Abstract
Holistic, integrated, and ongoing post-diagnostic care is essential to address the progressive cognitive and functional decline experienced by people living with dementia, as well as the psychosocial and emotional needs of their care partners. Despite growing recognition of its importance, post-diagnostic care remains fragmented and challenging to access in Australia. This study aimed to examine care partners’ experiences and perceptions of the accessibility and adequacy of post-diagnostic support services in the Australian Capital Territory. An explanatory sequential mixed-methods design was implemented. A cross-sectional survey of care partners (n = 86; 75.6% female) assessed service accessibility, availability, and responsiveness. Subsequently, purposive semi-structured interviews (n = 13; 53.8% female) elicited detailed perspectives on system navigation and lived experiences. Qualitative data underwent inductive content analysis and were triangulated with quantitative findings. Nearly 70% (n = 50) of care partners reported difficulties navigating dementia care and support with the person living with dementia. Reported barriers included extended wait times, funding limitations, service inflexibility, and convoluted referral pathways, with service adequacy diminishing as dementia progressed. While early-stage interventions were valued, participants highlighted inconsistent availability and inadequate flexibility of services to adapt to evolving needs. The findings align with the objectives of Australia’s National Dementia Action Plan 2024-34, highlighting the need for affordable, integrated and person-centred care models, strengthened referral and care navigation pathways, supported by ongoing workforce development.
Background
Dementia represents a major global public health challenge, with prevalence projected to increase substantially, intensifying pressure on health and social care systems (Nichols et al., 2022; World Health Organization, 2021). In Australia, the number of people living with dementia is expected to more than double by 2058, rising from approximately 433,000 to nearly 849,300, largely driven by population ageing (Australian Institute of Health and Welfare, 2024). This demographic shift poses challenges for care partners, over half of whom provide more than 60 hours of care weekly (Australian Institute of Health and Welfare, 2024), resulting in considerable emotional, physical, and financial burden, including increased psychological distress and social isolation (Kishita et al., 2020; Lindeza et al., 2024; Livingston et al., 2024). Fragmented and inadequate formal support services further exacerbate these challenges (Sorrentino et al., 2025).
The progressive cognitive, functional, and behavioural decline associated with dementia requires a holistic approach integrating pharmacological and non-pharmacological interventions, along with coordinated and responsive care (Bamford et al., 2021; Mansfield et al., 2022). Effective care coordination and access to dementia-specific services are central to maintaining quality of life and functional independence for people living with dementia, while mitigating care partner burden (Lee et al., 2025; Quinn et al., 2022). In this context, post-diagnostic support should be inclusive and culturally responsive, recognising the diverse needs and contexts of people living with dementia and their care partners (World Health Organization, 2021). A partnership-oriented model of care, grounded in shared decision-making, adaptive goal-setting, and tailored access to services, is therefore essential to ensure that care remains responsive to evolving dyadic preferences (Aworinde et al., 2022; World Health Organization, 2021). Despite the recognised importance of such support, access remains constrained by individual factors, including low awareness, stigma, and care partner burden, and by systemic barriers, such as fragmented pathways, inconsistent services, workforce shortages, and limited specialised care (Kårelind et al., 2024; Pavković et al., 2025). These barriers leave care partners and people living with dementia inadequately supported, highlighting a mismatch between care needs and service provision (Robertson et al., 2022). Notably, empirical evidence regarding how support services are perceived and experienced by people living with dementia and their care partners remains limited.
The Australian Capital Territory, home to approximately 480,000 residents, is served by two public hospitals that also provide care to neighbouring regions of New South Wales. Previous research has identified gaps in dementia-specific services in the Australian Capital Territory, including workforce gaps in dementia education and training, inadequate funding, and insufficient respite and community-based support (D'Cunha et al., 2025; Tabatabaei-Jafari et al., 2024). Although situated within a regional context, these challenges reflect global difficulties in delivering sustainable, coordinated, and person-centred post-diagnostic support (King et al., 2024; Lin et al., 2025). Building on this evidence, this study examined care partners’ perceptions of post-diagnostic support in the Australian Capital Territory to inform policy and practice aimed at strengthening support for people affected by dementia.
Materials and Methods
Study Design
This study adopted an explanatory sequential mixed-methods design (Creswell et al., 2011; Fetters et al., 2013) to explore care partners’ perceptions of post-diagnostic support in the Australian Capital Territory. Quantitative data captured service utilisation patterns, referral processes, and access timelines, which were then integrated with qualitative data to explore contextual influences on service engagement. Qualitative reporting followed the Consolidated Criteria for Reporting Qualitative Research (COREQ) (Tong et al., 2007). Ethics approval was granted by the University of Canberra Human Research Ethics Committee (HREC-13385). Data collection took place between July 2024 and March 2025.
Quantitative Phase: Cross-Sectional Survey
Participants and Recruitment
Eligible participants were current or recent (within five years) care partners of people living with dementia residing in the Australian Capital Territory or neighbouring New South Wales regions, including those who had relocated after accessing services in the Australian Capital Territory. Recruitment occurred through professional networks, social media, community newsletters, and snowball sampling. The target population was an estimated 3,820 care partners in the Australian Capital Territory region in 2022 (AIHW, 2024).
Survey
The survey was informed by findings from previous work in the Australian Capital Territory on dementia-specific services (D'Cunha et al., 2025; Tabatabaei-Jafari et al., 2024). To assess clarity and validity, survey development occurred in three stages: (1) a rapid review of peer-reviewed and grey literature to map existing post-diagnostic pathways and identify service gaps within the Australian Capital Territory dementia care system; (2) consultation with a multidisciplinary panel (dementia specialists, dietitian, occupational therapist) to identify domains of accessibility, availability, and responsiveness; and (3) expert review and pilot testing by academics with combined dementia expertise and lived experience. The 69-item survey assessed socio-demographics, service access, finances, and barriers using Likert-scale items, multiple-choice, open-text, matrix tables, and yes/no responses. Skip logic was used to ensure relevance of exposure. The survey was administered both online via Qualtrics (Qualtrics LLC, Provo, UT, USA) and in paper format. Paper surveys were distributed through dementia support group meetings and mailed to retirement and independent-living communities, while the online link was disseminated via organisational newsletters and social media platforms.
