Abstract
Purpose
Development of renal artery stenosis (RAS) can lead to renal function impairment, blood pressure elevation, and myocardial remodeling. However, the detail mechanism, the treatments using medicines and stents were still blurred. In order to study RAS, animal models are badly needed. This study aimed to establish RAS model based on the novel “two kidneys, one clip” approach for renal hypertension (2K1C-RHR) modeling in rats.
Methods
Sprague Dawley (SD) rats were selected for model building. Using the approach of 2K1C, the RAS model was built with delayed removal of clips. Animal models were evaluated using systolic blood pressure (SBP) and creatinine (Cr) at the time points for the stenosis recording. Data were expressed as mean ± standard deviation and one-way ANOVA was used for comparisons among groups.
Results
The SBP levels and the Cr concentrations were significantly increased after the establishment of RAS model (P < 0.05) using 2K1C method with delay of 1 to 3 weeks, blood pressures increased from 85.9 ± 5.9 mmHg to 131.5 ± 16.7 mmHg in model groups and especially obvious in after the establishment model of two weeks (from 87.4 ± 6.9 mmHg to 145.6 ± 11.8 mmHg). Cr increased from 19.2 ± 4.1 μmol/L to 40.5 ± 11.8 μmol/L in model groups and especially obvious in after the establishment model of three weeks (from 18.8 ± 4.5 μmol/L to 51.4 ± 9.7 μmol/L). When the clips were removed from the stenosis models, 3 days from removal of clips, the SBP levels and the Cr concentrations decreased.
Conclusion
Based on 2K1C approach, rat RAS model would be established and the reversible effects were observed which would be used for kidney function restore and the blood pressure control.
Keywords
Introduction
Renal artery stenosis (RAS) is the most common cause of renovascular hypertension (RVH), 1 accounting for 1–2% of all patients with hypertension and 5.8% of patients with secondary hypertension. 2 Among them, atherosclerotic renal artery stenosis (ARAS) is the most common cause of RAS, accounting for about 90% of patients with RAS,3,4 followed by fibrous muscular dysplasia (FMD). 5 Inflammations of the aorta are other causes of RAS, as well as renal artery entrapment or infarction, and exogenous compression of the renal arteries.6,7 In ARAS, the renal arteries were cramped, fluid flows were blocked and the kidneys blood supply were influenced. Further development of RAS leads to renal ischemia and cardiovascular dysfunction. 8 Studies showed that ARAS usually has poor prognosis. 9 Oxidative stress, inflammation, and fibrosis are exacerbated in the narrowed kidneys of ARAS compared to other causes of RAS.10,11
RAS activates the Renin angiotensin aldosterone system (RASS), which leads to sodium retention and reduced glomerular filtration rate. 12 HTN induced by RAS can appear in the presence of unilateral or bilateral stenosis as well as isolated RAS, which can subsequently lead to renal insufficiency (e.g. ischemic renal disease) or pulmonary edema. 13 Renal arteries blood flow restoration can decrease high blood pressure, even in some patients blood pressure may return to the normal. It is reported that the blood pressure of 2K1C or single-kidney, single-clamped (1K1C) rats fell rapidly after the clamp was released.14,15 Thus the hope of reversible RAS was still existed.
More than 50 animal models have been developed for hypertension study. The criteria for the establishment of these models were mainly based on a stable increase in systolic blood pressure (SBP). 16 One of the most widely used is the “two kidneys, one clip” (2K1C) Goldblatt model, in which Goldblatt and colleagues narrowed the renal arteries of dogs using silver clips. The SBP in rats increased after a period of renal artery narrowing, thus inducing the first animal model of renal vascular hypertension. 17 The establishment of the 2K1C model laid on the foundation for the following study of the RASS. Due to the high expenses of large animals and the financial requirements for experiments, some researchers have tried to do modeling with small animals and obtained more satisfactory results. In the study of Lorenz et al., the success rate of inducing hypertension using traditional u-shaped silver clips was 40–60%. 18 Warner et al. and Lorenz et al. successfully used polyurethane tubing to produce a mouse RAS secondary to hypertension mode.18,19 However, none of the above experiments lifted the RAS after SBP elevation and could not study the effect of renal artery lumen restoration on rats. Therefore, we need to make further improvements on the basis of the previous experiments.
