Abstract
Objective:
To investigate the effects of the angiotensin-(1-7) signaling pathway and the possible role of atrial natriuretic peptide (ANP) on atrial electrical remodeling in canines with acute atrial tachycardia.
Methods:
Forty dogs were randomly assigned to eight groups (five dogs/group): sham, paced control, paced + angiotensin-(1-7), paced + angiotensin-(1-7) + Mas inhibitor, paced + angiotensin-(1-7) + Akt inhibitor, paced + angiotensin-(1-7) + PI3K inhibitor, paced + angiotensin-(1-7) + nitric oxide (NO) inhibitor, and paced + angiotensin-(1-7) + A-71915 (ANP receptor antagonist). Rapid atrial pacing was maintained at 600 bpm for 2 h for all groups, except the sham group, and angiotensin-(1-7) (6 μg kg−1 h−1), Mas inhibitor (5.83 μg kg−1 h−1), Akt inhibitor (2.14 μg kg−1 h−1), PI3K inhibitor (2.86 μg kg−1 h−1), NO synthase inhibitor (180 μg kg−1h−1), or A-71915 (0.30 μg kg−1 h−1) were administered intravenously. Atrial effective refractory periods, inducibility, and duration of atrial fibrillation (pacing cycle lengths: 300, 250, and 200 ms), and left atrial ANP concentrations were measured.
Results:
After pacing, the atrial effective refractory periods at the six sites shortened with increased inducibility and duration of atrial fibrillation, which was attenuated by angiotensin-(1-7), and increased ANP concentrations, which was promoted by angiotensin-(1-7) (paced control vs. sham; P < 0.05). All inhibitors and A-71915 blocked the electrophysiological effects of angiotensin-(1-7). ANP secretion induced by angiotensin-(1-7) was also blocked by all inhibitors.
Conclusion:
Angiotensin-(1-7) prevented acute electrical remodeling in canines with acute atrial tachycardia via the angiotensin-(1-7)/Mas/PI3K/Akt/NO signaling pathway. ANP was related to the anti-arrhythmic effects of angiotensin-(1-7).
Introduction
The rennin–angiotensin system (RAS) has been proven to be involved with several cardiovascular diseases, especially in atrial fibrillation (AF). 1 Angiotensin-II (Ang-II), the member of the RAS that was first recognized to increase the risk of cardiac arrhythmia, is related to the development of AF.2,3 Recently, the focus has shifted away from Ang-II after the study by Schiavone et al., 4 which confirmed Ang-(1-7) as a novel member of the RAS that counterbalances Ang-II.5,6 Recently, the essential metabolic pathway of Ang-(1-7) has also been clarified. Ang-(1-7) is mainly produced by Ang-II and is also converted from angiotensin-I (Ang-I) by neutral endopeptidase. 7 Alternatively, Ang-(1-7) can be produced directly from Ang-II by the carboxypeptidase angiotensin converting enzyme 2 (ACE2), a homolog of ACE. 8 In a recent study, Ang-(1-7) was initially associated with increased atrial natriuretic peptide (ANP) secretion from the signaling pathway involving the Mas/PI3K/Akt/NO axis to decrease cardiac hypertrophy in vitro. 9
The purpose of this study was to investigate the effects of Ang-(1-7) signaling pathway on acute atrial electrical remodeling and the potential role of ANP in dogs with rapid atrial pacing. Our work is helpful to understand the exact mechanisms on the protective effects of Ang-(1-7) in vivo. It may provide novel evidence for the application of Ang-(1-7) to treat or prevent AF in clinical practice.
Methods and materials
Preparation of the canine model
Forty mongrel dogs of either sex, weighing 12–17 kg, were randomly assigned to the following groups (five dogs/group): sham (group 1), paced control (group 2), paced + Ang-(1-7) (group 3), paced + Ang-(1-7) + A-779 (group 4), paced + Ang-(1-7) + API-2 (group 5), paced + Ang-(1-7) + wortmannin (group 6), paced + Ang-(1-7) + L-NAME (group 7), and paced + Ang-(1-7) + A-71915 (group 8). Approval for conducting this study was obtained from the Experimental Animal Administration Committee of Tianjin Medical University and Tianjin Municipal Commission for Experimental Animal Control.
