Abstract
Light at dawn and dusk is the key signal for the entrainment of the circadian clock. Light at dusk delays the clock. Light at dawn advances the clock. The threshold for human entrainment requires relatively bright light for a long duration, but the precise irradiance/duration relationships for photoentrainment have yet to be fully defined. Photoentrainment is achieved by a network of photosensitive retinal ganglion cells (pRGCs) which utilise the short-wavelength light-sensitive photopigment, melanopsin. Although rods and cones are not required, they do play a role in photoentrainment, by projecting to and modulating the endogenous photosensitivity of the pRGCs, but in a manner that remains poorly understood. It is also important to emphasise that the age and prior light exposure of an individual will modify the efficacy of entrainment stimuli. Because of the complexity of photoreceptor interactions, attempts to develop evidence-based human centric lighting are not straightforward. We need to study how humans respond to dynamic light exposure in the ‘real world’ where light intensity, duration, spectral quality and the time of exposure vary greatly. Defining these parameters will allow the development of electric lighting systems that will enhance human circadian entrainment.
1. Background
1.1 The circadian clockwork
Almost all life on earth uses an internal biological clock to anticipate the profound changes that result from the Earth’s rotation upon its axis. Physiology and behaviour are ‘fine-tuned’ to the varied, yet predictable, demands of the day/night cycle. We effectively ‘know’ the time of day, and these internally generated daily cycles are called ‘circadian rhythms’ which comes from the Latin circa (about) and dies (day).
1
In addition to the alignment of the internal and external day, a circadian clock also ensures that biological processes occur in the appropriate temporal sequence. Human physiology is organised around the daily cycle of activity and sleep. In the active phase, when energy expenditure is higher and food and water are consumed, organs need to be prepared for the intake, processing and uptake of nutrients. The activity of organs such as the stomach, liver, small intestine and pancreas and the blood supply to these organs needs internal synchronisation, which a clock can provide. During sleep, although energy expenditure and digestive processes decrease, many truly essential activities occur including cellular repair, toxin clearance, memory consolidation and information processing. Indeed, our ability to function during the day is dependent upon the quality of our sleep. While night shift work is the most obvious disruptor of circadian rhythms and sleep, many of us experience shortened sleep as we try and squeeze more and more work and leisure activities into a daily schedule that is already over-packed. So, we push these activities into the night. The overall consequences of sleep and circadian rhythm disruption upon our emotional, cognitive and physiological health are summarised in Figure 1. The impact of sleep and circadian rhythms disruption (SCRD) upon human biology. The impact of SCRD on our acute emotional responses, acute cognitive responses and the chronic impact on physiology and health are shown. These effects are often due to the activation of the stress axis and the increased release of cortisol and adrenaline. Note that even short-term (acute) sleep loss (a few days) can have a big impact upon emotional performance and brain function. Longer-term (chronic) sleep loss (months and years) as experienced by some shift workers has been shown to increase the risk of several key diseases including cancer and cardiovascular disease. Such associations have long been a concern for shift workers, who suffer from extreme forms of sleep and circadian rhythm disruption. Citations: Fluctuations in mood,
