Abstract
Sexual responses are thought to be controlled by a brain module called the sexual module. Sexual strategies of males and females vary to a great extent, and sexual responses of males and females may be affected by their sexual strategies. However, the current view of the sexual module is that of a unisex module. This might be questionable since brain modules are defined as evolved cognitive mechanisms to solve adaptive problems which are different for males and females. We hypothesize that the sexual module responds differently in the presence of complex (high-order) contextual cues that are related to gender-dimorphic sexual strategies in males and females.
We conducted a priming experiment in which stimuli related to sexual strategies were disentangled from their sexual meaning. Nonsexual priming pictures related to either economic resources or social interactions preceded a sexual-target picture in order to test whether the primes were able to modulate the subjective sexual response to the sexual target. In a control condition, priming pictures without relation to mating preferences but with similar emotional impact were presented. In males, sexual responses were similar in the experimental and control conditions. In females, however, primes related to economic resources or social interactions modulated sexual arousal significantly more than the control primes. Our findings suggest that brain modules dedicated to process the experimental primes were functionally connected with the sexual module in females more than in males, making females’ sexual responses more prone to the impact of high-order cultural cues than males’ sexual responses. A gender-dimorphic connectivity of the sexual module may be the way in which gender-dimorphic sexual strategies are implemented in the human mind.
Keywords
Parental investment theory states that in species with sex-specific differences in obligatory parental investment, the higher investing sex (typically the female) will be more selective in its choice of mating partners (Trivers, 1972). If in these species males sometimes contribute resources to offspring, females’ selection will be based in part on the ability and willingness of males to invest in their offspring (Buss & Schmitt, 1993). Accordingly, in humans, sexual strategies are found to differ between sexes to a great extent, with females showing a higher preference than males for mates who possess resources and are willing and interested in investing in children, and males showing a higher preference for physically attractive and young mates than females (Brase, 2006; Conroy-Beam, Buss, Pham, & Shackelford, 2015; Janssen & Bancroft, 2007; N. P. Li, Bailey, Kenrick, & Linsenmeier, 2002; Roney, Hanson, Durante, & Maestripieri, 2006). Furthermore, women are repeatedly found to be less prone to short-time sex than men (Clark & Hatfield, 1989; Gueguen, 2011; Schmitt, 2005; Voracek, Hofhansl, & Fisher, 2005). Female-typical choosiness is an adaptation to sex-typical selection forces.
At the level of individual brain function, sexual behavior is controlled by an evolved cognitive mechanism, the sexual module (Spiering & Everaerd, 2007). Several studies showed a functionally specialized, automatic, and adaptive processing of sexual stimuli (Both et al., 2008; Gillath, Mikulincer, Birnbaum, & Shaver, 2007; Janssen, Everaerd, Spiering, & Janssen, 2000; Ponseti & Bosinski, 2010; Spiering, Everaerd, Karsdorp, Both, & Brauer, 2006), which is in accordance with the concept of an evolved cognitive mechanism (Barrett & Kurzban, 2006; Carruthers, 2006; Fodor, 1983; Sperber, 2005). In the following, we will shortly describe how sex research currently describes at a proximate level the functioning of the sexual module and what, from an evolutionary psychology point of view, is missing in that model.
Consistently, influential models of sexual stimulus processing assume that an internal representation of a (potentially sexual) stimulus undergoes several stages of pattern recognition (Janssen et al., 2000; Spiering & Everaerd, 2007; Stoleru, Fonteille, Cornelis, Joyal, & Moulier, 2012). Essentially, this process is seen as a stimulus-driven process (bottom-up). However, any type of meaning cannot be activated unless the stimulus representation matches a memory code. So far, some type of top-down processing has always been assumed when meaningful stimulus processing takes place. Memory codes can represent either explicit knowledge or implicit knowledge such as response tendencies. Successful matching between encoded stimulus representations and memory codes is also referred to as stimulus appraisal. If the activated memory codes are “sexual,” sexual responses are activated. Generally, sexual responses comprise motivational, emotional, and autonomic (genital and endocrine) aspects.
