Abstract
Hypothesis:
Single nucleotide polymorphisms and altered gene expression of components of the renin–angiotensin system (RAS) are associated with neurodegenerative diseases.
Introduction:
Drugs that interact with the RAS have been shown to affect the course of neurodegenerative disease, suggesting that abnormalities in the RAS may contribute to neurodegenerative disease.
Materials and methods:
A meta-analysis of genome-wide association studies and gene expression data for 14 RAS-related proteins was carried out for five neurodegenerative diseases: Alzheimer’s disease, Parkinson’s disease, narcolepsy, amyotrophic lateral sclerosis and multiple sclerosis.
Results:
No single nucleotide polymorphisms in any of the 14 RAS-related protein genes were significantly associated with the five neurodegenerative diseases investigated. There was an inverse association between expression of ATP6AP2, which encodes the (pro)renin receptor, and multiple sclerosis, Alzheimer’s disease and Parkinson’s disease. An association of AGTR, which encodes the AT1 angiotensin II receptor, and Parkinson’s disease and Alzheimer’s disease was also observed.
Conclusions:
To date, no single nucleotide polymorphisms in components of the RAS can be definitively linked to the neurodegenerative diseases evaluated in this study. However, altered gene expression of several components of the RAS is associated with several neurodegenerative diseases, which may indicate that the RAS contributes to the pathology of these diseases.
Keywords
Introduction
Neurodegenerative diseases present an increasingly pressing health issue. Alzheimer’s disease (AD) alone affects more than 44 million people globally, with billions of dollars in healthcare costs. 1 This is highly critical especially as AD and many other neurodegenerative disorders are still largely idiopathic. Intriguingly, recent research suggests the possible involvement of the brain renin–angiotensin system (RAS) in the aetiology of AD,2–4 amyotrophic lateral sclerosis (ALS), 5 multiple sclerosis (MS)6,7 and other neurodegenerative conditions.8,9 However, it is not entirely certain whether changes in RAS components in the brain in neurodegenerative diseases are causal or compensatory.
Much of the interest in the involvement of the RAS in neurodegenerative disease focuses on the involvement of angiotensin-converting enzyme (ACE) and AD. 4 Several reports describe an association of an insertion/deletion (I/D) polymorphism in the ACE gene and the incidence of AD.10–13 The II genotype which displays lower ACE activity in plasma, 14 but not in brain tissue, 15 than the DD and ID genotypes was associated with a reduced AD risk compared to the DD genotype.10–12 One study has indicated that homozygous DD or II showed a higher AD risk than the ID genotype 13 while some other studies have observed no association between the ACE I/D polymorphism and AD (see reviews).4,16 The potential for conflicting effects of ACE to decrease Aβ toxicity 17 contrasted with its ability to increase angiotensin II (Ang II) formation and attendant AT1 Ang II receptor mediated neurotoxicities 18 could explain the discordant observations.
In addition, there are several single nucleotide polymorphisms (SNPs) in the ACE gene that have been studied with respect to a possible association with AD, although the results are ambiguous. The SNPs rs4343, rs4291 and rs1799752 were shown to be associated with AD in Caucasians over 73 years of age, 19 and rs4343, rs4291 and rs1800764 were shown to be associated with age of onset of AD. 20 The association of the rs4291 SNP with AD was also found for sporadic AD, 21 however another meta-analysis did not show an association of rs4343, rs4291 and rs1800764 with late onset AD. 2 Four SNPs related to the ACE gene and ACE protein expression were reported to be associated with an altered risk of AD: rs4968782, rs4343, rs4316 and rs4459609. 22 The authors suggest that increased ACE activity in the brain clears Aβ42 decreasing AD risk. However, this runs contrary to studies suggesting that ACE inhibitors reduce AD risk. 23 Of note, Qiu et al. 23 only saw a reduction in AD risk with ACE inhibitors in populations that did not carry the Apo E4 allele that is associated with an increased risk of AD. However, in a subsequent study of an elderly population, an increase in AD risk was observed in ApoE4 allele carriers treated with ACE inhibitors. 24
Indeed, while the RAS works primarily to regulate cardiovascular functions, the receptors and hormones that make up the RAS in the brain may play roles at both the local and cellular level, 25 affecting not only autonomic control of blood pressure but also neuroinflammation, neurodegeneration and/or neuroprotection. This study attempts to evaluate the relationship between the expression of the genes encoding components of the RAS and related hormones (as well as the SNPs in those genes) and the occurrence of several neurodegenerative diseases through the incorporation and analysis of publicly available published datasets from genome-wide association studies (GWASs) and gene expression data in relation to neurodegenerative diseases.
