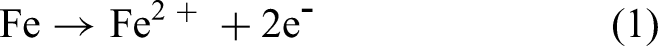Abstract
Elevated concentrations of dissolved inorganic nitrogen (DIN) that stimulate microbiologically influenced corrosion (MIC) likely much increase longer-term seawater immersion corrosion of mild and low alloy steels. In contrast, in shorter-term exposures, DIN less than about 0.25 mgN/L concentration usually has little or no effect. This is illustrated with data for 12 independent cases with corrosion loss trending interpreted using the bimodal model. Its early part is predominantly under oxygenated conditions suitable primarily for iron and sulphur oxidising bacteria that are less aggressive for corrosion. These conditions also are suitable for the nitrogenous components of DIN to act as corrosion inhibitors. Longer-term exposure conditions predominantly are anaerobic suitable for sulphate-reducing microorganisms, typically the main actors in MIC. The results demonstrate it is problematic to extrapolate short-term (days – months) observations to long-term corrosion, particularly when MIC or DIN are involved. The results also highlight that the change from oxygen reduction as the critical cathodic reaction in shorter exposures to water reduction (or hydrogen evolution) as the critical cathodic reaction in extended exposures has a profound effect on the effect of DIN concentration and its effect on corrosion.
Introduction
The corrosion loss and the pitting of bare steels in seawaters remain matters of practical interest, in part because protective coatings and cathodic protection are not always feasible or economic versus short-term or long-term deterioration of an infrastructure asset. 1 Use of unprotected steel implies a need for a sacrificial corrosion allowance and thus a need to predict the amount of general corrosion or pitting likely for the exposure period(s) of interest. For this, mathematical models can be used, ideally based on data from experiments sufficiently long to relate to the expected lifetime of the application, rather than short-term empirical or laboratory data.2–5 The potential for microbiologically influenced corrosion (MIC) must be considered, particularly for seawater exposures.6–10 The contribution of MIC to overall corrosion depends on the rate of metabolism of the microorganisms involved. Classically, these are the sulphate-reducing bacteria, although many others, usually considered less aggressive, have been identified.9,11 The rate of bacterial metabolism, and by implication, the contribution to corrosion depends on the availability of critical nutrients,12–15 including for exposures in seawater, ferrous ions (Fe2+). 16 For the corrosion of steels, this is readily available as a result of the dissolution of ferrous iron as part of the corrosion process:
Ferrous ions in seawater also have been proposed as a by-product of aeolian dust from terrestrial wind erosion deposited on water surfaces.17,18 Other nutrients required for bacterial metabolism in seawaters, including sulphates, phosphates, calcium and organic carbon, mostly are readily available in seawaters. 12 However, the rate-limiting nutrient usually is inorganic nitrogen, the only form of nitrogen that is biologically accessible. 19 In the oceans and coastal and other waters, it occurs as dissolved inorganic nitrogen (DIN), comprising ammonia and nitrites and nitrates, mainly of calcium. Its concentration for many locations is very low, even in surface waters 20 unless there is anthropogenic pollution. 21 Typically, this occurs in river mouths, estuaries and enclosed waters.
The link between the concentration of nutrients, in particular DIN, and the increased amount or rate of corrosion is well-established for steel specimens in laboratory solutions. Typically, these involve high nutrient concentrations and mono-bacterial species.9,11,22,23 In natural seawaters with a diversity of bacterial and other species, the link between DIN concentration and the severity of corrosion losses over longer-term exposures (years, decades) is well-established, with positive correlations between corrosion loss and DIN concentration and average seawater temperature.8,24 However, for shorter-term corrosion losses in natural seawater environments, there are cases for which corrosion losses are little or no more than in natural seawater, despite the relatively high DIN concentrations, while in other cases, there is some corrosion loss. The reason(s) for this apparent inconsistency are explored herein, using data from 12 cases of field exposure, all drawn from the existing literature but supplemented, in some cases, with additional information. These cases are described further below.
To add consistency to the data sets, they are interpreted using the framework of the bimodal model for the development of corrosion loss with continued exposure. That model and its main features including its corrosion rate-controlling mechanisms are reviewed in the next section. The section that follows describes 12 cases and the information that is available about their location and their potential for elevated DIN concentration at each exposure site. Where information about likely (average) DIN concentration was not available it was estimated by working backwards from earlier correlations between longer-term corrosion loss and DIN concentration. 8 The process involved is explained in the next section. It was used also to check the consistency between such predictions and the DIN concentrations where those were reported in the literature.
‘Discussion’ section considers observations and inferences that may be drawn from the cases presented. These include (a) the low effect DIN appears to have had for early corrosion in some cases but not others and (b) the apparent time-wise contraction of the bimodal model with increased DIN. ‘Discussion’ section also considers the likely reasons for those observations. It is proposed that the lower corrosion loss in the early stages is the result of reduced biological production of metabolites as well as the nitrogenous components of DIN (nitrites, nitrates and ammonia) acting as corrosion inhibitors. These are considered new interpretations. They fill the knowledge gap between corrosion results obtained from short-term laboratory tests and what can be expected for actual steels exposed for the extended periods typical for practical structures.
‘Conclusion’ section notes that the present results reinforce the point that short-term observations of corrosion loss under DIN conditions should not be used to make predictions about longer-term corrosion behaviour. This is because (a) the development of corrosion is not a uniform continuous process but rather one representable by the bimodal model, (b) this means that the governing corrosion process changes from control by oxygen diffusion and the oxygen reduction cathodic reaction early on to control by hydrogen evolution under the water reduction cathodic reaction later, (c) that this change influences the effect DIN has on the co-existing microorganisms and hence on MIC and (d) the corrosion inhibiting effects of the nitrogenous components of DIN in the early stages of corrosion.
Background and approach
The development of corrosion loss (and pit depth) as a function of increased exposure time is best represented by the so-called bimodal model (Figure 1). It was developed from observations on steels in seawater 2 and extended to a range of metals in a variety of environments.5,25 The model can be characterised using the parameters r0, ta, ca, ra, cs and rs as shown in Figure 1. Mean values of these parameters are available from calibration to worldwide corrosion data for low-pollution coastal conditions.2,26 There are also some estimates of their variability. 3 In all cases, mean temperature T is a critical influencing factor and is accounted for in these parameters.2,26 Local water velocity wave and action have limited long-term influence 5 but are not considered specifically herein.
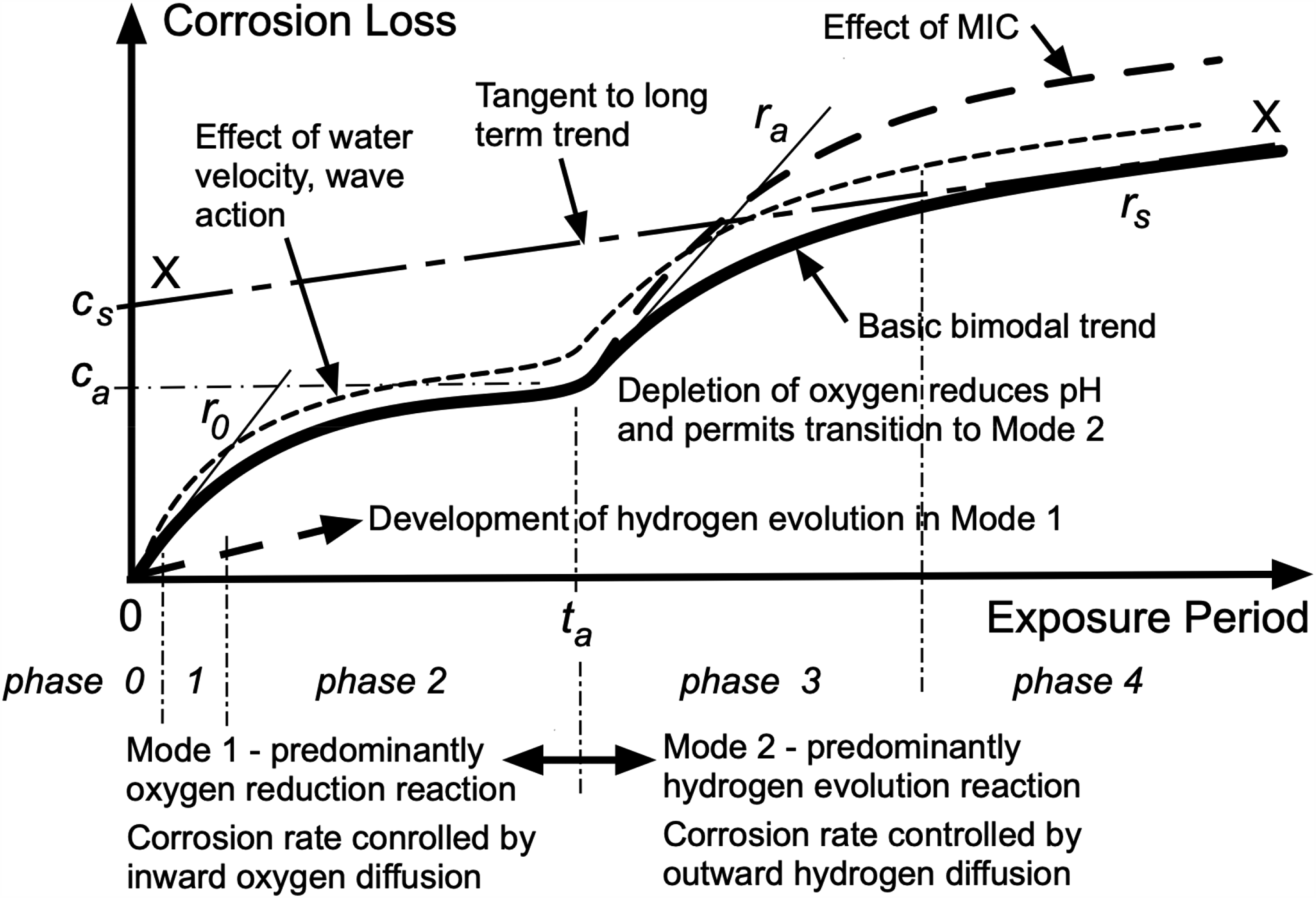
Bimodal model for corrosion loss development with increased exposure period. 2 For Mode 1, oxygen diffusion is the rate-controlling mechanism for the cathodic oxygen reduction reaction governing corrosion. For Mode 2, outward hydrogen diffusion is the rate-controlling mechanism for the cathodic hydrogen evolution (water reduction) reaction. Note that the hydrogen evolution cathodic reaction is present also in Mode 1. The parameters used to quantify the model are shown, including the linearisation used to represent the long-term effect of corrosion in Mode 2. The effects of microbiologically influenced corrosion are shown schematically.5,6,8
As shown in Figure 1, the main effect of DIN on corrosion loss is in Mode 2.8,27 This is consistent with Mode 2 being dominated by corrosion under anaerobic conditions. Its effect on longer-term corrosion can be approximated and evaluated through a linearisation (XX) of Mode 2 and parametrised by the variables cs and rs. Both are functions of T and DIN. 8
Only a relatively small number of reports are available in the corrosion literature for corrosion losses in seawaters considered to have some level of pollution. Examples include Peterson and Waldron, 28 Arup and Glantz, 29 Southwell et al. 30 and Edyvean and Videla. 31 Most provide corrosion loss observations at only a single point in time, typically after many years of exposure. Several more comprehensive cases, with corrosion loss data at a number of exposure periods, are used herein. Dissolved inorganic nitrogen estimates were available for some but for others, estimates of DIN concentrations were made using the calibrated results obtained earlier 8 as described below. The process relies on the linearisation XX of Mode 2 (Figure 1) and the parameters cs and rs, both, as noted, functions of T and DIN concentration. 8 The estimate starts with fitting the bimodal model to the data set for corrosion loss and knowledge (or an estimate) of the average seawater temperature at the site of interest. A trial linear tangent XX is then fitted to Mode 2 (Figure 1), giving trial values of cs and rs. Each of these then gives a trial value of DIN using the plots in Melchers. 8 The trial values of DIN will coincide when the correct linear function is fitted by trial and error. Because of the limited data sources underlying those in, 8 formal estimates of uncertainty in DIN so obtained a = cannot be obtained but is considered to be less than ±10%.
