Abstract
Drug-induced aseptic meningitis (DIAM) or chemical meningitis following spinal anaesthesia has rarely been reported. DIAM is caused by meningeal inflammation due to intrathecally administered drugs or secondary to systemic immunological hypersensitivity. We hereby present a case of a young adult with aseptic meningitis following neuraxial anaesthesia possibly provoked by bupivacaine. The initial cerebrospinal fluid (CSF) picture revealed neutrophilic pleocytosis and normal glycorrhachia. CSF culture was negative. The patient was put on invasive mechanical ventilation and started on intravenous antibiotics. There was a rapid improvement in clinical condition without any residual neurological deficit within the next few days. Aseptic meningitis following neuraxial anaesthesia can be prevented by strict aseptic protocols and careful inspection of visible impurities while administering the intrathecal drug. Detailed history taking, clinical examination, and focused investigations can distinguish between bacterial and chemical meningitis. Appropriate diagnosis of this entity may guide the treatment regimen, reducing hospital stay and cost.
Introduction
Aseptic Meningitis is a broad term used to indicate meningeal inflammation (cerebrospinal fluid (CSF) pleocytosis of ⩾5 cells/mm³) in absence of any infectious process.1,2 There are mainly three types of aseptic meningitis: immune-mediated disorders with meningeal involvement, drug-induced aseptic meningitis (DIAM) and neoplastic meningitis. Meningitis following neuraxial anaesthesia is very rare (incidence of <0.01%), and the incidence of aseptic meningitis is even rarer. 3 Delay in recognition of this entity and delayed initiation of treatment may lead to fatal consequences. Herein, we present a case of a young adult with suspected aseptic meningitis following spinal anaesthesia. He was diagnosed at the earliest, treated successfully in critical care unit and discharged in stable condition 10 days after admission.
Case
An otherwise healthy 22-years-old male patient presented to the critical care unit in a drowsy state with active seizures. He had undergone spinal anaesthesia for an elective femoral titanium nail fixation for closed fracture of neck of left femur eight hours ago. The patient fell from a tree while climbing up the same, resulting in fracture of neck of left femur three days earlier. He was taking paracetamol and tramadol tablets for pain relief over this period. Computed tomography (CT) scan of brain was not done at that time as there was no history or clinical features suggestive of head injury.
The patient’s symptoms started four hours after the operative procedure was completed. There was gradual deterioration of consciousness along with vomiting, headache and eventually stupor. He was given analgesics and anti-emetics in post-operative observation ward. Spinal anaesthesia was administered at 10 AM, completed at 11:20 AM; first onset of symptoms was at 2 PM and the patient was reviewed by critical care team at 6 PM. There was no history of fever or vaccination in the recent past. On presentation to the critical care unit, physical and neurological examination revealed bilateral extensor plantar responses, constricted and slowly reacting pupils. The Glasgow Coma Scale (GCS) score was 6/15 (Eye opening response–2; Verbal response–1; Motor response–3). Neck stiffness was absent. On discussion with the concerned anaesthesiology team, it was found that the intrathecal spinal anaesthesia was administered using strict aseptic protocol by an experienced anaesthesiologist; using 2.8 ml bupivacaine (0.5% heavy) along with 25 μg fentanyl in single shot at L3–L4 inter-space in sitting position. The skin of the patient’s back was appropriately cleaned with antiseptic solution containing isopropyl alcohol (70%) and chlorhexidine (0.5% Chlorhexidine gluconate in alcohol). Injection Cefazolin 1 gm, a cephalosporin with comparatively low blood-brain barrier penetration, was administered prior to surgery, as part of the standard protocol.
Intraoperatively there was one very brief period of hypotension (86/56 mm Hg non-invasive blood pressure on monitor for about 1 min) which was managed with intravenous bolus dose of 50 µg phenylephrine. The patient was shifted from post-anaesthesia care unit after acceptable reversal of motor blockade.
