Abstract
Objective:
With the development of genome-wide association studies (GWAS) concerning hypertension, a growing number of susceptibility genes related to hypertension have been revealed. Subsequently, several studies have investigated the association between CYP17A1 rs1004467 heritable variation and hypertension; however, the results have been inconsistent. In this study, a meta-analysis was performed to assess the association between the CYP17A1 rs1004467 polymorphism and hypertension risk.
Methods:
The PubMed, ISI Web of Science and Embase databases as well as China Wanfang, Weipu and the Chinese Journal Full-text Database were used to retrieve all publications from 2005 to 2013 related to case-control studies that reported a link between the risk factors for hypertension and the CYP17A1 polymorphism. All association studies were identified, and a meta-analysis was conducted using the RevMan 5.0 estimate for odds ratios (ORs) to determine whether the A allele predicts hypertension outcomes.
Results:
Three articles including five studies (totaling 4495 patients and 3529 controls) were identified. The overall effect suggested that rs1004467 was significantly associated with hypertension (OR=1.22, 95%CI 1.08–1.38, p=0.001).
Conclusions:
The present meta-analysis confirmed the significant association between a polymorphism of the CYP17A1 gene and hypertension susceptibility. The CYP17A1 A allele should be considered a risk factor for hypertension.
Introduction
Hypertension affects one-quarter of adults worldwide, and this proportion is expected to increase to one-third by 2025. 1 Hypertension is one of the most important risk factors for cardiovascular diseases, and the most important risk factor for morbidity and mortality. 2 Each year, approximately half of all cases of stroke and myocardial ischemia worldwide are caused by hypertension. 3 More than 40 years of clinical trials have shown that drug treatments to reduce blood pressure decrease the incidence of cardiovascular events. Hypertension is a complex disease that is affected by environmental factors, genetic factors, and their interaction. Over the last decade, scientists have found many gene loci or chromosomal regions associated with blood pressure or hypertension using the candidate gene and genome linkage analysis method. Furthermore, twin studies have provided evidence that variations in blood pressure have a heritability factor of approximately 50%. 4
Genome-wide association studies (GWASs) can screen for the gene polymorphism loci associated with hypertension. 5 For example, in 2009 Levy et al. 6 identified numerous loci associated with systolic blood pressure (SBP) levels and hypertension. They replicated genome-wide significant single nucleotide polymorphisms (SNPs) in 34,433 independent participants from the Global BPgen Consortium, and they found 11 genome-wide significant associations related to blood pressure, including four loci for systolic blood pressure (ATP2B1, CYP17A1, PLEKHA7, and SH2B3). The CYP17A1 rs1004467 gene is a common intronic variant associated with a rare Mendelian form of hypertension that emerged as a genome-wide significant locus in the meta-analysis results from both consortia. The relationship of this locus with hypertension was verified in a population-based cohort of Chinese Hans in 2010 when Liu et al. 7 genotyped eight variants that were confirmed to be associated with hypertension. Logistic regression and generalized linear analyses were used to test for an association between hypertension risk and blood pressure across each variant, and CYP17A1 rs1004467 was significantly associated with increased systolic blood pressure (p=0.005), diastolic blood pressure (p=0.01) and hypertension risk (p=0.0009). In addition, Tabara et al. 8 indicated that this locus is significantly correlated with high blood pressure. However, Yang et al. 9 have a different opinion regarding the relationship between the CYP17A1 gene polymorphism and hypertension.
The above studies have small sample sizes and substantial differences; furthermore, the results are inconsistent, so it is difficult to estimate an average effect. Meta-analysis is a statistical tool to combine the data of individual research projects, thereby increasing statistical power and improving the accuracy of the quantitative evaluation indicators to comprehensively analyze the relationship between rs1004467 polymorphism of the CYP17A1 gene and high blood pressure. We applied a meta-analysis for the integrated quantitative evaluation of the previous research results to provide a basis for hypertension prediction, prevention, and control, as well as to obtain reliable evidence of the relationship between the rs1004467 polymorphism and the incidence of hypertension.