In the following section, the cases for which sufficient corrosion loss data and information about DIN concentrations were available (or could be estimated as noted above) are shown arranged from background DIN concentration to successively higher DIN values.
Corrosion-loss data and trends
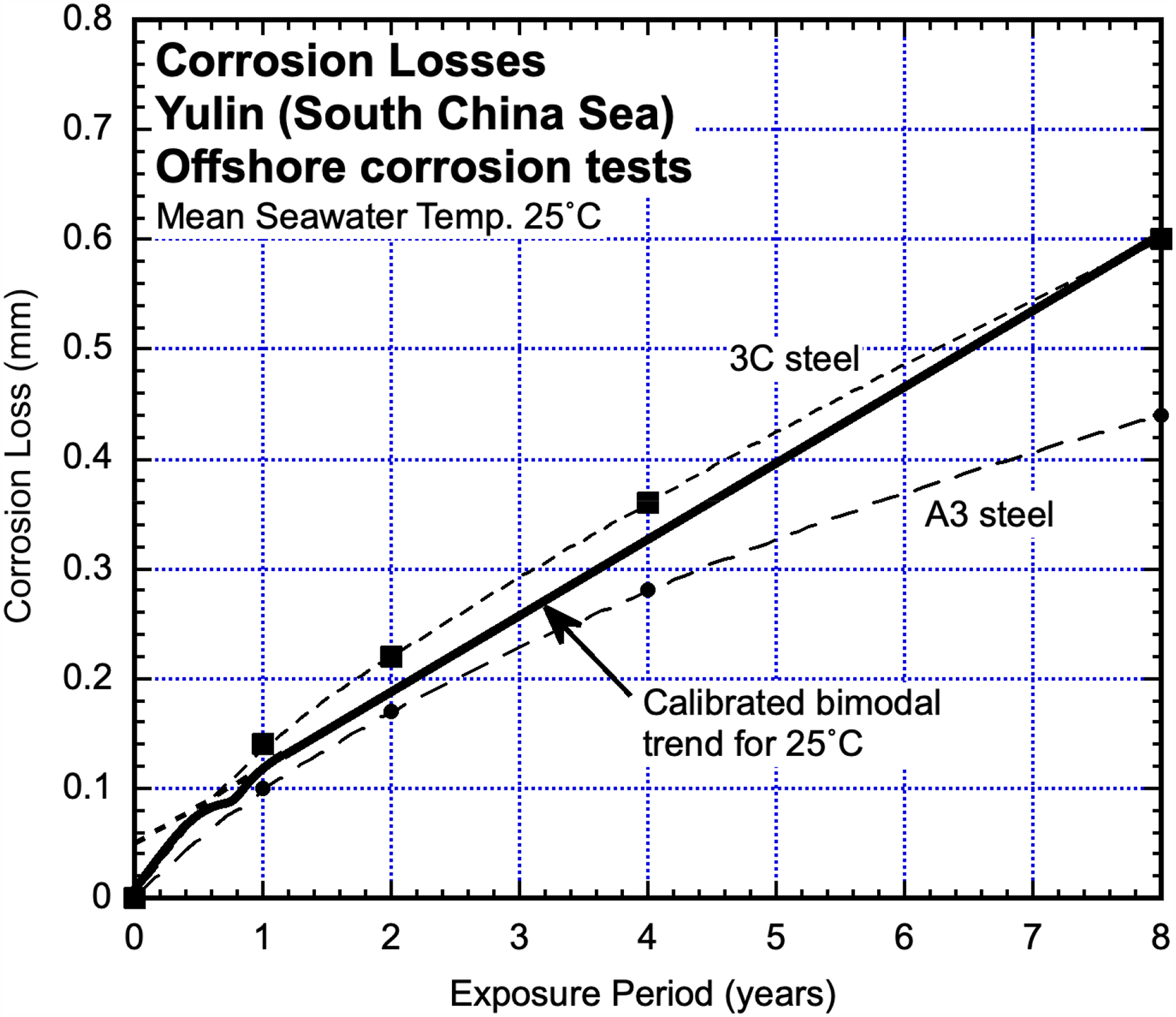
Yulin, SE China
Two steels A3 (0.2C, 0.55Mn, 0.009S, 0.015P wt%) and 3C (0.14C, 0.92Mn, 0.027S, 0.035P wt%) immersion tested for up to 8 years in the offshore zone off the coast of SE China near Yulin yielded corrosion loss trends as shown on Figure 2 from the data reported by Sun et al. 32 Dissolved inorganic nitrogen concentrations were not reported but have been derived from estimates of cs and rs made from the plots in Figure 2, with DIN concentration estimated using the calibrated data in Melchers. 8 This produced very low estimates of DIN concentration, typical of seawaters well away from anthropocentric pollution such as at river mouths and in coastal zones. The calibrated bimodal trend for essentially unpolluted seawaters at 25 °C is shown also.
Coffs Harbour Marina, NSW, Australia
The corrosion test site used at Coffs Harbour Marina is on the eastern Australia Pacific seaboard. The (measured) average seawater temperature is 21 °C. Steel test coupons were recovered and subject to corrosion mass loss observations at about 3-month intervals over some 1.8 years. The corrosion losses and the interpreted trend are shown in Figure 3. 27 The site has been subject to some degree of anthropocentric pollution.6,27 Quantitative data for DIN concentration was obtained from water samples taken at regular 3-month intervals, from which the average DIN concentration of 0.025 mgN/L was determined. As shown in Figure 3, this was checked against an estimate based on using the parameters cs and rs for the linearised version of Mode 2. It is seen that in Mode 1 there is only a small divergence of corrosion loss trends towards the end of Mode 1 but that, as expected, the divergence is much larger in Mode 2.
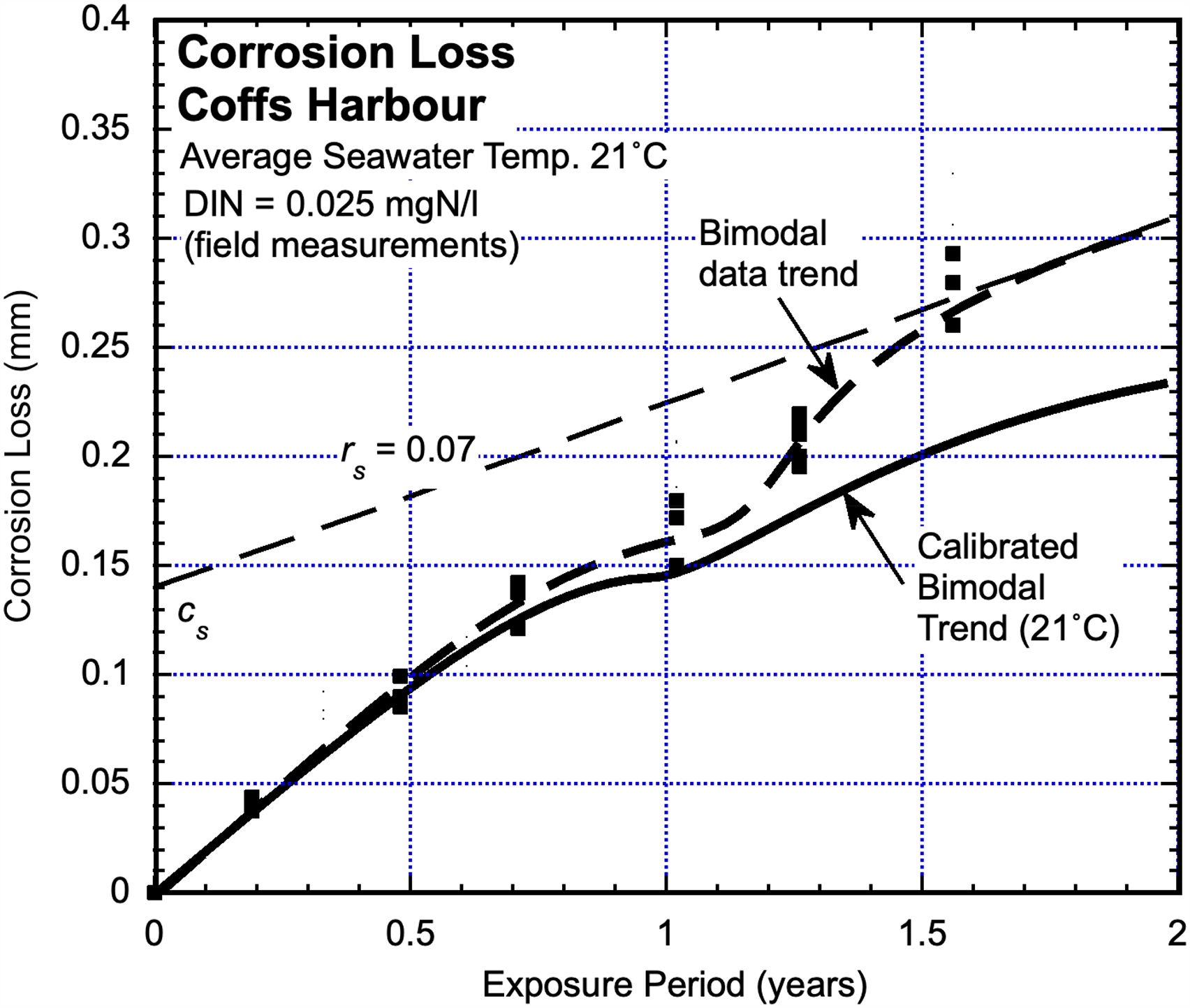
Corrosion loss data with best-fit trend for corrosion of steel coupons at Coffs Harbour Marina together with the trend from Figure 1 for corrosion loss in essentially unpolluted coastal seawaters at 21 °C mean temperature. 27
Xiamen, China
The corrosion site in this case lies in the outer reaches of the Jiulongjiang River on the East China Sea. It is subject to water velocities of around 0.7 m/s and possibly high concentrations of nutrients likely brought in by the river. The abundance of marine fouling at the site supports this qualitative assessment. The reported corrosion losses 32 over a period of 8 years are plotted in Figure 4 together with the bimodal trends through the data. The effect of DIN concentration is not particularly evident for corrosion in Mode 1 but is evident for the extended corrosion in Mode 2. The steel grades were A3 (0.2C, 0.55Mn, 0.009S, 0.015P wt%) and 3C (0.14C, 0.92Mn, 0.027S, 0.035P wt%).