Qualitative Phase: Semi-structured Interviews
Sample
Upon completing the survey, participants were asked whether they were interested in participating in a follow-up semi-structured interview. Consenting participants were purposively sampled to ensure diversity in demographics, care contexts, and satisfaction with post-diagnostic support. This strategy was employed to capture a broad range of perspectives, identifying factors influencing perceived adequacy and accessibility of services, and maximise informational richness (Patton, 2015). The final sample size was guided by the principle of information power rather than the absolute number of participants (Malterud et al., 2016).
Interview Procedures
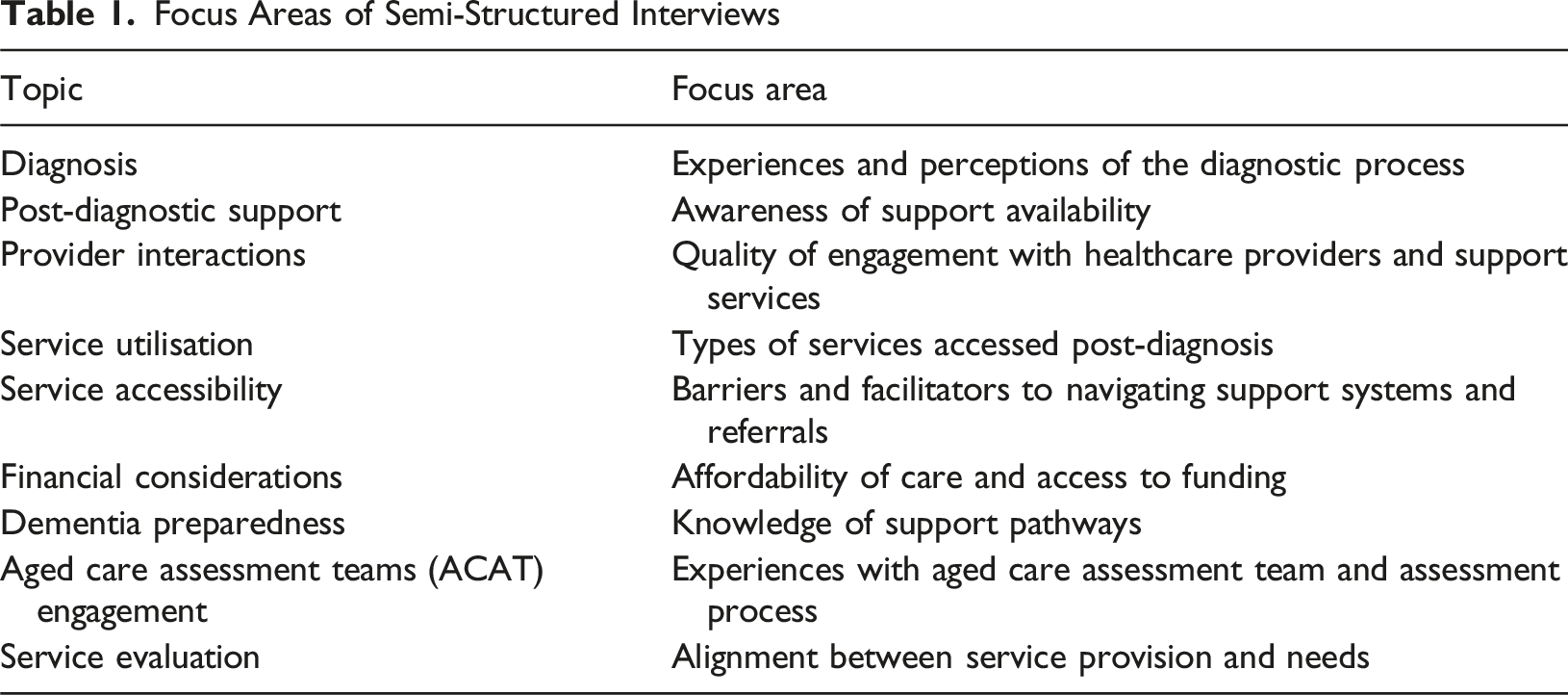
Focus Areas of Semi-Structured Interviews
Data Analysis
Survey data were checked for completeness and exported to IBM SPSS Statistics (v30.0). Participants completing at least one item beyond the socio-demographic section were included in the analytic sample. Descriptive analyses, including means and standard deviations for continuous variables and frequencies and percentages for categorical variables, were performed. Given variable response rates and contingent items, percentages reflect the number of eligible respondents. Open-ended survey responses were systematically coded and analysed using Inductive Content Analysis, a qualitative method well-suited to health research and emerging fields for generating practical, context-specific insights (Vears & Gillam, 2022).
All interviews were audio-recorded, transcribed verbatim, and anonymised using numerical identifiers and gender markers (F/M) for contextual clarity. Inductive Content Analysis guided qualitative analysis to identify themes through iterative coding, constant comparison, and data reduction, enabling an in-depth understanding of participants’ experiences. Two researchers (A.L. and N.M.D.) independently coded the data, refining categories through iterative analysis of response depth and coherence to develop clear, narrative-driven themes. Final themes were reviewed and validated through discussion with senior qualitative researchers (S.I. and J.K.).
Integration of Quantitative and Qualitative Data
Quantitative survey findings informed both interview questions and participant selection, while qualitative data were used to contextualise and extend survey patterns. Data integration was guided by triangulation and complementarity, enabling systematic comparison and synthesis of quantitative and qualitative findings (O'Cathain et al., 2010). Survey analysis identified service utilisation trends and barriers, while interviews provided nuanced insights into care partners’ lived experiences. Themes derived from qualitative analysis were examined alongside survey results to assess convergence, divergence, and elaboration. This mixed-methods integration facilitated a comprehensive understanding of service access, highlighting areas where qualitative insights clarified, reinforced, or contrasted with quantitative patterns.
Results
Survey Findings
Of the 109 care partners who started the survey, 86 (78.9%) progressed beyond the socio-demographic section, and 78 (71.6%) completed the survey in its entirety. Most responses were submitted online (n = 67), with a median completion time of 25 min.
