Abstract

Further discussion in response to MS Rea, The law of reciprocity holds (more or less) for circadian-effective lighting
The typical human indoor light environment strongly deviates from the natural light–dark cycle outdoors, both in terms of spectrum and amount of light exposure. The ubiquitous availability of electric light enables us to spend large parts of our day indoors, in conditions with limited, or sometimes even without, any natural daylight. Across daytime, we therefore expose ourselves to light conditions that are relatively dim, with daytime illuminances that frequently do not exceed civil twilight on a semi-overcast day, 1 while during the evening and at nighttime the abundant use of electric light deprives us of natural darkness. Consequently, in our 24/7 society, we are exposed to dimmer days, brighter nights and lower day–night contrasts as compared to the natural light–dark cycle outdoors.2–4 This has negative consequences for our mental and physical health, sleep and performance.5–7
The 24-hour light–dark cycle and its light exposure regulate our circadian rhythms and affect our mood, daytime functioning and nighttime sleep. These effects are strongly mediated by a (melanopsin based) photoreceptor that, in humans, is maximally sensitive to the short-wavelength portion of the visible spectrum around 480 nm. This melanopsin-based photoreceptor (often denoted as ipRGC, a shorthand for intrinsically photosensitive Retinal Ganglion Cell) is known to combine its own melanopsin-mediated (i.e. melanopic) response to light with (extrinsic) signals from rod and cone photoreceptors. 8 In 2018, an internationally balloted consensus metrology has been standardized 9 to provide a systematic SI-compliant framework to assess and characterize light levels based on the degree to which they activate each of the five different (α-opic) photoreceptor types (i.e. three kinds of cones, rods and ipRGCs) in the human retina. The metrology comprises five α-opic irradiances and five α-opic equivalent daylight illuminances (α-opic EDIs) that have a direct linear relationship with the luminous and/or radiant flux of a light source. The metrology allows to systematically investigate the extent to which a particular circadian, neuroendocrine or neurobehavioral response to light is driven by a single photoreceptor or by a combination of photoreceptors, and whether this depends, for instance, on the amount, duration or timing of the light exposure.10,11
Recently, an international expert workshop on circadian and neurophysiological photometry published a set of light recommendations to best support human physiology, sleep and wakefulness within indoor settings. 12 The workshop concluded that under most practically relevant situations, the spectral sensitivity of non-visual responses to light can be well described by the intrinsic, melanopsin-based, spectral sensitivity of ipRGCs. Consequently, the workshop recommendations were expressed in terms of the melanopic EDI, measured at the eye position of the user (with a detector orientation that corresponds to the dominant direction of gaze):
Throughout the daytime, the recommended minimum melanopic EDI is 250 lx.
During the evening, starting at least 3 hours before bedtime, the recommended maximum melanopic EDI is 10 lx.
The sleep environment should be as dark as possible with a recommended maximum melanopic EDI of 1 lx and 10 lx in case unavoidable activities during the nighttime require vision.
The recommendations provide highly needed additional considerations and guidance to successfully accomplish integrative lighting solutions. They are intended for healthy adults (18–55 years) with a day-active schedule, without the intention to supersede existing guidelines and regulations relating to for instance, visual function, comfort and energy consumption.
In this issue of LRT, Rea introduces and adopts an extension of the circadian stimulus (CS) model13,14 to specify recommendations for circadian-effective lighting. Both the CS model and its extension express the amount of circadian-effective light within a light stimulus in terms of a non-SI compliant CLA2.0 parameter, defined by means of the spectral irradiance and a non-linear expression that adopts several spectral sensitivity functions. The expression is derived from an early computational model for spectrally opponent circadian phototransduction 15 and has a peak sensitivity at 460 nm for short-wavelength-dominated light exposures (b – y > 0), and at 485 nm for long-wavelength-dominated light exposures (b – y ⩽ 0). A fixed dose–response relationship is used to convert the CLA2.0 parameter into an instantaneous ‘effective magnitude of neural signals for the circadian system’, that is, the CS. The CS value expresses the level of melatonin suppression that is expected to result from a nighttime light stimulus (with a particular CLA2.0 value). A CS value of 0.1 or 0.3 corresponds to 10% or 30% melatonin suppression, respectively.