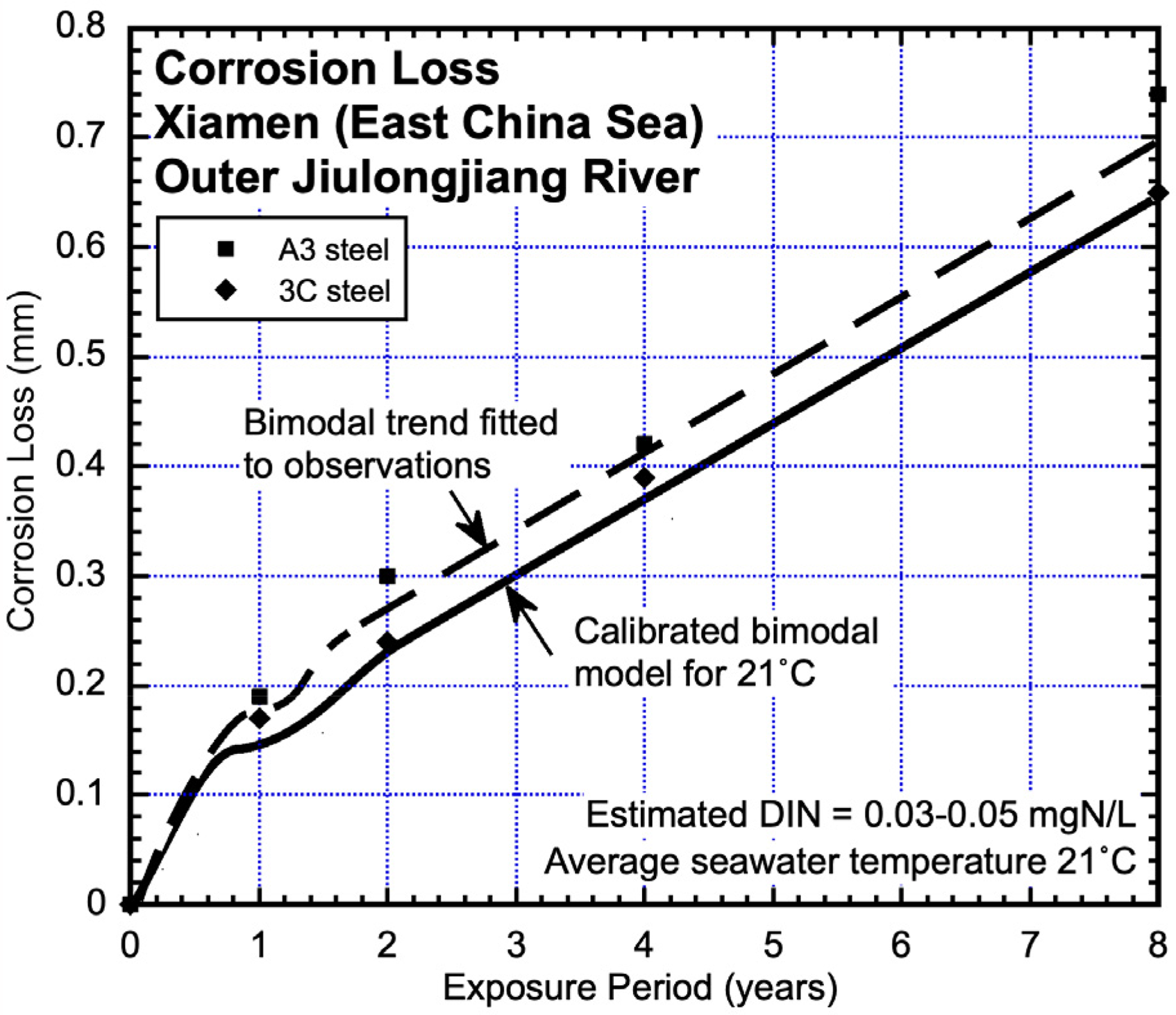
Corrosion losses (derived from Sun et al. 32 ) for general corrosion of A3 and 3C steels at Xiamen, with (subjectively) inferred bimodal trend with higher corrosion losses in Mode 2. The calibrated bimodal trend at 21 °C for unpolluted seawaters is shown. To estimate the parameters cs and rs Mode 2 was linearised as before for estimating the average concentration of dissolved inorganic nitrogen (DIN) at 0.03–0.05 mgN/L. No allowance was made for the effect of seawater velocity.
Kawasaki Bay (Japan)
Kawasaki Bay is part of the greater port region around Tokyo. Corrosion loss measurements have been reported by Tamada et al. 33 and are summarised in Figure 5, together with the best fit bimodal trend. The seawater averages about 17 °C. Although seawater was considered to be highly polluted in various ways, no detailed information for this site could be ascertained in the open literature. An estimate of the average DIN concentration was made using the long-term linear trend as shown with parameters cs and rs for Mode 2 as before. This provided an estimate for DIN concentration of 0.12 mgN/L.
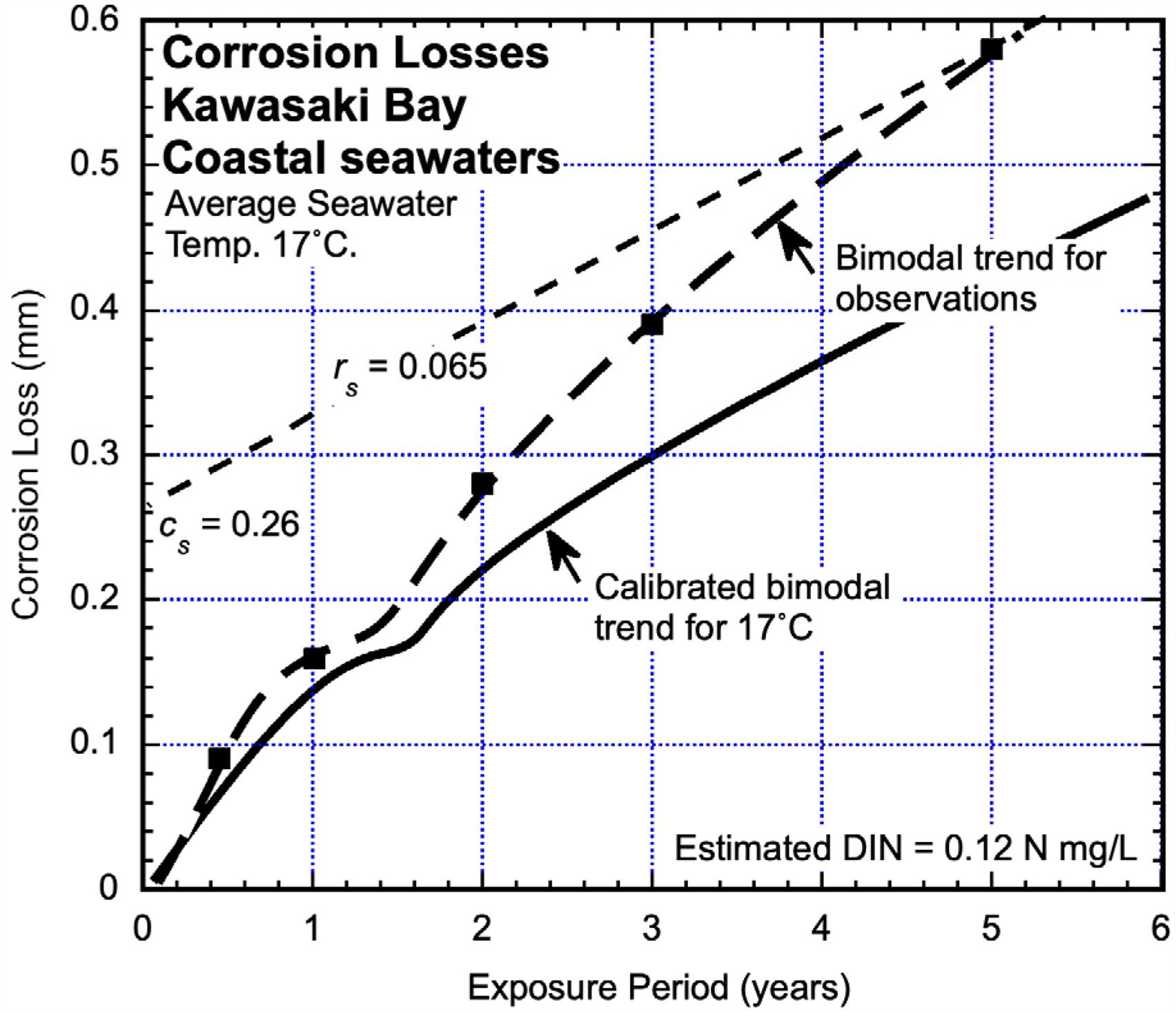
Corrosion loss trend based on numerical data reported by Tamada et al. 33 for Kawasaki Bay waters averaging 17 °C. Also shown is the calibrated bimodal corrosion loss trend for essentially unpolluted coastal seawater.
The effect of elevated DIN can be seen in Figure 5 also by comparing the data trend with the trend for corrosion loss expected in unpolluted coastal seawaters at 17 °C. 2 There is only a small difference between the trends for the period corresponding to Mode 1 but as before, there is considerable divergence between the trends for the period corresponding to Mode 2.
Langstone Harbour, UK
Langstone Harbour in the southern UK is open at the south to English Channel waters and although termed a ‘harbour’ it is essentially a bay subject to the inflow of coastal seawater and river inflows as well as outfall from a sewage plant. Maximum DIN concentration was estimated to be up to around 0.4 mgN/L 27 but over a 10-year period was about 0.15 mgN/L. 34 The mean seawater temperature is 12.5 °C. The corrosion losses reported by Phull et al. 35 for steel coupons are shown in Figure 6, together with the inferred bimodal trend through the data and the calibrated bimodal trend for essentially unpolluted coastal seawater at the same mean temperature. 4 There is little divergence between the trends in Mode 1 but a considerable divergence in Mode 2. Separate estimates for DIN concentration based on parameters cs and rs using the linearised version of Mode 2 support the field measurements for DIN concentration.
