Abstract
We report two cases of myxoedema coma that presented to the acute medical take with severe bradycardia. These patients were initially misdiagnosed as bradyarrhythmia of primary cardiac origin. They were then diverted to the cardiology service at another district general hospital (DGH) for admissions. Both cases were subsequently diagnosed with myxoedema coma having screened thyroid function tests on arrival at the cardiology unit. Despite being treated for myxoedema coma, both patients unfortunately succumbed to the disease and later died in the hospital. These cases highlight that clinical suspicion and recognition of myxoedema coma remain significant challenges in a developed world despite readily available and highly sensitive thyroid hormone assays.
Introduction
Myxoedema coma is a form of decompensated hypothyroidism characterised by marked impairment of the central nervous system, cardiovascular function and thermoregulation. Although it is a relatively rare occurrence nowadays in the United Kingdom given the widespread availability of thyroid-stimulating hormone (TSH) assays and frequent monitoring by primary care physicians, it still carries a high mortality in the elderly. We discussed about two elderly gentlemen with no prior history of hypothyroidism presented to accident and emergency (A&E) with severe bradycardia and hypothermia. With these two case stories, we hope to draw the attention of acute physicians to have myxoedema coma as a possible differential in mind in elderly patients with severe bradycardia.
Case presentations
Patient A
An 82-year-old man presented to acute medical take with dry cough, reduced mobility and confusion for 4 days. He was normally independent in activities of daily living and lived alone. His past medical history includes hypertension and ischaemic heart disease. The initial vital signs on arrival were as follows: Glasgow coma scale (GCS) 14, tympanic temperature 34.8°C (rectal temperature 28.3°C), blood pressure (BP) 104/70 mmHg, heart rate (HR) 29/min and oxygen saturation (SpO2) 97% on 4L nasal cannula. Physical examination was remarkable for macroglossia with lower limbs oedema up to his thighs. His heart sounds were dual and not muffled to auscultation. His electrocardiogram (ECG) revealed junctional bradycardia with HR 30/min with a known right bundle branch block (Figure 1). His Chest X-ray (CXR) showed infective changes on the right lung, so he was treated with IV amoxicillin for pneumonia. Five doses of intravenous (IV) atropine 0.6 mg were given followed by an intravenous isoprenaline infusion for severe bradycardia. He was then transferred to another DGH with cardiology service for admission.
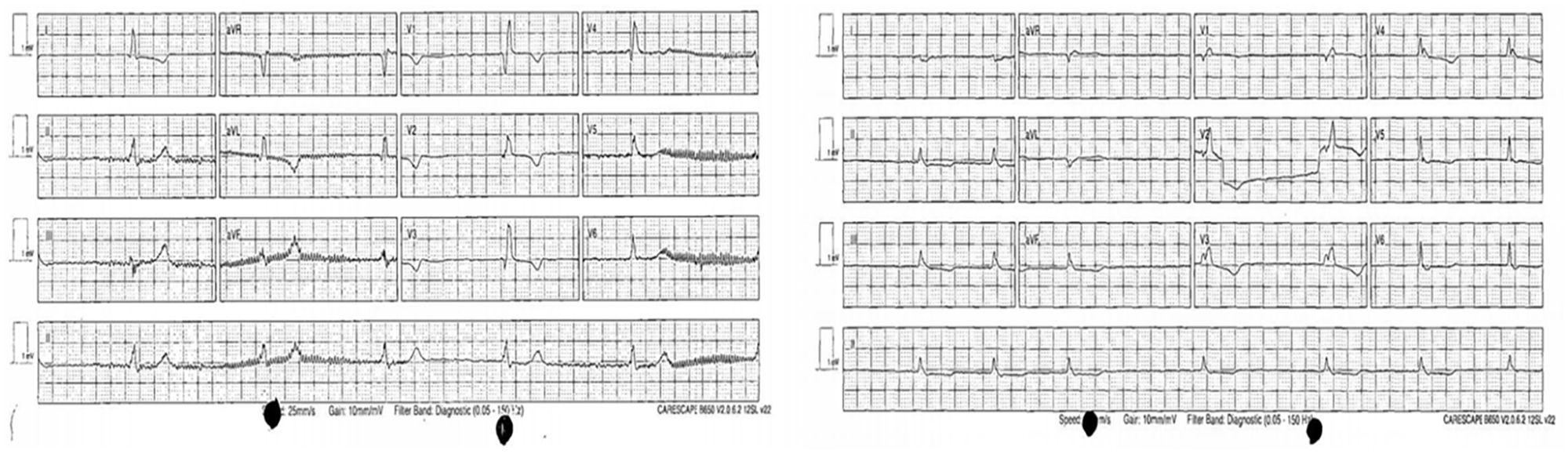
ECG of patient A (left) and patient B (right) was taken at the A&E resuscitation room.