Materials and methods
Literature and search strategy
Literature databases including PubMed, ISI Web of Science and Embase as well as China Wan fang, Weipu and Chinese Journal Full-text were searched for publications from 2005 to 2013. We sought to identify publications related to case-control studies that reported a link between the risk factors of hypertension and the CYP17A1 polymorphism using the keywords CYP17A1 AND polymorphism AND blood pressure OR hypertension. The list of retrieved articles was manually searched. The literature retrieval was limited to English-language articles.
Inclusion criteria and data extraction
The studies included in the meta-analysis met all of the following inclusion criteria: (1) They evaluated the association between the CYP17A1 rs1004467 polymorphism and hypertension; (2) They used a case-control or cohort design, where the control group included healthy or relatively healthy people without hypertension; (3) They provided an odds ratio (OR) with 95% confidence intervals (CIs) under the additive model or the genotype frequency for calculating ORs and 95% CIs; (4) Hypertension was defined as the presence of a systolic blood pressure measurement of at least 140 mmHg, a diastolic blood pressure measurement of at least 90 mmHg, a previous diagnosis of hypertension or the use of antihypertensive medication; (5) If more than one article was published using the same sample, then only the study with largest sample size was included; and (6) The control groups conformed to the Hardy–Weinberg equilibrium (p>0.05). The following information was extracted from each study: (1) the name of the first author; (2) year of publication; (3) sample ethnicity; (4) gender ratio; (5) sample mean age; (6) genotyping methods; (7) the numbers of patients with hypertension and controls; and (8) ORs with 95% CIs under the additive model. Two authors independently assessed articles for compliance with the inclusion/exclusion criteria. A third person decided whether to include an article when the two authors significantly disagreed. The detailed characteristics of the included studies are listed in Table 1.
Characteristics of included studies included in the meta-analysis.

OR: odds ratio; CI: confidence interval; HWE: Hardy–Weinberg equilibrium; PCR: Polymerase Chain Reaction.
Taqman is a real-time PCR method.
Statistical analyses
The association between the CYP17A1 rs1004467 polymorphism and hypertension was estimated by calculating the pooled OR and its associated 95% CIs. The pooled OR was determined via a Z-test (p<0.05 was considered significant). A Q test was performed to examine whether the variation was related to heterogeneity. If an article had an absence of heterogeneity (I2⩾50%), the Mantel–Haenszel method 10 was used to calculate pooled OR and 95% CIs. Alternatively, the DerSimonian–Laird method 11 was used to calculate pooled OR and 95% CIs when the articles presented heterogeneity (I2<50%). A sensitivity analysis was performed to assess the stability of the results after excluding one study at a time. A funnel plot was used as a visual tool to investigate publication and other types of bias or systematic heterogeneity. All analyses were conducted using RevMan 5.0.
Results
Characteristics of included studies
Thirty potentially relevant articles were identified through the literature search. Of these, 22 were excluded because they did not investigate the association between CYP17A1 rs1004467 and hypertension. Three studies were excluded because they investigated Chinese children.12–14 Two studies were excluded because they did not provide sufficient data. Therefore, five studies from three articles7–9 were included in the final meta-analysis. The flow diagram in Figure 1 demonstrates how the articles were included and excluded. Liu et al. 7 defined hypertension as having an SBP/DBP⩾140/90 mmHg, the use of antihypertensive medications or a previous diagnosis of hypertension. Normotension was defined as having an SBP/DBP⩽120/80 mmHg and the absence of antihypertensive medication use. In one study, Tabara et al. 8 demonstrated that participants with hypertension had parents, siblings, or both with hypertension, had been previously diagnosed with hypertension and were either receiving BP-lowering therapy or had an SBP/DBP of >160/100 mmHg. Normotensive controls had no family history of hypertension, had never been treated with antihypertensive medications, and their SBP/DBP was <120/80 mmHg. The selection criteria of the participants with hypertension in another study were as follows: either antihypertensive medication use or an SBP/DBP of >160/90 mmHg; normotensive participants were selected if they did not have a history of cardiovascular disease and had a SBP/DBP of <120/80 mmHg, did not currently use antihypertensive medication, or both. Yang et al. 9 diagnosed hypertension as the current use of antihypertensive medications, a history of hypertension, or an SBP/DBP of ⩾140/90 mmHg; otherwise, participants were included in the control group. The characteristics of the included studies are listed in Table 1.