Laboratory investigations revealed neutrophilic leukocytosis (total leukocyte count of 14,300/mm³ with 82% neutrophils), raised C-reactive protein (54 mg/L; reference range 0–10) and elevated lactate (7.01 mmol/L; reference range 0.5–1). Non-contrast CT scan of brain revealed generalised sulcal effacement suggestive of diffuse cerebral oedema along with posterior horn bleed (Figure 1). Magnetic resonance imaging (MRI) brain done 12 h after admission was corroborative with the CT images (Figure 2); MR venogram and angiography did not reveal any abnormality. There was contrast enhancement on MRI brain. Electroencephalogram done on postoperative day one was compatible with diffuse encephalopathic changes without any ictal activity. CSF study revealed increased cell count (1,100/mm³; reference range 0–5) with neutrophilic predominance (85%), increased protein (92 mg/dl; reference range 15–60) and normal glucose (73 mg/dl; corresponding blood glucose was 104 mg/dl). Gram-staining and Ziehl-Neelsen staining were negative. Polymerase chain reaction tests for Herpes simplex virus 1 and 2, cytomegalovirus, human herpesvirus 6 and enterovirus; serological tests for Epstein-Barr virus, varicella-zoster virus and Japanese encephalitis virus were negative.Vasculitis and autoimmune encephalitis panel were negative. CSF from the first lumbar puncture (prior to administration of any antibiotic) revealed no growth on culture. Blood culture and urine culture showed no growth. Rest of the initial investigations including chest radiography, CT scan of thorax and abdomen, and electrocardiogram were within normal limits. Reverse-transcriptase polymerase chain reaction for SARS-CoV 2 from the nasopharyngeal swab done both in pre-operative and post-operative period was negative.
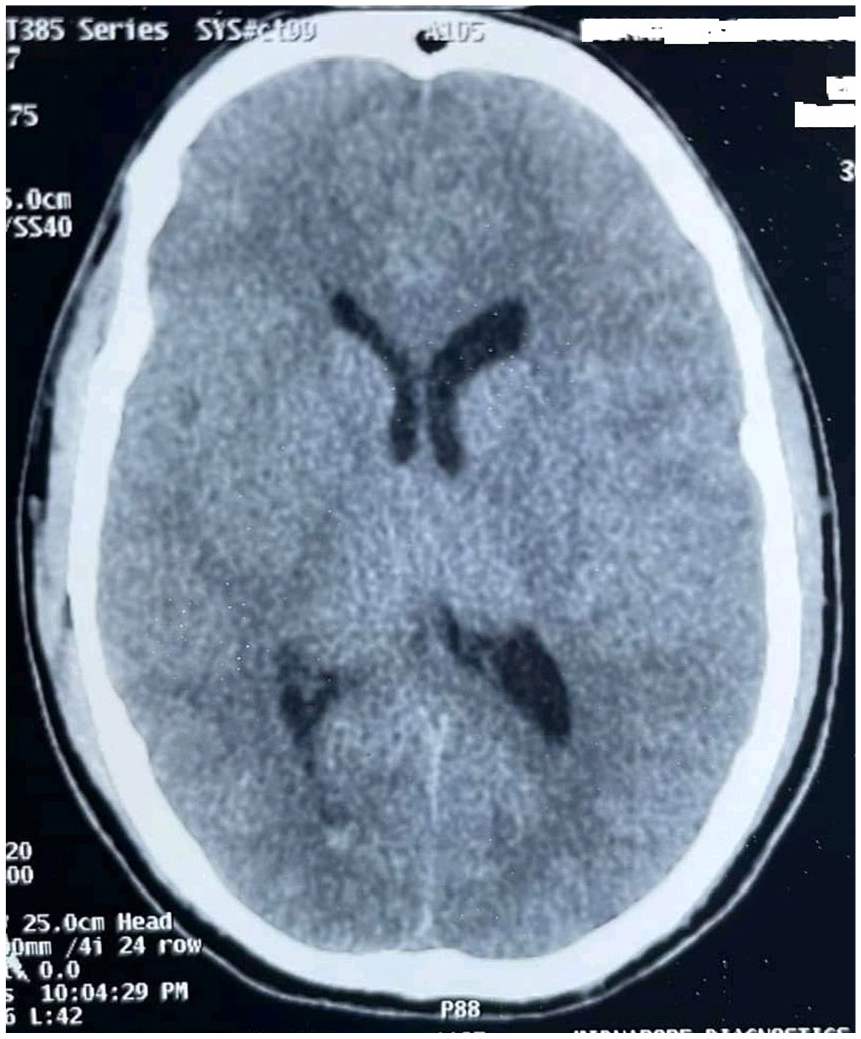
Non-contrast CT scan of brain showing generalised sulcal effacement suggestive of diffuse cerebral oedema, and posterior horn bleed.