Sociodemographic Characteristics of Survey Respondents (n = 86)

Services Utilisation and Timeliness of Access Following Referral
People Living with Dementia
Among 85 respondents, 71.8% (n = 61) reported that the person living with dementia had been referred to a health or social care service, with 95.1% (n = 58) of these subsequently accessing the service. Among those not referred (28.2%; n = 24), only 12.5% (n = 3) independently sought such services. As shown in Figure 1, most services were accessed within three months of referral, with a maximum wait of six months, except for respite care, which experienced longer delays. The most frequently accessed service was personal care support (e.g., home cleaning, gardening, meal preparation, and assistance with daily activities), used by 62.3% (n = 38), followed by driving assessment (55.7%; n = 34), and both respite care and allied health services (each 50.8%; n = 31). Utilisation of support services by people living with dementia and time from referral to access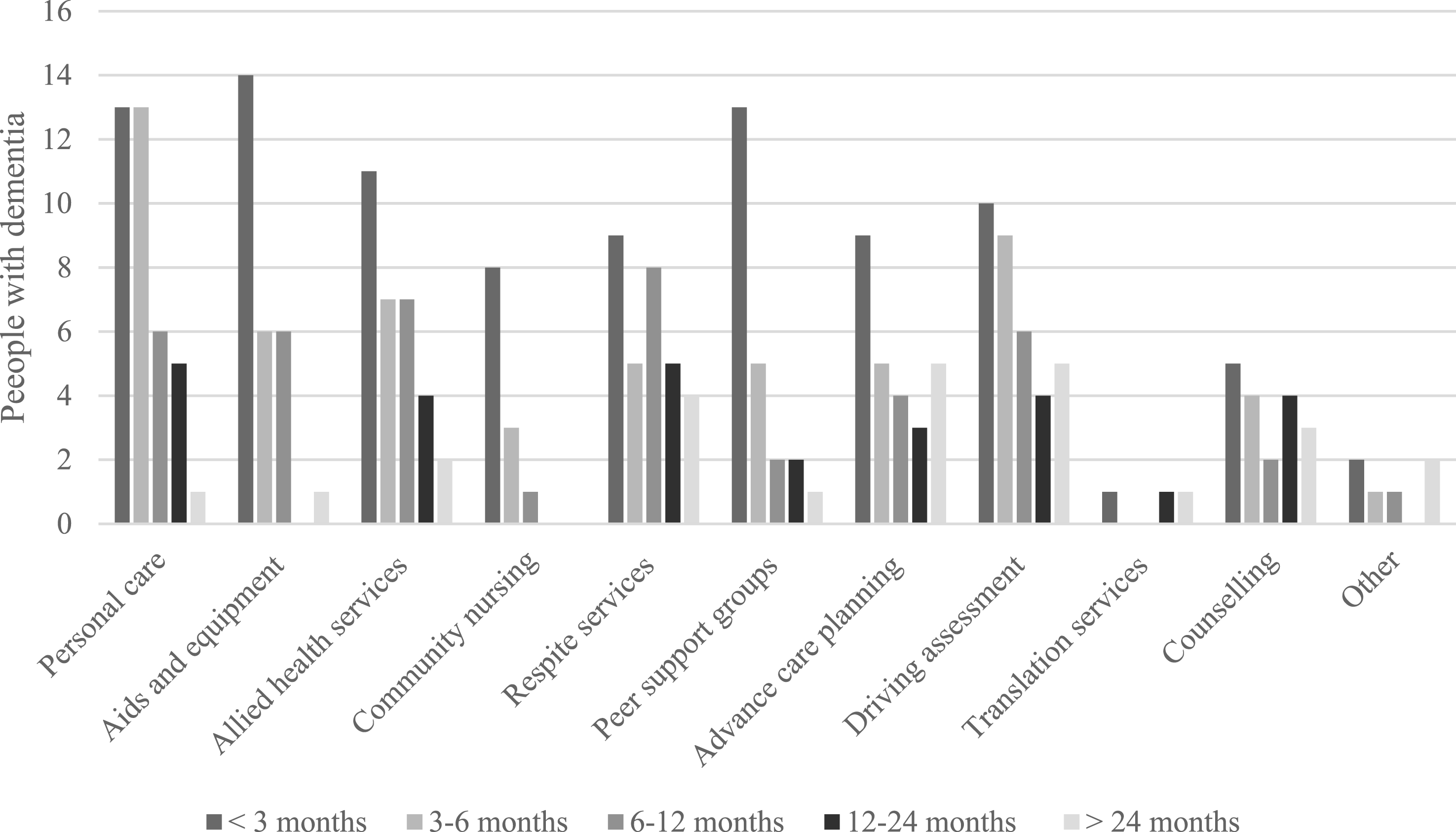
Care Partners
Of 84 respondents, 56.0% (n = 47) reported receiving a referral to support services, and 89.4% (n = 42) subsequently accessed at least one service. Among those not referred (42.9%; n = 36), only 11.1% (n = 4) independently accessed services. One respondent could not recall whether they had received a referral but had accessed services and was therefore included in service access analyses. Overall, 56.0% (n = 47) of respondents accessed at least one service. Among service users, 61.7% (n = 29) accessed counselling and psychological support, and 46.8% (n = 22) joined peer support groups. Use of other services was limited, including community transport (19.6%), legal services (12.8%), financial services (12.8%), other service types (10.6%), or translation services (6.4%). Most care partners accessed these services within six months of referral (Figure 2). Utilisation of support services by care partners and time from referral to access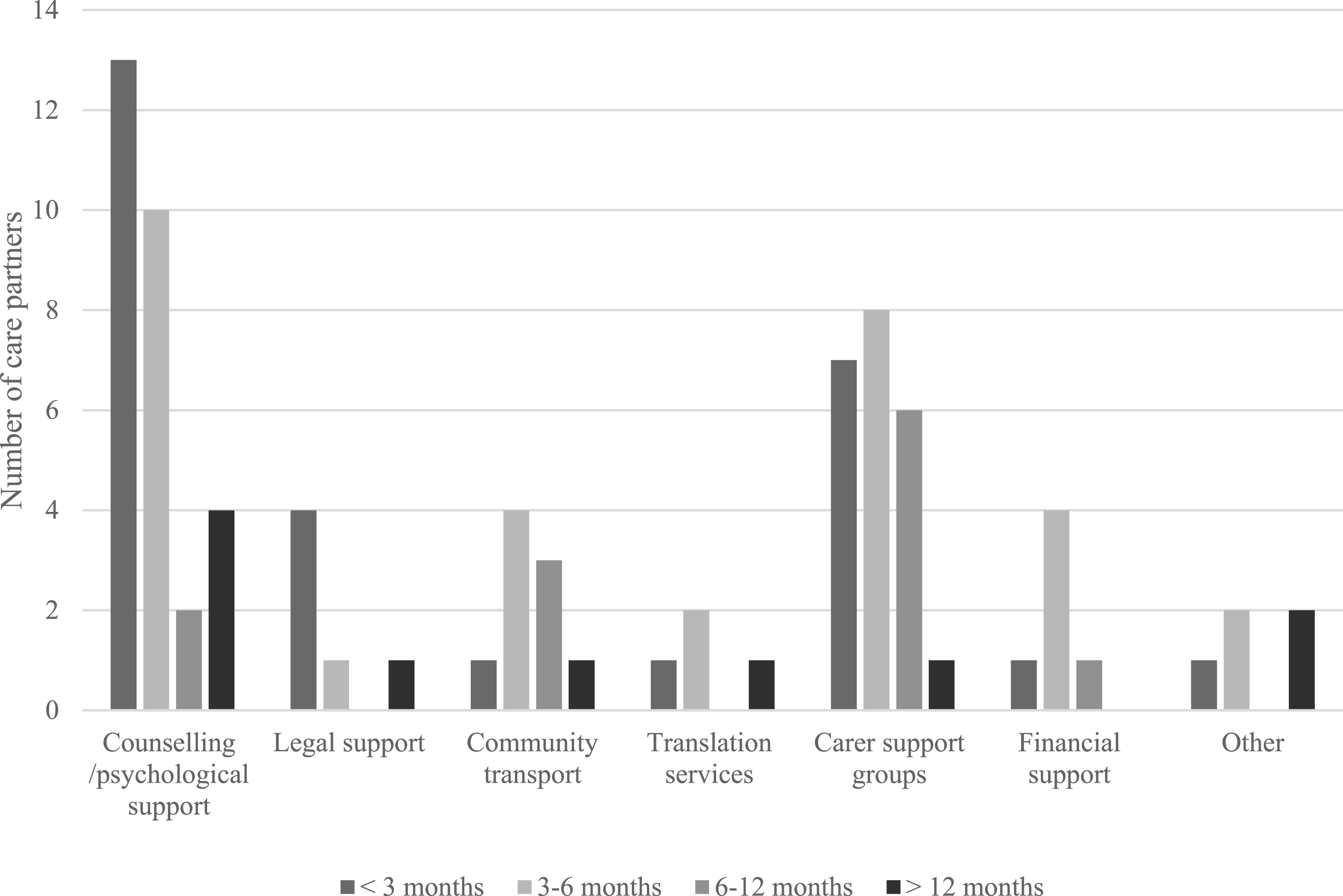
Mixed-Methods Integration
Integrated Quantitative and Qualitative Findings on Barriers and Support Needs Among Dementia Care Partners

Theme 1: Barriers to Service Access
Diagnostic Delays
Among 86 survey respondents, 70.9% (n = 61) received a diagnosis within one year of the initial consultation with a specialist (Table 2). Care partners described the diagnostic process as complex and protracted, involving multiple professionals: (M2, F4, M5, F6, F8): “It's just the whole process, sort of going to the GP, getting the geriatrician sort of appointment, sort of the length of time taken there, and then the neuropsychologist coming in. […] that whole process probably takes about 12 months. (F8)
These delays were perceived to hinder timely access to care and limit opportunities for early care planning (M2, F4, M5): “It's in retrospect, probably two years [to get a diagnosis] […] and we could have got onto it earlier. We could have taken some of those early-stage things. (M5)
Financial Barriers and Funding Constraints
Financial barriers were widespread among participants. Support services were considered unaffordable by 75% (n = 57) of 76 respondents (Table 3), with out-of-pocket costs cited by 20 of 59 respondents (33.9%). Interviews identified respite care as expensive, particularly without formal funding (M2, M5, F8, M11): “I wanted some respite, and I don't get any funding for that. It's quite expensive. Yeah, you want to go, and I think it's $60 an hour or something.” (M2)
Affordability concerns extended to services supporting physical activity and wellbeing. Care partners described limited access to affordable, personalised options F1, F7, F8, M11): “I tried to find any kind of support where he could have one-on-one, somebody who would come take him to the gym, stay with him, help him through it, or come to the house […]. So, if there was any way to keep somebody's fitness at a good level without having to outlay extraordinarily large amounts of money, that would be wonderful. (F7)
Funding limitations were a recurrent challenge. Fifty-two of 84 care partners (61.9%) did not receive payments or financial assistance (Table 3), and 32 of 63 (50.8%) experienced challenges accessing funding. Even when approved, allocations were perceived as inadequate, with higher-level packages described as slow and unresponsive (F1, M11): “You get high priority for the [Level 4 Home Care Package] because you've maxed out the [Level 2 Home Care Package], and clearly you need the extra work. […] But now we're waiting for the [Level 4 Home Care Package], […], they're not organising that quickly enough.” (F1)
