Abstract
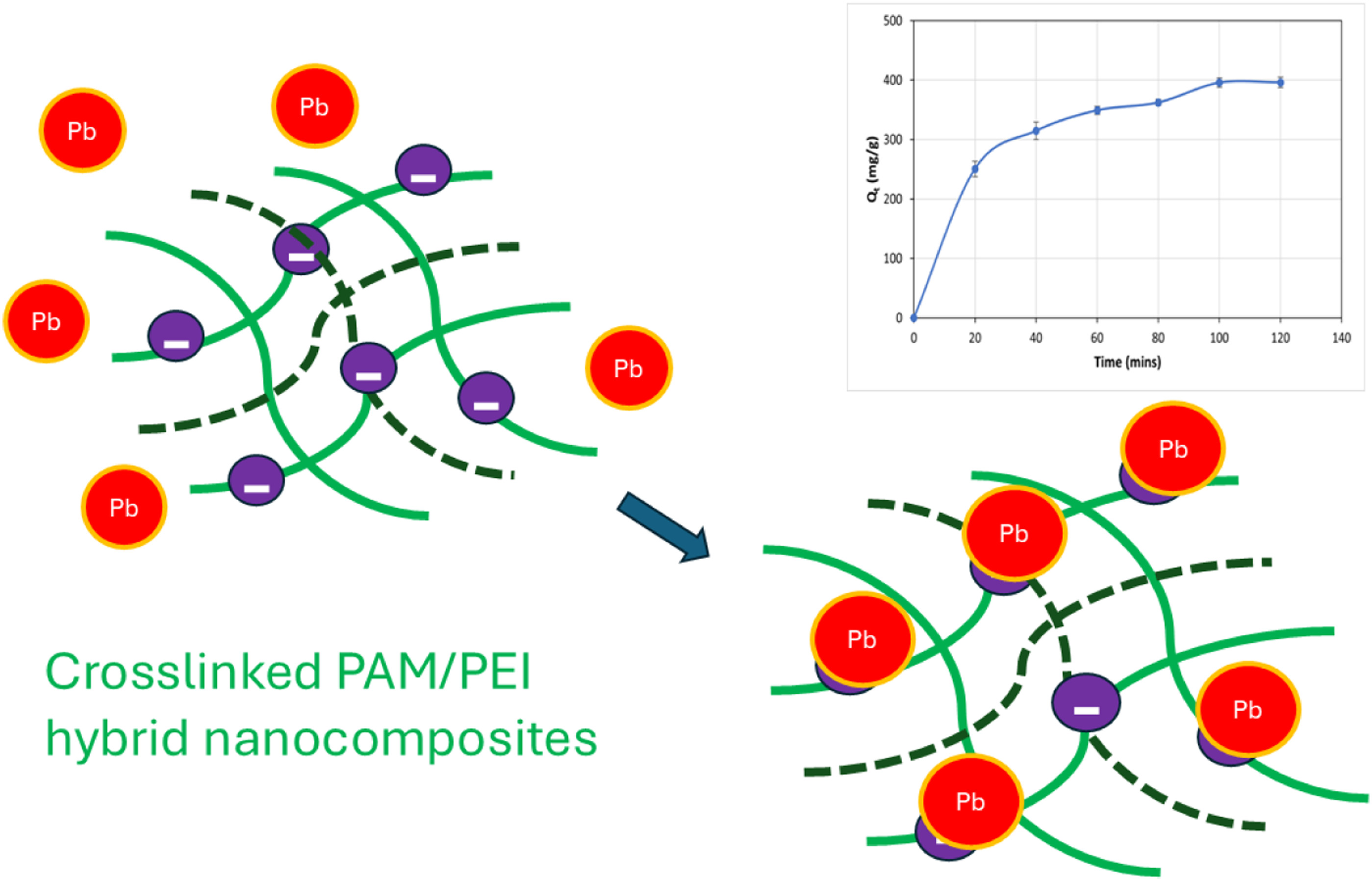
The development of a heavy metal adsorbent was successfully achieved through a crosslinked polymer nanocomposite system consisting of 70 wt% polyacrylamide (PAM), 30 wt% polyethylenimine (PEI), 3 phr nano-hydroxyapatite (nHA), and 15 phr cellulose. The nanocomposite was crosslinked using 10 phr N,N-methylenebisacrylamide (MBA) and prepared via solution casting. This study investigated the effects of the initial Pb2+ concentration and contact time on the adsorption capacity. This specific combination of dual fillers (cellulose + nHA) and crosslinking provided a synergistic enhancement in structural stability, active site accessibility, and Pb2+ adsorption efficiency, achieving a maximum adsorption capacity of 404.5 mg/g (at pH 5 with an initial Pb2+ concentration of 100 ppm and a contact time of 2 h using 10 mg of the adsorbent). The findings revealed that the PAM/PEI/Cel/nHA/MBA-10 nanocomposite followed the Freundlich isotherm and exhibited pseudo-second-order kinetics, indicating that the adsorption process is primarily governed by multilayer chemisorption. The findings demonstrate a distinct improvement over previously reported PAM/PEI or single-filler systems, highlighting the novelty and potential of PAM/PEI/Cel/nHA/MBA-10 for heavy metal (e.g., Pb2+) removal applications.
Get full access to this article
View all access options for this article.