Patient B
A 97-year-old man with a past medical history of congestive cardiac failure, atrial fibrillation, stroke, hypertension and chronic lymphocytic leukaemia presented to A&E with progressive breathlessness and bilateral lower limb swelling. He lived with his son who noticed a gradual deterioration in his general well-being for about 10 days. His vital signs at triage revealed a GCS of 15, a temperature of 34.3°C (rectal temperature of 27.6°C), a BP of 97/52 mmHg, an HR of 34/min and a SpO2 of 96% on 2L nasal cannula. There were signs of congestive cardiac failure with elevated jugular venous pressure (JVP), bilateral basal crepitations and generalised oedema of the extremities on examination. No goitre was palpated, and no surgical scar was seen on the neck. ECG showed slow atrial fibrillation (Figure 1). A septic screen with blood and urine cultures was taken. The decision was made to refer him to a dedicated cardiology unit considering a significant burden of cardiovascular disease and severe bradycardia.
Progress and outcomes
Patient A
An urgent thyroid function test (TFT) done at the cardiology triage revealed severe primary hypothyroidism with free T3 1.9 pmol/L (2.63–5.7 pmol/L), free T4 < 5.15 pmol/L (9.01–19.05 pmol/L) and TSH 41.3 mIU/L (0.35–4.94 mIU/L). His initial blood examination showed elevated white cell count and acute kidney injury (Table 1). His thyroid peroxidase (TPO) antibody was positive confirming autoimmune thyroiditis. The diagnosis of myxoedema coma was made based on the patient’s clinical presentation and a score of 85 on the diagnostic scoring system (Table 2). He was admitted to a high dependency unit (HDU) and started on a high dose p.o. levothyroxine 200 mcg together with IV hydrocortisone 100 mg immediately followed by oral levothyroxine (T4) 125 mcg daily and oral liothyronine (T3) 5 mcg four times daily. His GCS had improved to 15/15 on day 2 of admission. His free T3 improved to 5.60 pmol/L and TSH decreased to 24.8 mIU/L. However, his free T4 level remained undetectable. A transthoracic echocardiogram (TTE) revealed moderately impaired systolic function with an ejection fraction of 40% and biatrial dilatation without evidence of pericardial effusion. In the HDU, he deteriorated rapidly with the development of type 2 respiratory failure secondary to progressive pneumonia and congestive cardiac failure. Non-invasive ventilation was started, but he continued to deteriorate and died on day 3 of admission.