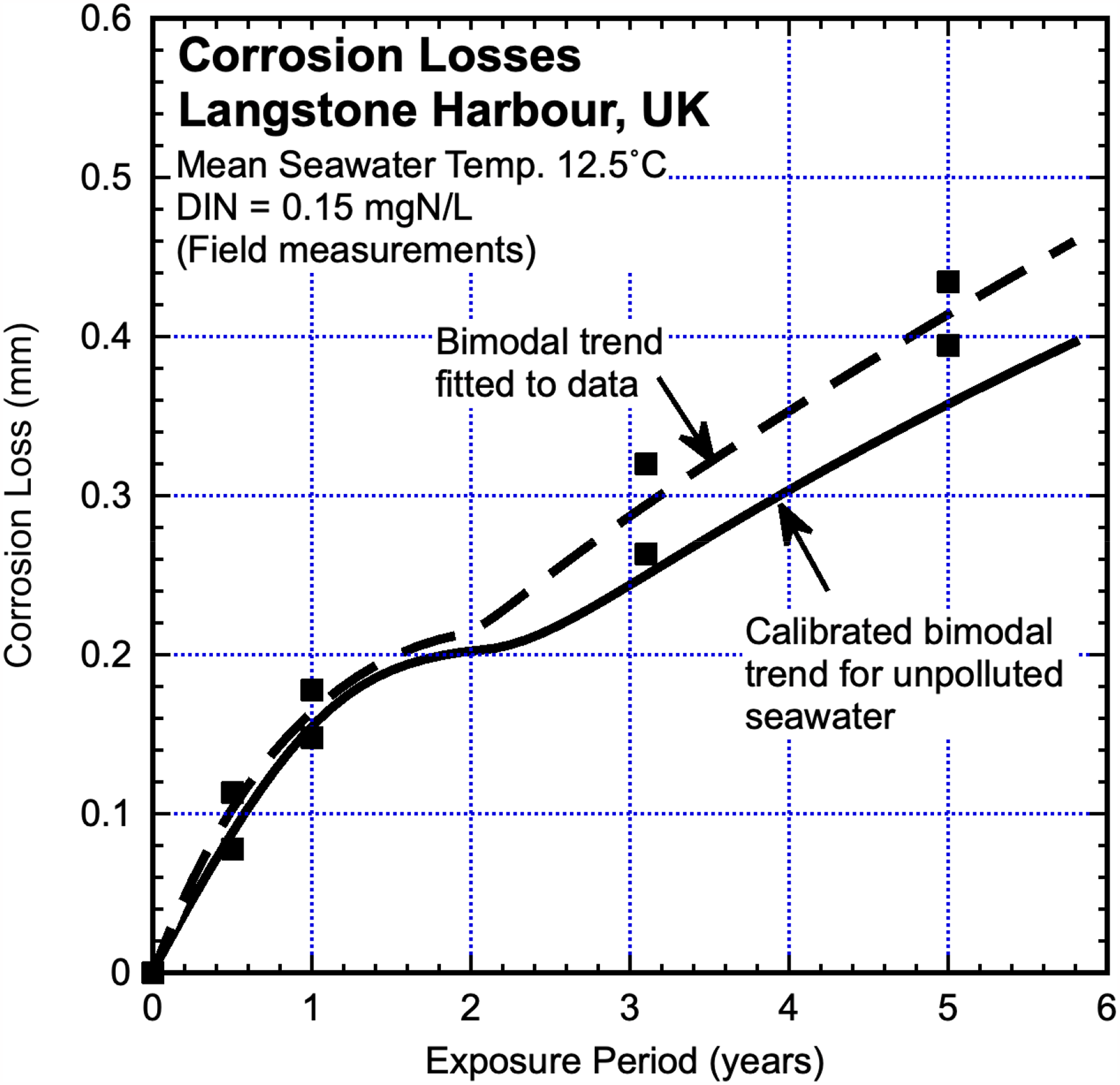
Corrosion loss data and inferred bi-modal trend, 27 for steel coupons exposed in Langstone Harbour seawater corrosion together with the trend from Figure 1 for corrosion loss in essentially unpolluted coastal seawaters at 12.5 °C mean seawater temperature.
Bombay High (Arabian Gulf coast)
The corrosion losses determined for mild steel coupons exposed at 2 m depth on a fixed offshore oil platform in the ‘Bombay High’ zone of the Arabian Gulf, some 170 km NNW offshore from Mumbai, are shown in Figure 7 based on numerical data reported by Sawant et al. 36 Similar corrosion loss values were reported for 22 m depth. The local water temperature varied from a minimum of 22.1 °C to 29.7 °C. This is consistent with other observations. 37 A bimodal trend line consistent with Figure 1 has been drawn through the data. The trend for corrosion loss in unpolluted seawaters at the same average water temperature (26 °C average) is as shown. The observed corrosion losses in what can be interpreted as in Mode 2 are very high. This was attributed37,38 to be the result of the considerable marine growth observed at the site. Another interpretation is that the high corrosion losses and the marine growth reflect high concentrations of nutrients in the seawater. Since DIN concentrations were not reported and did not appear to be available from other sources or nearby waters, they were estimated from Mode 2 using the parameters cs and rs. This gave an estimate of around 0.19 mgN/L. There is some uncertainty about the exact trending for corrosion loss in the period corresponding to mode 1 but it is clear that it is only marginally greater than for unpolluted seawater.
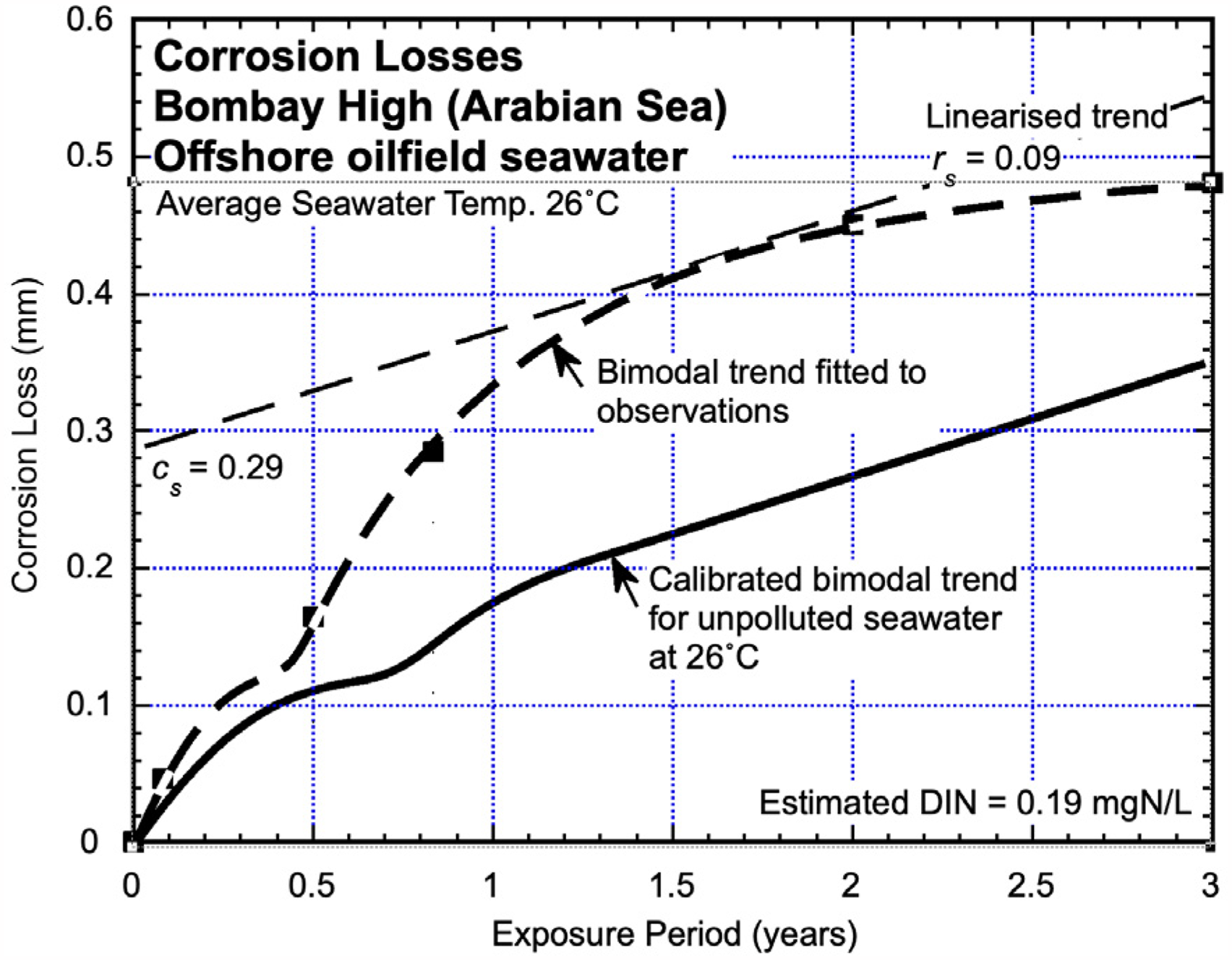
Corrosion loss data and inferred trend, for nutrient-polluted seawater corrosion together with the calibrated bimodal trend for corrosion loss in essentially unpolluted coastal seawaters at the same mean seawater temperature. 36 Only the data for corrosion losses at 2 m depth are shown. Similar corrosion loss values were reported for 22 m depth.
Port Hueneme, CA, USA (1950s)
The US Naval establishment at Port Hueneme (CA) is situated on the southern Californian coast some 100 km northwest of Los Angeles. It has along with history of marine exposure testing, mainly at a site near the harbour entrance. Corrosion data for mild steel coupons tested in the 1950s 39 is shown in Figure 8. Compared to the trend derived from the calibrated corrosion loss model 2 for unpolluted seawaters at 15 °C mean seawater temperature shows similarity for the early exposure period but a growing and significant departure for exposure periods longer than about 1.5 years (Figure 8). In this case, the corrosion loss trend corresponding to Mode 1 (Figure 1) is similar for both the DIN-polluted seawater environment and the natural unpolluted environment after allowing for mean seawater temperature. In Mode 2, however, the corrosion loss trend for the 1950s data is much higher.
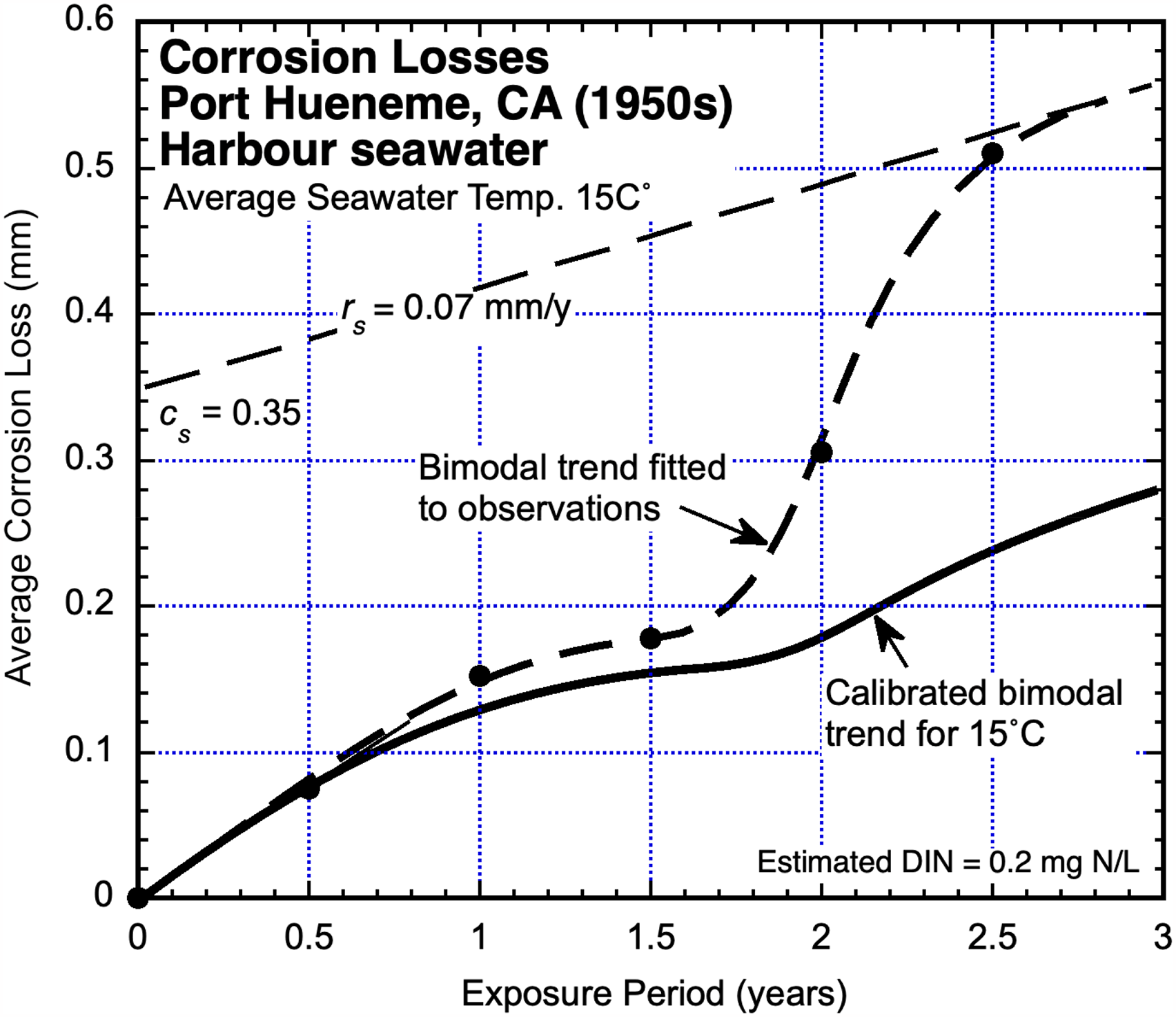
Corrosion loss data reported for the seawaters immediately off Port Hueneme (CA) 39 with inferred bimodal trend, based on the corrosion loss model with dissolved inorganic nitrogen (DIN) pollution influence in Mode 2 of Figure 1, compared with the bimodal trend for unpolluted coastal seawater at the same average water temperature.
