Abstract
Research in nonhuman animals (including insects, birds, and primates) suggests a trade-off in males between investment in competitive traits and investment in ejaculate quality. Previous research reported a negative association between perceived strength and ejaculate quality, suggesting that this trade-off also applies to human males. We conducted novel analyses of data secured as part of a larger project to assess the relationship between competitive traits (shoulder-to-hip ratio, handgrip strength, and height) and ejaculate quality (indexed by sperm morphology, sperm motility, and sperm concentration) in a sample of 45 men (ages ranging 18–33 years; M = 23.30, SD = 3.60). By self-report, participants had not had a vasectomy and had never sought treatment for infertility. We controlled for several covariates known to affect ejaculate quality (e.g., abstinence duration before providing an ejaculate) and found no statistically significant relationships between competitive traits and ejaculate quality; our findings therefore do not accord with previous research on humans. We highlight the need for additional research to clarify whether there is a trade-off between investment in competitive traits and investment in ejaculate quality in humans.
Research across the animal kingdom suggests a negative relationship between investment in competitive traits and investment in ejaculate quality (Kelly, 2008; Supriya et al., 2019). According to this “trade-off hypothesis,” individual males have limited metabolic energy, and investment across the life span in mate acquisition (e.g., via production and maintenance of intrasexually selected weapons or sexual ornaments or, more generally, competitive traits) occurs at the expense of fertilization success—a consequence of decreased investment in testes and, hence, ejaculate quality (Parker et al., 2013; Simmons et al., 2017).
Evidence consistent with the trade-off hypothesis has been found in several animals, notably in insects. In dung beetles (Onthophagus taurus), for example, there is a negative association between “critical body size” (a composite measure of pronotum width and horn length) and testes mass (Simmons et al., 1999) and between horn length (an intrasexual weapon) and testes mass (after controlling for body length, i.e., relative testes mass) in hissing cockroaches (Gromphadorhina oblongonota and Aeluropoda insignis; Durrant et al., 2016). Taken together, these insect studies document that horn size and testes mass are negatively related. In animals other than insects, magpie (Pica pica) tail length (a sexual ornament) is negatively associated with testes volume (after controlling for variables related to body condition, including body mass and spleen volume; Blanco & de la Puente, 2002). Thus, magpies with longer tails have lower testes volume. And across 63 nonhuman primate species, there is a negative association between sexual ornament production (e.g., tufts/beards and swelled/colored fleshy patches) and relative testes mass (Lüpold et al., 2019). These findings corroborate research in other animals (e.g., nonprimates), with sexual ornament size and quality being negatively associated with testes mass.
Support for the trade-off hypothesis also has been secured from experimental designs; for example, removal of the sexually selected hind leg of the leaf-footed cactus bug (Narnia femorata) is followed by an increase in testes mass, after controlling for pronotum width as a proxy for body size (Joseph et al., 2018). This experimental evidence supports the notion that this trade-off is a function of individual phenotypic plasticity that responds to short-term changes in the environment. This is the same principle by which operational sex ratio affects parental investment and intrasexual competition (see Kokko & Jennions, 2008).
Guided by findings in the animal literature, Foo et al. (2018) investigated the trade-off between investment in competitive traits and investment in ejaculate quality in humans. In their study, investment in competitive traits was operationalized as perceived physical strength, and investment in ejaculate quality was operationalized as sperm morphology, sperm motility, and sperm concentration in masturbatory ejaculates. The researchers secured full-body photographs and masturbatory ejaculates from participants. The photographs were rated for physical strength by a different sample of participants online. After controlling for several variables known to affect ejaculate quality (e.g., abstinence duration: time since most recent previous ejaculation), ratings of perceived physical strength were negatively associated with sperm concentration. Foo et al. concluded that, despite the benefits to precopulatory sexual selection (i.e., male–male competition and/or female choice), greater physical strength may come at a cost to sperm competitiveness. This research therefore provided corroborative support in humans for the trade-off hypothesis previously tested in nonhuman animals.