Methods
The study consisted of in silico analyses of five neurodegenerative diseases: narcolepsy, Parkinson’s disease, AD, ALS and MS. For each disease, a meta-analysis of GWASs and a survey of gene expression data were completed for several RAS-related genes (see Table 1). The GWASs that were included in the meta-analysis were sourced from <www.gwascentral.org>. The number of studies that were included varied for each disease, but each represented at least several hundred SNPs found to be significant (see Table 2).
Renin–angiotensin system component genes and functionality of the components in relation to neurodegenerative conditions.

Genome-wide association studies included in meta-analysis.

The SNPs present in various RAS-related genes were sourced from the UCSC Gene Browser, which in turn sources its data from RefSeq. These SNPs were then cross-referenced with the collations of GWAS data. Any significant SNPs would ideally match multiple times to indicate a repeated finding of significance. The gene expression data were sourced from NCBI’s GEO datasets, and reflected several different cell types (see Table 3). These datasets were analysed through both the built-in GEO2R utility and through visual interpretation of graphs of the varying levels of gene expression. The GEO2R utility performed statistical calculations automatically, which assisted in identifying significant differences in expression.
List of data used in expression comparison in neurodegenerative diseases.

Genes in bold type were significant after Bonferroni correction for multiple comparisons.
The data were corrected for multiple comparison using the Bonferroni multiple comparisons test with α=0.05 and m=14 (14 genes were studied). This resulted in a corrected significance threshold of 0.0036. However, several different signals which were observed to be significantly associated by individual analysis did not survive the correction for multiple comparisons. Because this correction placed such a high stringency on the analyses, we report all data that showed a significant association that did not survive the multiple comparison in Table 2, but place our focus on the associations that remained statistically significant after the multiple comparison correction: i.e. p<0.0036, which is indicated in bold font in Table 2.
We used random-effect meta-analyses as the primary method of discovery and SPSS (v. 17) to do a secondary meta-analysis for heterogeneity. This second analysis is useful to estimate the possible effect of study heterogeneity on results and qualitatively to infer the effect of study heterogeneity on generalisability for similar sample series. We calculated χ2 tests for heterogeneity (Cochran’s Q) with SPSS and we generated I2 estimates using the equation:

where Q is the Cochran’s Q test estimate, and df is the degrees of freedom=13.
Results
In our current analysis using the publicly available datasets for GWAS, we found no SNP matches for any of the RAS-related genes in association with AD or any other neurodegenerative diseases.
We next examined genome-wide transcriptional studies to determine if there was any correlation between mRNA levels of the RAS components and neurodegenerative diseases. Gene expression data showed multiple significant (p<0.05, after correction for multiple comparisons to a significance level of p<0.0036) differences in RAS-related gene expression. The most significant association overall was the decreased expression of the gene ATP6AP2, which encodes the (pro)renin receptor 26 in mononuclear blood cells of MS patients (p=3.52E-9). Expression of the ATP6AP2 gene was also significantly decreased in the brains of AD patients (p<0.000403). Another gene that exhibited connections with neurodegenerative diseases was AGTR1, which encodes the primary Ang II receptor subtype (AT1). Expression of AGTR1 was decreased in postmortem substantia nigra samples of Parkinson’s disease patients (p=1.56E-5) and showed a near significant positive correlation with AD severity (p=0.00394). Expression of the gene LNPEP (which encodes cysteinyl aminopeptidase, a protein that binds a 6 amino acid carboxy terminal fragment of Ang II and has an alternative nomenclature as the angiotensin IV (AT4) receptor was significantly increased (p=0.000039) in mononuclear blood cells from MS patients and showed a trend towards increased expression with narcolepsy (p=0.00743). Expression of AGT and MAS1 were decreased in Alzheimer’s diseased brains: (p=7.93E-6) and (p=0.000521). Expression of MME, which encodes neprilysin, also known as neutral endopeptidase, was increased in mononuclear blood cells of MS patients (P=0.0023). Several other RAS genes were found to be significant for one or more neurodegenerative diseases, but did not survive the correction for multiple comparisons (Table 2). In sum, various RAS genes appear to be expressed at significantly different levels in samples taken from subjects with neurodegenerative diseases.
