Abstract
Chylothorax is a lymphatic chylous pleural effusion typically associated with traumatic (iatrogenic, non-iatrogenic) and non-traumatic (infections, malignancy, lymphatic disorders) aetiologies. Drug-induced chylothorax is uncommon and mostly reported in association with BCR-ABL tyrosine kinase inhibitor therapy.
Keywords
Introduction
Chylothorax refers to an abnormal accumulation of lymphatic fluid in the pleural space, caused by disruption to the thoracic duct. It is usually associated with traumatic (iatrogenic, non-iatrogenic) and non-traumatic (infections, malignancy, lymphatic disorders) aetiologies. However, drug-induced chylothorax is uncommon, with several reported cases described in association with second-generation Bcr-Abl tyrosine kinase inhibitor (TKI) therapy. We report a case of dasatinib-induced chylothorax in a high-risk chronic myeloid leukaemia (CML) patient, and provide a comprehensive review of case literature to discuss the current clinical understanding and management strategies for this condition.
Case
A 63-year-old Chinese male, who has a history of chronic phase-CML with high-risk Sokal score treated with dasatinib therapy, presented to our practice with a 1-week duration of exertional dyspnoea and productive cough with occasional blood-stained sputum. On examination, the patient was afebrile and hemodynamically stable. Chest examination revealed reduced breath sounds and stony dullness over the left lower zone. There was no cervical lymphadenopathy.
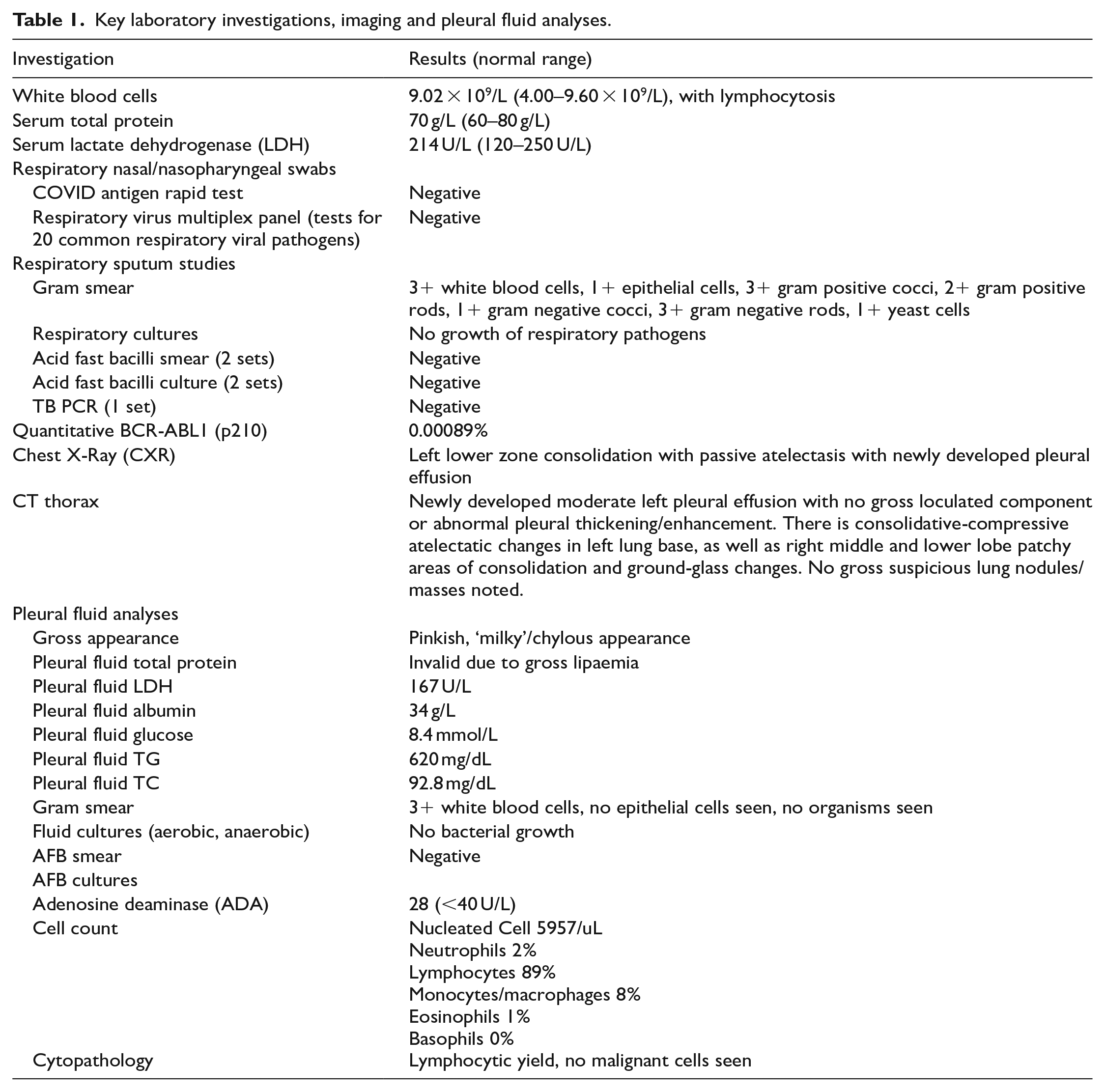
Initial blood investigations were largely normal except for peripheral lymphocytosis. Respiratory viral swabs, sputum cultures and acid-fast bacilli tests were negative. Chest radiograph showed left lower zone moderate-sized pleural effusion with surrounding consolidative changes (Figure 1). Subsequent computed tomography (CT) scan of the thorax revealed a moderate left-sided pleural effusion, bibasal consolidative changes, with no hilar/mediastinal lymphadenopathy. Diagnostic thoracocentesis demonstrated a ‘milky’ pinkish pleural aspirate (Figure 2), with fluid analysis suggestive of a lymphocytic exudative effusion with elevated pleural fluid triglyceride (TG) levels at 