This research presents an additional test of the trade-off hypothesis in humans. We operationalized competitive traits with measures of shoulder-to-hip ratio (SHR), handgrip strength (HGS), and height. These measures provide valid cues of actual (rather than perceived) physical strength (e.g., HGS provides an index of overall muscularity; see Gallup & Fink, 2018; Gallup et al., 2007; Manning et al., 2010; Montoya, 2007; Pawlowski et al., 2000; Sell et al., 2009; Swami et al., 2007). The use of actual strength might increase the validity of this type of research as it is based on objective measurements and not subjective ratings, the latter that represent a psychological construct. Additionally, height, HGS, and upper body muscularity are all influenced by testosterone and thus defensibly reflect overall competitiveness (Manning et al., 2010; Shoup & Gallup, 2008). We operationalized ejaculate quality with three measures (following Foo et al., 2018): sperm morphology, sperm motility, and sperm concentration. We also assessed several covariates known to affect ejaculate quality: age, body mass index (BMI), and abstinence duration. Some ejaculate parameters vary over the life span—for example, the number of sperm in an ejaculate decreases with age (Cooper et al., 2010; Ng et al., 2004). Additionally, obesity is associated with infertility in men; BMI, for example, is negatively associated with sperm count (Eisenberg et al., 2013). Moreover, some ejaculate parameters are affected by abstinence duration prior to ejaculation; for example, rapid and repeated ejaculation reduces sperm number in subsequent ejaculates (Hopkins et al., 2017).
Method
Participants
This study reports novel analyses of a subset of data from a larger project (Pham et al., 2018). The original data set included responses from 66 men, with ages ranging 18–34 years (M = 22.77; SD = 3.83). Although 66 men completed the intake survey, errors in coding, ejaculate sample processing, and participant compliance produced a final sample of 45 men who completed the intake session and both ejaculate sessions. Thus, the final sample included men attending a university in the Midwestern United States, with ages ranging 18–33 years (M = 23.30; SD = 3.60; 71.1% of the sample was Caucasian, with Asian representing the second most commonly reported ethnicity at 6.7%; see Pham et al., 2018). By self-report, participants had not had a vasectomy, had never sought treatment for infertility, and were currently in a committed, heterosexual, and sexually active relationship for at least 6 months (range 6–123 months; M = 35.50; SD = 26.80).
Measures
Competitive traits
We operationalized competitive traits with measures of SHR, HGS, and height. We used (1) a digital scale with a height-measuring rod (Detecto, Webb City, MO) to assess height (cm) and weight (kg), (2) a tape measure (SE MT9R double-sided tailor’s measuring tape, New York City, NY) to assess shoulder girth and hip girth in centimeters (to calculate SHR = shoulder girth/hip girth), and (3) a digital dynamometer (Camry Scale, South El Monte, CA) to assess HGS (kgf) which allowed the adjustment of the handle according to hand size. Each participant completed three trials for each hand. Following Fink et al. (2007), we averaged the three scores per hand to construct composite right and left HGS variables.
Ejaculate quality
Ejaculate quality was assessed using the Semen Quality Analyzer (SQA-V; Medical Electronic Systems, Los Angeles, CA), a fully automated machine that analyzes ejaculates along several clinical parameters (see Pham et al., 2018, for details). Sperm morphology is the percentage of progressive normally shaped sperm, sperm motility is the number of progressive motile sperm (in M/ml of ejaculate), and sperm concentration is the number of sperm (in M/ml of ejaculate). Upon receipt of the participant’s masturbatory ejaculate, the ejaculate was syringed into a proprietary measurement capillary, which was inserted into a chamber in the SQA-V for automatic analysis. After completion of the automated analysis, all materials that directly contacted the ejaculate were discarded in a biohazard waste container.