The CS model extension, CS t , adds the exposure duration t to the CS model, using the law of reciprocity as a first approximation: for exposure durations between 30 minutes and up to 3 hours a lower level of circadian-effective light can be compensated by a longer exposure duration as to result in the same CS. Rea combines this CS t model with the UL24480 Design Guideline recommendation for daytime light exposure (i.e. at least 2 hours with CS = 0.30) to yield a minimum dose of circadian-effective light CS d of 0.43 for day-active and night-inactive building occupants. By 8 PM, the UL24480 Design Guideline recommends to use light that produces less than 10% melatonin suppression (i.e. CS ⩽ 0.10).
There are several important concerns that need reflection when using CS-based models to describe light conditions and lighting designs:
(1) The CLA2.0 parameter and other CS-related measures are not SI compliant, while for international guidelines and traceable measurements in light and lighting a metrology that complies with the International System of Units (SI) 16 is essential. 17
(2) The CS models are based on two spectral sensitivity functions that combine input from the melanopsin-based photoreceptor with rod and cone inputs. There is no scientific evidence that such a complex interplay of photoreceptors is needed to describe nocturnal melatonin suppression. An extensive body of research from a large number of independent research groups suggests that for most practically relevant circumstances, the spectral sensitivity of melatonin suppression and phase shifting responses to light in humans can be well approximated by the spectral sensitivity of the melanopsin-based photoreceptor.8,11,12,18–23
(3) The fixed CLA2.0–CS (dose–response) relationship as used in the CS model is primarily derived from two melatonin suppression studies for narrowband nocturnal light exposures in people with pharmacologically dilated pupils. The model has been proven to effectively describe data from a somewhat wider selection (see Rea in this LRT issue) of melatonin suppression studies with and without pupil dilator. The CS model does not discriminate between cases with or without pharmacological pupil dilation. In contrast, melanopic EDI-based recommendations and predictive models typically differentiate between cases with and without pharmacological pupil dilation, as pharmacological pupil dilation is known to reduce the thresholds for circadian responses to light.11,12,24
(4) The CS models have their maximum value set to 0.7 (i.e. 70% melatonin suppression), which means that they are unable to account for more than 70% melatonin suppression. A recent laboratory study 25 has found near to full (>99%) suppression of melatonin on 61 nights while exposing 55 individuals to a wide range of evening light conditions (10–2000 lx, 4100 K, for 5 hours, starting 2 hours before dim light melatonin onset). In about 80% of the tested individuals, an illuminance of 100 lx already produced a melatonin suppression of at least 75%.
(5) The CS model predictions have been verified and tested using data from a limited number of studies (see Rea in this LRT issue), while today’s literature provides a much larger collection of studies with data on melatonin suppression and other circadian responses to light.11,20,21,25 The melanopic EDI-based recommendations and predictions11,12 have been derived and validated using a more extensive data set featuring a larger diversity and number of studies as compared to their CS-based counterparts.
In view of the above concerns, it is important to make a careful and balanced comparison between CS-based and melanopic EDI-based healthy light recommendations and predictions of nighttime light-induced melatonin suppression. A recently published generalized model used a machine learning approach and data from 29 peer-reviewed publications to predict nocturnal melatonin suppression by means of three light exposure characteristics: melanopic EDI, exposure duration and the use of a pharmacological pupil dilator (y/n). Figure 1 displays a simulation of this generalized model and compares its predictions to the corresponding CS t model predictions. The figure clearly displays that both models predict more melatonin suppression for longer exposure durations. In contrast to the melanopic EDI-based model, the CS model cannot account for more than 70% melatonin suppression; neither does the CS model differentiate between cases with or without pharmacological pupil dilation.