The dogs were anesthetized with intravenous pentobarbital sodium (30 mg kg−1). After intubation and mechanical ventilation, a median sternotomy was performed using sterile techniques. Six pairs of electrodes (diameter: 1.5 mm; distance between poles: 1.5 mm) were stitched on the left and right atrial epicardium in each dog. The corresponding sites were at the high left and right atria (HLA and HRA, respectively), left and right atrial appendages (LAA and RAA, respectively), and low left and right atria (LLA and LRA, respectively). A jugular vein catheter was inserted for intravenous infusion, while a catheter was inserted into the left femoral artery of each dog to monitor systemic blood pressure.
Interventions for each group included the following (Figure 1): group 1, normal saline infusion without atrial pacing; group 2, atrial pacing with normal saline infusion; group 3, atrial pacing with intravenous Ang-(1-7) (6 μg kg−1 h−1); group 4, atrial pacing with intravenous Ang-(1-7) (6 μg kg−1 h−1) and A-779 (Mas inhibitor; 5.83 μg kg−1 h−1); group 5, atrial pacing with intravenous Ang-(1-7) (6 μg kg−1 h−1) and API-2 (Akt inhibitor; 2.14 μg kg−1 h−1); group 6, atrial pacing with intravenous Ang-(1-7) (6 μg kg−1 h−1) and wortmannin (PI3K inhibitor; 2.86 μg kg− 1h−1); group 7, atrial pacing with intravenous Ang-(1-7) (6 μg kg−1 h−1) and L-NAME (NO synthase inhibitor 180 μg kg−1 h−1), group 8, A-71915 (ANP receptor antagonist; 0.30 μg kg−1 h−1). Atrial pacing was performed at 600 bpm (100 ms cycle length) for 2 h using 2 ms square-wave pulses at twice the threshold current of the electrodes at the HRA. Direct systolic blood pressure was measured during anesthesia at baseline and after 2 h of atrial pacing. Surface electrocardiograph (ECG) and blood pressures were recorded by a multi-channel physiological recorder (Model P4B533-K, China). A dose of 6 μg kg−1 h−1 was selected for Ang-(1-7) because it was the highest dose at which blood pressure was not affected in our preliminary study (before and 2 h after continuous Ang-(1-7) infusion the systolic blood pressures were 137 ± 13 mmHg vs. 134 ± 15 mmHg, respectively; P > 0.05; n = 10). The ratio of antagonists (A-779, API-2, wortmannin, or L-NAME) to Ang-(1-7) was based on a previous study in which an increase in the dose of antagonists blocked ANP secretion induced by Ang-(1-7) in isolated rat atria with rapid pacing. 9 The ratio of A-71915 to Ang-(1-7) used in this study has been previously proven to optimally block the cardiac anti-hypertrophy of Ang-(1-7). 9

Ang-(1-7) was initially thought to increase ANP secretion through the signaling pathway involving the Mas/PI3K/Akt/NO axis to decrease cardiac hypertrophy.
Measurement of atrial effective refractory periods and atrial fibrillation induction
The changes in atrial effective refractory periods (ERPs) and the inducibility and duration of AF at basic pacing cycle lengths (BCLs) of 300 ms (BCL300), 250 ms (BCL250), and 200 ms (BCL200) at the six aforementioned sites were observed. When the atrial ERP was measured, the coupling interval of the extra stimulus (S2) at various BCLs was shortened by 2 ms steps, and the longest coupling interval that failed to capture the atrium was defined as the ERP. AF induction was defined as P-wave disappearance and rapid atrial activation with irregular ventricular response on atrial ECG after atrial programmed stimulation (S1–S2), which was attempted three times at each site. The AF induction site was defined as the site where AF persisted for >1 s. The sites with AF induction were excluded from atrial ERP analyses in each dog. The duration of induced AF was also recorded.
Atrial natriuretic peptide concentration assays
After electrophysiological studies, the left atria of the dogs in all groups (except group 8) were removed. Specimens were immediately frozen using liquid nitrogen and stored separately at −80°C for further analysis. The tissue samples were extracted with 0.1 M phosphate-buffered saline (pH = 7.4) containing 1% Triton X-100 and centrifuged at 14,000g for 20 min. The supernatant was collected and the ANP concentration (ng/mL) was determined using ELISA (Abcam, UK).