For this site field data for DIN concentrations were not available. Instead, the average DIN concentration was inferred from the plot of Figure 8 using the parameters cs and rs estimated for the linearised Mode 2, and from Melchers. 8 This indicated a value of 0.2 mgN/L.
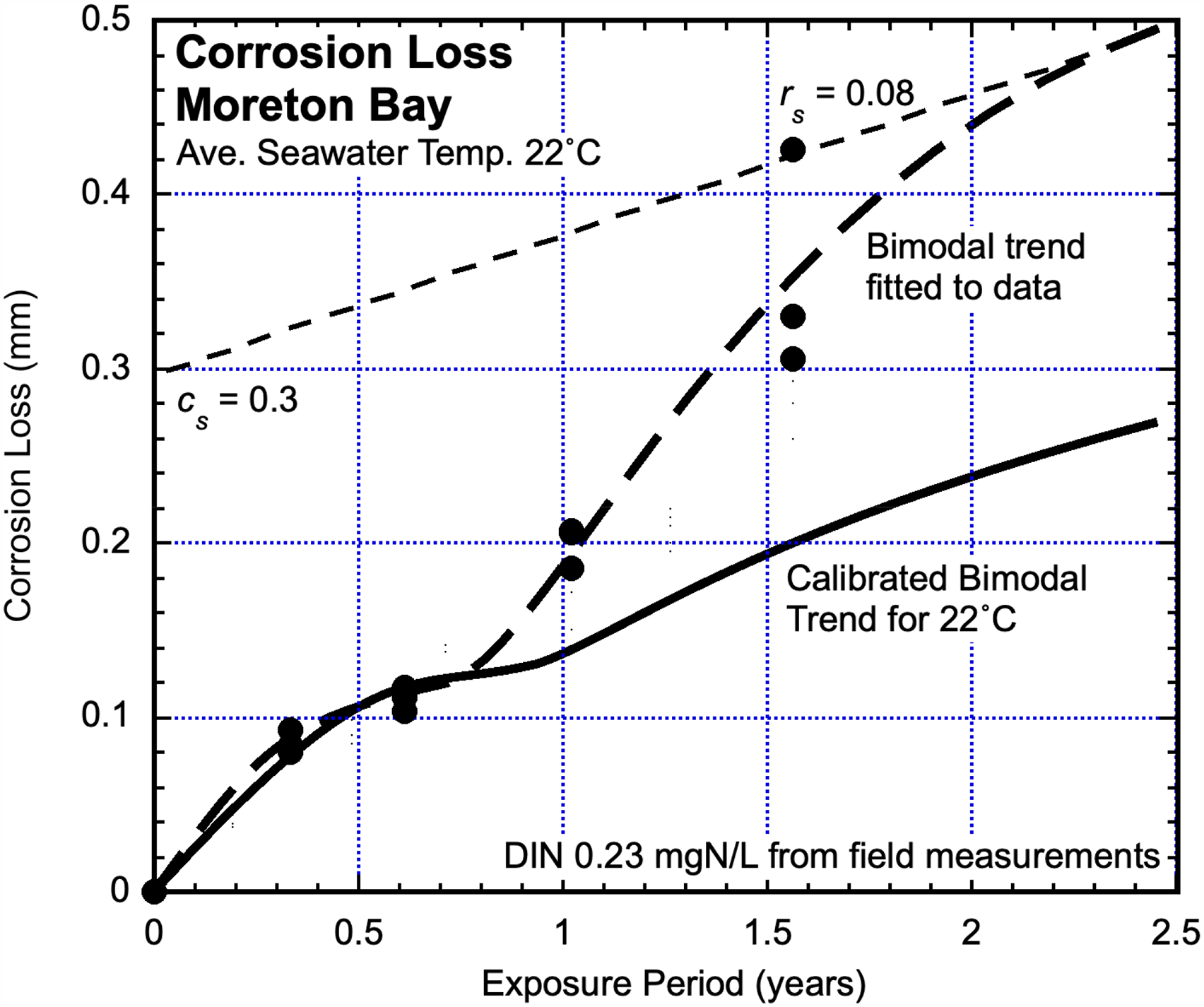
Moreton Bay, Queensland, Australia
The corrosion losses measured over some 2 years at a depth of 1.2 m at an exposure site within Moreton Bay6,27 are shown in Figure 9 together with the fitted bimodal trend. The bimodal trend for unpolluted natural seawater at the same average temperature is shown also. There is little divergence between the trends for Mode 1 but considerable divergence for Mode 2. Water quality investigations showed that the high DIN concentration for Moreton Bay coastal waters could be attributed to elevated concentrations of nitrates and phosphates caused by runoff of agricultural fertilisers used for local banana plantations draining into the bay and dispersing through the local Pacific Ocean coastal seawaters. 27 Field investigations showed that the average DIN concentration was 0.23 mgN/L. This was confirmed by using the linearised trend for Mode 2 (Figure 9) and the parameters cs and rs as before.
Qingdao oil exploration area (China)
Two steels A3 (0.2C, 0.55Mn, 0.009S, 0.015P wt%) and 3C (0.14C, 0.92Mn, 0.027S, 0.035P wt%) immersion tested for up to 8 years in the oil exploration area near Qingdao, China, 32 produced the corrosion losses shown in Figure 10. Additional test data for up to 3 years of exposure, and thus mainly in Mode 1, were reported by Li et al. 40 They are consistent with the trends shown.
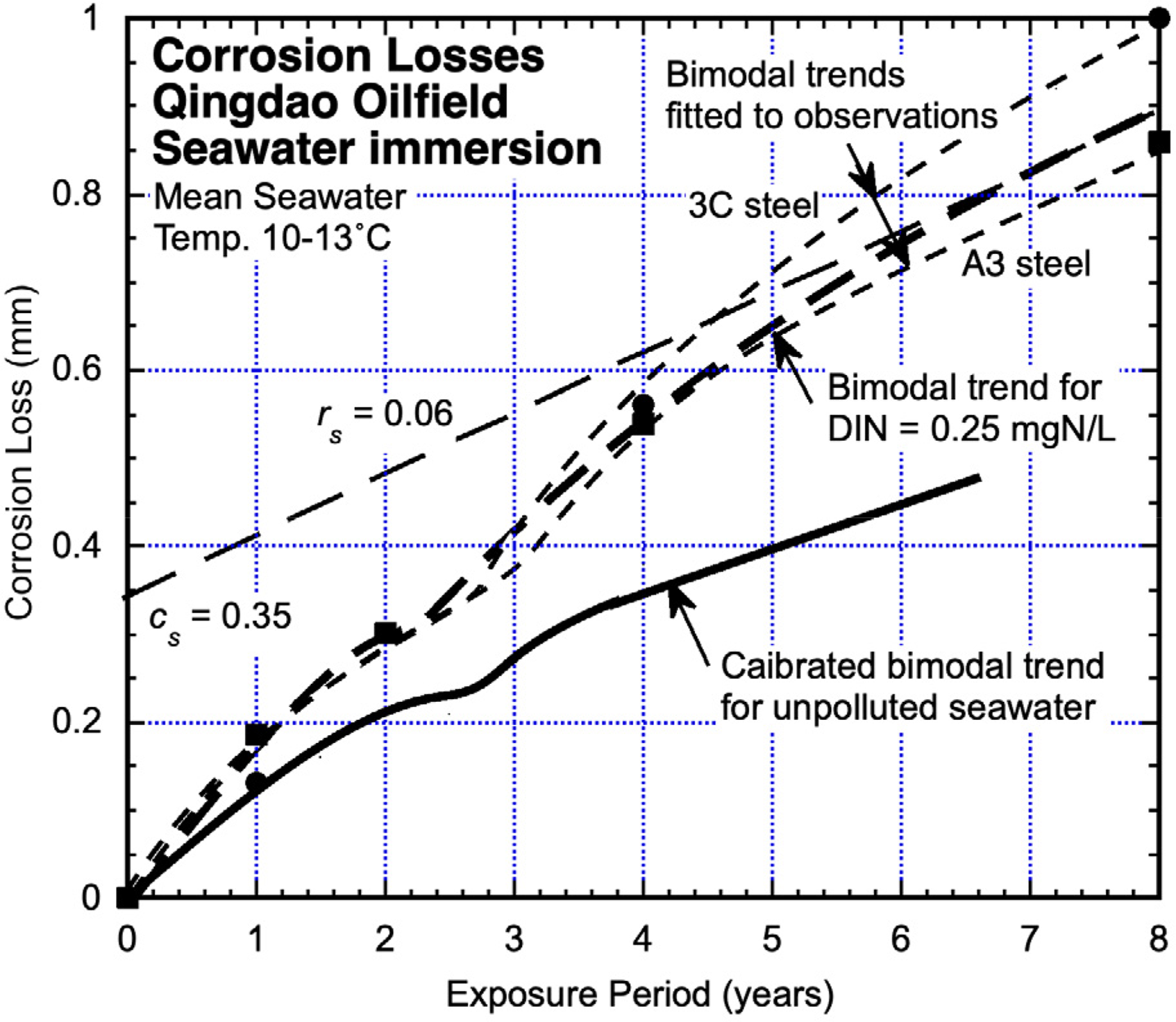
Corrosion loss data (derived from Sun et al. 32 ) for general corrosion loss for A3 and 3C steels at Qingdao, with inferred bimodal trends and the calibrated bimodal trend for essentially unpolluted seawaters at 11 °C.