2
–
5
depression and psychosis,
6
–
9
anxiety, irritability, loss of empathy, frustration
10
–
12
risk-taking and impulsivity,
13
–
16
negative salience,
17
stimulant, sedative and alcohol abuse,
18
–
22
illegal drug use,
23
impaired cognitive performance and the ability to multi-task,
24
–
26
memory, attention and concentration,
27
–
30
communication and decision-making,20,31–
34
creativity and productivity,
35
–
38
motor performance,26,39 social connectivity and increased social detachment,40,41 day time sleepiness, micro-sleeps, unintended sleep,
42
–
45
altered stress response,46,47 altered sensory thresholds,
48
–
50
impaired immunity and infection,51,52 cancer,
53
–
55
metabolic abnormalities and diabetes II,
56
–
60
cardiovascular disease61,62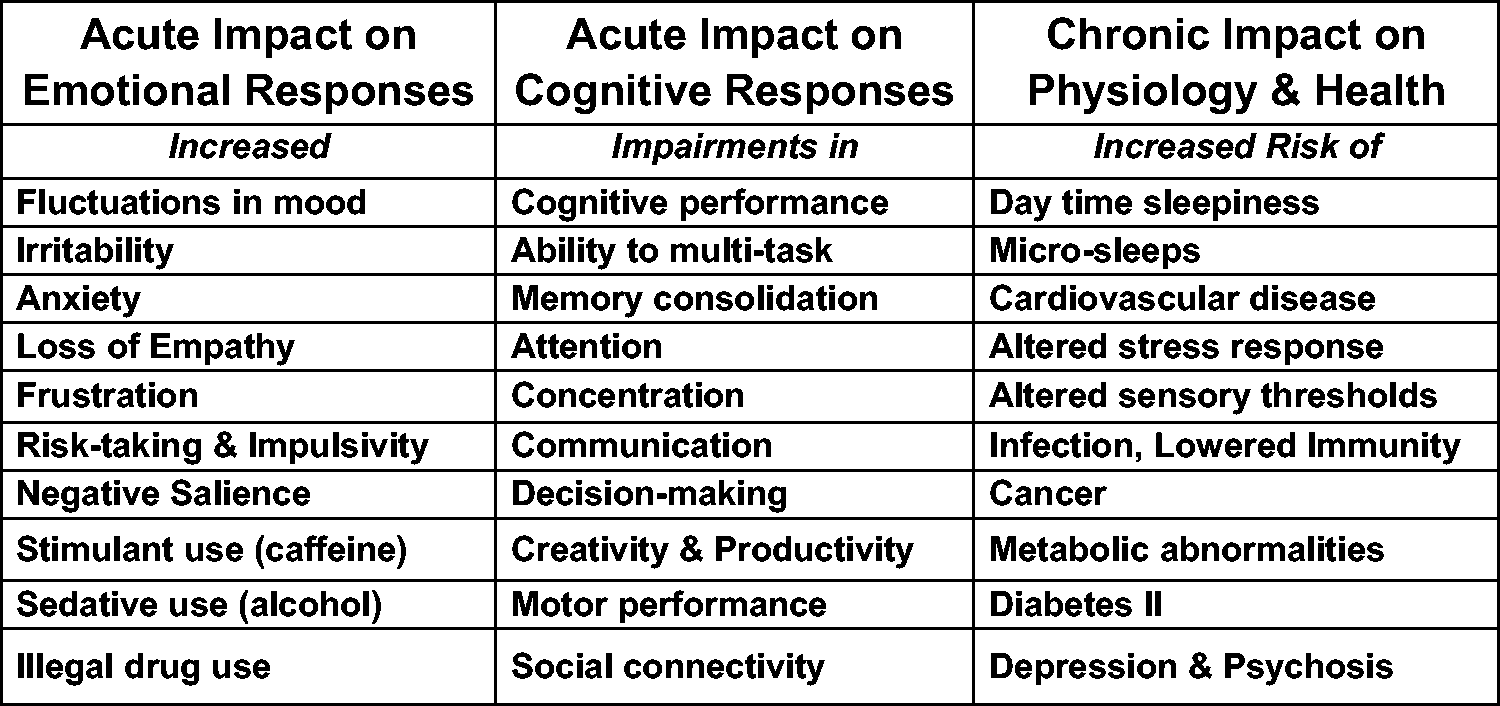
Our occupation of the night has been possible as a result of the widespread commercialisation of electric light. This extraordinary resource has allowed us to declare war upon the night, and without really appreciating what we have done, our sleep and circadian organisation have been the luckless victim. 63
At the heart of the circadian system is a paired structure located deep within the hypothalamus called the ‘suprachiasmatic nuclei’ or SCN. The SCN of humans is comprised of about 50,000 cellular circadian ‘clocks’ that are coupled together to produce a ‘master clock’. The generation of circadian rhythms within a cell arises from a complex interaction between key clock genes and their protein products. Subtle changes in the clock genes contribute to an individual’s tendency to be a morning person (‘lark’), evening person (‘owl’) or intermediate chronotype. 63 Although the SCN is the ‘master clock’ in mammals, it is not the only clock. There are cellular clocks, using essentially the same molecular mechanisms as SCN neurones, within the liver, muscles, pancreas, adipose tissue and probably in every organ and tissue of the body. The SCN acts as a pacemaker to coordinate, but not drive, the circadian activity of billions of individual peripheral circadian oscillators throughout the tissues and organs of the body. The result is a complex circadian network that coordinates rhythmic physiology and behaviour. 63
1.2 Shedding light on the clock
One very striking feature of circadian rhythms, across all life, is that they do not run at exactly 24 hours and tick a little faster or a little slower. The human circadian system, on average, is longer than 24 hours, so without daily resetting we would get up and go to bed about 10 minutes later each day. In this way, circadian rhythms resemble an old mechanical grandfather clock which needs a slight daily adjustment to make sure the clock is set to the ‘real’ astronomical day. The most important signal that aligns (entrains) the internal day to the external world is light, and especially light at sunrise and sunset. The process of daily resetting is called ‘photoentrainment’. In us, and other mammals, eye loss prevents photoentrainment, and individuals who have lost their eyes, or are exposed to insufficient light, drift through time and experience something similar to jet lag.