Statistical analysis
All data were expressed as mean ± SD. For atrial ERPs, left atrial ANP levels and the duration of induced AF were compared among groups using analysis of variance (ANOVA). Subsequently, all significant data were analyzed with a t-test with Student–Newman–Keuls correction or a Dunnett’s test to evaluate the differences between individual mean values. Inducible rates of AF were analyzed with the exact probability test. A P < 0.05 was considered statistically significant.
Results
Hemodynamic parameters
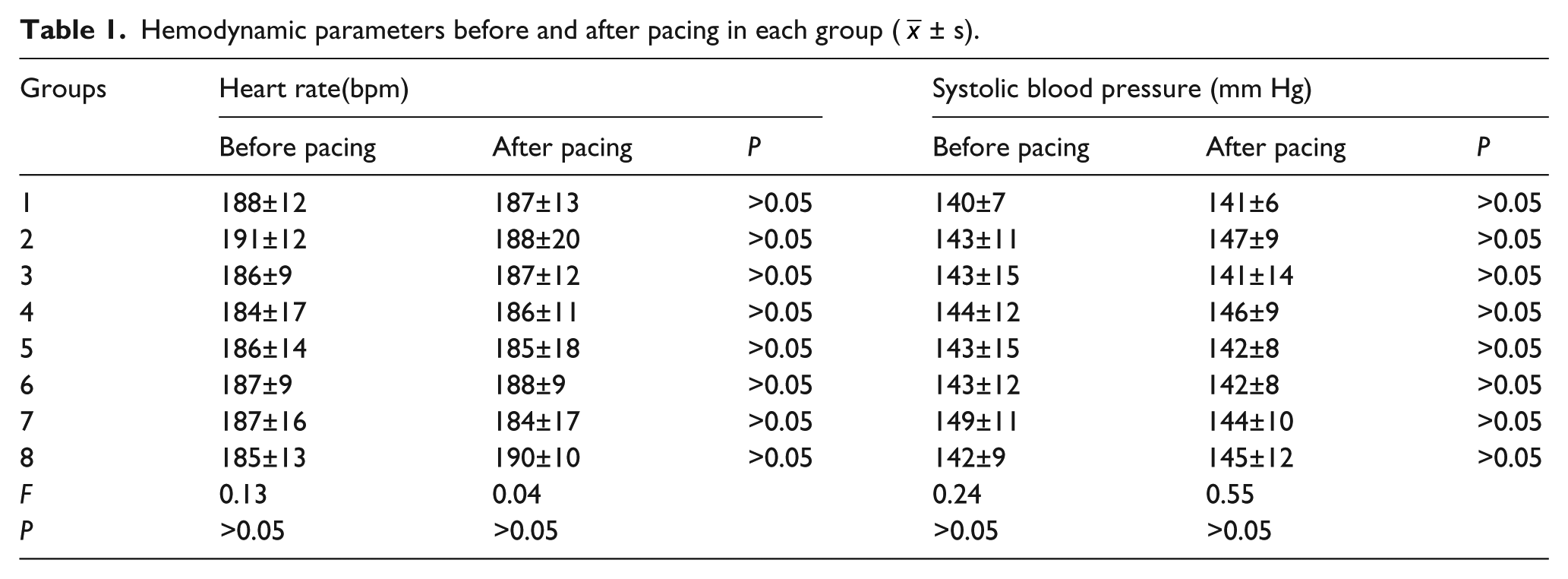
As summarized in Table 1, there were no differences in the ventricular rate or systolic blood pressure between all eight groups at baseline (P > 0.05). Also, the heart rate or blood pressure did not change due to pacing among all groups (P > 0.05).