The current theory of a sexual module is an important achievement for various reasons: First, it comprises descriptions of stimulus processing that are in accordance with basic theories of emotional processing (LeDoux, 1996; Öhman, Flykt, & Lundqvist, 2000); second, it is compatible with detailed neurofunctional (Georgiadis & Kringelbach, 2012; Stoleru et al., 2012) and neuroendocrine characterizations (Pfaus, 2009); and third, it acknowledges its evolutionary foundation. However, the multiplicity of sex-dimorphic responses, behaviors, and preferences in the realm of mating is not accounted for in the current characterization of the sexual module. This appears to be a theoretical contradiction, or at least a major gap, given that a sexual module is assumed to solve adaptive problems of reproduction that are of course different for males and females. Thus, it is questionable whether males and females process sexual stimuli by means of a unisex sexual module.
To be more specific, particularly in females, the sexual attractiveness of a potential mate can be altered by additional information regarding his status or his fondness for children (Dunn & Searle, 2010; La Cerra, 1995; Townsend & Levy, 1990) or by the potential risk of male–female aggression (Y. Li et al., 2014). Females’ attitudes toward sexual stimuli can even be altered by a context of luxuriousness (Vohs, Sengupta, & Dahl, 2014). With regard to sexual stimulus processing, these examples suggest (1) that the sexual salience of a stimulus can be modulated by complex contextual cues and (2) that this modulation can be influenced by the sexual strategies of the observer. Neither of these assumptions is accounted for by the current view of the sexual module. And in fact, these assumptions are not easy to reconcile with the conceptualization of brain modules, in particular with the assumptions of domain specificity and encapsulation (i.e., its relative imperviousness to cognitive control). If the sexual module is activated preferentially by sexual stimuli, sexual stimulus processing (including response generation) should run relatively independently of contextual stimuli. Similarly, a snake might trigger more or less the same fear response independently of whether a person is confronted with the snake in a forest or in a modern shopping mall. One possibility to reconcile the contextual modulation of (female) sexual responses with the assumption of an evolutionary-shaped sexual module could be to assume that the processing of the sexual module is somehow modulated by the processing of other brain modules, for instance, modules dedicated to object recognition or social processing. The impact of other brain modules on the sexual module could be realized by varying, and possibly sex-dimorphic, degrees of connectivity.
In order to better understand the functioning of the sexual module in regard to sexual strategies, we wanted to test two assumptions: First, that sexual responses can be modulated by nonsexual contextual cues, and second, that such a modulation can be sex dimorphic. To this end, we disentangled cues that are relevant for mating preferences from sexual meaning and tested their impact on sexual stimuli by means of a priming experiment. We used pictures depicting either abundant resources (economic primes positive [EP+]) or a lack of resources (EP−) as priming stimuli to test whether the response to a subsequent sexual-target picture could be modulated. Similarly, we tested the impact of priming pictures that showed either opposite-sex adults who were friendly looking or caretaking toward children (social primes [SP+]) or aggressive looking or aggressive behaving (social primes negative [SP−]) on the response to sexual-target pictures. For females, the facial expression of an adult male has been found to be a proxy for his fondness for children (Roney et al., 2006).
We expected the priming pictures to modulate the response to the sexual-target pictures in females more than in males because we assumed that a positive economic or social context would trigger functional connections to the sexual module of females in particular. However, emotionally positively valued circumstances might enhance sexual response independently from evolutionary-selected, sex-dimorphic mating preferences simply because a positive mood might be more compatible with a sexual response than a negative mood (Klauer & Musch, 2003). To control for such unspecific priming effects, we additionally used priming stimuli without the social or economic meaning of EP+/− and SP+/− but with similar characteristics in terms of emotional valence and arousal.