The test site lies towards the north of the Yellow Sea and has a strong influence on tidal conditions, perhaps accounting for the reported low incidence of marine growth even though the site is located within an oil exploration area. Observations regarding the bacterial species involved in the early exposure period have been presented by Duo et al. 41
It is evident that the divergence between the trend for nominally unpolluted seawaters and that for the Qingdao seawaters increases during Mode 1 and is considerable in Mode 2. This is consistent with an earlier assessment of the environmental conditions at this site. 42
No information about DIN concentrations for this site appears to have been reported but other oil exploration fields suggest high values. Using the parameters cs and rs and the tangent to long-term corrosion in Mode 2 as before, an estimate was made of average DIN concentration. This produced 0.25 mgN/L. In this case, corrosion in both Mode 1 and Mode 2 is considerably higher than for unpolluted seawaters at the same mean seawater temperature.
North Sea
Data for corrosion loss derived from mass loss for low-alloy steel I4 in the North Sea at 1.6, 2.7, 4 and 7.2 years of exposure, all at 10 °C average seawater temperature, is shown in Figure 11 2 derived from date reported by Blekkenhorst et al. 43 All data points are the average of five individual observations. The corrosion losses for various other low-alloy steels of slightly different compositions tested at the site were very similar to those shown. A best-fit trend is shown fitted to the data. Also shown is the trend expected for unpolluted seawater, based on calibration data for the bimodal model (Figure 1). The trends are closely similar for the part of the trend corresponding to Mode 1 but for Mode 2 the corrosion losses are much greater than predicted by the calibrated model. Independent water quality assessments show elevated concentrations of DIN throughout the year at the exposure site in the North Sea (OSPAR 2000) with average DIN about 0.27 mgN/L. 27 This value was independently confirmed through the approach of measurements of the parameters cs and rs for the linearised long-term tangent corrosion loss trend for Mode 2.
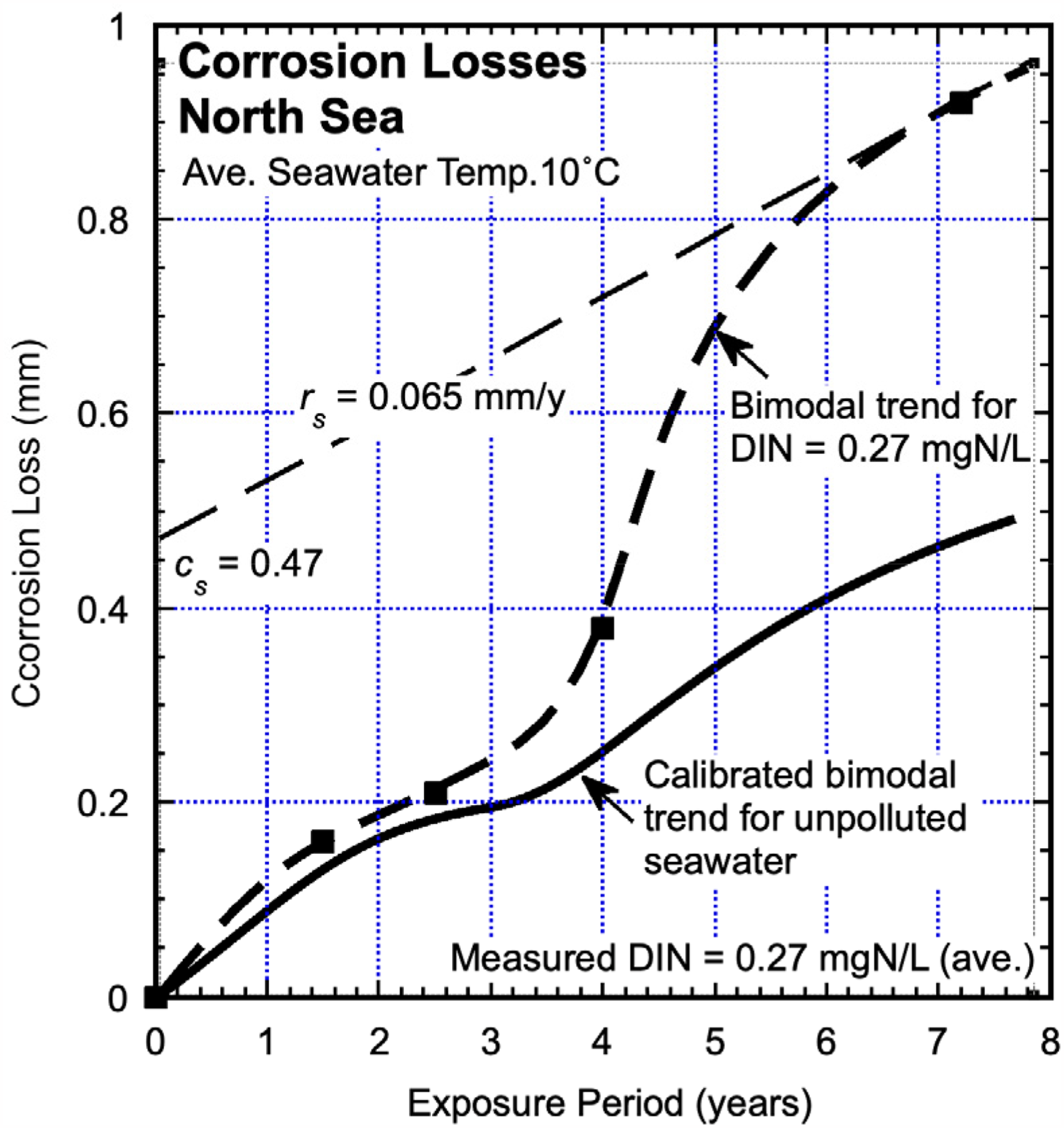
Corrosion loss observations for low alloy steels (represented by alloy I4) in the North Sea off the Scottish coast, averaging about 10 °C, showing also the expected trends for corrosion in such seawaters according to the calibrated bimodal model for natural seawater.
Port Hueneme, CA, USA (1990s)
A later exposure program at the Port Hueneme site, also using steel coupons 35 produced data shown in Figure 12. The water temperature at the site would have the same mean (15 °C) as in the 1950s program (Figure 8) but as evident in Figure 12, the corrosion losses for both Mode 1 and Mode 2 are higher than those shown in Figure 8. This suggests there was an elevated degree of seawater pollution during the test program for the data for Figure 12 (i.e. early 1990s). But quantitative data to support this supposition could not be ascertained. However, there is some anecdotal information 6 along with the lines that local surf-riders claimed (in online chats) to have observed periodic ‘brown coloured effluent’ apparently from a local waste-water treatment plant. That information has since disappeared from the web, an independent estimate of average DIN concentration related to the data in Figure 12 was made using the long-term linear tangent to the corrosion trend in Mode 2 and the parameters cs and rs as before. This produced an estimate of about 0.4 mgN/L.
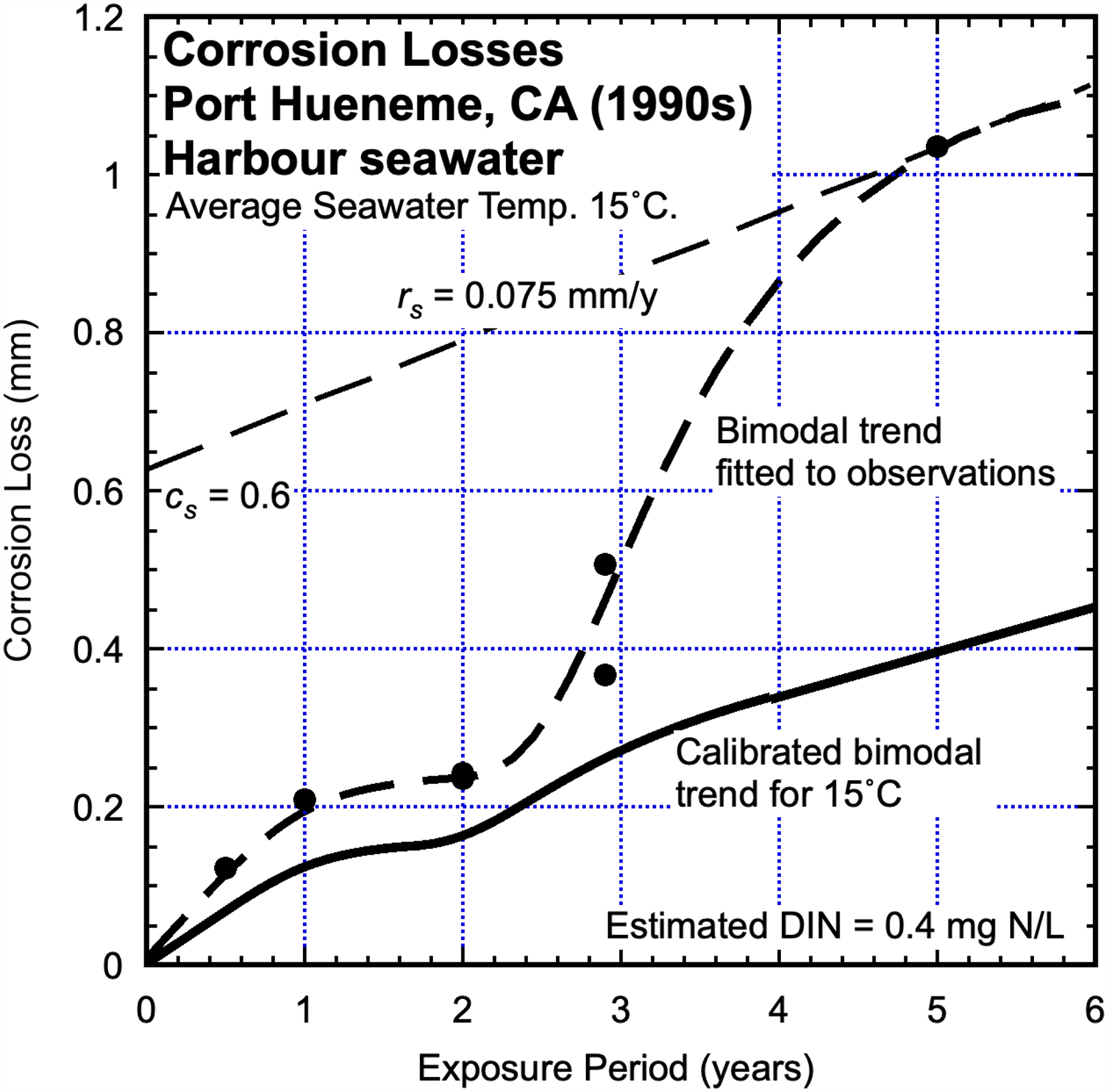
Corrosion losses for steel coupons as reported by Phull et al. 35 for the seawaters immediately off Port Hueneme (CA) with bimodal trend added to the data. Also shown is the calibrated bimodal trend for similar steel in essentially unpolluted coastal seawater.