Government funding was reported for the person living with dementia by 67.1% (n = 57) of 85 respondents, while the remaining 32.9% (n = 28) indicated that no such funding had been received (Table 3). Among those receiving funding, Home Care Packages (n = 30; 52.6%) and the Commonwealth Home Support Program (n = 19; 33.3%) were most commonly reported, whereas few respondents reported National Disability Insurance Scheme (NDIS) funding (n = 4; 7.0%). Participants described funding as inflexible (F1, M2, M5, F6, F7, F8, M11), resulting in rationed care hours and substantial out-of-pocket costs (F1, M11): “I have to find someone to come and stay with [her]. I just don't think it's worthwhile for the cost involved. […] Because I've got to take it out of my care hours. They cost about 90 plus dollars an hour, and I've already fully utilised our level four package. In fact, I pay over $1,000 a month more, simply to buy more hours.” (M11)
Service models further failed to accommodate dementia care’s unpredictable, ongoing nature, making packages impractical for emergencies (F6, F7): “I’ve never accessed their packages, because none of them were going to give me the help any time of the day or night that I needed. It would only be appointments at their specific times within their working hours, and that doesn't work if you have someone who falls over in the middle of the night.” (F6)
Extended Waiting times
Waiting times were commonly reported as a barrier across the dementia care pathway, with 31.3% of 67 respondents (n = 21) identifying delays as a significant impediment to access. Within three months post-diagnosis, 52.2% of 67 respondents (n = 35) reported consulting a GP, 40.5% of 74 respondents (n = 30) saw a geriatrician, and only 10.6% of 47 respondents (n = 5) accessed an allied health professional (Table 3). Care partners noted these waits resulted in missed opportunities for timely intervention (F7, F10): “A physio is a long wait through the public health system […]. If we could have done something about that a few years ago, when my husband was able to engage in doing the rehab, it would have been more beneficial. (F7)
Similarly, 36.1% (n = 22) of 61 respondents waited more than six months for an Aged Care Assessment Team review (Table 3), and among 56 respondents, 33.9% (n = 19) experienced funding approval delays exceeding six months, with 17.8% (n = 10) receiving no approval. While some care partners (F3, F7, F6, F10) noted hospitalisation unexpectedly facilitated access by fast-tracking aged care eligibility assessments during inpatient admissions, most interview accounts described administrative errors, lengthy application processes, and limited specialist availability as compounding these systemic delays (F1, M2, M5, F7, F10, M11, M12): “That's taken us ages to find out, work our way through that, and we found out that we, probably six or eight months ago, weren't even on the list [for an Aged Care Assessment Team review] even though I had spoken to someone on the phone about it.” (M2)
Reluctance of the Person Living with Dementia
Interviews highlighted reluctance among people living with dementia to engage with available services, frequently leading to declined support. Several care partners (F1, F6, F7, F10, M12, M13) described refusals, with one noting this discouraged subsequent help-seeking: “It doesn't matter what information I get. If [she] won't cooperate, it's pointless. She won't go anywhere for respite, won't let me have time off, won't accept a carer.” (M13)
Even when initial engagement occurred, participation may be short-lived or discontinued unexpectedly (F7, F10, M13): “Post her hospital visit, she visited a rehabilitation home […]. They had physios and occupational therapists and people talking to them and having proper meals […], it was all very lovely. And it was fantastic for myself as well. But then one day, she said, I'm done.” (F10)
Theme 2: Navigating the System
Mismatch Between Expectations and Experiences
Difficulties navigating the aged care system were commonly reported, with 50 of 72 respondents (69.4%) identifying it as a considerable barrier to accessing appropriate support (Figure 3). Among 81 respondents, 51.8% (n = 42) reported a referral to care planning services. Only 41.0% (n = 34) of 83 respondents believed they had received adequate information to facilitate service access (Table 3). During interviews, participants reported limited practical assistance, an absence of proactive guidance, and challenges navigating complex health and social care systems (M2, F8, F10, M11, M13): “I would have thought we would’ve been given more help in terms of trying to work our way through the system. […]. It's so hard to navigate.” (M2) Perceived barriers to accessing support services reported by care partners in the Australian Capital Territory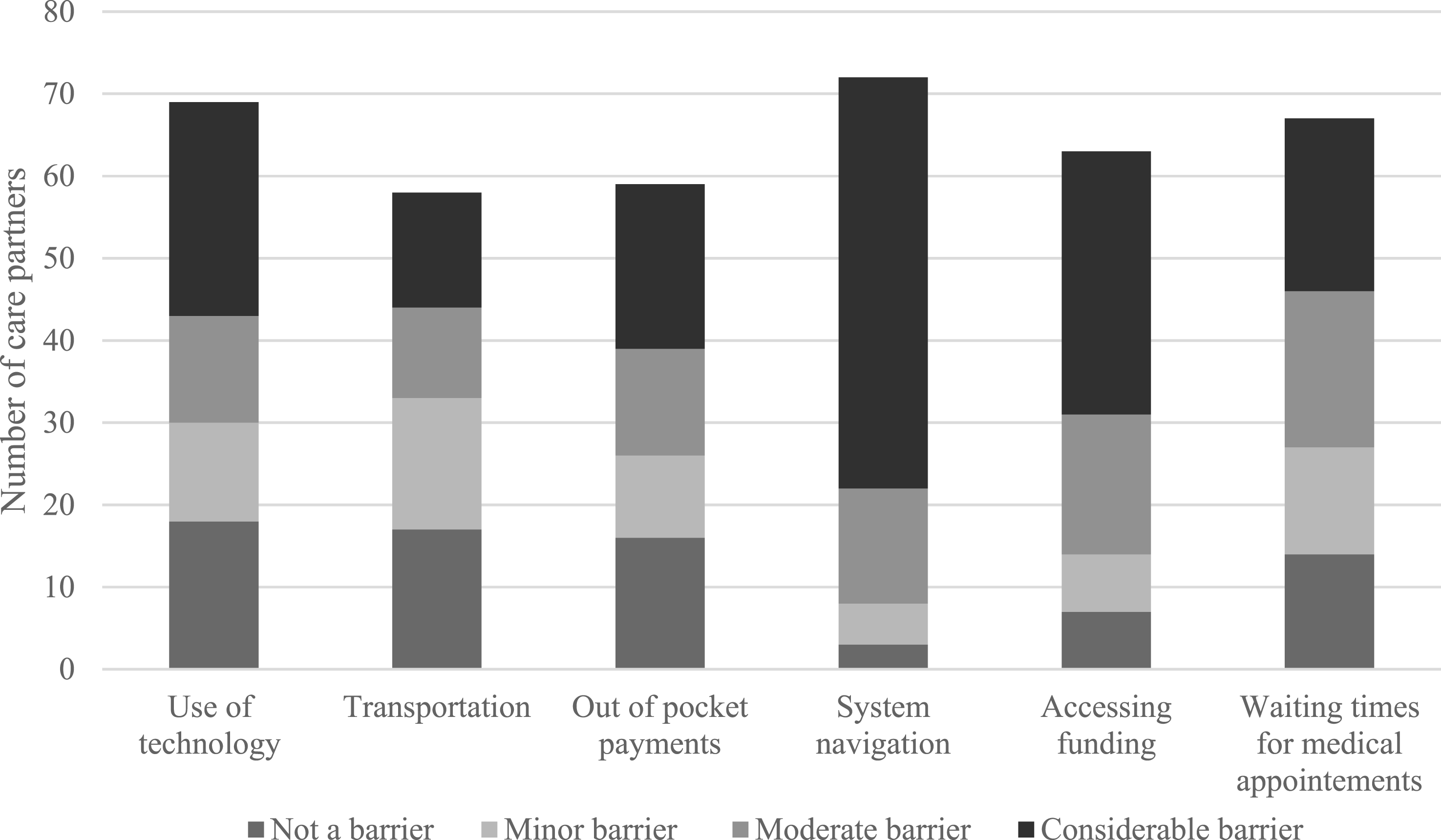