Although the 2K1C model has been widely established, there is no requirement to unblock RAS in previous studies. Our inspiration for our experiments originated from the controversy related to the treatment of renal artery stent placement. Establishing a small animal model that can restore renal artery blood flow is our initial goal. Building on the research conducted by our predecessors, we will try to establish a new “2K1C” rat model that can restore blood flow in the renal arteries. The modeling success will be assessed by the changes in blood flow in the renal artery lumen, renal artery pulsation and pressure. We will also assess the effects of releasing RAS on rats at different times by measuring the corresponding indexes.
Materials and methods
Silicone tube for the experiment
In this study, a silicone tube with a smooth surface will be used as the shrinking renal artery material. It was purchased from Shanghai Beifut Rubber & Plastic Products Co. The silicone tube (Figure 1) has an inner diameter of 0.3 mm, an outer diameter of 4 mm, and a length of 5 mm (the length is cut according to your needs). The Silicone Tubing has a smooth rounded inner wall and a thicker outer diameter that is supportive and can ensure the consistency of the inner diameter. To ensure the sterile environment, sterilizing the silicone tubing is necessary. Besides, we will use sterilized instruments to slice the silicone tube to be the short segment of approximately 5 mm in length and slice a fissure in the side of the tube as the entry into the tube.

The size of silicone tube.
Animals
Thirty-two SPF-grade Sprague Dawley (SD) male rats weighing 230g-250 g were obtained from Sprague Dawley (Beijing) Biotechnology Co. Ltd (License No.: SCXK (Beijing) 2019-0010). Before the experiment, the rats were acclimatized and fed for at least 1 week with access to food and water ad libitumin the Binhai New Area Medical Animal Experiment Center. The rats were randomly divided into the control (sham) and the model group. In the model group, according to time, three subgroups were classified including 1-week, 2-week, and 3-week subgroups (the subgroups were defined according to the model time) and marked at the tail. This work was approved by the Ethics Committee of TEDA International Cardiovascular Hospital (Resolution No. TICH-JY-20180824-2). This sample size was calculated and considered feasible by the statistical expert Dr Wei.
Construction of animal model
All rats were fed with aspirin enteric-coated tablets (9 mg/kg, 1 time/d) by gavage for 3 days before surgery. All instruments and silicone tubes were sterilized before surgical operation, and the animals were fasted for 12 h. Inhalation anesthesia (isoflurane) was administered to rats using a small animal anesthesia machine. The anesthetized rats were placed on a thermostatic heating pad at 37 °C and the limbs were fixed with tape. The abdominal rat feathers were shaved and sterilized with iodophor. A longitudinal incision of 2–3 cm on the middle abdomen was made. After exposing the abdominal cavity adequately, the colon and small intestine on the left side were pushed to the right side. Then, the intestinal tube was covered with a wet gauze soaked in saline. After the left kidney was uncovered, the left renal artery and renal vein were carefully separated. A silicone tube with an inner diameter of 0.30 mm, an outer diameter of 4 mm and a length of about 5 mm was looped into the proximal part of the renal artery near the abdominal aorta. The physiologic position of the bowel was restored after the ooze was disposed of and the abdominal cavity was carefully inspected for bleeding and bowel injury. The peritoneum, muscles and skin were closed with 2-0 silk sutures. The sham control group of rats were opened the abdominal cavity and separated the renal arteries without putting the silicone tube into the renal arteries, and all other operations were the same as those in the model group (Figure 2). The rats in the 1-week, 2-week, and 3-week groups were operated to reopen the abdominal cavity and remove the silicone tubes at 1, 2, and 3 weeks postoperatively, respectively (Figure 3). After release of RAS, feeding was continued for 3 days. In order to block thrombosis, aspirin (same dose as before) was continued to be given after operation until the end of the experiment (Figure 3). The post-operative intramuscular injection of cefuroxime sodium (0.2 g/kg, 3 times/d) was used continuously for 3d to prevent and control postoperative infections in rats.

Flowchart of the experiment.

Renal artery before and after stenosis and after release of stenosis.
SBP measurement
Measurements of SBP were conducted before modeling of renal artery stenosis (preoperation), after renal artery stenosis (postoperation), and the 3 days after release of renal artery stenosis. Measuring the SBP of the rat requires keeping the rat awake. The temperature of the thermal barrel was needed to regulate to induce the rat to enter the thermal barrel where is a warm and dark environment. Then wrap the barrel in a rat bag. After waiting for the rats to be quiet for at least 10 min, a manometric airbag was inserted into the root of the rat's tail, and the rat's blood pressure was measured by clicking the pressurization measurement button on the manometer. SBP was measured three times, and the mean SBP was recorded.