Material and Method
Stimulus Preparation
For each of the four priming conditions, 21 colored images were preselected by the experimenters: (1) pictures indicating an abundance of resources, such as expensive houses, expensive cars, and jewelery (EP+); (2) pictures indicating a shortage of resources, such as barracks, humble furniture, and broken cars (EP−), with no persons visible in either the EP+ or EP− images; (3) pictures depicting either one friendly looking or caretaking adult or two (male and female) friendly looking or friendly behaving adults (sometimes taking care of a child; SP+, social primes positive); and (4) pictures depicting either one aggressive-looking or -shouting adult or two adults (male and female) fighting or aggressively shouting (sometimes with a child visible; SP−). None of the (21 × 4 =) 84 images of the four priming conditions displayed any sexual content.
Although most of the images were used equally for male and female participants in the main experiment, some images varied according to the gender of the participant. In pictures in which only one adult was visible (in the SP+ or the SP− condition), participants were exposed to images of an opposite-sex adult. In some images of the SP− condition that displayed fighting adults, one adult was depicted as the aggressor while the other appeared rather as the victim of the aggression. In these cases, participants in the main experiment were exposed to images with an opposite-sex aggressor and same-sex victim. While all images in the EP− condition were identical for male and female participants, five of the EP+ images were different for the male and female participants (showing in these cases gender-typical luxuries—e.g., shop for expensive clothes for women or shop for expensive clothes for men, respectively). Taken together, a male and female stimulus set of 84 priming stimuli was used, which was largely identical and, in those cases where it was not identical, it was analogous.
Thirty-one males rated the 84 images of the four priming conditions (male set) and an additional 298 images in terms of valence and arousal (Bradley, Greenwald, & Hamm, 1993). We preselected the 298 images with the aim that they would not be perceived as related to one of the four priming conditions (EP+, EP−, SP+, and SP−). The 298 images depicted a multiplicity of topics, like animals, landscapes, and objects, but not humans. Of the 298 images, we aimed to select 84 images that closely matched the valence and arousal ratings of the 84 priming images in order to serve as control stimuli. However, because we did