Hemodynamic parameters before and after pacing in each group (
Atrial effective refractory periods at various basic pacing cycle lengths
In the paced control group, ERPs of the six atrial sites at BCL300, BCL250, and BCL200 were shortened compared to the sham group. However, the shortening of atrial ERPs in most of the sites at various BCLs was attenuated by Ang-(1-7) treatment in group 3 compared to the paced control group (P < 0.05). The atrial ERPs of dogs treated by A-779, API-2, wortmannin, L-NAME, or A-71915 were not different compared to the paced control group (P > 0.05; Table 2; Figure 2).
The atrial ERPs of the six sites on left atria in each group at various BCLs (

At BCL300, BCL250, and BCL200, ERPs of the six atrial sites were shortened in the paced control group compared to the sham group (†P < 0.05). The shortening of atrial ERPs in most of the sites at various BCLs was attenuated by Ang-(1-7) treatment in group 3 compared to the paced control group (*P < 0.05). The atrial ERPs of dogs treated by A-779, API-2, wortmannin, L-NAME, or A-71915 were not different compared to the paced control group (P > 0.05). HRA indicates high right atrium; RAA, right atrial appendage; LRA, low right atrium; HLA, high left atrium; LAA, left atrial appendage; LLA, low left atrium; and BCL, basic pacing cycle length. The numbers of the sites without AF induction for atrial ERPs analysis were recorded in the brackets.
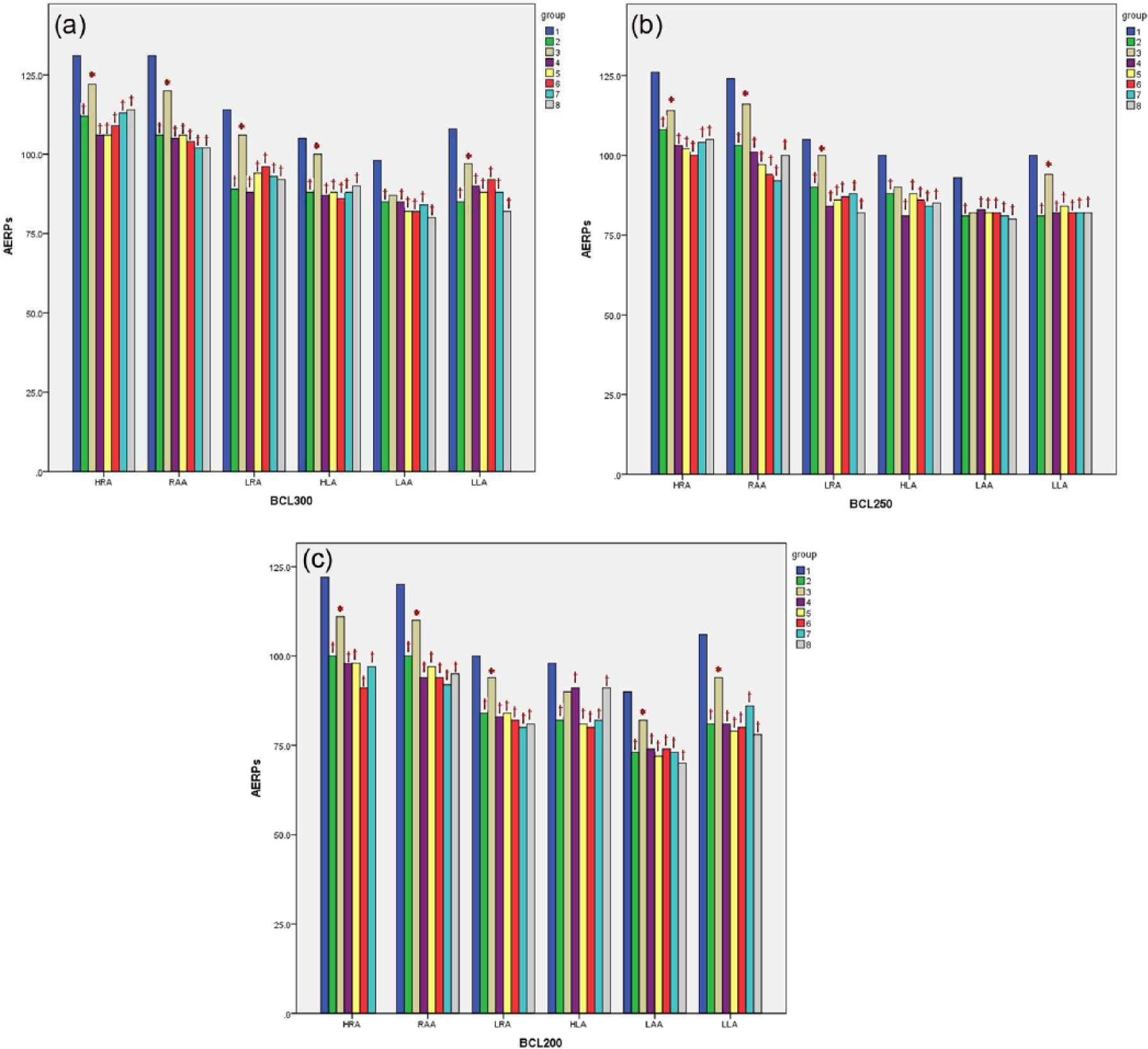
(a) At BCL300, the atrial ERPs of six atrial sites were shortened in the paced control group compared to the sham group (†P < 0.05). The shortening of atrial ERPs in Ang-(1-7) treated dogs was attenuated, except at LAA, compared to the paced control group (*P < 0.05). The atrial ERPs of dogs treated by A-779, API-2, wortmannin, L-NAME, or A-71915 showed no differences compared to the paced control