Participants also noted a need for more proactive engagement from healthcare professionals, particularly GPs (F3, F4, F7, M12): “The GP should be more aware, and instead of waiting for me to ask, they should be more proactive. (F4)
Absence of Service Coordination and Fragmentation of Care Delivery
Survey findings revealed that 25 of 56 respondents (44.6%) were dissatisfied with the level of support coordination for managing their funding packages (Table 3). This lack of coordination extended beyond individual experiences, as interview data revealed that navigation challenges affected the entire health and social care system (F1, M2, F3, M5, F8, M9, F10, M11), with participants describing it as fragmented, overwhelming, and lacking both guidance and a central point of contact (F1, M2, F8): “…It's like all these places to go to and no way to sort of channel it. […] that's the issue for me […]. I do think that it's the coordination of all of those things. It's just about how we are guided along the way, and how they interact with one another. (F8)
Communication gaps between services further exacerbated delays and confusion (M5, F10) “It did take another 12 months for the next appointment […]. [Service provider] told us if you've got questions, you can ask her GP. But her GP says, oh, didn't she have an aged care assessment? Probably just wait for that assessment.” (F10)
One participant observed that siloed services overlooked the interdependent needs of the care partner and person living with dementia: “The structure separates [her] from me […]. But in reality, dementia and caring is happening at the same time, as a couple, but they decouple-ise you […] because it's siloed, and that causes a lot of problems. (M9)
Theme 3: Support Needs Across Dementia Stages
Satisfaction with Early Stages of Support
In response to an open-ended survey question on support satisfaction, 27 of 74 respondents reported positive experiences with dementia organisations and support programs, with ten specifically highlighting their effectiveness during early dementia stages: “[Support organisation] was very good for the initial stages. “Some of the support has been fantastic for individuals in early-mid stages of dementia
Care partners reinforced these findings during interviews, describing initial supports as high quality and beneficial (F1, F4, M5, F6, F7, F8, F10, M12, M13): “The information they started to give us through the post-diagnosis support appointments - every time we went to those - he was quite happy to be there. My husband would discuss things. She [support worker] would say, now, this is something maybe you would like to look at. And I would go away, and I would look at it. (F7)
One support organisation was commended for its day respite and educational programs (F1, F4, F6, F8, M12), which promoted engagement while offering respite and training for care partners: “I was put on to them fairly early, and they had a day respite program that suited him really well. (F6)
Early access to personal care services was similarly identified as critical for maintaining independence, particularly among people living with dementia residing alone: “So, it's hard to know, because everything is important. But it was to make sure that Mum was looking after herself. So, getting that personal care, I think, was really useful. Getting someone to get her medications, because she lives on her own. (F1)
However, several interviewees reported a decline in the intensity and relevance of support over time (M5, F10, M11): “Initially, they were really good. You know, they did online courses and webinars, and there was a dementia talk online, but they've just faded into …. They send me emails now, but it's like a lunch here or a Christmas party, you know, but no, no real physical talks or help.” (F10)
Later Stages: Inadequate Support and Lack of Respite
