Saos-2 osteoblast-like cells, with cell viability values remaining above 80% throughout the test period (Figure 18(d) and Table 7).
Several groups demonstrated a proliferative response, with viability surpassing control values. The Mg1Ag1 group exhibited viability levels close to the control, suggesting that the initial cytotoxicity observed at earlier time points diminished as the material stabilised. At day 1, HA groups (especially Mg1Ag10HAS, Mg1Ag15HAS, Mg1Ag5HAN, and Mg1Ag10HAN) show even higher values (>130–150%), indicating early proliferative response. Notably, composites containing HAS showed significantly higher viability values compared to the control, with Mg1Ag15HAS and Mg1Ag10HAS reaching 163.32 ± 11.13 and 177.78 ± 2.43% at day 5, respectively. This enhanced response is likely due to the bioactive nature of HA, which improves cell adhesion and proliferation by buffering the local environment and promoting favourable cell–material interactions. HA's bioactive properties clearly enhance cell proliferation. Immersion studies in SBF revealed greater Ca/P phase mineral deposition on the composites compared to pure Mg. 35 Regarding day 7, the viability remained high for Mg1Ag5HAS ∼153% and Mg1Ag10HAS ∼120%, indicating sustained compatibility and possibly osteogenic stimulation. HAN-containing groups (e.g. Mg1Ag10HAN) also exhibited elevated viability levels (up to 171.02 ± 8.52%), though slightly lower than HAS counterparts, suggesting differences in their physicochemical properties. In contrast, Mg1Ag15HAN showed moderate viability values, indicating that higher HAN content may influence surface characteristics and ion release behaviour.
Conclusion
Synthetic and natural doped Mg–Ag composites with different ratios were produced using mechanical mixing and sintering. Micro-structural, mechanical, corrosion, and in vitro assays were performed on the composites. The main findings are summarised below:
The HAS promotes superior micro-structural refinement, densification, and homogeneity compared to natural fish bone-derived hydroxyapatite (HAN), particularly at lower reinforcement levels. However, excessive addition of either type of HA increases porosity and particle clustering, suggesting that the optimal reinforcement content must be carefully controlled to achieve desirable mechanical properties in Mg1Ag composites. The HAS is more effective than HAN in enhancing the hardness of Mg1Ag composites, with 10 wt-% HAS addition yielding the most favourable mechanical performance. In general, the addition of HAS at a lower content (5 wt-%) effectively improved compressive strength and ductility, whereas higher HAS and HAN contents performed brittleness and lower mechanical stability under compressive loads. the trade-off between strength and ductility, where HAN reinforcement – especially at 10% – enhances stiffness and strength but compromises deformability, while HAS favours ductile behaviour but provides limited reinforcement at higher mechanical loads. Mg1Ag15HAS demonstrated the best corrosion resistance among HAS composites, with the most positive Ecorr and lowest icorr. In contrast, Mg1Ag5HAS and Mg1Ag10HAS were more susceptible to corrosion. The addition of HAN slightly increased icorr but improved Ecorr, especially in Mg15HAN, suggesting enhanced stability due to passivation. The improvement is attributed to the barrier effect of HA nano-particles, which likely reduce the penetration of chloride ions and stabilise the surface film. The synergistic effect of Ag, HAS, and HAN appears to be most effective at 15 wt-%, making Mg1Ag15HAS and Mg1Ag15HAN the most corrosion-resistant composition among the tested samples. All Mg–Ag–HA composites showed good cytocompatibility with Saos-2 cells, maintaining or exceeding control viability after 72 h with direct MTT assay. Initial reductions in viability at 24 h were mitigated over time, indicating cell adaptation to the ionic environment. Composites containing HAN exhibited slightly better cell responses, highlighting their enhanced biocompatibility potential. The best-performing group is Mg1Ag15HAN, showing the highest and most stable viability across the release period. The indirect MTT assay showed that all Mg–Ag–HA composites demonstrated non-cytotoxic behaviour, maintaining cell viability above 50% in accordance with ISO 10993-5. Incorporation of HA, particularly HAN, significantly enhanced Saos-2 cell viability and metabolic activity compared to Mg1Ag alone. Mg1Ag10HAN consistently exhibited the highest viability, indicating an optimal balance between degradation rate, ion release, and bioactivity. The indirect MTT results confirm that Mg–Ag–HAN composites possess excellent long-term cytocompatibility, making them strong candidates for biodegradable bone implant applications.
Footnotes
ORCID iDs
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by MÖÖ, NÖK, BDC, NB, DK, and KA. The first draft of the manuscript was written by MÖÖ, NÖK, BDC, and KA, and all authors have commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author upon request.