Initial investigations on arrival at A&E.
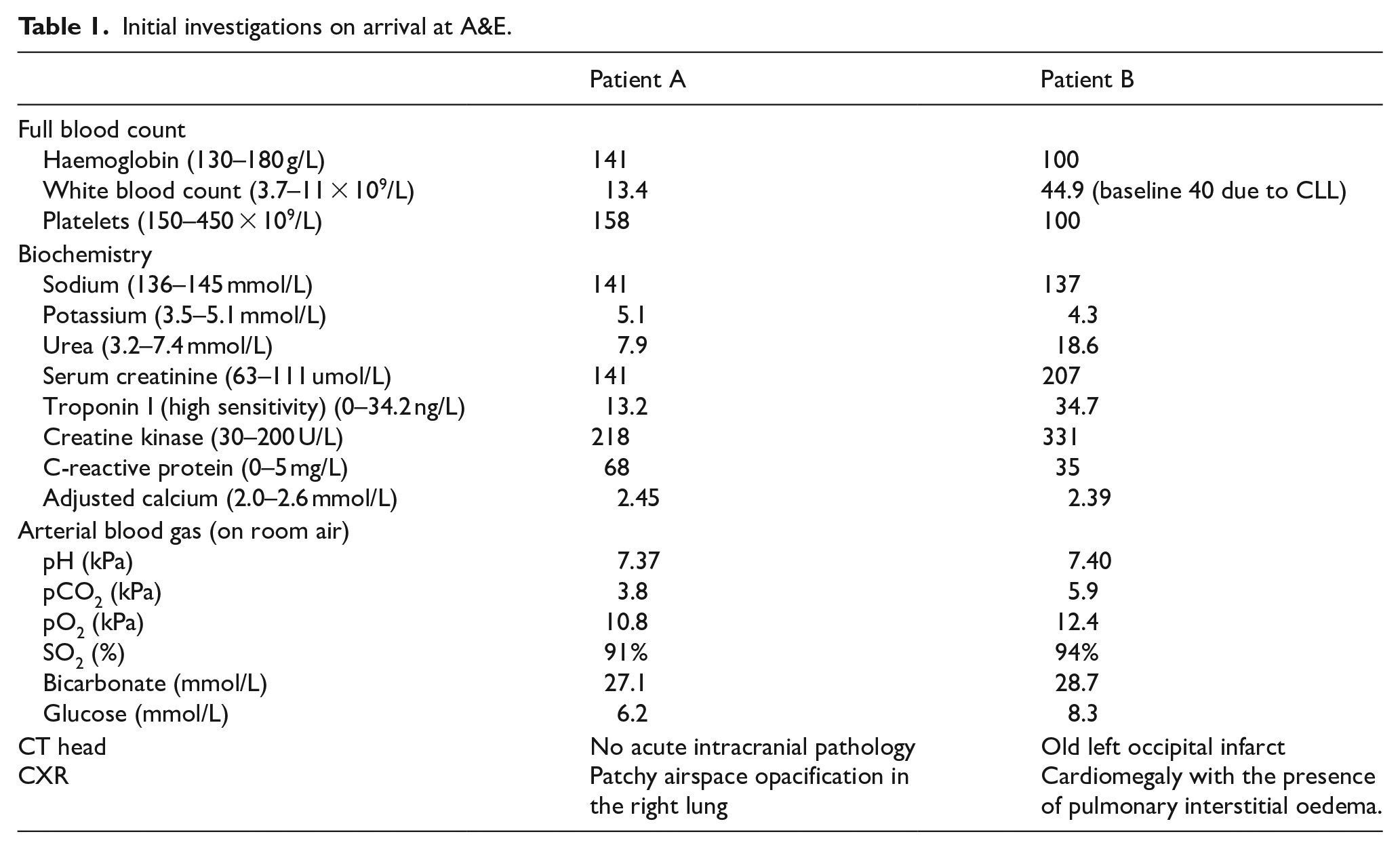
Diagnostic scoring for myxoedema coma.
Management of myxoedema coma. 4

Patient B
Hypothyroidism was later confirmed at the cardiology unit with undetectable thyroid hormones and markedly elevated TSH (free T4 <5.15 pmol/L and TSH 54.2 mIU/L). TPO antibody was negative. There was no prior history of neck surgery or radioactive iodine treatment. His myxoedema coma score was 95, highly suggestive of the diagnosis. He was seen by an endocrinologist and adopted a conservative dose of levothyroxine 250 mcg orally for loading due to his advanced age with high cardiovascular risk. A maintenance dose of p.o. levothyroxine 100 mcg daily with p.o. liothyronine 10 mcg three times daily were initiated. IV hydrocortisone 100 mg Q8 hourly was also given while continuing other supportive treatments including intravenous diuretics, passive rewarming and empirical broad-spectrum intravenous antibiotics. TTE showed a severely dilated atrium with tricuspid regurgitation and mild left ventricular systolic dysfunction. There was no evidence of pericardial effusion. His bradycardia and hypothermia resolved on day 3. Repeat TFT done 48 h later showed an improving thyroid hormone profile with free T3 2.30 pmol/L, free T4 7.5 pmol/L and TSH 15.5 mIU/L. Unfortunately, he acquired a nosocomial COVID-19 infection and progressively more lethargic, confused and hypoxic. He subsequently died despite treatment of COVID-19 infection with dexamethasone and IV remdesivir.
Discussion
Myxoedema coma is a rare but severe life-threatening form of decompensated hypothyroidism. The term ‘myxoedema coma’ is a misnomer. The textbook description of myxoedema (nonpitting oedema) or coma is absent in the overwhelming majority of patients. Instead, the cardinal manifestation is altered mental status. It is significantly more common in females, the elderly and during winter months. 1 It is a medical emergency with a reported mortality of up to 60%. 1 The predictors of poor outcomes include older age, cardiac complications, reduced GCS, sepsis, requirement for mechanical ventilation and persistent hypothermia. 2 Both of our patients had at least three (advanced age, severe bradycardia and hypothermia) poor prognosticators at their initial presentations.