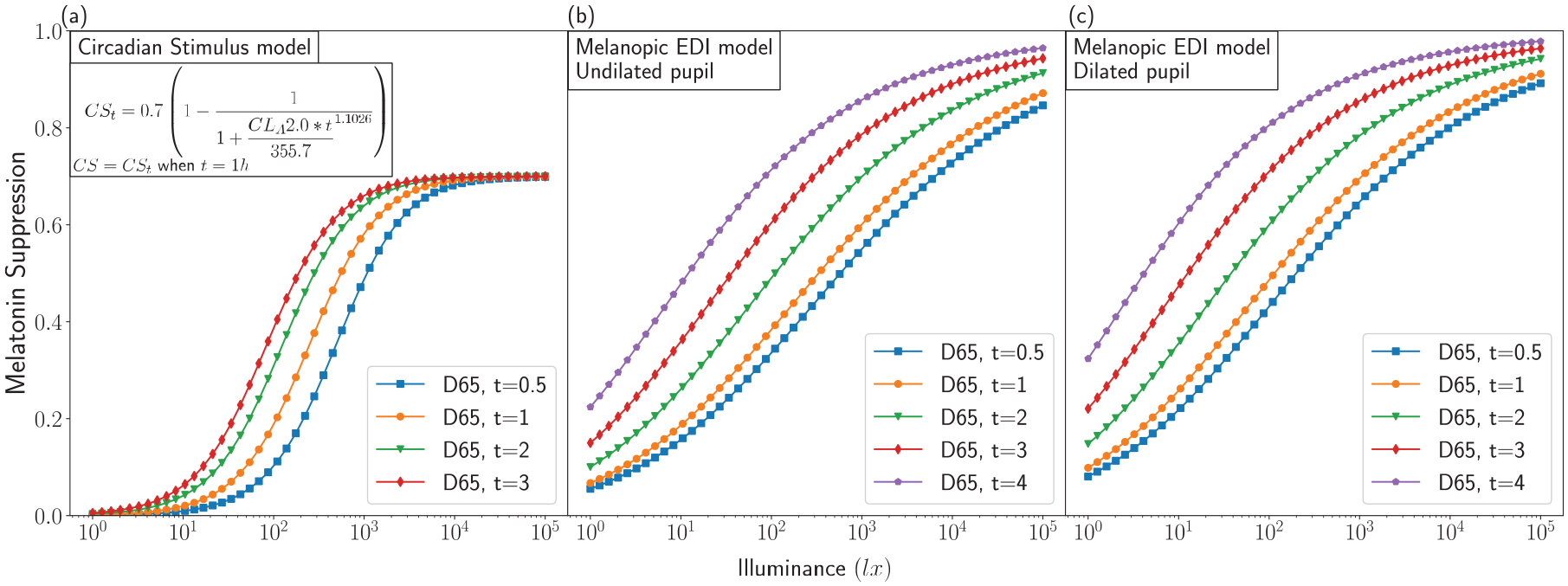
The % melatonin suppression (with 1 corresponding to 100%) as a function of the (photopic) illuminance of CIE standard illuminant D65 for a nocturnal light exposure with an exposure duration, t, of 0.5, 1, 2, 3 and 4 hours, (a) as predicted by the CLA2.0-based CS t model, here t = 4 hours is not included since the CS t model has only been tested for durations between 0.5 and 3 hours, (b) as predicted by the melanopic EDI-based logistic model from Giménez et al. 11 for a situation with and without pharmacological pupil dilation (c). For D65, the photopic illuminance equals the melanopic EDI.9,26 The calculations were implemented using the open-source LuxPy Phyton Toolbox for Lighting and Color Science v1.9.6. 27
In Figure 2, the CS-based daytime light recommendation from the UL Design Guideline (at least a CS of 0.3 for 2 hours during daytime, which corresponds to CLA2.0 = 274 for 2 hours, and according to Rea to CS d = 0.43) is converted into the corresponding photopic illuminance and melanopic EDI values for 1495 white light-emitting diode (LED) sources28,29 with a wide range of correlated colour temperatures (CCTs).