The SCN receives a major projection from the eye; however, the visual cells of the retina, the rods and cones, are not required for the detection of the dawn/dusk signal. There exists a third class of light sensitive cell (photoreceptor) within the eye. Our studies in the 1990s showed that mice lacking rods and cones and visually blind, either as a result of naturally occurring mutations or genetic modification, could still regulate their circadian rhythms to light perfectly normally. But when the eyes were covered, this ability to regulate the clock was lost. These experiments showed that there had to be another photoreceptor within the eye. With these critical data, our studies, and the work of several other groups, then demonstrated that the retina contains a population (around 10,000 cells) of photosensitive retinal ganglion cells (pRGCs) that use the short-wavelength sensitive photopigment call ‘melanopsin’
1
(Figure 2). This photopigment molecule (opsin) was originally isolated from light sensitive pigment cells, called ‘melanophores’, found in the skin of frogs and toads. The name ‘melanopsin’ was used to describe this photopigment and is often confused with ‘melatonin’ from the pineal (see below), but the two molecules are entirely unrelated. The term melanopsin is slowly being replaced with ‘OPN4’, partly for this reason. The retina of the eye. The rods and cones convey visual information to the retinal ganglion cells via the second-order neurones of the inner retina – the bipolar (BC) and amacrine (AC) neurons. The optic nerve is formed from the axons of all the ganglion cells and this large nerve takes light information into the brain. A subset of photosensitive retinal ganglion cells (pRGC – shown in blue) detects light directly by using the short-wavelength sensitive photopigment called melanopsin or OPN4. Thus, photodetection in the retina occurs in three types of cell: the rods, cones and pRGCs. The pRGCs receive indirect signals from the rods and cones, via inner retinal neurons, and these visual cells modulate light responses by the pRGCs. It is also important to note that at least five different subclasses of pRGC have been identified to date, suggesting that different pRGCs mediate different responses to light.
1
Counter-intuitively, light reaches the rods, cones and pRGCs by passing through the outer to the inner retina. AC: amacrine cells; BC: bipolar cells; H: horizontal cells; MWS – green: medium wavelength sensitive cones; LWS – red: long wavelength sensitive cones; rod – grey: rod photoreceptors ; SWS – purple: short wavelength sensitive cones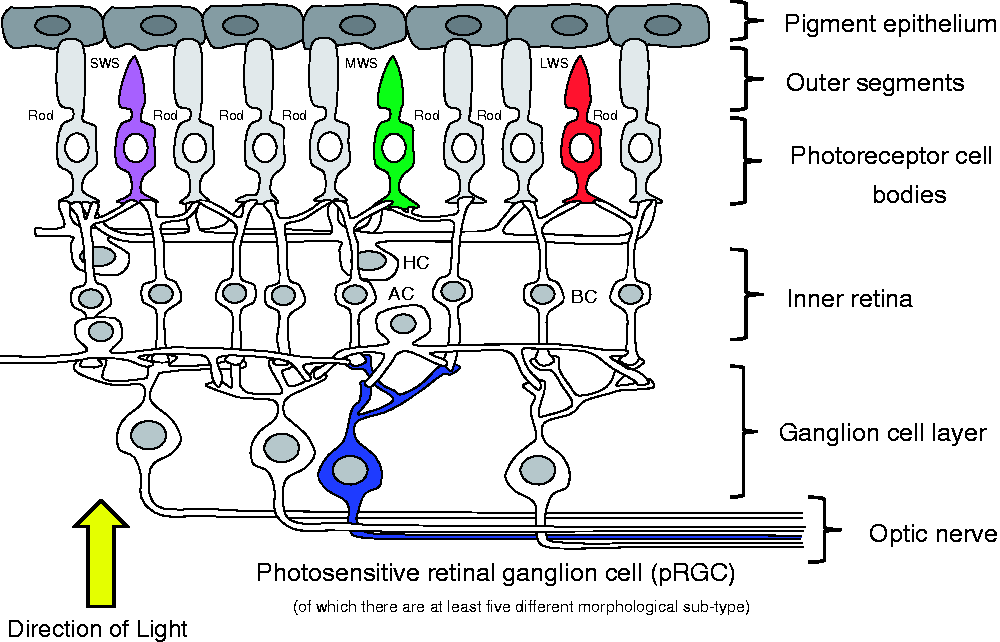
Following the studies in mice, we and others were able to show that humans also possess an OPN4-based short-wavelength sensitive pRGC system. Note – pRGCs are also called ipRGCs (intrinsically sensitive photosensitive retinal ganglion cells) or mRGCs (melanopsin retinal ganglion cells). It is also important to note that although the rods and cones are not needed for the regulation of the clock, we now know that they project to the pRGCs and can play an important role in circadian photoentrainment, as discussed below. Finally, in addition to circadian entrainment, multiple ‘brightness detection tasks’ are regulated by the pRGCs (see below), which in humans includes levels of alertness, the state of pupil constriction and the regulation of pineal melatonin. 64
1.3 A role for melatonin?
Melatonin is often confusingly termed the ‘sleep hormone’, and this is misleading. Melatonin is made mainly in the pineal gland, and the pineal is regulated by the SCN to produce a pattern of melatonin release, with levels rising at dusk, peaking in the blood around 02.00–04.00 a.m. and then declining around dawn. Bright light, detected by the pRGCs, also acts to stop melatonin production. Thus, melatonin acts as a biological marker of dark exposure. 65 While melatonin production occurs at night during sleep in day-active animals such as us, nocturnal animals like mice and rats also produce melatonin at night when they are active. So, if melatonin is not a ‘sleep hormone’, what does it do? Certainly, the tendency to sleep in humans is closely correlated with the melatonin profile, but this may be correlation and not causation. Indeed, individuals who do not produce melatonin (e.g. tetraplegic individuals, people on beta-blockers, or people who lack a pineal) still exhibit circadian sleep/wake rhythms with only very minor changes in sleep. So what is melatonin actually doing?