Plasma Cr measurement
After each measurement of SBP, 1.5 mL of blood was taken immediately at the medial canthus with a capillary tube in an EP tube with anticoagulant. The sample was kept at room for 30 min, and centrifuged at 3000 rpm for 15 min. Supernatant was removed to a new EP and stored at −80°C for finial measurement using a Japan Toshiba automatic biochemistry analyzer (TBA-40FR ACCUTE, Beijing Furui Runze Biotechnology Co.).
Statistical analysis
Mean ± standard deviation was used to express SBP levels and Cr concentration. One-way ANOVA was used for comparison among groups. Data were processed using SPSS 25.0 statistical software (Armonk, NY: IBM Corp), and graphs were drawn using Graphpad Prism (version 9.5, GraphPad Software, San Diego, California USA), in which P < 0.05 was statistically significant difference.
Results
Visualization of the renal arteries
A strongly pulsatile renal artery with thick walls and light color was seen before surgery. When placing the silicone tube, the blood flow reduced and removal of the tube, renal artery immediately filled up and the lumen springs back and blood flow recovered.
SBP and Cr changes
Obviously, without SBP fluctuates in the sham group was observed during the research period. Yet, increased SBP were observed in the model groups after the establishment of RAS instead of the time cost for the model. Before operation, blood pressure were about 85.9 ± 5.9 mmHg in rats. However, after operations, SBP increased from 85.9 ± 5.9 mmHg to 131.5 ± 16.7 mmHg in model groups and especially obvious in after the establishment model of 2 weeks (from 87.4 ± 6.9 mmHg to 145.6 ± 11.8 mmHg). After removing the clamp from the stenosis model, a decrease in blood pressure was observed in all model groups. Rats in the model group had higher SBP after renal artery stenosis than before stenosis (1-week group,2-week group, and 3-week group: Before vs. After P < 0.001), especially in the 2-week group, which had the highest SBP elevation. The SBP in the model group was significantly lower than that after renal artery stenosis in the sham control group 3 days after release of renal artery stenosis (1-week group, 2-week group: After vs. After 3 days P < 0.001,3-week group: After vs. After 3 days P < 0.05).(Figure 4).

Changes in SBP before renal artery stenosis, after renal artery stenosis and 3 days after release of renal artery stenosis.
The trends of Cr concentration were the same as the changes of SBP. That is, before operation, Cr were about 19.2 ± 4.1 μmol/L in rats. However, after operations, Cr increased from19.2 ± 4.1 μmol/L to 40.5 ± 11.8 μmol/L in model groups and especially obvious in after the establishment model of three weeks (from 18.8 ± 4.5 μmol/L to 51.4 ± 9.7 μmol/L). After removing the clamp from the stenosis model, a decrease in blood pressure was observed in all model groups. The Cr after renal artery stenosis was significantly higher than that before stenosis in rats of the model group (1-week group,2-week group, and 3-week group: Before vs. After P < 0.001). The longer the duration of stenosis, the more pronounced the elevation of Cr was, and the highest Cr after renal artery stenosis was seen in the rats in the 3-week group. The Cr of rats in the model group, the 3 days after release renal artery stenosis, was significantly lower than that after renal artery stenosis (1-week group,2-week group, After 3 days: P < 0.01) (Figure 5).