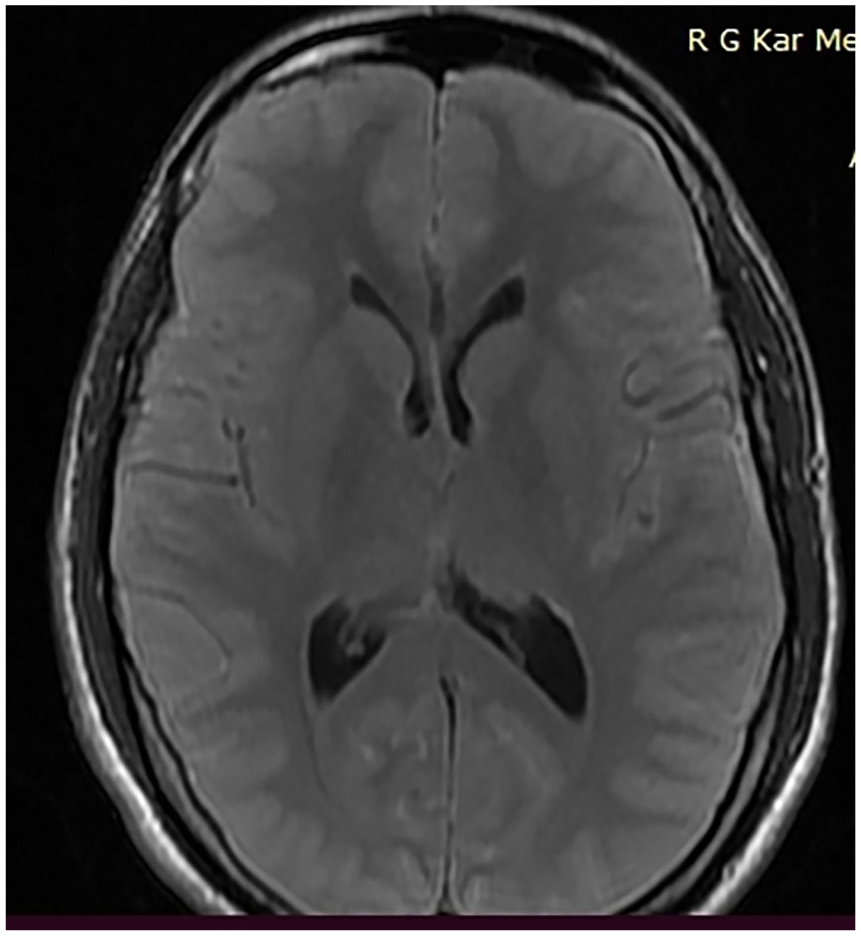
MRI of brain (T2 flair axial imaging) showing generalised sulcal effacement suggestive of diffuse cerebral oedema.
He was suspected to have developed aseptic meningitis following neuraxial anaesthesia possibly provoked by bupivacaine. As the initial CSF picture could not completely exclude the possibility of acute bacterial meningitis, he was started on empirical antibiotics. However, the abrupt onset of symptoms following the procedure, absence of fever or neck rigidity, normal glycorrhachia, sterile cultures and a very prompt recovery without any neurological sequelae in our patient favoured the diagnosis of DIAM. No fat globule was detected in urine and mini-bronchoalveolar lavage. MRI and clinical findings ruled out Fat Embolism Syndrome according to Takahashi grading and Gurd’s criteria respectively. 4 Perioperative episode of hypotension was only for a brief period of around 1 minute, which was well managed. Such a brief episode of hypotension was unlikely to cause hypoxic brain injury resulting in diffuse cerebral oedema.
The patient was haemodynamically stable during admission. Due to low GCS score, he was immediately intubated and put on invasive mechanical ventilation. Empirical meningitis coverage regimen (Ceftriaxone 2 gm intravenously twice a day; Vancomycin 1 gm intravenously twice a day) was started along with anti-seizure medications (injection levetiracitum 500 mg thrice daily through intravenous route). Raised intracranial pressure (ICP) goals were maintained with Infusion mannitol (100 ml, 20%w/v thrice daily), 30–45° head end elevation, maintenance of normocapnia along with temperature, seizures and glycemic control. Routine ICU protocols of feeding, analgesia, sedation, thromboprophylaxis and stress ulcer prophylaxis were maintained while the patient was on ventilation. Propofol infusion (30 mg/h) was administered as part of ICU sedation protocol following Richmond Agitation Sedation Scale. 5 On Day 3 of admission, patient’s level of consciousness improved and total leukocyte count started to decrease (9,900/mm³). On Day 5, patient was conscious and following commands. He was maintaining oxygenation on spontaneous pressure support ventilation. He was extubated successfully after passing spontaneous breathing trials. Repeat CSF study sent 5 days after admission was within normal limits (cell count of 2–3/mm3, all lymphocytes). Antibiotics were stopped and he was transferred back to surgical follow-up ward. The patient was discharged in a clinically stable condition on day 10.