Flow chart of meta-analysis for exclusion /inclusion of individual studies.
Meta-analysis results
Five studies of the CYP17A1 rs1004467 polymorphism were identified that included 4495 patients and 3529 controls. The meta-analysis results suggested the presence of significant between-study heterogeneity (χ2=8.26, p=0.08, I2=52%); therefore, a random effects model was used to calculate the pooled OR and 95% CIs. 11 In the forest plot, if the 95% confidence intervals of the pooled OR show no relationship with the midline, then the study was significant. In our study, the additive model frequency showed significant difference between the rs1004467 polymorphism and hypertension risk (OR=1.22, 95% CIs=1.08–1.38, p=0.001, Figure 2). The dominant model and recessive mode frequency also showed significant difference between the rs1004467 polymorphism and hypertension risk (OR=1.16, 95% CIs=1.04–1.29, p=0.007, Figure 3, OR=0.76, 95% CIs=0.63–0.90, p=0.002, Figure 4, respectively). This indicated that the CYP17A1 rs1004467 A allele should be considered a risk factor for hypertension.

Forest plot of the meta-analysis of the association between CYP17A1 rs1004467 and hypertension risk. Rs1004467 with increased risk of hypertension under an additive genetic model.

Forest plot of the meta-analysis of the association between CYP17A1 rs1004467 and hypertension risk. Rsl004467 with increased risk of hypertension under a dominant genetic model.

Forest plot of the meta-analysis of the association between CYP17A1 rs1004467 and hypertension risk. Rs1004467 with increased risk of hypertension under a recessive genetic model.
Sensitivity analysis
A sensitivity analysis was performed by excluding one study at a time to repeatedly calculate the effect size and compare it with the pooled OR of the meta-analysis before it has been excluded. If the results do not significantly change after exclusion, then the study has low sensitivity. The results confirmed that a significant association exists between the CYP17A1 rs1004467 polymorphism and hypertension susceptibility.
Publication bias
A funnel plot is a visual tool for investigating publication and other types of bias in meta-analysis. Smaller sample sizes are associated with more dispersed distributions, whereas larger sample sizes are associated with more concentrated distributions. The funnel plots of normal distributions are symmetrical, and asymmetric plots indicate publication bias. In our study, the symmetry of the funnel plot was symmetrical; thus, no significant publication bias was detected (Figure 5).