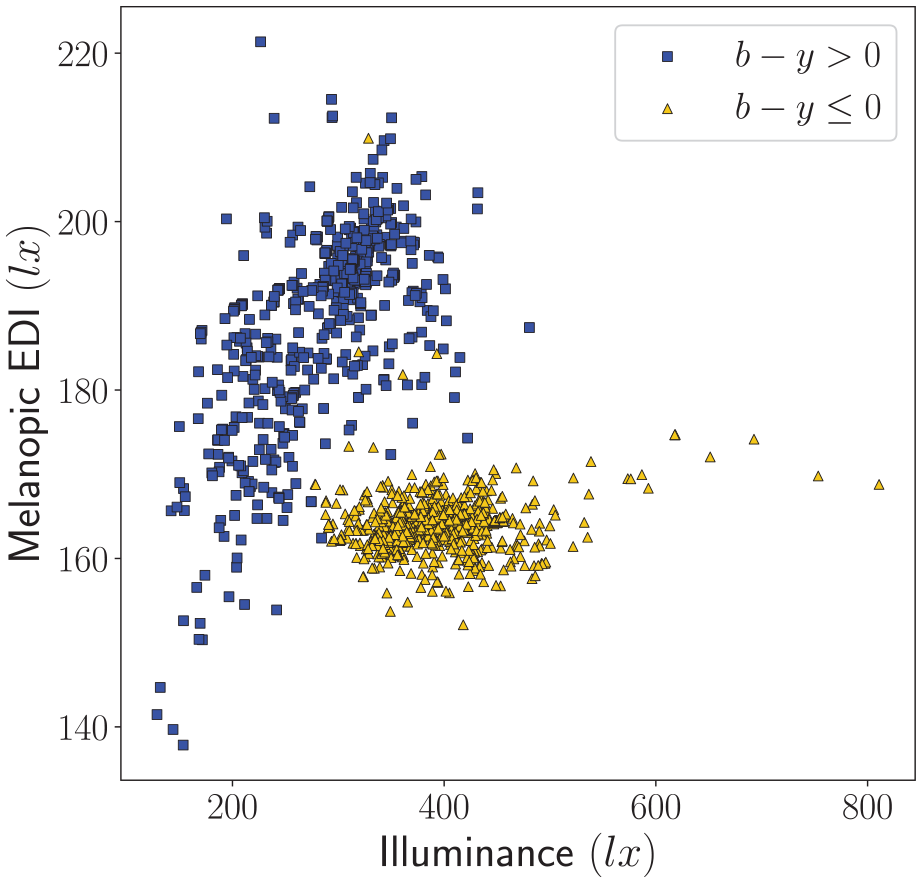
The photopic illuminance and melanopic EDI that corresponds to CS = 0.3 (i.e. CLA2.0 = 274) for 1495 white LED sources28,29 with different SPDs across a wide range of CCTs. Each point in the figure has a CLA2.0 of 274, and for t = 0.5, 1, 2 and 3 hours this corresponds to a CS t of 0.18, 0.30, 0.43 and 0.50, respectively. The blue squares denote SPDs for which b − y > 0 and the yellow triangles denote SPDs for which b − y ⩽ 0. For visualization purposes, 3 out of the 1495 light sources are not included in the figure: their CCT was below 1725 K (i.e. b − y ⩽ 0) and although their melanopic EDI was in the normal range, their illuminance was at least twice the illuminance of the rightmost point in the figure. The calculations were implemented using the open-source LuxPy Phyton Toolbox for Lighting and Color Science v1.9.6 27
All melanopic EDIs in Figure 2 are below the minimum of 250 lx melanopic EDI, as recommended for daytime light exposure by the expert workshop. 12 This indicates that the CS-based recommendation for healthy daytime light exposure is on the conservative side, and for all of the investigated LED sources insufficient to meet the recommendations from the international expert workshop.