group (P > 0.05). (b) At BCL250, the atrial ERPs of six atrial sites were shortened in the paced control group compared to the sham group (†P < 0.05). The shortening of atrial ERPs in Ang-(1-7) treated dogs was attenuated, except at HLA and LAA, compared to the paced control group (*P < 0.05). The atrial ERPs of dogs treated by A-779, API-2, wortmannin, L-NAME, or A-71915 showed no differences compared to the paced control group (P > 0.05). (c) At BCL200, the atrial ERPs of six atrial sites were shortened in the paced control group compared to the sham group (†P < 0.05). The shortening of atrial ERPs in Ang-(1-7) treated dogs was attenuated, except at HLA, compared to the paced control group (*P < 0.05). The atrial ERPs of dogs treated by A-779, API-2, wortmannin, L-NAME, or A-71915 were not different from those for the paced control group (P > 0.05). Abbreviations as in Table 2.
Inducibility and duration of atrial fibrillation
Overall, 30 atrial sites were used to induce AF because six atrial sites were used for the five dogs in each group. Compared with the sham group, the inducible rate of AF increased at various BCLs in the paced control group (P < 0.05). However, the increased inducibility and duration of AF was attenuated in dogs treated with Ang-(1-7) at various BCLs compared with the paced control group (P < 0.05). There was no statistical difference between the control and antagonists + Ang-(1-7) treated groups (P > 0.05). The inducibility and duration of AF was not different among groups treated with A-779, API-2, wortmannin, L-NAME, or A-71915 compared to the paced control group (P > 0.05; Table 3).
The inducibility and duration of AF in each group at various BCLs.
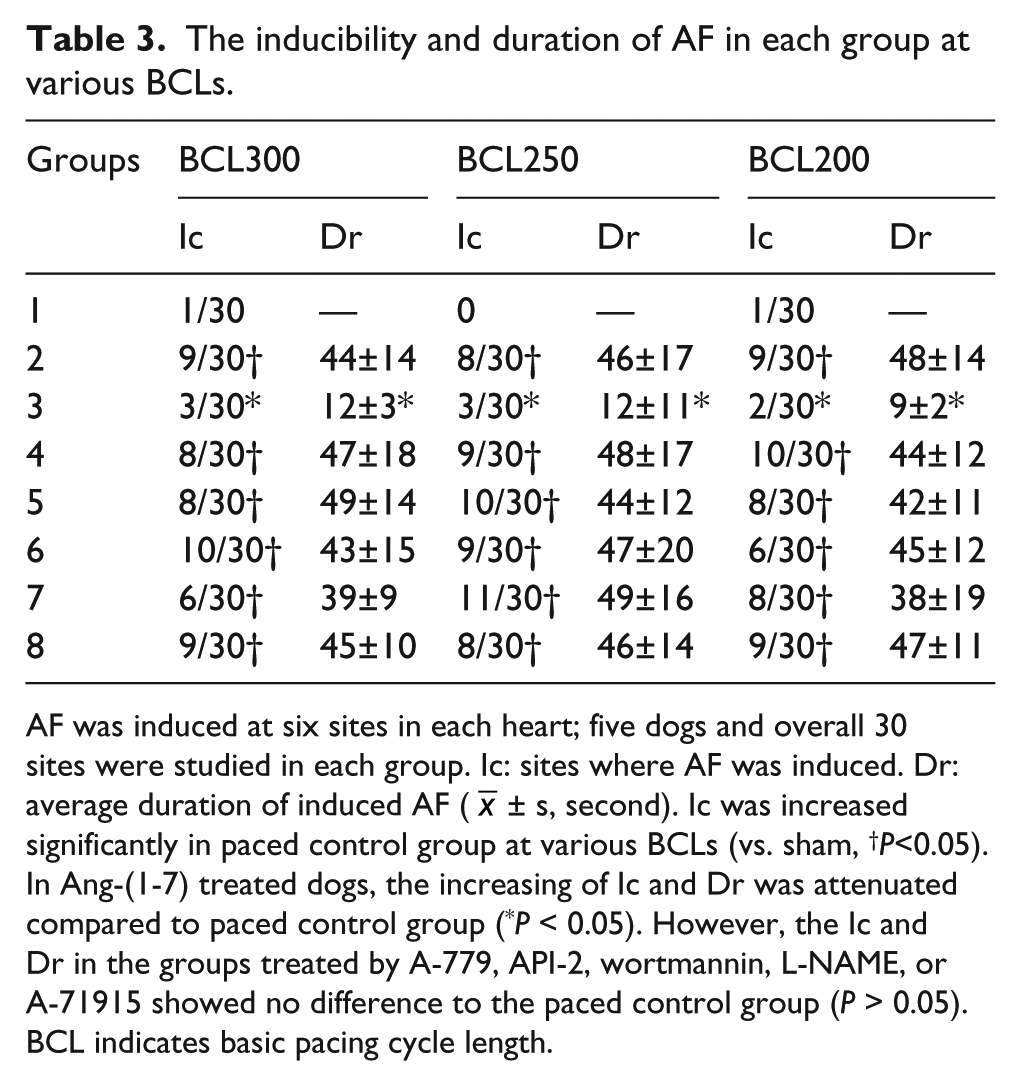
AF was induced at six sites in each heart; five dogs and overall 30 sites were studied in each group. Ic: sites where AF was induced. Dr: average duration of induced AF (