In a previous study, the two ejaculate samples from each participant were produced in experimental (more sexually arousing) and control (less sexually arousing) conditions, then examined for differences using a Wilcoxon signed-rank test, which revealed no significant differences in any ejaculate parameter (see Pham et al., 2018). We correlated each parameter across the two ejaculates provided by each participant. Each parameter was moderately correlated across the two ejaculates (average Spearman’s ρ = .44; p < .05). We correlated ejaculate quality parameters with standings on the competitive traits separately for each of the two ejaculates provided by the participant. These results did not differ from those produced when we used average values for parameters across the two ejaculates (see Results section). Therefore, for parsimony and reportorial efficiency, we retained for analyses the averaged parameters estimated from the two ejaculates for each participant (produced in the experimental and control conditions in Pham et al., 2018). We verified that all ejaculate parameters were within the reference values for fertile ejaculate characteristics provided by the World Health Organization (see Pham et al., 2018, for details).
Procedures
All procedures were approved by the institutional review board of the university where data were collected. Participants were recruited via advertisements posted on bulletin boards on the campus of a Midwestern University in the United States. Participants contacted the laboratory to schedule three in-person sessions. In Session 1, participants were escorted to a private room and completed a survey containing several self-report questionnaires unrelated to the current report (e.g., measures of personality and relationship satisfaction). Then, the researchers collected several anthropometric measurements, including shoulder and hip girth (to calculate SHR), HGS, and height (see Measures subsection). At the conclusion of Session 1, participants received materials required to collect and transport two masturbatory ejaculates in two scheduled sessions (i.e., Sessions 2 and 3). The materials included a nonlatex, nonspermicidal condom, a plastic twist tie, a screw-top specimen container, a biohazard Ziploc bag, and an aluminum foil.
Participants were instructed to abstain from ejaculating for at least 48 hr prior to each masturbatory session following World Health Organization (2010) guidelines. Participants were asked to masturbate without the help of their partner and to not use any materials that we did not provide (e.g., pornography, lubricant). Participants masturbated to ejaculation in a private location of their choosing while wearing the provided condom. After ejaculation, participants sealed the condom and delivered it (within 1 hr of ejaculation) to the laboratory. Participants provided written consent, were told the purpose of the study at Session 1, and received US$25 at the conclusion of each session.
Results
Table 1 displays the descriptive statistics for the competitive traits and for the measures of ejaculate quality. We first conducted three hierarchical multiple regression analyses in which each model included one of the three ejaculate parameters as the dependent variable (sperm morphology, sperm motility, and sperm concentration). In all three models, the first step included only the covariates of age, abstinence period in days, ethnicity, and BMI. The second step in all three models included these covariates and the three competitive traits (SHR, HGS, and height). We identified just a single significant effect, for Step 1 (covariates only) of the model predicting sperm motility (see Table 2). The results of this full model, presented in Table 3, show that the only significant predictor of sperm motility is abstinence period, with longer abstinence duration positively predicting sperm motility. Once the competitive traits were added in the second step, the overall model was nonsignificant; however, the abstinence period remained as a significant predictor.
Descriptive Statistics for Competitive Traits and Measures of Ejaculate Quality.

Note. See text for the definition of each measure. SHR = shoulder:hip ratio; LHGS = left handgrip strength; RHGS = right handgrip strength.
Hierarchical Multiple Regression Models Predicting Ejaculate Quality From Competitive Traits.

Note. See text for the definition of each measure. Covariates = age, abstinence period, body mass index, and ethnicity. Competitive traits = shoulder:hip ratio, left handgrip strength, right handgrip strength, and height.
*p < .05.
Hierarchical Regression Results for Full Model Predicting Sperm Motility, Controlling for Age, Abstinence Period, BMI, and Ethnicity.