not find enough images that closely matched the 84 priming stimuli, we did a second rating with 20 males who rated an additional 130 images. By this, we finally gathered 84 images that matched the priming stimuli. These 84 matched images served as control stimuli (EPC+, EPC−, SPC+, and SPC−). Similarly, 24 females rated the 84 images of the four priming conditions (female set) and an additional 300 images (again, preselected, aiming not to relate to the four priming conditions) to serve as control images in terms of valence and arousal. Of the 300 images, we selected 84 images that closely matched the 84 priming images with regard to the female valence and arousal ratings. Two-sided paired t tests revealed no significant differences between the valence and arousal ratings of the experimental primes and corresponding control primes. Mean valence and arousal ratings of the priming stimuli of the four experimental and the four control conditions and p values are listed in Table 1.
Mean Valence and Arousal Ratings and Standard Deviations (SD) for the Four Priming Conditions and the Corresponding Control Primes (21 Images in Each Condition) by Male and Female Raters.
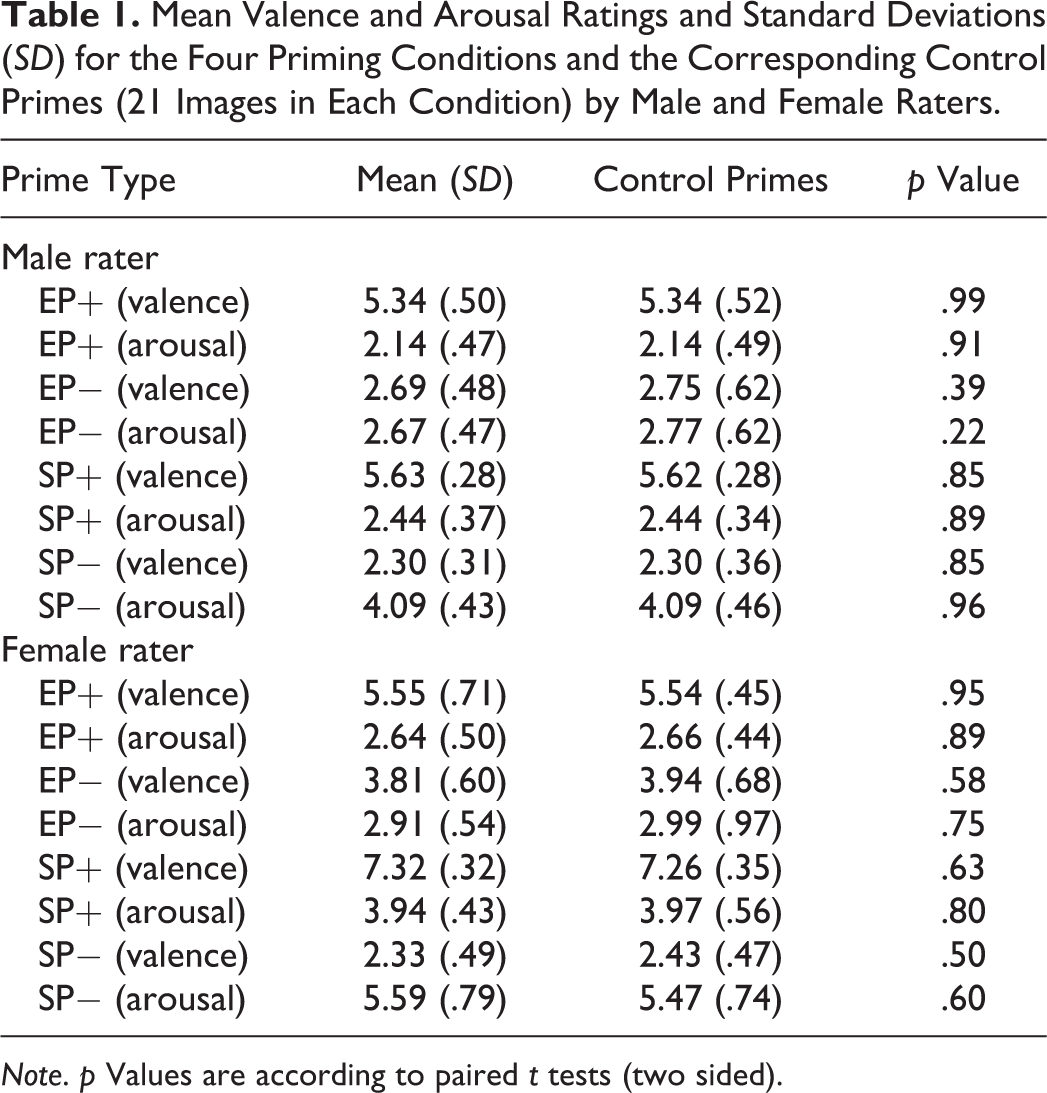
Note. p Values are according to paired t tests (two sided).
Finally, we preselected 15 sexual-target images. These images depicted heterosexual coitus. In order to prevent any social or face processing, in these images neither faces, heads, nor any meaningful background was visible. The visible background was mostly blank in light grey.
After the main experiment, all priming images (EP+, EP−, SP+, SP−, EPC+, EPC−, SPC+, SPC−, and male and female sets) were presented to nine additional raters (three males) in order to validate whether the experimental priming stimuli were perceived according to the intended priming conditions and to exclude the possibility that the control priming stimuli were perceived according to the experimental priming stimuli. To this end, raters were asked to indicate whether a depicted image displayed “economic abundance” (EP+), “economic shortage” (EP−), “friendly looking or caretaking adults” (SP+), “aggressive looking or behaving adults” (SP−), “destruction,” “mourning,” “death,” or “something else.” The experimental priming images were rated in almost all cases according to the stimulus category (confirming ratings per category: EP+ = 99.6%, EP− = 96%, SP+ = 99.2%, and SP− = 96.1%). Similarly, control priming stimuli were rated in 92.4% of the cases as a nonexperimental category (“destruction,” “mourning,” “death,” or “something else”).