Atrial natriuretic peptide concentration of left atrium
ANP concentrations in the left atrial tissue increased significantly after 2 h of pacing in group 2, compared to those in the sham group (P < 0.05). The elevation of ANP was significantly augmented by Ang-(1-7) in group 3 compared to that in the paced control group (P < 0.05). However, the atrial ANP concentrations were not different among dogs treated with A-779, API-2, wortmannin, or L-NAME compared to the paced control group (Figure 3).

The ANP concentration (ng/mL) of the left atria in each group except group 8. ANP concentrations increased markedly in the paced control group compared to the sham group (P < 0.05). The increased ANP concentration was significantly augmented in group 3 treated by Ang-(1-7), compared to that in the paced control group (P < 0.05). The ANP concentrations in dogs treated by A-779, API-2, wortmannin, or L-NAME were similar to the paced control group (P > 0.05).
Discussion
According to recent research, Ang-II induces the up-regulation of connective tissue growth factors, which can lead to atrial fibrosis in the canine AF model. 10 In the last two decades, Ang-(1-7), which is another heptapeptide formed from Ang-I and/or Ang-II, became a novel focus in the RAS because of its protective roles in the cardiovascular system (i.e. vasodilation and antifibrotic effects).11,12 Furthermore, Ang-(1-7) is known to induce protective signaling in cardiomyocytes. According to a recent study, Ang-(1-7) may stimulate ANP secretion in vitro through a signaling pathway involving the Mas/PI3K/Akt/NO axis. 9 However, the study was performed in isolated, perfused, and beating atria of rats; it is still unknown whether the increased ANP secretion mediates the effects of Ang-(1-7) on atrial electrical remodelling induced by AF in vivo.