As care needs intensified, service adequacy and flexibility often failed to meet families’ demands. Among 52 respondents, 53.8% (n = 28) were uncertain whether available services met their needs, while 28.9% (n = 15) considered them inadequate. Only 9 of 52 respondents (17.3%) considered existing support adequate for optimal care. Reflecting these concerns, 69.7% (n = 46) of 66 care partners identified a need for additional support (Table 3). Qualitative accounts mirrored these findings, describing limited adaptability in service responses and difficulties obtaining timely or appropriate assistance (F1, M5, F6, F7, F8, M9, F10, M11), even among those receiving high-level packages (F1, F6): “He couldn't continue to go to that social respite day program […], he just couldn't mobilise very well. […] we needed something that we could call on at any time […]. I’ve never accessed their packages [Government organisation], because none of them were going to give me the help any time of the day or night that I needed.” (F6)
A lack of individualised support also emerged, with 60.8% (n = 31) of 51 respondents reporting insufficient assistance with everyday living, mobility, and engagement (Table 3). Qualitative findings reinforced this, with care partners describing systemic rigidity and limited capacity to tailor services to changing needs (F1, F7, F8): “You're basically locked into a particular service provider who will give you what they have.” (F1)
Similarly, 48.1% (n = 39) of 81 respondents indicated limited care planning options, underscoring gaps in collaborative, adaptive, and continuous care approaches. Qualitative data supported this concern, revealing participants’ desire for ongoing, flexible planning that adjusted to changing circumstances. Service misalignment was particularly evident in allied health provision, with participants citing unmet functional needs relating to exercise and stability (F6, F8, M11): “Well, they [physiotherapists] weren't doing what I thought they should do. It's, I think they [people living with dementia] need more exercise rather than physio - you want more stability, and exercise flexibility.” (M11)
System-level constraints further undermined personalisation, with participants reporting inconsistent engagement and insufficient meaningful activity. Poor stimulation and social interaction were linked to distress and disengagement (F3, M13): “I said, look, if I'm going to be going off talking to these other people, you must keep her occupied. You must play games with her. You must talk to her. […]. They didn't engage her, and they just left her out there.” (M13)
Respite care also reflected this lack of adaptability. While 36.1% (n = 31) of 86 respondents accessed respite, many found it inflexible or unsuitable for higher care needs (M5, F8, M9, M10, M11, M13): “Speaking with them [Service provider], when it became obvious that they'd have to have someone really keeping an eye on [her] all the time and doing this sort of stuff… They said, no, um, probably only on a Friday.” (M5)
These limitations collectively shaped care trajectories, with many care partners identifying residential care as the only viable option when home-based care became unsustainable (F1, M5, F7, M9): “But then you realise, […] no, there's really nothing, and that's all. […] They've got to go into residential aged care. That's really where they can be looked after when they get to the advanced or severe stage.” (M5)
Staffing Turnover and Continuity of Care
High staff turnover undermined the stability and person-centredness of dementia care, increasing care partner involvement (F1, M5, M9, M11). Participants described this as detrimental to trust, continuity, and quality of care due to poor understanding of individual preferences, routines, and support needs (F1, M9, M11): “But this one in [Service provider], the person running it knows, knows mum, has worked with her, knows the issues […]. That's not the case for a lot of people. They have a lot of trouble with different support providers because they don't turn up, […] and they keep changing the worker. So, you don't have that, you don't have that continuity.” (F1)
To mitigate these challenges, some adopted proactive measures, such as retaining the same provider despite staff changes (M11) or arranging for their personal support worker to supplement formal services (M5): “I actually got one of our support ladies to go with [her] to that [Service provider], and which the [staff] appreciated too, because they another person there [with high care needs]. (M5)
Theme 4: The Critical Role of Peer Support
Among 47 care partners (56.0%) accessing support, counselling was the most commonly used (61.7%, n = 29), followed by peer support (46.8%, n = 22) (Figure 2). Of 50 respondents evaluating the service’s impact on well-being, most were uncertain (36.0%; n = 18) or reported no perceived benefit (26.0%; n = 13).