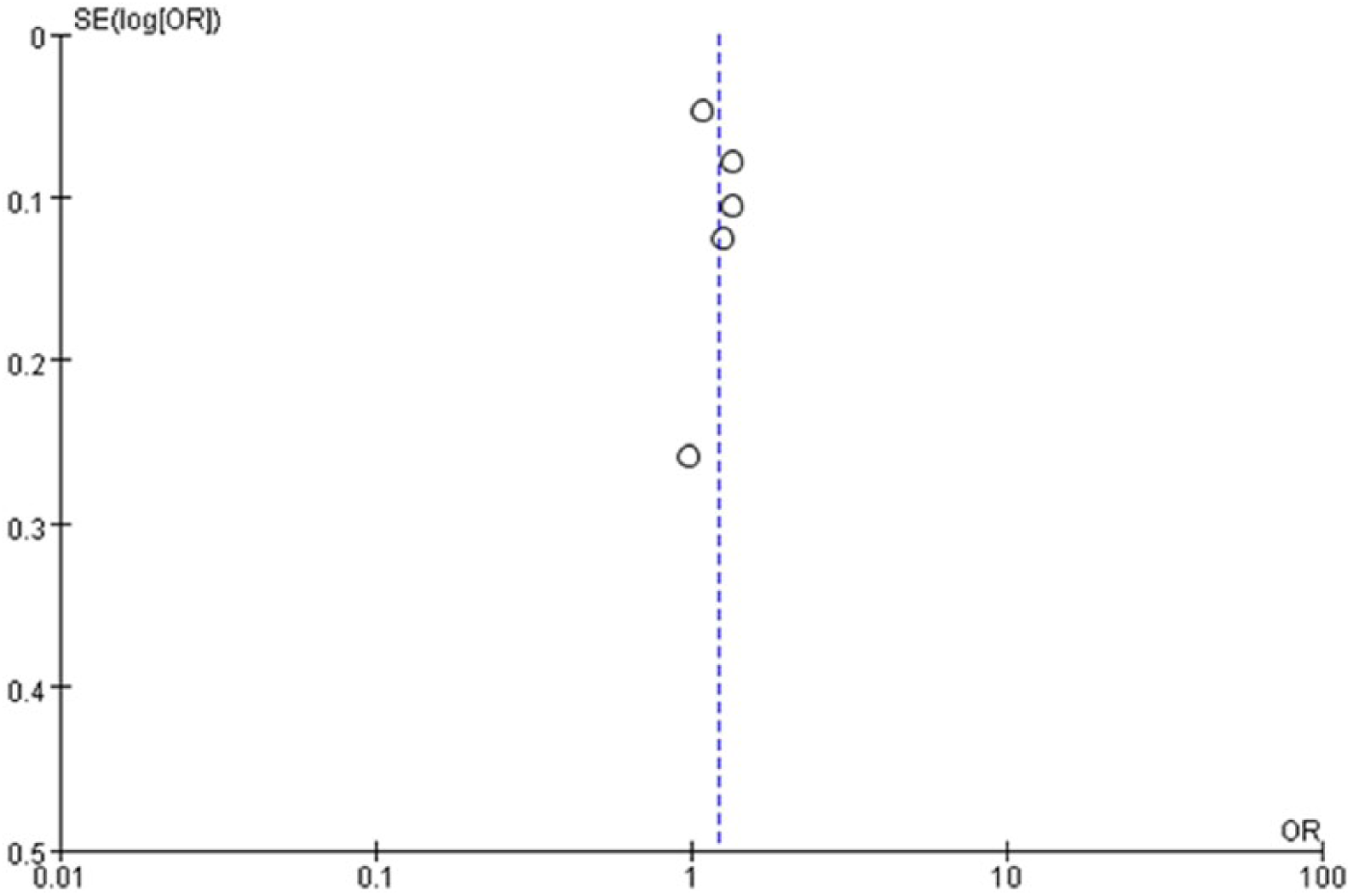
Funnel plot of the association between CYP17A1 rs1004467 and hypertension risk; the symmetry of the funnel plot was symmetrical, no significant publication bias was detected.
Discussion
This meta-analysis showed that the CYP17A1 rs1004467 polymorphism was significantly associated with the risk of hypertension, and this association was independent of conventional environmental factors. The incidence of hypertension among carriers of the A allele was significantly greater than that of carriers of the G allele.