We also conducted Cochran’s Q test for heterogeneity and calculated the I2 value. The Cochran’s Q test for χ2(14) was 24.1, p=0.03. The I2 value was 46.0±4.6%, indicating low levels of heterogeneity in the results of the studies considered in this analysis. However, this level of heterogeneity is not unexpected because the same pooled treatment estimate for the entire study dataset was considered with a random effects model. The heterogeneity present in the study could also be attributed to the different types of tissues included in the study along with the use of different types of genomic studies with various types of genome-wide analysis technological platforms.
Discussion
There has been considerable speculation regarding the involvement of the brain RAS in neurodegenerative disease,21,27 however there has not been a systematic survey of all the current GWASs evaluating all genetic alterations in the genes of the various proteins that comprise the RAS, including newly discovered elements such as the prorenin receptor, ACE-2, and other enzymes that metabolise angiotensin peptides. 28 There was no evidence of SNPs in any RAS-associated genes surveyed that are significantly associated with any of the neurodegenerative diseases analysed in this study. However, this analysis is based upon publicly available databases that were hosted at http://www.gwascentral.org, and does not preclude the discovery of significant RAS-associated gene alteration associations in proprietary databases, or specific ethnic group subsets, or in more comprehensive datasets that are yet to be populated with larger numbers of patients.
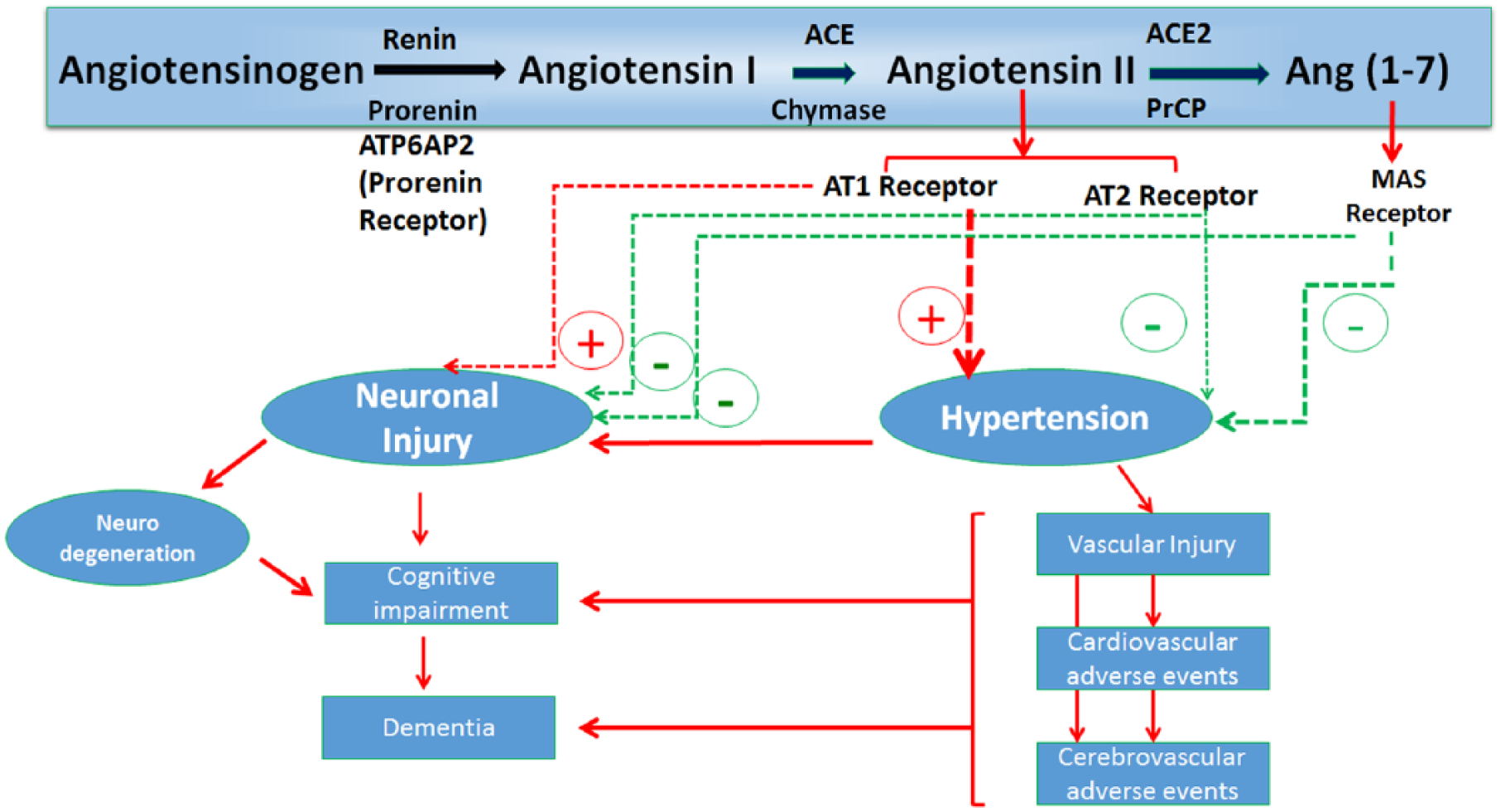
Potential Mechanistic Links Between Renin-Angiotensin System and Neurodegeneration.
In contrast, the gene expression data demonstrate a strong link between RAS gene expression and several neurodegenerative diseases. Of note, the two components of the RAS which show the greatest association with neurodegenerative diseases are receptors for RAS hormones: the AT1 receptor subtype for Ang II (the primary hormone and pathogenic component of the RAS) and the prorenin receptor, which is now recognised to signal via mitogen activated protein kinases (MAPKs) in response to prorenin and renin, thereby paralleling some of the pathophysiological effects of Ang II at the AT1 receptor. 