Its prevalence has considerably declined in developed countries due to improved diagnosis and earlier treatment. This could perhaps lead to acute medicine physicians being less likely to consider it as a differential diagnosis due to its rarity and atypical presentations. Furthermore, myxoedema coma remained a clinical diagnosis. A high index of suspicion is therefore required for a prompt diagnosis and treatment. The myxoedema coma diagnostic scoring system (Table 2) might be helpful if the diagnosis is uncertain. 3 TSH is an appropriate initial test to screen for suspected cases as the majority are caused by primary hypothyroidism, Hashimoto’s thyroiditis, in iodine-sufficient areas. 1 None of our patients had a history of thyroid disease or radiation therapy to the neck. It is worth noting that myxoedema coma could be an initial presentation of undiagnosed hypothyroidism in elderly patients.
Our patients were admitted to the HDU/intensive treatment unit (ITU), with closer monitoring of clinical and laboratory parameters after loading doses of levothyroxine. 4 Although thyroid hormone replacement is the mainstay of treatment, it is still controversial on the dose, route of administration and regimen (combined T4 and T3 vs T4 therapy only). IV levothyroxine is usually preferred to avoid impaired gastrointestinal absorption secondary to ileus. 4 However, IV levothyroxine is not available in our institution. There is evidence to suggest that IV and oral levothyroxine are similar in terms of efficacy. 5 On the other hand, T3 is an active hormone with a quicker onset of action than T4. Hence, we decided to treat both patients with both oral T4 and T3 combination treatment while monitoring their BP, ECG, and symptoms of myocardial ischaemia. The risk of arrhythmias, myocardial infarction and mortality with larger loading doses in the elderly made us hesitant to use considerably large doses of T4 replacement recommended in the literature.
Random cortisol levels were taken, and our patients were started on intravenous hydrocortisone regularly to prevent adrenal crisis caused by increased metabolism of endogenous cortisol in the context of coexisting adrenal insufficiency. 4 Other supportive measures such as passive rewarming, correction of fluid and electrolyte imbalances and prevention of hypoglycaemia were instituted concurrently. 4 Core body temperatures were confirmed to be low in both patients on admission. Hence, we have used regular blankets to gradually rewarm. Active rewarming with heated blankets was avoided as it could lead to hypotension caused by peripheral vasodilatation. Serial capillary glucose profiles did not show any hypoglycaemia. Meticulous attention to fluid and electrolyte monitoring was also given to correct metabolic derangements early.
Decompensation into myxoedema crisis often occurs when homeostatic mechanisms are disrupted by a precipitating factor. It is therefore important to identify the trigger and treat it appropriately. Infections, acute coronary syndrome, stroke and sedative drugs are common triggers for myxoedema coma. 4 Appropriate investigations to search for precipitating causes should be requested based on clinical presentation. 4 Empiric therapy with broad-spectrum antibiotics can be considered until infection has been effectively ruled out.
In summary, we highlighted these two elderly patients to raise the awareness and importance of screening TFT in patients presenting with severe bradycardia to the acute unselected medical take. Neither of these patients had a prior history of hypothyroidism. Despite presenting with typical clinical signs of severe hypothyroidism, the diagnoses were unfortunately delayed with a subsequent diversion to the cardiology unit. These cases also illustrate that the mortality from myxoedema coma remains high despite appropriate treatments in a first-world nation.
Conclusion
Myxoedema coma is a rare but potentially fatal complication of hypothyroidism especially in elderly patients.
A high index of suspicion is essential for early diagnosis and appropriate treatment.
TSH screening should be performed in all patients presented with severe bradycardia.
An endocrinology consult should be sought if the diagnosis is uncertain.
The four key pillars of management include thyroid hormone replacement, glucocorticoids, supportive measures and treatment of precipitating causes.
Footnotes
Acknowledgements
We would like to thank our patients and their loved ones for allowing us to share for contribution towards medical literature.
Authors contributions
Aye Chan Maung wrote the manuscript; Sarbpreet Sihota, Henrietta Brain and Chitrabhanu Ballav edited the manuscript. All authors were involved in the management of patients. All authors have reviewed the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