Despite mixed quantitative findings, qualitative data highlighted peer support as particularly valuable. Care partners described peer networks as essential sources of emotional, informational, and practical support (F1, M2, F4, F7, F8, M11, M12, M13), particularly for alleviating the emotional strain of providing care (M2, F4, F7, F8): “[Support] organisations have been really good and really helpful. So, I've got myself into a carer support group. Yeah, so, normally they're my sounding board for things, because they're dealing with their own partners themselves who have dementia.” (F8)
Peer support groups also functioned as key facilitators of system navigation, enhancing awareness of available services and providing practical guidance on access pathways through peer exchange (F1, M2, F8): But, you know, having the group from [Support program], being able to talk to them about what they have and what [services] they use, that does help.” (F1)
These interactions further enabled experience-sharing, strategy comparison, and collaborative problem-solving (F1, M2, F8, M11, M12, M13): “That [Service provider] has been great, […], the carers were able to compare notes, swap stories, tell them what worked, what didn't work, and we learned a hell of a lot between us.” (M13)
Peer support was also considered beneficial for people living with dementia by care partners, fostering acceptance and a sense of normalcy through connection with others facing similar challenges (F4, F7, M12): “So, we went to this [Service provider], we found that people were having the same problems [as] her. […] So, yeah, it really helped with accepting that she wasn't alone in this world.” (M12)
Discussion
Persistent difficulties in accessing and navigating post-diagnostic support within the Australian Capital Territory reflect systemic fragmentation and unmet care needs. Care partners described protracted waiting times, limited funding, and complex referral pathways that collectively constrained timely access to support and compromised community living for people living with dementia. These findings align with international evidence showing similar systemic barriers across the United States, Europe, and Asia, indicating that challenges observed in the Australian Capital Territory are emblematic of globally recognised issues in dementia care (Jeon et al., 2024; Kern et al., 2024; Sorrentino et al., 2025). This study presents findings from care partners in an Australian city, with insights that may apply to diverse health systems seeking to strengthen integrated post-diagnostic support throughout the dementia care continuum.
In this study, barriers to the delivery of dementia care were reported before diagnosis, with care partners reporting limited guidance and assistance throughout the diagnostic process. Delays in diagnosis hinder timely service engagement and reduce opportunities for early intervention, both of which are critical for effective care planning (Molvik et al., 2025). In our sample, the median interval from referral to specialist consultation and subsequent formal diagnosis was 5.8 months, exceeding the national median of 4.2 months (Lin et al., 2024). In an international context, a recent meta-analysis reported a median interval of 3.5 years from symptom onset to formal diagnosis (Kusoro et al., 2025), underscoring broader systemic inefficiencies that exacerbate barriers to care (Giebel, 2024; Kusoro et al., 2025). Post-diagnosis, gaps in support remained evident: one-third of people living with dementia and almost half of care partners in our sample did not receive referrals to post-diagnostic support services. The absence of onward referral restricts access to timely guidance and early interventions, leading to suboptimal medical and social support that compromises independence and quality of life for people living with dementia (D'Cunha et al., 2025; Pimouguet et al., 2016). Moreover, care partners may be left particularly vulnerable to social isolation, psychological distress, and unmet needs (Duplantier & Williamson, 2023; Lindeza et al., 2024). Where referrals were provided, however, service uptake was high, in contrast to findings from an Australian qualitative study in which care partners were less likely to act on referrals (Pavković et al., 2025). In the Australian Capital Territory, initiatives such as the Sustainable Personalised Interventions for Cognition, Care, and Engagement (SPICE) Program have contributed to modest improvements in referral activity, reflecting growing recognition of and engagement with dementia rehabilitation (Gibson et al., 2025). Nevertheless, variability in referral patterns persists, highlighting systemic barriers that limit timely and consistent access to post-diagnostic support for both people living with dementia and their care partners.
Financial and administrative constraints negatively affected service uptake. Access to government-funded support packages, used by approximately two-thirds of people living with dementia, was frequently delayed by protracted assessment procedures, with waiting times reported as long as twelve months. These protracted processes, compounded by extended delays in accessing allied health professionals, restrict opportunities for early therapeutic and rehabilitative interventions (Layton et al., 2024). Despite strong evidence supporting their effectiveness, allied health services remain underutilised due to restrictive funding arrangements, fragmented care models with high administrative burdens, and inadequate integrated care approaches (Robson et al., 2025). Financial inaccessibility further exacerbated these challenges, with approximately three-quarters of care partners perceiving support services, particularly respite care, as unaffordable despite formal funding. The high cost of care, combined with delays in subsidy approvals, intensifies care partner burden and deters sustained service engagement, particularly among socioeconomically vulnerable families (Ramakrishnan & Malhotra, 2025). For people living with young onset dementia, participation in and uptake of services is similarly constrained by rigid eligibility criteria and complex administrative processes associated with National Disability Insurance Scheme funding applications (Loi et al., 2023).