The SCN has receptors that detect melatonin and so, the night time rise in melatonin may provide an additional signal to set (entrain) the clock to the environment. Perhaps melatonin provides an additional signal to tell the SCN how long it has been dark, reinforcing the light signals from the eyes. However, it is important to appreciate that the melatonin signal is not very powerful, and the clock will always respond to light in preference to melatonin. There are also studies suggesting that melatonin may help reduce the time it takes to get to sleep and increase total sleep time. While the effects of exogenous melatonin on sleep have been shown to be significant in the majority (but not all) studies, the effects were relatively minor, shortening the time to fall asleep by less than 10 minutes. 66 For more discussion see Foster et al. 1 . In conclusion, and on the basis of the data available, melatonin seems to be a mild modulator, rather than a driver, of sleep.
2. The dynamics of circadian light detection
This section considers the key features of the light environment necessary for photoentrainment, specifically the timing, intensity, duration and wavelength of light, which can be all be modulated by an individual’s age and light exposure history. It is perhaps worth noting that until 1987 the assumption was that human circadian rhythms are primarily entrained by social cues, 67 with little if any role for light. Work on rodents had shown that the circadian system of mice is exquisitely sensitive to light. A light/dark cycle of 12 hours of light and 12 hours of darkness (L:D 12:12) using light intensities as low as 0.01–0.1 lux will photoentrain mice. 68 Light at such levels is completely ineffective in humans, 69 and the first clear evidence of photic entrainment in humans used a L:D cycle of 5000 lux. 70 We will discuss the light intensities needed for circadian entrainment in more detail below, but let us start with the timing of light exposure.
2.1 The timing of light exposure
In 1800, most of human society in Europe and the USA worked outside and was exposed to the natural cycle of light and dark. Today, in the UK, the proportion of workers in agriculture and fishing is 1% of the working population. This illustrates how the workforce, which means most of us, and particularly in the developed and developing nations, has become rapidly and profoundly detached from environmental light. And as a result, we have attenuated one of the most important signals that regulate our sleep/wake cycle. A critical, and frequently overlooked, feature of photoentrainment is the timing of light exposure. In brief, light around dusk and in the early evening will delay the circadian clock, and make us go to bed later and get up later the next day. By contrast, early morning light will have the opposite effect, advancing the clock, making us go to bed earlier and get up earlier. When we spent most of our time outside, we experienced a symmetrical exposure to dawn and dusk, with morning advances of the clock balanced by evening delays of the clock. 1
Today, dawn/dusk exposure can be very asymmetrical. As mentioned above, while our genes are an important part of our chronotype, when we are exposed to light can also be critical. Our recent study on university students from around the world showed that when the late types (‘owls’) were exposed to light in the evening (delaying light) but experienced little light in the morning (advancing light), the net effect was to shift the body clock to a later time. 71 In another study, sleep/wake timing was studied in individuals after one week of maintaining daily routines of work, school, social activities and exposure to electrical-lighting and then compared to one week of outdoor camping in tents and exposed to natural light. After the week of exposure to natural lighting, circadian timing and sleep/wake cycles had advanced by 2 hours. 72 These two examples illustrate both the importance of natural light, and that the timing of light exposure needs to be considered when designing artificial lighting systems.
2.2 The intensity, duration, wavelength and complexity of light detection
To provide some context to the discussion in this section, the approximate light levels within different environments and the sensitivities of the different photoreceptor classes have been illustrated in Figure 3. The human retina functions over a very wide range of light intensities. For reference, the familiar terms ‘scotopic’, ‘photopic’ and ‘mesopic’ have been used in this diagram. They are less than ideal measures of light intensity and may shortly be replaced by ‘alpha opic illuminances’.