Participants
A total of 93 participants participated in the experiment. The study was performed in three parts with three groups (Group I: 30 females; Group II: 34 females; and Group III: 29 males). Groups were matched for age, Group I: M = 23.5, SD = 2.6 years; Group II: M = 23.6, SD = 4.3 years; Group III: M = 25, SD = 4.2 years; one-way analysis of variance (ANOVA) F(2, 92) = 1.45, p = .24, and education level (indicated on a 5-point scale with 1 = no degree and 5 = high school; highest academic degree: Group I: 90% high school, 10% college; Group II: 91% high school, 9% college; Group III: 76% high school, 24% college; Kruskal–Wallis test, p = .17). Oral contraceptives were used by 18 participants of Group I and 19 participants of Group II.
Recruitment was accomplished through announcements on the campus of the Kiel University (Germany) and in the community that asked for participation in a study on the psychological processing of sexual stimuli. The participants of our mixed student-community sample received payment or course credits for their participation. We prescreened the participants by means of an inventory to verify that they were not mentally distressed (Löwe, Spitzer, Zipfel, & Herzog, 2002). By means of a self-constructed questionnaire, we verified that the participants were heterosexual (i.e., Kinsey rating of fantasy and behavior of 0 and 1; Kirk, Bailey, Dunne, & Martin, 2000) and had no history of sexual dysfunction or any other sexual disorder.
Procedure
Participants were assessed individually or in small groups of up to five individuals. Following a general introduction and completion of the questionnaires, participants were seated in front of a computer monitor and keyboard.
One experimental trial consisted of 10 priming pictures (of the same stimulus condition) and a sexual-target picture presented consecutively. Each priming picture was visible for 1.2 s with no gaps in between. Five seconds after the target picture onset, a visual analog scale appeared on the computer screen (ranging from 0% to 100%) and prompted the subject to rate his or her actual sexual arousal by means of a mouse click on the visual analog scale. Three seconds after the mouse click, the sexual-target picture disappeared and was followed by a fixation dot (3 s) indicating the beginning of the following trial. Figure 1 shows the general design of the priming experiment.

Schematic description and time course of the eight priming conditions. The upper four rows represent the four experimental priming conditions that are aimed at depicting a context with relevance for mating preferences. These contexts are absent in the four priming trials of the control conditions in the lower four rows. After target onset, the participants were required to indicate their sexual arousal. The priming images depicted in the figure correspond to the female stimulus set. Please note, for copyright reasons, the images included in this figure are not identical (but similar) to those images that were used in the experiment. EP+ = economic primes positive; EP− = economic primes negative; SP+ = social primes positive; SP− = social primes negative; correspondingly, for the control (C) conditions: EPC+, EPC−, SPC+, and SPC−.
In the first part, the females of Group I were exposed to the priming conditions EP+, EP−, SP+, and SP− (female set). In each trial, 10 primes of one condition were randomly selected (of 21 primes), followed by one of the 15 sexual-target stimuli. Each condition was repeated 15 times, each time with a different target stimulus. Correspondingly, each target picture was shown in every condition once. Priming conditions were presented in random order. In total, one subject was exposed to 15 Trials Per Condition × 4 Conditions = 60 trials, corresponding to about ([12 s + 5 s + 3 s] × 60 =) 20 min’ stimulus exposure time and the additional time the subject needed to rate her actual sexual arousal after each target. In the second part, the females of Group II were exposed to the priming conditions EPC+, EPC−, SPC+, and SPC− (female set). The remaining experimental conditions (number of trials, trial order, stimulus exposure times, target stimuli, and rating of sexual arousal) were identical to the first part. In the third part, the males of Group III were exposed to the priming conditions EP+, EP−, SP+, SP−, EPC+, EPC−, SPC+, and SPC− (male set). In order to shorten the total duration time of the experiment in the third part (given the double number of conditions in comparison to the first and second parts), we made some minor changes to the third part. While the total number of trials per condition, the number of priming stimuli within one trial, the presentation time of each prime (1.2 s), and the intertrial interval (3 s) remained unchanged, the visual analog scale appeared immediately with the sexual-target stimulus and disappeared immediately together with the sexual target after the subject indicated his actual arousal with a mouse click. The male participants in the third part were exposed to a total of 120 trials, corresponding to about ([12 s + 3 s] × 120 =) 30 min’ stimulus exposure time and the additional time the subject needed to rate his actual sexual arousal after each target. The eight priming conditions of Part III were presented in random order. For each participant, mean response values were calculated for each priming condition to which he or she was subjected.