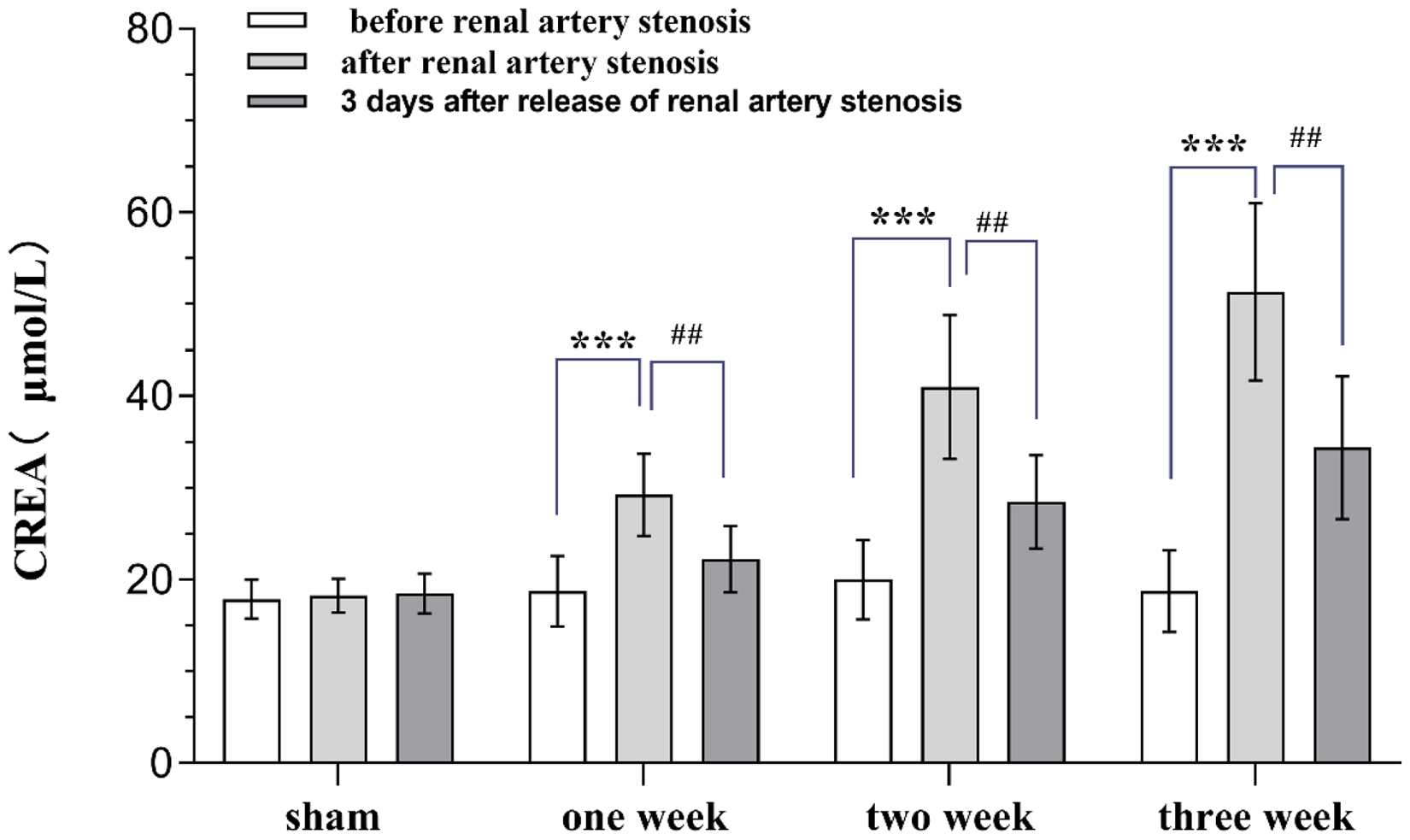
Changes in Cr before renal artery stenosis, after renal artery stenosis and 3 days after release of renal artery stenosis.
Discussion
Basic treatment for ARAS includes blood pressure control, lipid lowering, antiplatelet agents and lifestyle improvement.20,21 Percutaneous angioplasty and revascularization may be further considered in severe cases or when basic treatment is not effective. The advent of endovascular therapy has benefited patients by alleviating the harm of open surgery, most RVH patients have improved hypertension after stenting, and some of them are even able to return to normal blood pressure levels. However, the results of some clinical randomized controlled trials have shown that stenting is not better than drug therapy in regulating blood pressure and improving renal function, such as ASTRAL, 22 STAR, 23 CORAL. 24 Considering the limitations of clinical studies, such as the inability to observe whether renal injury from ischemia in patients can be improved after restoring blood flow by pathology, the bias in inclusion and exclusion criteria, the fact that some patients have hypertension due to RAS and some have elevated blood pressure before RAS, and the pathophysiology of the different conditions may be different. Therefore, the establishment of an animal model of RAS that restores renal artery blood flow is particularly important, and may provide a basis for future studies on the effects of drug therapy and renal artery blood flow recirculation.