73
Scotopic vision is defined as light detection by the rod photoreceptors of the eye under low light conditions. In the human eye, cone photoreceptors are nonfunctional in low light and rods mediate scotopic vision but extend into the mesopic range. Photopic vision is light detection by the cone photoreceptors of the eye under bright light conditions. In humans and many other animals, photopic vision allows colour perception which allows higher visual acuity and temporal resolution compared to scotopic vision. Mesopic vision is a combination of photopic vision and scotopic vision in low but not quite dim lighting situations and involves an input from both rod and cone photoreceptors. As light levels increase, and as rods become saturated, melanopsin photoreception is activated. While this diagram gives some sense of the sensory thresholds for the different photoreceptor classes under white light, it is also a little misleading in that it fails to take into consideration the differences in effective stimulus durations and wavelength sensitivities for the rods, cones and melanopsin-pRGCs. Rods and cones detect light within milliseconds while melanopsin-based pRGCs require long duration exposure (many minutes) to light in order to elicit a biological response. See text for details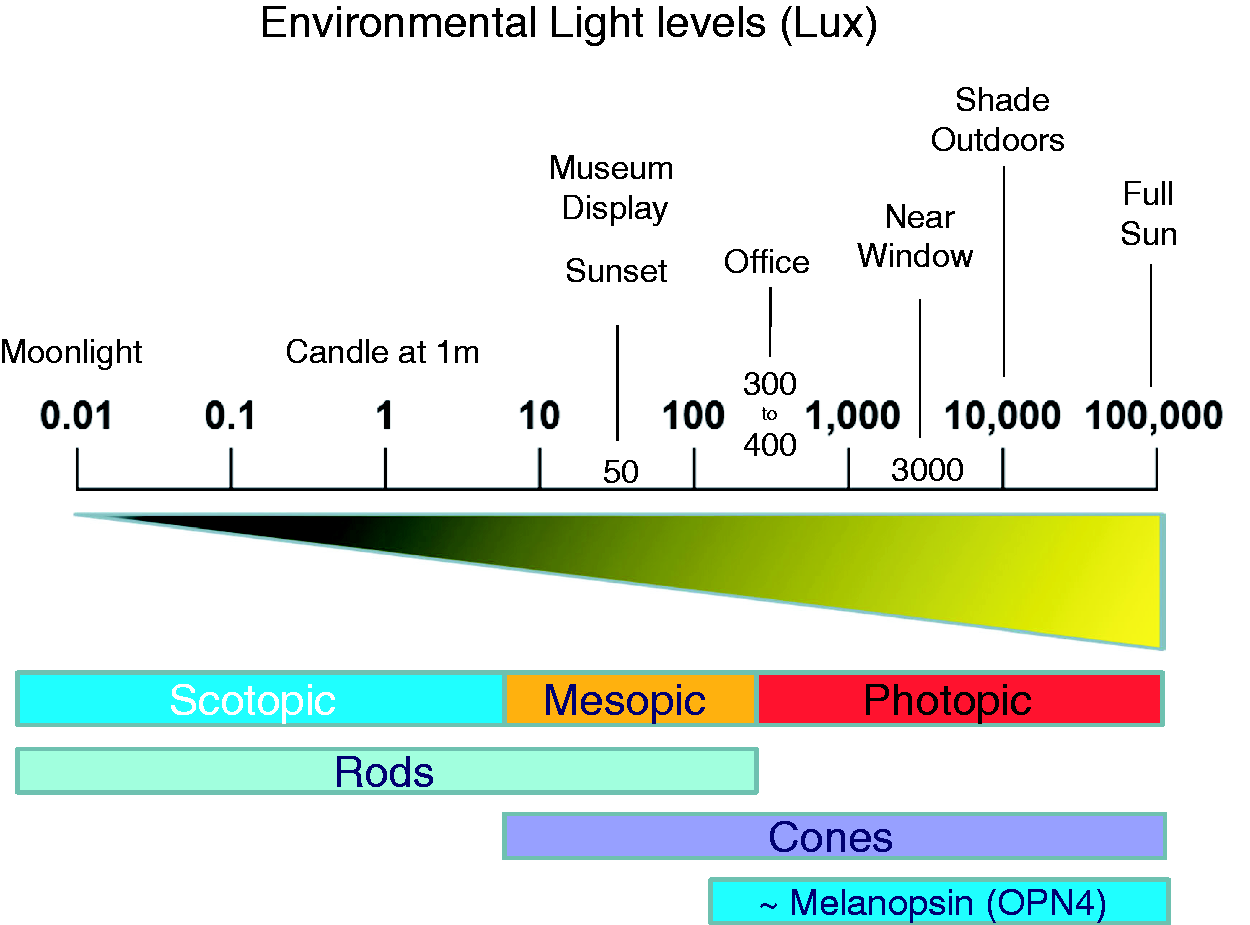
The early studies on the impact of light on the human circadian system used bright white artificial light (∼5000 to 10,000 lux) of a long duration (∼ 3 to 6 h) to simply demonstrate that light was capable of entraining human circadian rhythms.70,74,75 But defining the threshold of entraining light stimuli by systematically varying the intensity and duration of the stimulus has been only undertaken rarely and never at the level of detail seen in animal models. The irradiance of light exposure will greatly influence the magnitude of the response. Such a relationship is called an ‘irradiant (intensity) response curve’ or IRC. 76 Perhaps the most complete IRC in humans was undertaken over 20 years ago. 77 In this study, a single light exposure of 6.5 hours and of varying irradiance (1 to 10,000 lux) was delivered in the early night (delaying the clock), and the effect of this stimulus on shifting circadian rhythms was assessed. The study suggested that the phase shifting response saturated around 550 lux. These researchers concluded that the human circadian system is much more sensitive to light than previously considered, and that even small changes in light exposure during the late evening hours can significantly shift the human circadian system. The limitation of this conclusion is that the duration of the stimulus was not also systematically varied in these studies. While approximately 550 lux can saturate phase shifting responses to light, this was achieved with a stimulus duration of 6.5 hours. Thus, simple statements relating to the sensitivity of the human circadian system, without mentioning stimulus duration, can be deeply misleading. It is perhaps worth mentioning that the melanopsin photopigment can absorb light as efficiently as the photopigments of the rods and cones, 78 but unlike the rod and cone photoreceptors, pRGCs lack specialised photopigment-concentrating structures, primarily the outer segments, to maximise the chance of absorbing a photon. Thus, the probability of absorbing a photon for a pRGC is more than 1 million times lower than in a rod or cone photoreceptor. 78
Despite early suggestions that human non-rod, non-cone responses to light are maximally sensitive to 460 nm of light,79,80 it is now clear that these responses are melanopsin-based and peak close to 480 nm,81,82 and in this respect, humans are very like mice. Indeed, an early study on an individual lacking functional rods and cones showed that non-visual responses to light are based upon a ∼480 nm melanopsin-based photopigment.