620 mg/dL (Table 1). In the absence of other secondary aetiologies, the patient was diagnosed with dasatinib-induced chylothorax. He was switched over from dasatinib to imatinib for CML treatment, and was given empirical Augmentin for 7 days. At presentation, the patient had been on dasatinib therapy for 45 months, and previously attained cytogenetic and deep molecular remission of CML. Upon cessation of dasatinib therapy, his symptoms resolved within a week, and repeat CXR at 4 months showed that the left-sided pleural effusion has largely resolved (Figure 1). After 6 months of imatinib therapy, patient tolerated treatment well and remained in deep molecular remission of CML.
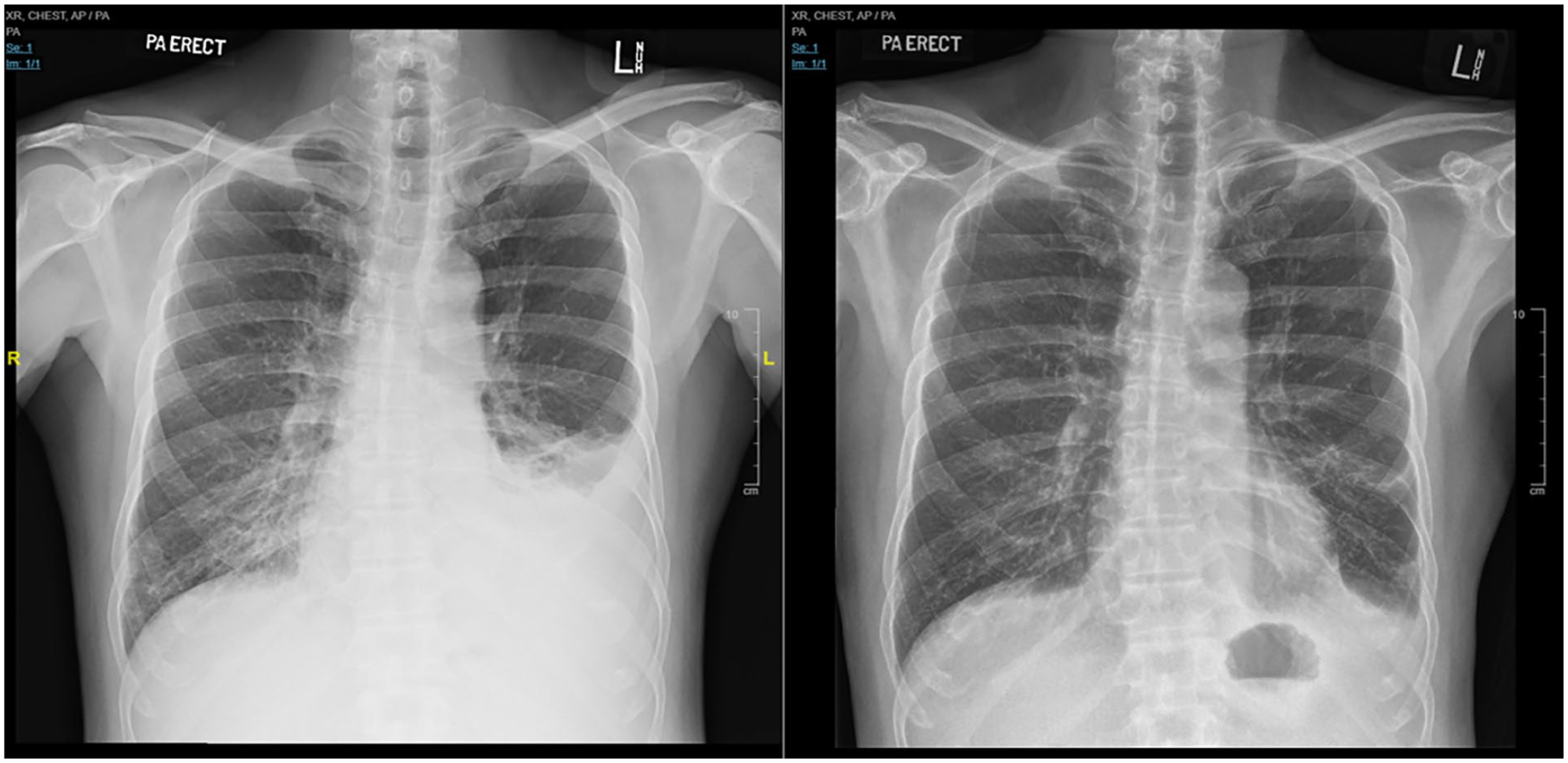
Initial chest radiograph and repeat chest radiograph 4 months after cessation of dasatinib therapy.
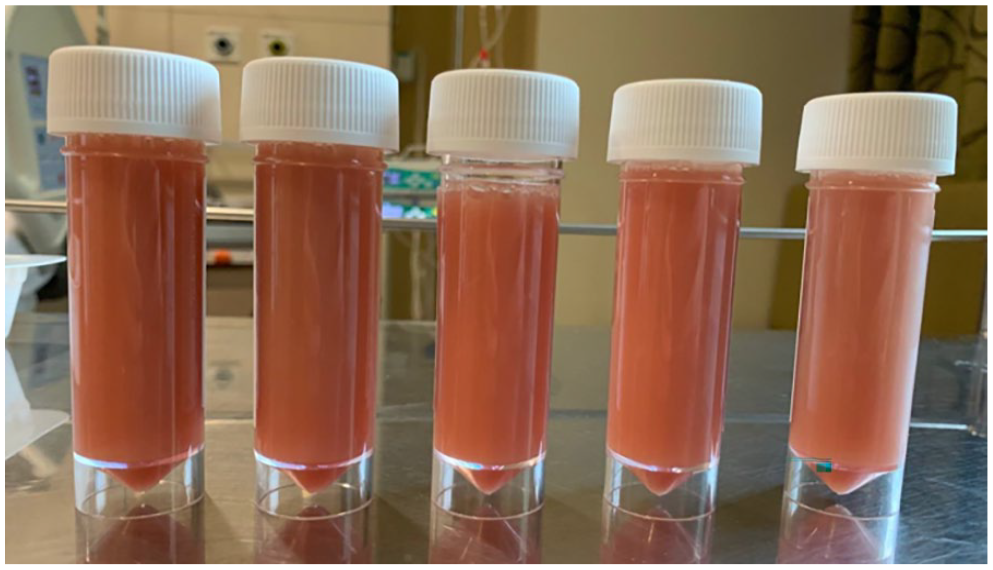
Clinical photo of pleural fluid aspirate.
Key laboratory investigations, imaging and pleural fluid analyses.
Discussion
We describe a case of dasatinib-induced chylothorax and performed a review of reported cases in literature (Supplemental Table).