The effects of the experimental conditions on the dependent variable (subjective sexual arousal) were calculated by means of two mixed-model ANOVAs. The first mixed-model ANOVA tested the effect of the EPs relative to the control condition. It was built by entering the following factors: a subject variable, gender (male vs. female) as a between-group factor, prime type (EP vs. control primes [EPC]), prime valence (positive primes [+] vs. negative primes [−]), and a covariable of no interest (total sexual arousal). The second mixed-model ANOVA tested the effect of the SPs relative to the control condition. It was built by entering the following factors: a subject variable, gender (male vs. female) as a between-group factor, prime type (SP vs. control primes [SPC]), prime valence (positive primes [+] vs. negative primes [−]), and a covariable of no interest (total sexual arousal).
Results
Over all conditions, male and female participants were moderately sexually aroused by the sexual targets. The three groups did not differ significantly in their reported sexual arousal, Group I: M = 43.1, SD = 16.8; Group II: M = 40.7, SD = 15; Group III: M = 42.7, SD = 22.8; one-way ANOVA F(2, 92) = .16, p = .85.
Mixed-model ANOVAs revealed a significant main effect of prime valence in both the EP’ analysis, F(1, 235) = 28.3, p < .001, and the SPs’ analysis, F(1, 235) = 70.4, p < .001, indicating that positive prime conditions (EP+, EPC+, SP+, and SPC+) triggered increased sexual arousal relative to the corresponding negative prime conditions (in both males and females). Both mixed-model ANOVAs revealed a two-way interaction between prime valence and prime type, EPs’ ANOVA: F(1, 235) = 8.5, p < .01; SPs’ ANOVA: F(1, 235) = 7.4, p < .01, indicating that the experimental prime conditions (EP+, SP+) were associated with an elevated sexual response relative to the corresponding control conditions (EPC+, SPC+). However, this effect appeared to be driven by an increased response of the female participants, as indicated by a three-way interaction: With regard to the EPs and the corresponding control condition, the mixed-model ANOVA revealed a significant 3-fold interaction among Gender × Prime Type × Prime Valence, F(1, 235) = 7.8, p < .01. The inspection of the corresponding means (Figure 2a) revealed that female sexual arousal was increased in the positive economic priming condition (EP+) relative to the negative economic priming condition (EP−) and that this response difference was not seen in the control conditions (EPC+ vs. EPC−). This differential responding was not seen in the male probands. A similar but even more pronounced effect was found with regard to the SPs: The mixed-model ANOVA revealed a significant 3-fold interaction among Gender × Prime Type × Prime Valence, F(1, 235) = 10.6, p < .001. Again, the corresponding means (Figure 2b) revealed that female but not male sexual arousal was increased by SP+ relative to SP− and that this effect did not happen in the control conditions.

Mean subjective sexual arousal values in the economic (a) and social (b) priming and control conditions of the female and male probands. EP = economic primes; SP = social primes; C = control primes. Error bars correspond to standard errors of mean.
Discussion
We conducted a priming study aimed at drawing inferences about the functional implementation of sex-dimorphic sexual strategies in the human mind. To this end, we tested whether priming stimuli that are nonsexual but relevant for sexual strategies can alter responses to core sexual stimuli (i.e., the functioning of the sexual module). To control for unspecific emotional effects, we additionally used priming stimuli without any relevance for sex-dimorphic sexual strategies but with similar emotional characteristics in terms of valence and arousal. As predicted, we found that the experimental primes (EP+ and SP+) enhanced sexual responses beyond an unspecific effect in females but not in males. Additionally, we found that an emotionally positively valued context (as represented by the EPC+ and SPC+) promoted sexual responses. We found that the effect of these nonspecific primes was similar for males and females.