83
These findings, and multiple others
84
–
88
all suggest that short-wavelength (‘blue’) light is significantly more effective for photoentrainment compared to longer wavelength of light, and is much more effective than broad spectrum white light. While these studies are all consistent with the involvement of melanopsin-based pRGCs, they do not, however, demonstrate that the pRGCs are the only photoreceptors mediating entrainment. We now know that the pRGCs receive indirect inputs from the rods and cones (Figure 4). The spectral sensitivities of the human photopigments. The spectral sensitivity of human melanopsin (OPN4) has a wavelength of maximum sensitivity (λmax) that is very close to 480 nm.82,83 The human rod photopigment – grey, has a λmax close to 498 nm, while the short wavelength sensitive cones (SWS) – purple, have a λmax close to 420 nm; medium wavelength sensitive cones (MWS) – green have a λmax close to 530 nm; and long wavelength sensitive cones (LWS) – red have a λmax close to 560 nm.
89
The fact that the rods and cones can modulate the responses of the pRGCs, and because of the high degree of overlap between the spectral responses of the different photopigments, the use of monochromatic light alone to stimulate just one class of photoreceptor is problematic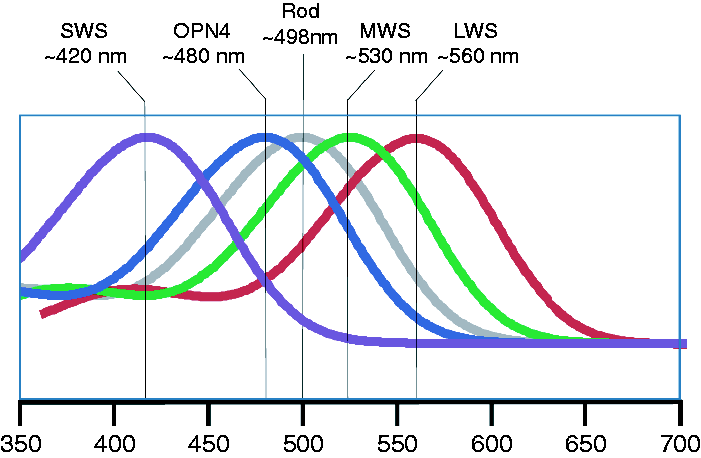
It seems that pRGC responses are the integrated product of endogenous melanopsin photoreception along with some level of input from rod and cone photoreceptors. 90 This means that the dynamic range of spectral responses for photoentrainment can, in theory, be very broad (Figure 4). However, the problem is that we do not know precisely the level of pRGC activations by the rods and cones. Indeed, inputs from SWS cones (Figure 4) may even inhibit pRGC activation. 90 As a result it remains unclear how ‘excitatory’ any light source is in regulating the circadian system, and in the absence of empirical physiological data, the current models that attempt to calculate the effectiveness of different light sources on shifting the clock 73 can, at best, only provide a very crude approximation under certain circumstances.