Chylothorax refers to chylous/lymphatic pleural effusion caused by disruption of lymphatic flow in the thoracic duct. 1 Diagnosis is established through thoracocentesis which typically reveals a ‘milky’ pleural aspirate, with elevated TG levels > 110 mg/dL or the presence of chylomicrons.2,3 Pseudochylothorax is a differential diagnosis for ‘milky’ pleural effusion, with cholesterol levels >250 mg/dL and TG < 110 mg/dL 3 . Causes of chylothorax can be classified into traumatic (iatrogenic (e.g. thoracic/head-and-neck surgery, mediastinal radiotherapy), non-iatrogenic (e.g. chest wall trauma, forceful coughing/emesis)) and non-traumatic (malignancies (e.g. lymphoproliferative disorders, lung cancer), infections (e.g. tuberculous lymphadenitis, parasitic infections), congenital/acquired lymphatic disorders (e.g. lymphangiectasis, lymphangiomatosis), and translocation of chylous ascites (e.g. heart failure, liver cirrhosis, nephrotic syndrome)) aetiologies. However, drug-induced chylothorax is uncommon and mainly described in association with second-generation Bcr-Abl TKI use.4,5
Dasatinib is a second-generation Bcr-Abl TKI currently approved for use in intermediate- to high-risk CP-CML and Philadelphia chromosome-positive (Ph+) acute lymphoblastic leukaemia (ALL). Its use in CP-CML was propelled by the landmark DASISION trial in 2010 which reported superior complete cytogenetic and major molecular response in CP-CML patients treated with dasatinib as compared to imatinib. 6 Despite its promising clinical efficacy, dasatinib use is associated with pulmonary toxicity, in particular, pleural effusion (19–29%) and pulmonary hypertension (<1–5%). 7
From our literature review (Supplemental Table), 63% of reported cases were males and 37% females, with a broad age range of 5–84 years. The main indication for dasatinib treatment was CML (92.9%), with the remaining cases treated for Ph-positive ALL. The average time to onset of chylothorax post-dasatinib initiation was 39 months (range: 2–168 months). In terms of site of chylothorax, 29.6% were right-sided, 18.5% left-sided and 51.9% bilateral. Interestingly, from the cases with available biochemical and cell count data, all the patients had lymphocytic-predominant exudative effusions. Reported pleural fluid TG levels range from 155 mg/dL to 4300 mg/dL.
It is known that chylothoraces do not always present with chylous-appearing pleural aspirate, with a previous study reporting only 44% of cases as having the classic ‘milky’ appearance. 1 This highlights the importance of having a high clinical index of suspicion for chylothorax based on known aetiologies to avoid misdiagnoses, given that pleural fluid TG levels are not routinely sent in diagnostic pleural studies. In addition, while general cases of chylothorax are frequently exudative (68–86%) and lymphocytic predominant (48–90%),1,8 all reported cases of dasatinib-induced chylothorax were uniformly lymphocytic exudates. There are two possible mechanisms that may explain this finding. Firstly, physiological chyle is a protein-rich fluid which contains immune cells like T-lymphocytes. 9 Secondly, approximately one-third of dasatinib-treated CP-CML patients have peripheral lymphocytosis, which was found to be associated with development of pleural effusion, 10 and may translate to pleural fluid lymphocytosis, as a previous study found that pleural fluid lymphocytes from dasatinib-related pleural effusions had similar phenotype and genotype to peripheral blood lymphocytes. 11
To date, the mechanism of dasatinib-induced chylothorax remains unclear, although it is postulated to be related to the inhibitory effect of dasatinib on PDGFR-β signalling, which regulates lymphangiogenesis. 4 Interestingly, dasatinib is found to be 67× more potent than the first-generation TKI imatinib in inhibiting PDGFR-β activity, 12 which may explain the absence of reports of imatinib-related chylothorax.
Management options for dasatinib-induced chylothorax have been highly heterogeneous. In general, dasatinib cessation/dose reduction is almost always warranted, with consideration of generic strategies to control chylous effusion, which includes dietary modification (e.g. medium chain fatty acid/low-fat diet, total parenteral nutrition), medications (e.g. steroids, octreotide, diuretics) and/or procedures (e.g. repeated thoracocenteses, thoracic duct ligation). The main treatment principles in chylothorax management involve reducing gastrointestinal fatty acid absorption, chyle formation, thoracic duct lymphatic flow and aborting the chylous leak.
Conclusion
In summary, dasatinib-induced chylothorax is an important clinical entity that clinicians should be privy to in view of dasatinib being increasingly used as first-line treatment in CML and Ph-positive ALL. Moreover, chylothorax is a potentially serious clinical condition that, if left untreated, can cause malnutrition and immunodeficiency. 9 Future research with multi-centre data would be helpful to identify clinical/biochemical markers that may predict dasatinib-related pulmonary complications such as chylothorax to identify patients in whom dasatinib should be avoided, as well as evaluate the efficacy of various treatment modalities used in dasatinib-induced chylothorax in order to develop evidence-based and targeted therapies for this condition.
Supplemental Material
sj-docx-1-rcp-10.1177_14782715241237577 – Supplemental material for Drug-induced chylothorax as a rare pleural complication in dasatinib therapy for chronic myeloid leukaemia
Supplemental material, sj-docx-1-rcp-10.1177_14782715241237577 for Drug-induced chylothorax as a rare pleural complication in dasatinib therapy for chronic myeloid leukaemia by Isaac KS Ng, Mamta Ruparel, Esther HL Chan and Kay Leong Khoo in Journal of the Royal College of Physicians of Edinburgh
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent
written consent obtained from patient.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