In Figure 3, the CS-based recommendation for evening and nighttime light from the UL Design Guideline (a CS of maximally 0.1, which corresponds to CLA2.0 = 70) is converted into the corresponding photopic illuminance and melanopic EDI for the same 1495 white LED sources as used in Figure 2.
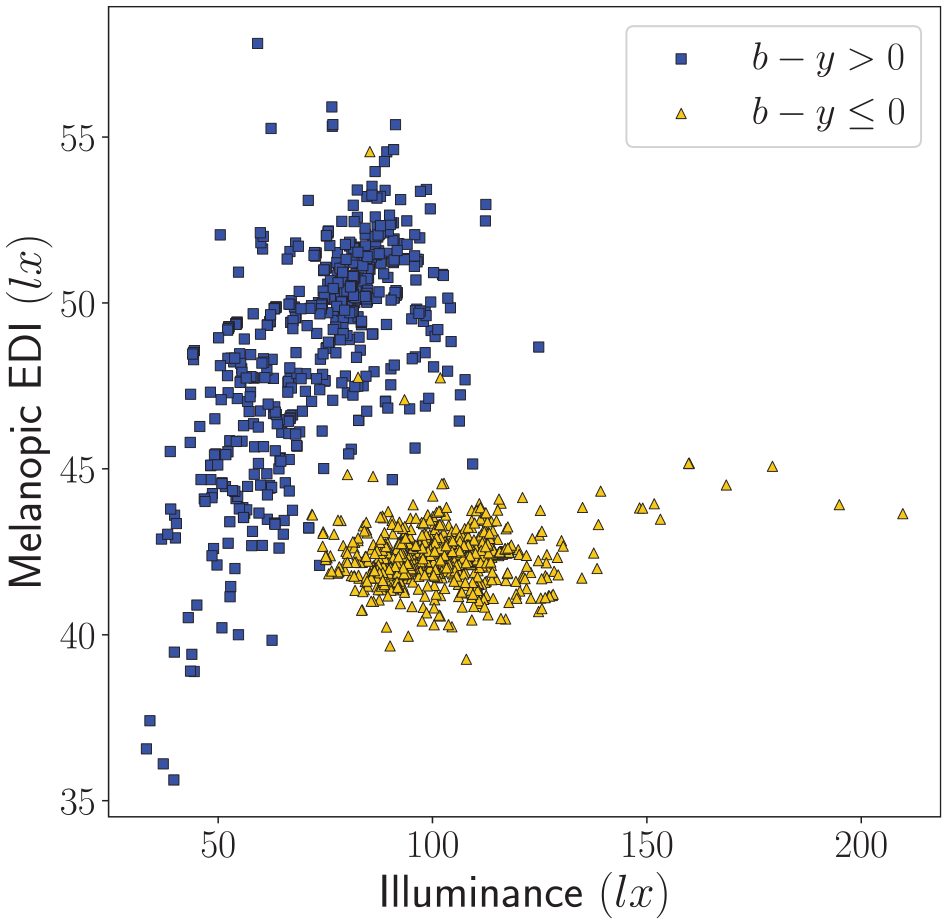
Comparison of the melanopic EDI and photopic illuminance that correspond to CS = 0.1 (i.e. when scaling all SPDs to CLA2.0 = 70) for a large database28,29 with SPDs from 1495 white LED sources. Each point in the figure has a CLA2.0 of 70, and for t = 0.5, 1, 2 and 3 hours this corresponds to a CS t of 0.05, 0.1, 0.18 and 0.25, respectively. The blue squares denote SPDs for which b − y > 0 and the yellow triangles denote SPDs for which b − y ⩽ 0. For visualization purposes, 3 out of the 1495 light sources are not included in the figure: their CCT was below 1725 K (i.e. b – y ⩽ 0) and although their melanopic EDI was in the normal range, their illuminance was at least twice the illuminance of the rightmost point in the figure. The calculations were implemented using the open-source LuxPy Phyton Toolbox for Lighting and Color Science v1.9.6 27
All melanopic EDIs in Figure 3 are above the maximum melanopic EDI of 10 lx for evening light exposure (starting at least 3 hours before bedtime), as recommended by the expert workshop. 12 This indicates that the CS-based recommendation for evening and nighttime light provides less (and potentially even insufficient) protection against the sleep- and circadian rhythm-disturbing effects of evening (and nighttime) light exposures than the recommendations from the international expert workshop.