So, what can we say about pRGC, rod and cone interactions? The working model is that there is an integration of light signals at the level of the pRGCs. The view is that the rods are employed for dim light detection; the cones are used for the detection of higher light intensities and for the detection and integration of intermittent light exposure; and the pRGCs provide information regarding higher levels of light over extended periods of time. Such a sensory task of extended light detection would be ill-suited to the more transient light detecting capabilities of rod and cone photoreception (Figure 3). This hypothesis helps explain why short-wavelength sensitive melanopsin-based responses can be isolated in individuals possessing all their photoreceptor classes (rods, cones and pRGCs) when using long duration light exposures, but not short exposures. Studies that use stimulus durations of more than 30 minutes, and certainly those stimuli that were of many hours, would preferentially activate the melanopsin channel, and so shift the overt sensitivity to a short-wavelength melanopsin response. In short, high levels of blue light of a long duration will preferentially drive melanopsin responses in the pRGCs. 91
Could there be another role for multiple photoreceptor inputs to the clock? At dawn and dusk, there are very precise spectral changes in the light environment, primarily an enrichment of the shorter wavelengths (< 500 nm) relative to the mid-long wavelengths (500–650 nm). If the circadian system was capable of comparing the outputs from different photoreceptors to measure changes in the relative amounts of short and long wavelength light, then the timing of dawn and dusk could be determined very accurately. We proposed this idea some years ago on theoretical grounds, 92 and interestingly, some empirical evidence has emerged for such a ‘twilight detector’. 93
Since their identification, pRGCs have been shown to contribute to a broad range of non-image forming (NIF) responses to light, 94 including: pupillary light response (PLR) 95 the acute suppression of locomotor activity (negative masking) 96 sleep induction 97 – 100 ; levels of alertness 101 – 103 ; light aversion104,105 and influencing mood-related behaviours such as levels of anxiety and cognitive function.101,102,106 More recently it has been discovered that melanopsin contributes not only to NIF responses to light but also visual pathways, challenging the previous model of separate image forming (IF) and non-image forming (NIF) systems. 107 – 109 For example, melanopsin-based pRGCs provide visual centres of the brain with information regarding overall levels of environmental light, and perform roles in irradiance coding and brightness discrimination,83,110– 112 contrast detection 113 and adaptation of visual responses, 114 while also possibly providing spatial information and potentially supporting basic pattern vision.115,116 While these observations have been made primarily in mice, I think it very likely that a similar situation will exist in us and that the pRGCs can be considered as a ‘generalised irradiance detector’ for the regulation of multiple aspects of physiology and behaviour.
In addition to the different physiological and behavioural responses mediated by the pRGCs, the pRGCs of both mice and humans do not comprise a uniform population of cells. Instead they form an anatomically, genetically and functionally distinct set of subtypes. At least five, and possibly six, pRGC subtypes have been identified in mice. 116 – 120 They differ in their morphology and retinal connections, and show light responses with distinct properties (for reviews see.108,121,122 In addition, pRGC subtypes seem to project to different brain regions 116 –118,123– 125 and as a result, the different subtypes are almost certain to mediate distinct physiological responses to light. For reviews see literature.120,122 A similar level of anatomical diversity has been discovered in humans. Particularly elegant studies by Hannibal et al.126 have identified five different pRGC subtypes. As in mice, these melanopsin subtypes are not uniformly distributed across the retina, suggesting a level of functional specialisation and diversity of roles.
The consensus emerging is that diverse melanopsin pRGC subtypes mediate different behavioural and physiological responses to light. In addition, these pRGCs receive inputs from inner retinal neurons, strongly suggesting that rod and cone photoreceptors communicate, and likely modulate the activity of the pRGCs, 127 perhaps even ‘fine-tuning’ their spectral responses to light, for which there is good evidence in mice. 128 Understanding the functional significance of this organisation will undoubtedly be important in lighting design, but our current understanding precludes any definitive recommendations or advice at this time.
2.3 Exposure to light-emitting electronic devices
The use of light-emitting devices immediately before bedtime has been a concern in some quarters because of the potential impact these devices may have on human circadian timing, not least because the light they emit is enriched in blue light.129,130 Indeed, in the popular press, the use of electronic devices before bedtime is ‘known’ to alter our circadian rhythms. Most of the studies have focused upon the impact of such devices on melatonin suppression, which does not equate directly to the circadian regulation of sleep/wake (see Section 1.3). For example, in one study, a 5-hour exposure to a (white) light-emitting diode (LED) backlit computer screen suppressed melatonin and enhanced performance compared to a non-LED backlit screen. 129 Similar small effects have been obtained in other studies, 130 showing that a 2-hour exposure to the light from a computer screen can induced a slight, but not statistically significant reduction in melatonin concentrations in college students. This study was then extended using Apple iPads 131 who showed that melatonin levels were not significantly suppressed after 1-hour exposure to the tablets but this difference reached significance after 2 hours. The most detailed study to-date compared reading an LE-eBook in dim room light for ∼4 h (18:00–22:00) before bedtime for five consecutive evenings versus reading a printed book under the same conditions. The light emitted from the LE-eBook was ∼31 lux while the light reflected from the printed book was ∼1 lux. The results showed that LE e-Book use delayed sleep onset by less than 10 minutes after five days compared to reading the print book. Although the results were statistically significant (just), a delay of 10 minute is almost meaningless. 132
A cross-platform computer program has been developed called ‘f.lux’ that adjusts a computer screen’s colour temperature according to location and time of day based upon local sunrise and sunset. The effect is to reduce the irradiance and blue light emission from a screen in the evening compared to the morning. The proponents of f.lux hypothesise that altering the colour temperature of the display to reduce the prominence of blue light at night will improve sleep and reduce circadian rhythm disruption. Although the developer provides a list of relevant research on the website: https://justgetflux.com/, the program itself has not been tested to determine its efficacy. In spite of this, f.lux has been widely and positively reviewed by technology journalists, bloggers and users.