Participants reported significant challenges in care coordination arising from fragmented and siloed service delivery, most notably the absence of a central point of contact. Care partners described dementia care as an inherently dyadic experience encompassing both the person living with dementia and the care partner. However, services within the Australian Capital Territory were perceived as fragmented and insufficiently equipped to address the holistic, interdependent needs of these dyads. These perceptions are consistent with research examining the views of health and aged care professionals in the Australian Capital Territory, which found that fragmented service delivery contributes to delays in care, confusion, and reduced access to essential interventions, including rehabilitation, respite services, and personalised care planning (D'Cunha et al., 2025). Similarly, Brookman et al. (2025) demonstrated that fragmented post-diagnostic support in Australia generates bureaucratic delays and information gaps that disrupt care continuity and undermine care quality by delaying access to essential assistive equipment and intensifying emotional distress, particularly among care partners from diverse backgrounds or with limited digital literacy. Collectively, these findings align with international evidence from the Western Pacific region and the United States, highlighting how under-resourced diagnostic pathways, siloed service structures, and inadequate care coordination continue to impede the delivery of sustainable, person-centred dementia care (Jeon et al., 2024; Kern et al., 2024).
Despite these systemic limitations, care partners in the present study initially reported high satisfaction with available support services, particularly community-based, non-clinical interventions such as informational resources, guidance, and personal care, that helped sustain independence and engagement. These early benefits align with international evidence indicating such supports can enhance quality of life and delay the need for intensive interventions, even within fragmented systems (Conlon et al., 2026; Harding et al., 2023; Yuan et al., 2025). However, as dementia progressed, perceived support adequacy and relevance declined, consistent with broader evidence that services fail to meet increasingly complex care needs (Brookman et al., 2025; Pavković et al., 2025). This gap may stem from the heterogeneity of dementia trajectories (Whitlatch & Orsulic-Jeras, 2018), limited forward planning, and inadequate anticipation of evolving needs (Saragosa et al., 2024; Wheatley et al., 2021), as well as limited specialised services (Tabatabaei-Jafari et al., 2024). Care partners also described inflexible service models and rigid funding arrangements that failed to adapt to their needs. This is consistent with Australian evidence linking such frameworks to constrained respite, inadequate crisis responses, and limited tailored supports (Engel et al., 2022; Hill, 2022). The lack of individualised care planning ultimately led some families to view residential care as the only viable option. Notably, peer support networks emerged as a critical source of emotional support, practical guidance, and connection, reinforcing previous findings (Pavković et al., 2025; Sullivan et al., 2022).
The findings of this study highlight the importance of person-centred care and emphasise the urgent need for policy measures that strengthen referral pathways, integrate dementia-specific education within primary care, and enhance cross-sector coordination (Gibson et al., 2025; Prince et al., 2016). Addressing these system-level barriers is essential to ensuring continuity of care and sustained support across the dementia trajectory, directly aligning with the strategic priorities outlined in Australia’s National Dementia Action Plan (NDAP) 2024-2034 and the World Health Organization’s Global Action Plan on the Public Health Response to Dementia 2017-2025 (Australian Government, 2024; World Health Organization, 2021). Flexible funding mechanisms and adaptive service models are critical for meeting the evolving and heterogeneous care needs of people living with dementia (Thapaliya et al., 2023). Improving access to allied health and respite services, alongside increased investment in specialised dementia care, may also prevent avoidable crises and delay or avert transitions into residential aged care (Livingston et al., 2024). At a population level, prioritising dementia-specific system reform through sustainable policy initiatives, workforce development, and community-based support can promote early engagement, social inclusion, and sustained wellbeing for people living with dementia and their families (Hung et al., 2021; Morton et al., 2021).
This study has several limitations. The use of self-report data introduces the potential for recall and response biases, while incomplete survey responses may have contributed to non-response bias, affecting the representativeness of the study sample. The sample was restricted to care partners who had engaged with services in the Australian Capital Territory and surrounding regions, limiting generalisability to other settings. Most participants were English-speaking, and there was limited representation of younger carers and culturally diverse populations. The absence of perspectives from people living with dementia constrained interpretive depth. Nonetheless, the mixed-methods design enabled methodological triangulation, providing a comprehensive and contextually grounded understanding of post-diagnostic support in this setting.
Conclusion
This study provides important insights into care partners’ experiences of post-diagnostic support in the Australian Capital Territory. While some services were accessible shortly after diagnosis, ongoing challenges were identified, including service fragmentation, funding limitations, and difficulties navigating complex care systems. These findings emphasise the need for integrated, flexible, and person-centred care models responsive to evolving needs across the dementia trajectory. Strengthening referral pathways and care coordination is essential to ensure continuity of care. These insights advocate for coordinated national reforms to improve dementia care and support for people living with dementia and their care partners.
Footnotes
Acknowledgments
The authors would like to sincerely thank all participants for their valuable time and contributions. We also acknowledge everyone who assisted in disseminating the study advertisement, facilitating broader participant recruitment.
Ethical Considerations
This study was conducted in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration. Ethical approval was granted by the Human Research Ethics Committee of the University of Canberra (HREC-13385) on June 14, 2024.
Consent to Participate
Written informed consent was obtained from all participants prior to their inclusion in the study. For the interview component, additional verbal consent was secured from participants immediately preceding the interview process.
Consent for Publication
Participants provided informed consent for the publication of research outcomes, with the explicit understanding that individual privacy and confidentiality would be rigorously maintained.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Nathan D’Cunha is supported by an ACT Health Research and Innovation Fellowship.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
De-identified quantitative data that support the findings of this study are available from the corresponding author upon reasonable request, subject to institutional data sharing policies and ethical considerations.