Consistent with the predictions of the parental investment theory and with many findings about sex-dimorphic mating preferences in humans, here we found that females’ but not males’ sexual responses were particularly enhanced in a context of high economic resources and social attachment. In real life, contextual cues often coincide with sexual stimuli and it is therefore difficult to draw clear conclusions about the way these stimuli are processed. By disentangling contextual stimuli from sexual meaning and by using core sexual stimuli that bear neither contextual information nor facial cues, some inferences about sexual processing can be drawn. Since the EP and SP lacked any sexual cues, it is not likely that the perception of these stimuli activated sexual stimulus processing (i.e., the sexual module). The EP and SP will rather have activated other brain modules designed to process emotions like fear, anger, social affection, or reward including functions of pattern recognition and memory recall that are required to process these stimuli. Most probably, the sexual module was activated in our experiment the moment the sexual target appeared on the screen (and the EP and SP had already disappeared). The enhanced sexual response of females to the core sexual-target stimuli (after being exposed to the SP+ and EP+) cannot be explained by the sexual module as it is currently described. Following the current concept of the sexual module, the response to the sexual-target stimuli would be expected to be similar in the experimental conditions and the control conditions and furthermore expected to be similar for males and females. Our data rather support the two hypotheses we made regarding the functioning of the sexual module: First, that the processing of the sexual module can be modulated by the output of other brain modules; and second, that this modulation can be sex dimorphic. This interpretation is in accordance with the view that brain modules may have inhibitory or enhancing effects on some other modules, thereby allowing the mind to be context sensitive (Barrett & Kurzban, 2006; Buss, 2008, p. 57; Sperber, 2005). We suppose that the brain modules that were activated by the EP+ and SP+ were functionally connected with the sexual module in females more than in males and that these connections provide processing resources that can be used for enhanced sexual responding in case of a succeeding sexual stimulus. The same holds for brain modules activated by EP− and SP− which could be connected by inhibiting connections to the sexual module in females more than in males.
With respect to the control primes, we interpret the enhancing effect of the EPC+ and SPC+ stimuli within the context of affective priming and automatic sexual stimulus processing: Positively valued primes preactivate memory codes of the sexual-target stimuli because they are affectively related (Janssen et al., 2000; Klauer & Musch, 2003). Although the co-occurrence of positive affect and subjective sexual arousal has been shown previously (Peterson & Janssen, 2007), we are not aware of studies to date that demonstrate the enhancing effect of positive affect on sexual arousal, as it was found in the present study. It is of interest that this basic effect is similar for males and females, as it could reflect a “unisex” aspect of the sexual module’s functioning.
Our data suggest that the sexual module’s processing is more controlled by nonsexual brain modules in females than in males, or, stated conversely, that sexual processing is less context-dependent and more stimulus-driven in males than in females. However, there might be contexts that would impact the sexual responses of males and females differently from what was found in the current study. For instance, in the case of long-term mating, males might evaluate complex behavioral aspects of their mates too. Similarly, differing impacts of contextual cues can be also expected in females, depending on whether short-term mating or long-term mating is involved. Effects of the menstrual cycle are also possible. Both genders pursue mixed reproductive strategies and perform cost-benefit analyses regarding whether short- or long-term mating will maximize their reproductive success (Davies & Shackelford, 2008). In our study, we used sexual stimuli that lacked facial cues, aiming to avoid identification of a potential mate in order to keep things as simple as possible and to enhance the potency of the preceding primes. Under these circumstances, without any information regarding long- or short-term mating, females’ sexual responses were found to be more context dependent. Roughly speaking, when everything else is equal, females’ sexual responses tend to be more context dependent (at least with respect to the context we provided with our priming stimuli). This view is in accordance with a recent two-level model of sexual processing that states low-level “straightforward (automatic) matching between the stimulus and an adaptive reaction” more in males, and high-level complex cognitive stimulus processing that “involves extrapolation beyond sensory input” more in females (Toates, 2017).