Figure 4 provides a different representation of the data shown in Figures 2 and 3. It displays the melanopic EDI and CLA2.0 value per lx for each of the 1495 light sources plotted as a function of the CCT of the sources. This figure is included to provide lighting designers and practitioners with some first-order practical guidance on how to adjust CCT and illuminance as to reach a particular melanopic EDI or CS threshold value (see figure caption for more details).
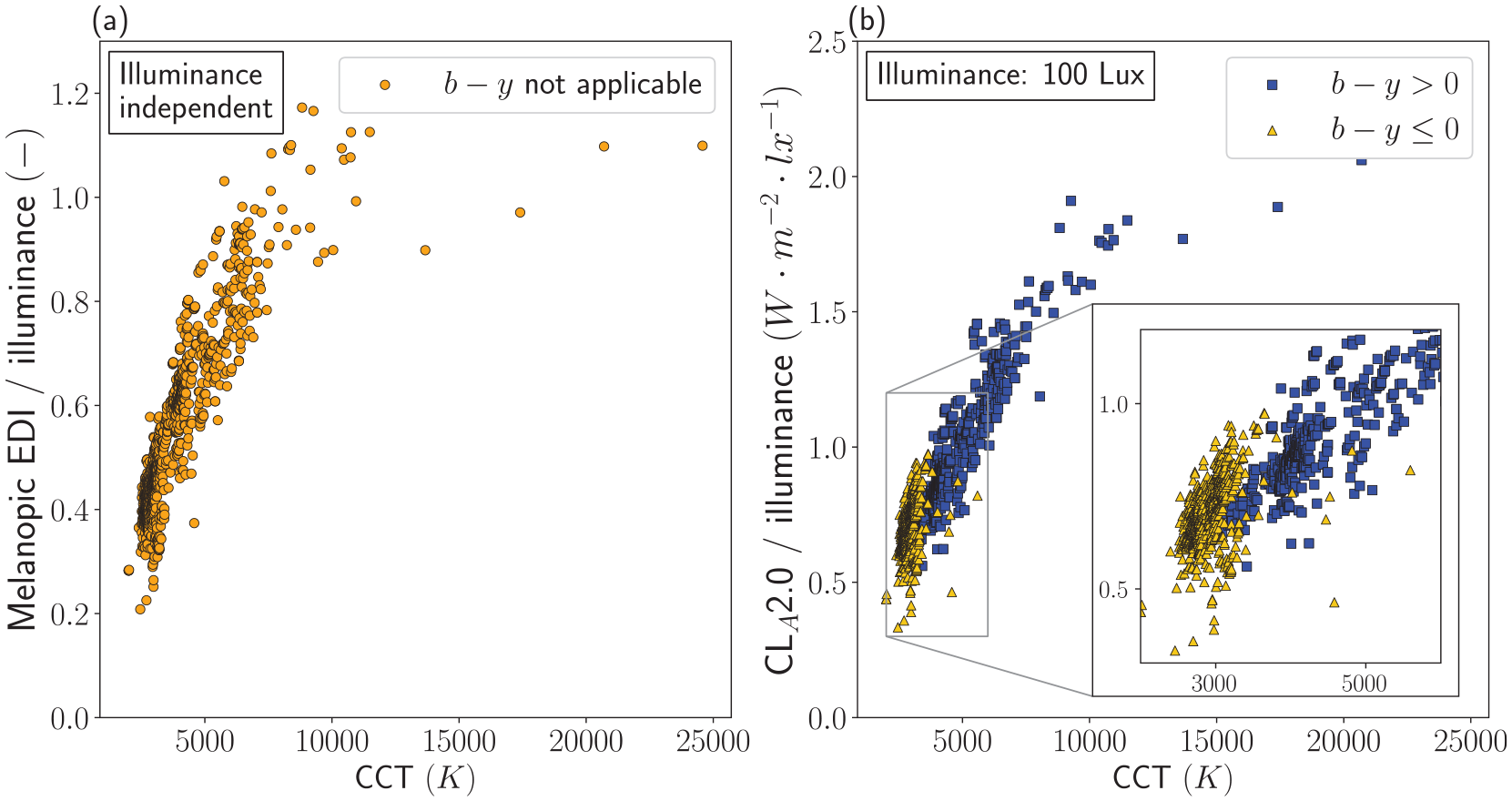
The illuminance-normalized melanopic EDI (a) and illuminance-normalized CLA2.0 values (b) for 1495 LED sources as a function of their CCT. The y-axes represent the melanopic EDI and the CLA2.0 value per lx of the source: 1 lx of the source produces a melanopic EDI or CLA2.0 that equals the value on the y-axis, while 100 lx produces 100 times that value. The term melanopic daylight efficacy ratio (melanopic DER) as defined in international standard CIE S026 9 is equivalent to the illuminance-normalized melanopic EDI (= melanopic EDI/illuminance) of the light source. 26 This feature represents a dimensionless ‘M/P ratio’ that is a light source characteristic, quite analogously to the S/P ratio (RSP) which is defined as the scotopic luminous output of a source divided by its (photopic) luminous output (see https://cie.co.at/eilvterm/17-21-113 and Schlangen and Price 26 ). In contrast to the illuminance-normalized melanopic EDI, the illuminance-normalized CLA2.0 (i.e. CLA2.0/illuminance) of a source is not fully constant across the illuminance range. However, the deviations are of little practical relevance: at an illuminance of 1 lx, the CLA2.0/illuminance has a mean, median, and largest difference (in %) from the values as plotted in the figure (which were calculated for an illuminance of 100 lx) of −0.26%, −0.14% and −0.9%, respectively. For an illuminance of 1000 lx, these differences are 2.26%, 1.26% and 7.59%, respectively, and for 2000 lx they are 4.54%, 2.56% and 14.9%. Overall, the illuminance-normalized values tend to increase with increasing CCT, yet the illluminance-normalized values can be quite different for a given CCT (as multiple SPDs can result in the same CCT30,31). The calculations were implemented using the open-source LuxPy Phyton Toolbox for Lighting and Color Science v1.9.6 27
In conclusion, the standardized α-opic metrology (CIE S 026/E:2018 9 ) and the melanopic EDI-based recommendations 12 provide a powerful and straightforward framework to inform light researchers, designers and other indoor professionals on light and lighting that optimally supports human health. They are (i) supported by a wide scientific consensus and evidence base, (ii) SI compliant and (iii) modular, thus enabling for future refinements while insights on the influence of other (α-opic) photoreceptors develop. The concerns and observations regarding the CS and CSt models as put forward in this work justify a further debate on whether these models can be considered an appropriate framework to assess ‘adequate circadian light exposure’ throughout daytime or nighttime. In addition, the CS-based recommendations as provided in the UL24480 Design Guideline seem insufficient to secure the merits of bright days and dim nights for integrative lighting solutions.
Footnotes
Acknowledgements
We thank Dr. Smet for his LuxPy package which has enabled a smooth and rapid implementation of the different calculations in this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work is (partially) supported by funds from the European Union and the nationals contributing in the context of the ECSEL Joint Undertaking programme (2021–2024) under the grant #101007319.