While it is probably sensible to minimise light exposure prior to bedtime to reduce levels of alertness, 133 the impact of light from digital devices on the circadian system needs a great deal of further study. Although the circadian impact of light exposure from screen use remains unresolved, we do know that late night use of technology-related behaviours (games, computer or phone use) does increase brain alertness and delays sleep. 134 This topic illustrates how misconceptions about light can be accepted as fact on the basis of very little or no evidence.
2.4 The impact of light history and age
Studies in humans provide evidence that the responses of the circadian system to light are influenced by prior exposure to light and darkness. For example, several studies have examined how the duration and relative intensity of prior light history affects the subsequent response to light.135,136 In one study, maintaining individuals on a dim versus bright L:D cycle resulted in significantly greater melatonin suppression and clock sensitivity in response to a 200 lux light stimulus.135,136 Overall the limited number of studies to date suggests that the light levels to which humans are exposed over the day will impact upon the sensitivity of the clock to light.
Age seems to be another important factor influencing the effects of light on the clock. In one study, melatonin suppression to low and moderate light levels in mid-pubertal (9.1–14.7 years) and late-pubertal (11.5–15.9 years) adolescents was compared. The results showed that the pre- to mid-pubertal group showed significantly greater melatonin suppression to light compared to the late-pubertal group. These results would support the hypothesis that early pubertal children show greater sensitivity to evening light compared to post-pubertal adolescents and that the increased sensitivity to evening light in younger adolescents could be particularly disruptive to sleep regulation for this group. 137 While there is evidence for increased photosensitivity of the circadian system during early adolescence, there is evidence for decreased photosensitivity in elderly humans. 138 One possibility for this decline in photosensitivity might be related the development of ocular problems such as cataracts. 139 This was tested by examining sleep quality before and after cataract surgery and using either clear (UV blocking) or blue blocking lens replacements. After surgery at six months, sleep quality had improved in both groups, suggesting that increased lens transmission had improved sleep/wake timing. There was no statistical difference between the two lens types, suggesting that the reduction in short-wavelength light transmission had not affected entrainment. 140 Based on mouse studies, there are also likely to be major changes in the circadian system as we age. For example, we showed that the retinohypothalamic tract diminishes in aged mice and that light-induced gene expression in the SCN is reduced by ∼50%. 141 Whatever the mechanism, increasing light exposure in the elderly seems to have positive benefits on sleep/wake timing. For example, long-term daily treatment with bright versus dim light in the elderly ameliorated symptoms of disturbed cognition, mood, behaviour, functional abilities, and sleep. These researchers concluded that the long-term application of bright light (∼1000 lux) should be considered for use in care facilities for elderly individuals to improve health and well-being. 142
3. The ecology of human light exposure
There is clearly a complex relationship between stimulus duration and intensity for photoentrainment. There is also evidence that the rods, cones and pRGCs interact for the regulation of entrainment, but precisely how remains unknown. The sensory threshold and spectral sensitivities of the different photopigments overlap (Figures 3 and 4). As a result, using monochromatic light to selectively stimulate a specific photoreceptor channel is not possible. In addition, we do not fully understand if the different photoreceptors interact in an additive or antagonistic manner. The timing of the stimulus is also absolutely critical. Light exposure at dusk will delay the circadian system (go to bed and get up later), while early evening light exposure will advance the circadian system (go to bed and get up earlier). For these reasons, and because so much essential information is lacking, including whether the direction of light is important, trying to develop artificial lighting within buildings to stabilise human sleep/wake cycles is not straightforward. Furthermore, trying to gather the missing information from laboratory studies would take both time and require very significant resources. Perhaps the way forward is to measure how humans respond to dynamic light exposure in the ‘real world’ where light intensity, duration, direction, spectral quality and the time of exposure vary greatly. Such an approach is now feasible because devices are available that measure non-invasively, continuously and over an extended period of time (weeks) light exposure (irradiance, duration, wavelength, time of exposure) and individual circadian timing (sleep/wake, and other measures of circadian phase), alertness and mood. Such measurements, in relatively large numbers of individuals, across a diverse demographic, would provide a means to identify how key features of natural light exposure interact to achieve entrainment (or not). I am strongly of the view that such natural light experiments will provide a key way forward for our understanding of how light is both detected by the human circadian system – and by extension – what is required for the development of truly human centric lighting.
Footnotes
Acknowledgements
This article was originally published in-part in Architectural Design, and thanks are given to Prof. Neil Spiller for permission to reproduce in a longer form here.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our research is supported by the Wellcome Trust.