We only assessed subjective sexual arousal. Therefore, our findings cannot be generalized to genital responses. Genital responding of (heterosexual) females is puzzling, since it is less correlated with subjective sexual arousal (Chivers, Seto, Lalumiere, Laan, & Grimbos, 2010) and because it is less gender specific as it is in males (Chivers, 2017). There is evidence supporting our notion of an increased context dependency in female genital responding. For instance, female orgasm frequency was found to be positive related to the male partner’s family income (Gallup, Ampel, Wedberg, & Pogosjan, 2014). However, there is conflicting evidence as well. In two studies, Chivers and Timmers (2012) and Timmers and Chivers (2017) exposed their participants to auditory narratives of sexual interactions that varied by relationship context (stranger, friend, and long-term relationship). The genital responses of both male and female participants were increased in the stranger and in the long-term relationship context relative to the friend context. In contrast to the present study, Chivers and Timmers did not vary the context independently from the sexual stimulus. It is therefore not possible to rule out the possibility that the participants imagined in case of the friend context less sexually attractive partners than in the other contexts (stranger, long-term relationship). Beside this limitation, Chivers and Timmers findings limit the notion that males’ sexual responding is less context depended as females’. Complex partner characteristics (e.g., the ability to provide resources) might be evaluated by females more than by males and therefore be able to modulate the sexual salience of a potential mate in females more than in males. Such evaluations reflect the sexual strategies of a person. Our findings indicate that these evaluations are realized at the proximate level by a network of varying connections between the sexual module and other brain modules. A certain relationship context, such as the one of “no relationship” (stranger) might be a context that preactivates sexual meaning in males and in females because it fits with the sexual strategies of both males and females (because of the male typical strategy of indiscriminative sex and females’ pursuit for “good genes” in case of an attractive stranger). The central aim of the present study was not to test whether high-order contextual cues modulate sexual responses in females more than in males in general but to provide a more detailed model of how sex-dimorphic mating strategies could be implemented in the human mind.
Recommendations for Future Research
The current paradigm could be used also to study clinical manifestations of human sexuality. Prevalences of sexual dysfunctions are highly sex dimorphic with sexual dysfunctions that are related to a decreased sexual response being elevated in females, like female sexual interest/arousal disorder, genito-pelvic pain/penetration disorder, or female orgasmic disorder. In contrast, sexual dysfunctions that are related to increased sexual response, like premature orgasm or hypersexual disorder are elevated in males (Laumann, Paik, & Rosen, 1999). This sex-dimorphic distribution of hyper- and hyposexual functioning is in accordance with differences in selectivity of mates as predicted by the parental investment theory (note: small differences between the means of male and female sex drive can lead to extended differences in outer parts of the distributions). That is, the elevated female selectivity might be realized at the expense of an elevated number of females suffering from sexual dysfunctions that are related to decrease sexual response. In light of our findings, we would suppose that in hyposexual females the functional connections between brain modules that process EP+ and SP+ and the sexual module would be less active than in sexually functioning females. Thus, after a successful replication of these preliminary findings, the current paradigm could be used to study particular manifestations of female sexuality and thereby enhance our understanding of sexual dysfunctions.
To sum up
Our experimental paradigm appears to be sensitive for sex-typical differences in sexual processing, with females’ sexual responses being more context dependent than males’ sexual responses. Our findings are in accordance with parental investment theory that predicts females’ mating behavior to be more selective than males. Furthermore, this study adds to the findings from numerous field studies in that females have stronger preferences for mates who possess resources and are willing to invest their resources in children. We replicated these findings under restricted laboratory conditions that allowed us, by disentangling nonsexual and sexual meaning, to study how sexual strategies impact sexual responses. Our findings are compatible with the concept of a core unisex sexual module. The activity of this sexual module however is most likely influenced by the output of other nonsexual modules in a sex-dimorphic manner.
Footnotes
Authors’ Note
The study was approved by the Ethics Committee of the Medical Faculty of Kiel University. All participants gave their written informed consent before participating in the experiment.
Acknowledgment
We are grateful to the GETRA society (Gesellschaft der Freunde und Förderer psychotraumatisierter Menschen in Europa) and in particularly to Prof. Dr. Wolf-Dieter Gerber who supported this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