It has been tried to increase blood pressure in experimental animals by various methods of damaging the kidneys, and researchers have used injections of nephrotoxic substances, x-ray irradiation of the kidneys, renal vein thrombosis, removal of a portion of the renal tissue, and ligation of unilateral or bilateral kidneys to cause persistent hypertension in the model. 25 It was first experimentally demonstrated that stenosis of the renal arteries could elevate blood pressure, and that recanalization of the renal arteries could reverse hypertension or ischemic nephropathy, in 1930. 26 However, some patients with prolonged renal ischemia are not always cured even if the renal artery blood supply is restored. The traditional 2K1C rat model usually uses silver clips as an auxiliary tool to constrict the renal arteries, but the success rate of the model is only 40% to 60%.26,27 Due to the ductile nature of silver and its easy to change shape, the inner diameter of the silver clip is very possible to change during the surgical operation, resulting in the stenotic renal arteries not being able to maintain the same inner diameter, which leads to a significant error in the final results. If the inner diameter of the silver clip becomes larger, the degree of stenosis will be smaller, and it will increase the chance of the silver clip falling off, leading to an increase in the failure rate of the model. Therefore, choosing a material that can keep the degree of RAS consistent, and is affordable and easy to obtain is one of the aims of our experiment.
Li LQ et al. contracted the renal artery using a titanium clip with an internal diameter of 0.23 mm. They clamped the renal artery and insulin needle together with the titanium clip and subsequently carefully withdrew the needle. 28 In fact, the titanium clip is triangular in shape when it is opened and its inner wall is not uniformly round. When it clamps both the renal artery and insulin needle, the renal artery may be displaced to the angle of the clamp, which may not reach the degree of constriction of the renal arterial wall, then the renal arterial diameter is unaffected by insulin needles, which may ultimately fail to cause a stenosis. We use a material that is not easily deformed and has a smooth inner diameter, which ensures the consistency of the model and improves the success rate of the model.
Before surgery, the renal arteries are strongly pulsating, their walls are thicker than the renal veins, and their internal diameters are smaller than those of the renal veins. We can see that the color of the artery is light red and the vein is dark red. After placing the silicone tube, we can clearly see that the blood flow in the lumen of the constricted renal artery is reduced, and the pulsation is weakened. The model group removed the silicone tube at 1, 2 and 3 weeks respectively, when the silicone tube was removed, we could see that the stenosed renal artery immediately filled up, the lumen rebounded, the blood flow in the renal artery was smooth and the renal artery pulsation was enhanced.
The inner diameter of 0.3 mm silicone tubing used in this study was explored by our group in the early stages. In the pre-experimental study of our group, we used silicone tubing with an inner diameter of 0.2 mm, 0.3 mm and 0.4 mm to narrow the renal arteries. However, we observed that using 0.2-mm inner diameter silicone tubes usually resulted in thrombus formation in the renal arteries of rats, which ultimately led to occlusion of the renal artery walls. Using a silicone tubing with an inner diameter of 0.4 mm was not sufficient to produce a significant change in the rat's SBP. However, a tubing inner diameter of 0.3 mm not only raises the SBP but also prevents the formation of thrombus in the renal artery of rats.
The animal model showed that SBP and Cr were significantly higher in the model group after renal artery stenosis compared with before renal artery stenosis. SBP and Cr in the group, the 3 days after release the stenosis, decreased compared with the group after renal artery stenosis. The above change indicated that the renal artery blood flow could be restored. The blood pressure increased with the extending of the time, and peaked at the second week. SBP is the blood pressure at which arterial blood pressure reaches its highest value in the middle of ventricular systole, the principle of which involves mainly the elastic saving effect of the large arteries and the aorta. Therefore, the peak of SBP at week 2 in the experimental results may be related to arterial elasticity and self-regulation. After releasing the RAS, the SBP of the rats in the 1-week and 2-week groups decreased more significantly than that of the rats in the 1-week group, and it was easier to return to the normal range. Although the SBP of rats does not increase indefinitely, the SBP was significantly lower after the release of renal artery stenosis than after renal artery stenosis. It indicates that renal artery stenosis lifting plays a crucial role in lowering blood pressure in rats with renal vascular hypertension. After renal artery stenosis in rats, Cr increased with the prolongation of stenosis time, indicating that renal artery stenosis caused renal injury, and the longer the stenosis time, the more severe the renal injury. The Cr of rats in the model group all decreased after the release of renal artery stenosis, considering the improvement of renal function after the successful restoration of renal artery blood flow.
X-ray irradiation of the kidney, injections of nephrotoxic substances, and excision of renal tissue can increase the blood pressure of rats, but they cannot realize the recirculation of renal artery blood flow. Moreover, all these methods will lead to occlusion of the renal artery and cannot narrow the lumen of the renal artery. As we know, renal artery occlusion and different degrees of stenosis not only affect the hemodynamics of the body, but also cause different degrees of damage to related organs. And our aim in the clinic is to study the effects on the organism after stent placement in patients with renal artery stenosis. Therefore, the above traditional experimental methods are not applicable to this experiment.