Wrightsville Beach, NC, USA
As part of the ASTM worldwide marine corrosion study steel coupons were exposed in Banks Channel near the river mouth and the then LaQue Corrosion Centre at Wrightsville Beach (NC). Banks Channel is part of the coastal seawater waterway along the North Carolina Atlantic coast. 35 Figure 13 shows the reported corrosion losses, together with the inferred bimodal trend through the data. 27 The calibrated bimodal trend for steel in natural seawater has been added to Figure 13. It is clear there is elevated corrosion loss both in Mode 1 and in Mode 2. This is consistent with estimates of the local DIN concentration at the site around 1.2 mgN/L and with other information that indicates issues with sewage treatment particularly during the vacation periods in summer and corresponding to the local influx of tourists. 6 However, interpretation from the corrosion loss plot for long-term corrosion in Mode 2 and using estimates for cs and rs indicates more likely DIN = 0.5 mgN/L on average.
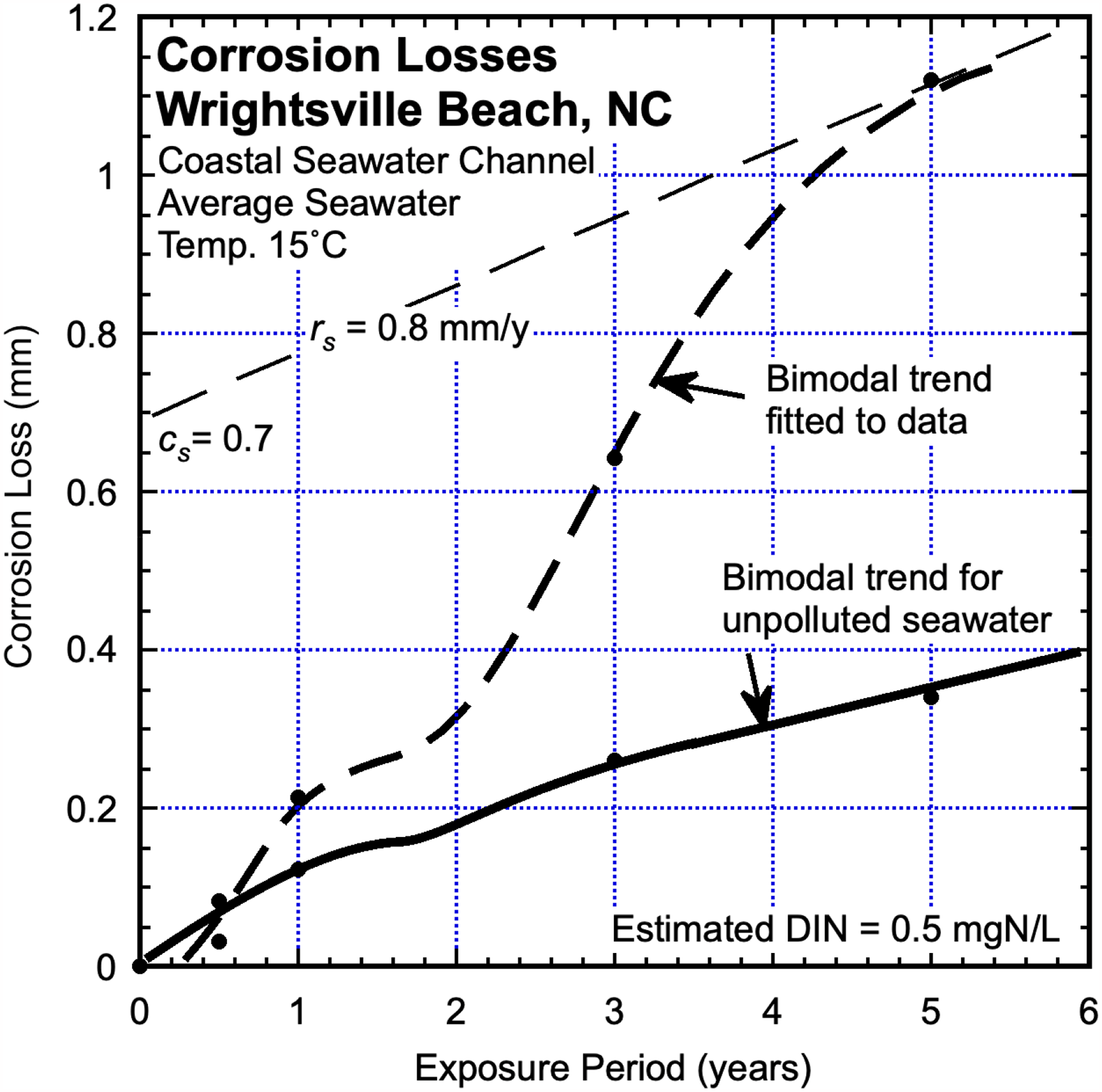
Corrosion loss data and bimodal trending for steel coupons exposed in nutrient-polluted seawater corrosion together with the bimodal trend from Figure 1 for corrosion loss in unpolluted coastal seawaters at the mean seawater temperature (15 °C). 27
Interpretation of observations
The trends shown in Figures 2–13 all show that relative to the corrosion loss trend for natural seawaters, that is, those with background or negligible DIN concentration, all show relatively much greater corrosion in the part of the trend corresponding to Mode 2 in Figure 1. Only a few cases (Figures 10–13) show a high degree of corrosion in Mode 1. These had DIN concentrations greater than about 0.25 mgN/L.
Careful examination of the cases shows that at a given interval after ta (see Figure 1), say at 1.5 ta, the increment in corrosion loss over and above that for natural seawater is approximately 80–100% for those cases with DIN concentration greater than about 0.2 mgN/L. For lower DIN concentrations, the increment in corrosion loss is much less and roughly proportional to DIN concentration. Only a small number of cases underpin these rather subjective observations but they nevertheless are as might be expected. Thus, increased DIN concentration can be expected to increase the rate of microbiological metabolism, but only up to a point, as noted earlier for field observations. 24
A second observation is that the length in time of Mode 1, that is, from time zero to the transition time ta appears to reduce slightly with increased DIN concentration. This can be seen by comparison of Figures 2–13. Such a reduction might be expected. While the dominant cathodic reaction in Mode 1 is oxygen reduction, there also is a gradual increase in participation by pitting corrosion and related hydrogen evolution from the increasing participation of the water reduction reaction within the anaerobic locations within pits as they develop. These locations provide increased venues for MIC as the availability (i.e. concentration) of DIN increases. These permit the onset of the anaerobic corrosion-dominated Mode 2 to be possible earlier, reflected in the corrosion development as ta occurring slightly earlier in time.
Apart from the small number of cases with sufficient data for the present analyses, any apparent inconsistencies may be the result of incomplete or inaccurate information about the DIN concentrations at the specific corrosion testing sites. As noted, the concentrations quoted herein and in the references were not obtained in the field at the time any of the corrosion experiments were carried out. Where specific information is quoted, in all cases the data were obtained by cross-reference to other sources, 27 and this adds the possibility of inaccuracy in the DIN estimates, particularly since available evidence shows considerable spatial and geographic variability in DIN concentrations (e.g. OSPAR 44 ). The immediate conclusion is that any future seawater corrosion testing programs that encompass the possibility of MIC involvement must include detailed and seasonal monitoring of DIN and in particular its components ammonia, nitrites and nitrates as well as potential corrosion inhibitors in the immediate neighbourhood of the steel at the exposure site.
Throughout the present analyses, it was assumed that the annual average DIN concentrations at an exposure site remained very similar throughout the whole period of exposure, at least for the relatively short periods of exposure considered herein. It is known from other observations that there has been a gradual increase in such concentrations with time (cf. OSPAR 44 ) but this is not considered as a significant factor for the cases considered herein.
Discussion
To throw some light on the behaviour observed in the above cases and in particular the observation of little effect of DIN on corrosion in Mode 1, several matters might be noted. The current conventional wisdom is that the microorganisms involved in MIC are ubiquitous, that environmental conditions and the availability of nutrients govern their metabolism and that this governs both their multiplication rate, and thus their numbers and the amount or severity of MIC. 9 Importantly, these considerations do not distinguish between species – they apply to all living things in both aquatic and terrestrial ecosystems 19 and thus to aerobic as well as anaerobic species. On this basis, without other considerations, it would be expected that corrosion would increase under higher DIN concentrations. This is irrespective of the dominant bacteria species involved and whether or not the conditions are aerobic or anaerobic. In turn, this indicates that the effect of DIN should be similar for both Modes 1 and 2 of Figure 1. The only difference is the aggressiveness of the microbial species for contributing to MIC. In the present case, the microorganism in Mode 1 includes species such as the iron oxidising bacteria. These usually are considered less aggressive than the sulphate-reducing bacteria mostly held responsible for contributing to MIC in Mode 2.
In the above, the bi-modal model trend for corrosion in natural seawater was used as the basis for comparisons. That model was originally derived for what were considered unpolluted natural seawaters. Subsequently, it was recognised that at most location used in that work, the concentration of DIN was at a level slightly higher than zero and that typically there is a ‘background’ concentration of DIN, even though at many locations this is close to zero. 20 Experience with data collection and availability shows that for practical and economic reasons most commercial testing of nitrates, nitrites and ammonia is limited to providing results with a lower limit typically given as <0.01 mgN/L. The question that arises is whether this is sufficiently accurate for assessing the effect of DIN concentration on corrosion, particularly on MIC in Mode 2. An estimate of this can be seen in results from laboratory tests using flow-through seawater with some background DIN concentration and the same seawater subjected in a separate flow through system to fine filtration and to along-flow UV radiation, the latter in an effort to eliminate live microorganisms and thus eliminate MIC. 45 The UV treatment has been shown to be very effective in almost completely killing microorganisms. 46 The comparison between the corrosion from the two streams over a period of 3 years is shown in Figure 14. There is little difference between the trends in Mode 1 and the trends in Mode 2 are quite close.