Discussion
Meningitis following spinal anaesthesia may be of two types: bacterial and aseptic. The latter is mostly attributed to drugs and is known as DIAM. Although rare, meningitis following spinal anaesthesia may have potentially serious complications. DIAM is mainly caused by either direct chemical irritation of meninges from intrathecal administration of drugs, or immunological hypersensitivity to the systemically administered drugs (such as non-steroidal anti-inflammatory drugs and antibiotics).3,6 The alleged chemical compound can breach the arachnoid membrane and enter into subarachnoid space and stimulate meningeal inflammation. 7 Aseptic meningitis provoked by anaesthetic agents used in spinal anaesthesia (such as bupivacaine and ropivacaine) has been reported in literature.3,8 Apart from them, corn starch powder in powdered surgical gloves, povidone iodine or chlorhexidine disinfectant solution used during pre-procedure skin antisepsis, and additives used during preparation of the intrathecal drug or contaminants can cause this complication rarely. Chlorhexidine 0.5% in alcohol was used in our patient for skin antisepsis taking all other precautions to prevent chlorhexidine from reaching the CSF as recommended by the Association of Anaesthetists of Great Britain and Ireland (AAGBI). 9 According to AAGBI guidelines, skin antisepsis for central neuraxial blockade using 0.5% chlorhexidine in alcohol provides the safest compromise between the risk of infection and the risk of neurotoxicity. Hence, according to published literature, with the correct recommended procedure undertaken of skin antisepsis, it is very unlikely that 0.5% Chlorhexidine could be responsible for meningitis. 10 One point cut is widely practised to prevent inadvertent mixing of outside particles with the intrathecal drug. 11 It was done in our patient too.
Acute bacterial meningitis is the closest differential of DIAM or chemical meningitis. 12 Most of the patients with DIAM, irrespective of the offending drug, present with fever, headache, meningismus and alteration of mental status. Lethargy, diplopia, periorbital oedema, seizures and confusion are some atypical symptoms which may be associated with aseptic meningitis. The incidence of cerebral oedema and elevated ICP is more common in bacterial meningitis than DIAM. But the clinical presentation does not help to differentiate DIAM from infectious meningitis. 13 Rapid onset of symptoms following the procedure, absence of fever, normal CSF glucose, and sterile cultures favour the diagnosis of chemical meningitis. Those differentiating features are not always confirmatory, thereby making this entity a diagnosis of exclusion. Most of the patients recover on their own. Nevertheless, prompt identification and truculent management can alleviate the symptoms, prevent unnecessary treatment (including antimicrobials which can lead to resistance) and curtail hospital stay. Residual neurological sequelae can also be prevented by this approach. After discussion with the concerned authorities, the particular batch of drug, needle, gloves and disinfectants were discarded from institutional usage.
Conclusion
We suspect that this case might be aseptic meningitis caused by spinal anaesthetic bupivacaine, which is the most widely used intrathecal local anaesthetic. Some impurities or potentially dangerous ingredients present accidentally in the ampoule might also be the cause. Although very rare, bupivacaine-induced aseptic meningitis has been reported in literature. While the product inserts of bupivacaine ampoules manufactured by all companies in India mention about central nervous system adverse effects ranging from anxiety to seizures, there is no mention of DIAM as a possible complication. As the diagnosis of bupivacaine-induced aseptic meningitis is one of exclusion, any early post-operative patient with signs of meningism after a spinal anaesthesia should have an immediate CSF examination and initiation of medical treatment as the aseptic meningitis may mimic bacterial meningitis at presentation. The relevant authorities should be officially notified and the entire process starting from bupivacaine manufacturing and packaging to the final administration of spinal anaesthesia should be reviewed at local and administrative levels to ensure total purity of the drug and immaculately sterile and correct procedure of administration of spinal anaesthesia by anaesthesia personnel.
Footnotes
Contributorship
AnC and AtC prepared the manuscript with adequate planning and execution. AC and SD contributed to patient management, review of literature, critical revision of content and final approval of manuscript. All authors are in agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient’s consent
An informed written consent was obtained from the patient after full explanation regarding his images being published for academic interest. The patient did not have any objection regarding use of his images and gave due permission to use them.