The CYP17A1 gene, which is located on chromosome 10q24.3, consists of eight exons and seven introns, and it is primarily expressed in the adrenal glands and gonads. This gene produces P450c17 proteins, which are members of the cytochrome P450 superfamily of enzymes; furthermore, they catalyze many reactions involved in drug metabolism and the synthesis of cholesterol, steroids and other lipids. P450c17 proteins have both 17alpha-hydroxylase and 17,20-lyase activities and are key enzymes in the steroidogenic pathway that produces steroids. Rs1004467 is a mutation in an intron. Several recent studies have found that CYP17A1 is associated with hypertension.15–18 Tabara et al. 8 performed a multiple regression analysis with possible covariates to show that CYP17A1 was independently associated with blood pressure traits and hypertension. Moreover, they confirmed that CYP17A1 independently determined both blood pressure traits and hypertension after adjusting for age, sex, body mass index (BMI), and drinking habits. At present, how the CYP17A1 gene leads to hypertension is unclear. One possible underlying mechanism might be that CYP17A1 gene mutations can cause mineralocorticoid over-expression, leading to high blood pressure. 19 CYP17A1 mutation status was evaluated with regarding rare hypertension among Chinese participants, 20 and two compound mutations were identified: the amino acid missense mutation at codons 436–438 on exon 6 and the deletion mutation at codons 487–489 on exon 8, which caused the deletion of three amino acids Asp-Ser-Phe. Levy et al. 6 argued that these data suggest that common variants of CYP17A1 might be associated with blood pressure by promoting mild forms of enzyme deficiency or dysfunction; however, CYP17A1 mutations that cause phenotypic 17a-hydroxylase deficiency are rare. High blood pressure can be caused by fat metabolism disorder, and fat distribution in the body is influenced by genetic factors.21,22 Many studies have reported that the susceptibility genes of hypertension are also associated with fat distribution.23,24 A significant association was found between the rs1004467 polymorphism and the reduction of visceral and subcutaneous fat among women.
CYP17A1 gene mutations affect the synthesis of steroids, which are the precursors of sex hormones. Animal experiments and clinical observations demonstrate that the occurrence of hypertension is related to sex hormone levels in the body. 25 Recently, other evidence has indicated that the levels of sex hormones can affect the development of cardiovascular and cerebrovascular diseases. 26 A clinical study 27 showed that testosterone levels play an important role in the progress of hypertension among elderly men, whereas lower testosterone levels promote high blood pressure. The incidence of hypertension differs by gender, and an epidemiological survey found that the incidence of hypertension among premenopausal women was significantly lower than that among men.28,29 Therefore, estrogen likely plays a protective role in the cardiovascular system. A lack of estrogen is one of the major reasons that the incidence of hypertension increases among woman. Although the specific action mechanism of sex hormones is controversial with regard to hypertension, the fact that CYP17A1 gene mutations decrease sex hormones might provide new ideas regarding the etiology of hypertension.
Our current meta-analysis has one advantage: the OR covariates and 95% CIs from each study were adjusted and used to calculate a pooled OR. Therefore, the accuracy of the effect estimate and the statistical power were improved with regard to the association between the CYP17A1 rs1004467 polymorphism. However, several limitations should also be noted. First, between-study heterogeneity is common in meta-analyses of genetic association studies, and performing the Mantel–Haenszel method would increase the odds of a type I error. 30 Second, the control group included healthy and relatively healthy people without hypertension; therefore, selective bias might exist. Third, gene–gene interaction effects were not included in our meta-analysis; moreover, the interaction between environmental and genetic factors might also affect susceptibility to hypertension. Fourth, positive results were more likely to have been included in our study because negative results are unlikely to have been published or not revealed by our literature search; thus, some publication bias is most likely present.
Conclusion
In conclusion, we expected to find a significant correlation between the CYP17A1 rs1004467 polymorphism and susceptibility to hypertension. However, this subject needs additional research to support our conclusion. Future studies should not only consider the independent effect of genes but also gene–gene and gene–environment interactions to evaluate the relationship between gene mutation status and hypertension.
Footnotes
Author contributions
CFD and XX conceived the study, participated in the design, collected the data, performed statistical analyses, and drafted the manuscript. YTM, YNY and QL XML conceived the study and revised manuscript. FL, BDC and MTG participated in the design, and helped to draft the manuscript. All authors read and approved the final manuscript.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by National Natural Science Foundation of China (81160017) and Xinjiang Science and Technology Projects (201491181).