The food grade silicone tubing used in this experimental study was able to keep the diameter of the tubing unchanged due to the thick wall of the silicone tubing as compared to the conventional silver clamps. On the one hand, it will not easily fall off from the renal artery due to deformation as silver clamps do, which can largely improve the success rate of the model and save the experimental cost. On the other hand, the inner diameter of the silicone tube can be kept consistent, unlike the silver clip, the inner diameter will not change easily, which can reduce the experimental error. Since the inner diameter of the silicone tube is morphologically regular and circular, it contracts uniformly against the wall of the renal artery. Compared with titanium clips, the use of silicone tubing avoids the uneven contraction of the renal artery wall due to the presence of pinch points, and somewhat reduces the deviation of the inconsistent degree of RAS in experimental rats.
The physiological cycle of rats is very different from that of humans, and the body's response and regulation to RAS are also different. When we constricted the renal artery to 0.3 mm, the SBP of most of the rats in the experimental group began to change at 1 week and reached a peak at about 2 weeks, and the SBP decreased overall at 3 weeks. We found that changes in SBP in rats and their organismal self-regulation are more sensitive than sensitive, and therefore chose to study only the series of changes during the narrowed three-week period. The limitation of this experiment is that the stenosis time of the experimental group of rats was only set for 3 groups, and the changes in the renal artery and SBP after 3 weeks are full of unknowns. Therefore, in future experiments, we consider extending the stenosis time of the renal artery to further increase the rigor of the experiment. At the same time, the stress response, temperature, and different measurement time may cause the SBP and Cr of rats to change, so the measurement index of this experiment has some limitations, and our group plans to not only extend the time of RAS in future experiments, but also to increase the research index.
Based on the successful establishment of this novel rat model of reversible RAS, we can carry out more basic research in conjunction with the clinic. Is stenting and revascularization necessary in patients with RAS leading to renal vascular hypertension? We believe that this experimental model can be used for more in-depth study. We can divide the experimental group into drug treatment group, release RAS group and drug plus release RAS group. The renal function, SBP, pathology, and protein test of the experimental subjects in each group will be tested separately, and then we can compare the degree of benefit brought to the experimental subjects by different treatments
In addition, aortic coarctation of the cumulative renal arteries, renal artery compression, and other conditions that result in damage or stenosis of the renal arteries can be studied using this model. Since the clinical research can only detect the patient's Cr, renin and other blood indexes, but can not obtain the kidney tissue for pathological testing, so for this type of research, we can use our model for relevant interventions. In addition to obtaining blood index tests, it is also possible to obtain the healthy and affected kidneys of experimental rats for HE and immunohistochemistry studies, which are more convincing.
However, the improved model still has some defects, ARAS is a slow process, and the stenosis in this model is different from the pathophysiological process of ARAS. The method of constricting the renal artery adopted in this experiment is more suitable for acute RAS diseases, such as arterial entrapment. The indexes such as SBP and Cr may be affected by the environment and the self-regulation of the body, which may cause experimental errors. In order to solve these problems, on the one hand, our group plans to develop a slowly expanding material to be placed on the renal arteries, and appropriately prolong the stenosis time of the renal arteries in the experimental group of rats, so as to realize the slow stenosis of the renal arteries. On the other hand, in the next experiments, we considered HE and immunohistochemistry of bilateral kidneys as the focus of experiments to further optimize the model and experimental studies.
Combining the above experimental results, the reversible RAS model can be successfully established. The establishment of this innovative model lays the foundation for the further basic experimental study on the of RAS.
Conclusion
Based on “2K1C” approach, rat RAS model would be established and the reversible effects were observed which would be used for kidney function restore and the blood pressure control.
Footnotes
Acknowledgements
This job was supported by Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-019A). We also thanked the staff members of TICH Basic Medical Research Center for helping us with our work. We also thanked Prof. Wei Maoti for his crucial advice and guidance on our article. In addition, I would like to thank the anonymous reviewers who have helped to improve the paper.
Authors’ contribution
Ms. Zhou Shuang ang Ms. Lin Yejia contributed equally to this work, they carried out lab study, data and sample collection and drawn the draft. Prof (Dr) Li Xuedong conceived the paper, carried out revising the draft and approved the article.
Data accessibility statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent statements
The Experimental Animal Welfare Ethics Review Committee of TEDA International Cardiovascular Disease Hospital evaluated that this experiment complies with the ethical protection of animal welfare.
Funding
This work was supported by the Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-019A).