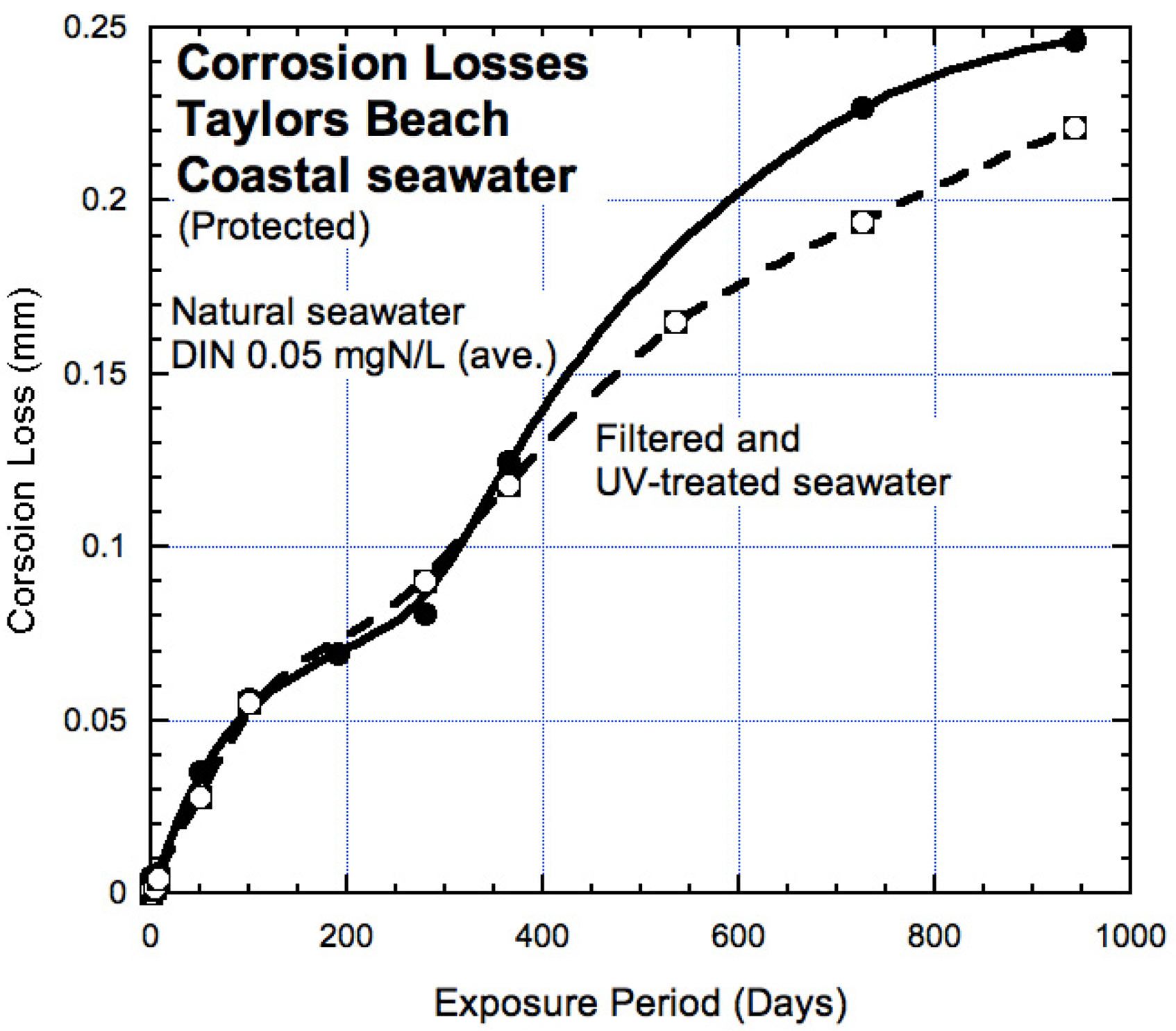
Corrosion loss from mass loss observations for steel coupons exposed to natural 20 °C (average) seawater and similar coupons exposed to filtered and UV-treated seawater, both at the Taylors Beach test site. The data for the latter have been reduced to allow for the slightly higher water temperature resulting from the UV treatment. 45
Apart from affecting the rate of metabolism of microorganisms, the ammonia, nitrite and nitrate components of DIN are known to have some capacity to act as corrosion inhibitors. This is particularly the case in aerated waters. Ammonia solutions have shown extremely low corrosion for a wide variety of metals and alloys during exposures up to 8 months in enclosed waters, an effect attributed to development of increased local pH typically adding corrosion products less permeable to oxygen 47 and relevant for corrosion in freshwater streams immediately downstream of outfalls from secondary treatment sewage plants and downstream of steel mills and ammonia plant discharging condensate waters. 48 However, in open waters such as coastal seawaters, ammonia as a corrosion inhibitor is much less relevant. In aerated waters ammonia oxidises, relatively quickly, to nitrite and then to nitrate, a process thought to be carried out exclusively by prokaryotes as part of the nitrification cycle. 49 In terms of the progression of corrosion as summarised in Figure 1, ammonia may have an influence during Mode 1 but in open waters this is likely short-term and only near the release of ammonia-contaminated waters.
Nitrites, in particular calcium nitrite Ca(NO2)2 are known corrosion inhibitors 50 and effective in inhibiting corrosion of low alloy and mild steels in chloride solutions, including seawater and for steel reinforcement in concretes 51 and stainless steels 52 as demonstrated in longer-term tests. 53 The conventional notion is that Ca(NO2)2 acts as anodic inhibitor, based on the observation that in the presence of oxygen and water, it facilitates the formation of ferric hydroxide (Fe(OH)3), magnetite (Fe2O3) and hydrated ferric nitrite (Fe(NO3)3) as additional corrosion products and that these are considered to reduce diffusion processes from anodic regions (pits).50,54 This assumes that anodic polarisation is the corrosion rate–controlling process. However, this does not accord with the conventional and longstanding interpretation that the earlier part of the corrosion process, in particular phases 1 and 2 in Figure 1 are rate-controlled by cathodic polarisation, specifically by the much slower rate of diffusion of oxygen from the external environment through the corrosion products to the corroding metal (steel).55–57 The observed formation of the additional corrosion products noted above would increasingly reduce oxygen diffusion, consistent with cathodic polarisation as the corrosion rate-controlling mechanism. In terms of the contribution of MIC to the corrosion process, it is noted that during the predominantly aerobic Mode 1 species such as iron oxidising or sulphur oxidising bacteria would be feasible and would be directly affected by the rate of oxygen supply. Thus, increased nitrite availability would, on the one hand, inhibit abiotic oxidation of the steel through the formation of additional corrosion products while on the other hand tend to increase the rate of metabolism of the oxidising bacteria.
The effectiveness of calcium nitrate Ca(NO3)2 as a corrosion inhibitor is less well-understood, but, like calcium nitrate, appears to work by forming additional corrosion products.58,59 These have been proposed as blocking or inhibiting anodic reactions such as pitting. Such deposits also have been proposed as modifying local electrical field concentrations, based on short-term 7-day immersion tests. In 10 mass% CaCl2 solution general corrosion of steel showed rather unclear inhibition as Ca(NO3)2 concentrations increased (0.1–10.0 mass%) but pit depth was found to decrease to give ‘flatter’ pits (Dong et al., 60 Figure 2(a) and (c)). However, it is doubtful whether these short-term tests can adequately represent longer-term exposures, mainly because the corrosion process develops along the lines represented in Figure 1, for which, after the early phase (phase 0), the rate-controlling step is cathodic polarisation through oxygen diffusion. Despite some uncertainty regarding the mechanisms involved, practical experience has shown that nitrates can be effective inhibitors.50,54
In view of the above comments, it is unlikely that the corrosion-inhibiting effect will be the same for ammonia, for nitrite and for nitrate. Hence, a broad categorisation such as concentration of DIN or total inorganic nitrogen is not sufficient to assess the degree of corrosion inhibition – the proportion of the components needs to be known. Also, the inhibition effect of each needs to be known – this is a clear area where further research is required. On the other hand, for the opposing effect of bacterial metabolism and consequent greater rate of corrosion, the concentration of DIN or of total inorganic nitrogen, is sufficient.
Turning now to the situation for corrosion in Mode 2, the oxygen diffusion corrosion rate-controlling effect for Mode 1 does not apply for the predominantly anaerobic corrosion in Mode 2. Instead, the rate-controlling process for Mode 2 is hydrogen gas diffusion outwards from the cathodic reaction, which is now the hydrogen evolution reaction. 25 For this reaction, the electron acceptor is hydroxide, from dissociation of water. 61 Since no oxygen is involved, it follows that there can be no inhibition effect associated with ammonia, nitrite or nitrate along the lines noted above. On the other hand, in the predominantly anaerobic conditions of Mode 2 the metabolism of anaerobic bacteria, such as the sulphate-reducing bacteria, is possible, provided energy and critical nutrients are available. This is clearly the case for the cases in Figures 7–13, with increased concentration of DIN causing considerable increased corrosion in Mode 2.
Regarding the contribution of bacterial metabolism to the early part of Mode 1 (phase 0), it has been shown previously 4 that, at least for a quite short period of time, high concentrations of anaerobic microorganisms such as SRB may exist and add to overall corrosion loss. 27 This observation was based on correlating detailed observations of corrosion with observed incidence of bacteria, apparently principally SRB. 41 Subsequent corrosion was found to be under the influence of oxidising bacteria (iron oxidising bacteria and sulphur oxidisers). Evidently, this short-term effect is not obvious in most of Figures 2–10 owing to the lack of corrosion date for the very early exposure period.
The above observations have an important implication for attempting to use laboratory experimental results to predict the likely effect of MIC in field exposures. Most studies of MIC are performed under closely controlled, rather artificial experimental conditions with monocultures and high concentrations of nutrients. In the main, corrosion effects of anaerobic microorganisms have been considered. 11 The above analysis shows, however, that for prediction of the likely practical outcomes, both the early and the later effects of microbial corrosion should be considered and placed in the context of the development of the corrosion conditions as in Figure 1. It shows the relationship between short-term corrosion behaviour and longer-term term corrosion in the same environment. Thus, the corrosion loss in Mode 1, including any inhibition, will carry over into Mode 2, reflected in parameter ca, and thus in parameter cs, irrespective if parameter rs. It is also now clear that in terms of the effect of DIN on MIC there is unlikely to be a one-to-one correspondence between the effect in Mode 1 and the effect in Mode 2.
In summary, the influence of DIN is not just through MIC but also through the potential to inhibit abiotic corrosion in the early stages, with carry-over effects to subsequent longer-term corrosion. This is most clearly seen through the effect of lowering the value of parameter ca (Figure 1) as a result of inhibition. From a practical perspective these considerations, supported by the trends in Figures 7–13, indicate that it is not feasible at present to extrapolate from Mode 1 behaviour to longer-term corrosion behaviour. As noted above, greater quantitative understanding of the inhibiting effects of the components of DIN is required to begin to make this possible – a matter that requires further research. In addition, a much improved, more extensive database for actual corrosion losses under different DIN scenarios and the nitrogenous components of DIN will be necessary for model calibration.
For practical applications and predictions, differentiation between the nitrogenous components of DIN can now be seen to be necessary also to assess their potential for corrosion inhibition in Mode 1 of Figure 1. Such differentiation likely will enable improved understanding of those factors most responsible for any extra corrosion loss in Mode 1. This likely has zero-cost implications since most water quality laboratories already analyse separately for nitrites, nitrates and ammonia so as to provide their estimate of inorganic DIN.
Conclusion
The development of MIC for steels in marine exposures does not follow a monotonic pattern with advancing exposure time but, after an initial short period with biofilm involvement, develops under aerobic conditions in Mode 1 of the bimodal model, involving oxidising bacteria, and after anaerobic conditions develop under rust deposition in Mode 2, involving the usual SRB (sulphate-reducing bacteria) and other anaerobic microorganisms.
Generally, the severity of MIC increases with temperature and with higher concentrations of DIN but for the predominantly aerobic Mode 1 of the bimodal model the ammonia, nitrites and nitrates constituting DIN may act as corrosion inhibitors and thus reduce the total corrosion in Mode 1 but not in Mode 2.
Abiotic corrosion mechanisms and the contributions of microorganisms differ between Modes 1 and 2. As a result, they contribute differently to the non-linear (or simple) development of corrosion loss with continued exposure, typically with corrosion loss development following the bimodal model. This means it is usually not feasible to predict longer-term corrosion development from trending and information for short-term observations.
To permit development of the capability to predict MIC for shorter-term exposures it is necessary to also measure and report separately the concentrations of ammonia, nitrites and nitrates close to the exposed steel and to perform calibrations against observed corrosion losses.
Footnotes
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.