26 It is noteworthy that the AT1 receptor signals via the activation of nicotinamide adenine dinucleotide phosphate, reduced form (NADPH) oxidase, thereby generating reactive oxygen species29,30 creating oxidative stress in neurons which could contribute to the neuronal cell death associated with neurodegenerative diseases. Indeed, AGTR1 expression has been linked to nucleic acid oxidation in models of Parkinson’s disease; 31 wherein 8-hydrodeoxyguanosine has been reported to be elevated under the influence of AGTR1 expression in Parkinson’s disease. This could be a possible contributing mechanism for the action of the RAS in neurodegenerative disease; however, a causal relationship is still not established.
While the gene expression data demonstrate a strong association between RAS gene expression and neurodegenerative diseases, some concerns must still be addressed. One of the expression datasets used as a basis for comparison, GSE5839, consisted of only four samples, which may have limited its ability to demonstrate the significance of the RAS’s connection to MS in the general population.
It is noteworthy to mention that there was an inherent low level of heterogeneity in our study results that could be attributed to the different types of tissues included in the study, along with the use of different types of genomic studies with various types of genome-wide analysis technological platforms.
Despite the study’s limitations, the differences in the expression of RAS genes in neurodegenerative diseases are still notable enough to warrant further study. A reasonable next step would be to conduct in vivo studies on animal models of various neurodegenerative diseases in order to determine if the differences in gene expression observed here are translated into differences in functional expression of these receptor proteins. Studies using AT1 receptor blockers and renin receptor antagonists could also be conducted in order to test whether activation of these receptors whose gene expression is increased causes increased oxidative stress or other pathophysiological effects that aggravate or cause characteristic neurodegenerations associated with specific neurodegenerative diseases.
Footnotes
Acknowledgements
The authors acknowledge GWAS Central and the NCBI GEO Database for making the data used in this study freely available. They thank Dr Jaime Tartar for statistical assistance.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