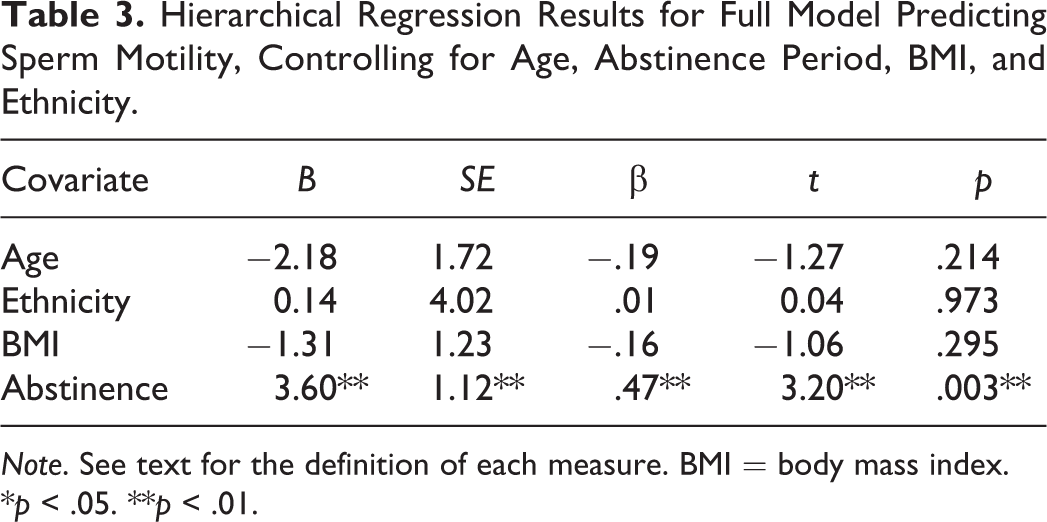
Note. See text for the definition of each measure. BMI = body mass index.
*p < .05. **p < .01.
Next, we calculated zero-order correlations between the competitive traits (SHR, HGS, and height) and each of the three measures of ejaculate quality (sperm morphology, sperm motility, and sperm concentration) using Spearman’s ρ (given nonnormal distributions of several variables; see Fowler, 1987). To adjust for Type I error, we reduced α from .05 to .01 (Tukey, 1977). As displayed in Table 4, none of the correlations reached statistical significance.
Finally, we calculated partial Spearman’s correlations between the competitive traits and the measures of ejaculate quality, controlling for abstinence duration before providing the ejaculate, age, ethnicity, and BMI. Because SPSS (IBM SPSS version 26) does not readily allow this function, we first transformed the variables of interest to ranked cases. Then, we entered the ranked cases into the partial correlation command for Pearson’s correlation coefficients. Once again, to adjust for Type I error, we reduced α from.05 to .01. As displayed in Table 4, none of the partial correlations reached statistical significance.
Zero-Order and (Parenthetical) Partial Correlations Between Measures of Competitive Traits and Measures of Ejaculate Quality.

Note. See text for the definition of each measure. Values outside of parentheses are zero-order Spearman’s correlation coefficients; parenthetical values are partial Spearman’s correlation coefficients, after controlling for the covariates of abstinence period, age, ethnicity, and body mass index. SHR = shoulder:hip ratio; LHGS = left handgrip strength; RHGS = right handgrip strength.
All ps > .01.
Discussion
We investigated the hypothesized trade-off between investment in competitive traits and investment in ejaculate quality in humans. Foo et al. (2018) reported a negative association between perceived male strength (measured as ratings of strength from full-body photographs) and sperm concentration, consistent with the trade-off hypothesis. We did not detect significant associations between competitive traits (operationalized as SHR, HGS, and height) and ejaculate quality parameters (i.e., sperm morphology, sperm motility, and sperm concentration). We note, however, that Foo et al. report a Pearson’s r of −.25 between perceived strength and sperm concentration, a small negative effect. Although the results of the current research did not reach statistical significance, the size of the effects (Spearman’s ρs ranged from −.06 to .13) of the associations between sperm concentration and competitive traits is even smaller than reported by Foo et al. Given that the established effect sizes are small, adequate power (achieved by appropriately large sample sizes) is especially important for detecting these effects. In addition, the use of different measures (i.e., perceived strength vs. actual strength) renders it questionable to make direct comparisons of effect size between studies.
Thus, there are several possible reasons why the current research failed to support the trade-off hypothesis. First, our measure of strength was not based on ratings provided to a static photograph as in Foo et al. (2018). Instead, we used direct assessments of strength, which should be more valid and, therefore, more likely to accurately reflect the relationship between strength and ejaculate quality. Previous research included a single ejaculate from each participant, whereas we secured two ejaculates from each participant. We then calculated the average for each parameter across the two ejaculates and used this average value as the data point for each parameter. Because ejaculate quality parameters are known to exhibit within-subject variability (Mallidis et al., 1991; Schwartz et al., 1979), it is possible that the null relationships we identified are attributable to the use of averaged and, therefore, more representative ejaculate parameters. That is, the relationships we report may reflect more accurately the true (null) relationship between investment in competitive traits and investment in ejaculate quality.
Another issue to note is that participants in this research masturbated to ejaculation while reading written vignettes rather than during exposure to erotic images or videos. It is possible that the written erotica did not generate adequate arousal to produce ejaculates of representative quality. We note, however, that all parameters for all ejaculates included in analyses were within normal range as defined by the World Health Organization (2010).
A limitation of this study is the relatively small sample of 45 men. This sample may have lacked statistical power to detect small effects (as the correlation coefficients suggested small to moderate effects). Additionally, small sample sizes increase the risk of both Type I and Type II errors. Unfortunately, small sample sizes are a recurrent limitation of psychological research investigating ejaculate quality (e.g., Baker & Bellis, 1989), perhaps due to difficulties recruiting participants outside a clinical setting. Future research would benefit from larger sample sizes in individual studies as well as combining these and future small-sample studies into meta-analyses that investigate the hypothesized trade-off between competitive traits and ejaculate quality in humans.
Alternatively, and more speculatively, there could be a theoretical explanation for why our results did not support the trade-off hypothesis. Despite some research supporting the trade-off hypothesis, other research suggests that there may be a phenotype-wide fitness factor such that fitness is signaled in multiple ways simultaneously, including through secondary sexual characteristics and ejaculate quality (Arden, Gottfredson, & Miller, 2009; Houle, 2000; Miller, 2000; Prokosch et al., 2005). According to this framework, positive correlations should be observed between ejaculate quality and many indicators of phenotypic quality. For example, Arden, Gottfredson, Miller, and Pierce (2009) documented a positive correlation between ejaculate quality (including sperm concentration and sperm motility, two of the measures used in the current study) and intelligence that was unaffected by covariates (e.g., age and abstinence duration). Positive correlations also have been documented between ejaculate quality and body symmetry (Manning et al., 1998) and mate value (operationalized as a composite of self-reported mate value and third-party ratings of attractiveness and dominance; Leivers et al., 2014). In the current study, although they did not reach statistical significance, the directions of the correlations between competitive traits and ejaculate quality were mostly positive, especially for correlations of higher magnitude. These positive correlations are opposite in direction to that predicted by the trade-off hypothesis; hence, these results may reflect the hypothesized phenotype-wide fitness factor. This is an area that needs more research, as even a meta-analysis on the relationship between male phenotypic quality and ejaculate quality was inconclusive due to the small number of studies of human participants (Jeffery et al., 2016).
In conclusion, we tested the hypothesized trade-off between investment in competitive traits and investment in ejaculate quality in humans. We did not find support for this hypothesis; instead, we identified no significant associations between competitive traits and ejaculate quality measures. Given the difficulties in recruiting large samples for this type of research, future researchers can incorporate these results and those of similar small-sample studies into meta-analyses to address these research questions more definitively.
Footnotes
Author's Note
Bernhard Fink is alo affiliated with Department of Evolutionary Anthropology, University of Vienna, Vienna, Austria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