Currently, only limited research studies have been conducted on the potential role of ANP in the Mas/PI3K/Akt/NO axis. Therefore, we established a canine model with a 2 h atrial rapid pacing to investigate the effects of the Ang-(1-7) signaling pathway on atrial electrical remodeling and the possible role of ANP. In our study, the electrophysiological parameters (i.e. atrial ERPs and AF inducibility) were determined in vivo, and therefore, the canine model was more appropriate than the murine model. In acute atrial tachycardia dogs, we observed the following: (a) Ang-(1-7) augmented the increases in the atrial ANP concentration induced by atrial pacing; (b) Ang-(1-7) attenuated the shortening of atrial ERPs and reduced the vulnerability of AF through the Mas/PI3K/Akt/NO signaling pathway; (c) ANP may be involved in the mechanism by which Ang-(1-7) prevents atrial tachycardia-induced electrical remodeling.
Effects of Ang-(1-7) on atrial effective refractory periods and atrial fibrillation vulnerability
The electrophysiological changes with AF in the acute atrial tachycardia canine model observed in this study were similar to those in previous studies. 13 In both short and chronic atrial remodeling models, it has been found that the shortening of atrial ERPs was related to the development and maintenance of AF.14,15 In an earlier report, it has been confirmed that in the 4 h rapid pacing canine model, atrial ERPs decreased significantly after 1 h of rapid atrial pacing, reached a minimum after 2 hours, and remained relatively stable thereafter. 14 Correspondingly, in this present study, rapid pacing for 2 h induced the shortening of atrial ERPs and increased AF vulnerability at different sites compared to the sham group. In our previous study, the effects of Ang-(1-7) on ion channels and atrial fibrosis contributed to the effects of Ang-(1-7) on AF vulnerability in dogs with chronic atrial tachycardia.15,16 The densities of ITO and ICaL and the related ion channel subunit gene expression were reduced in paced atria, while Ang-(1-7) prevented decrease in ITO, ICaL, and Kv4.3 (ITO channel subunits) mRNA expression. 15 Ang-(1-7) attenuated the shortening of atrial ERPs and reduced the vulnerability of AF after atrial pacing compared to the paced control group in this study. However, the effects of Ang-(1-7) were abolished in groups treated by different antagonists of the Ang-(1-7) signaling pathway. Therefore, we concluded that Ang-(1-7) prevented atrial electrical remodeling induced by atrial pacing through the Mas/PI3K/Akt/NO signaling pathway.
Ang-(1-7) signaling and the possible role of atrial natriuretic peptide in preventing atrial electrical remodeling
In this study, atrial ANP secretion was increased in the paced control group following atrial pacing for 2 h. Our findings were in accordance with several clinical reports. In a recent study, it was confirmed that atrial and circulating ANP concentrations increased in patients with paroxysmal and persistent AF. 17 ANP was observed to be predictive of AF recurrence up to 12 months post-cardioversion. 18 Therefore, increase in ANP secretion related to atrial tachycardia was observed in both animal and human experiments.
It has been known that the increased atrial wall tension induced by hemodynamic changes (i.e. atrial arrhythmias or heart failure) was the principal stimulus for ANP secretion from atrial cardiomyocytes. 19 Although the hemodynamic parameters (i.e. heart rate and blood pressure) were unchanged by atrial rapid pacing in our study, the increased left atrial volume may be induced by atrial tachycardia. In a latest clinical study, the left atrial volume was enlarged and left atrial function was impaired in patients with paroxysmal AF. 20 In addition, the promotional effects of Ang-(1-7) on ANP secretion after atrial pacing were independent from hemodynamic changes in the present study. Correspondingly, Mercure et al. confirmed that increase in cardiac Ang-(1-7) led to a significant increase in ventricular ANP gene expression in normotensive rats. However, they found that cardiac Ang-(1-7) decreased ANP expression in the hypertensive rat induced by Ang-II infusion, which contradicted with our results. 21 There are two possible reasons to explain this contradiction. First, the expression of ANP mRNA was detected in the rat ventricle in a previous study, 21 while the concentration of ANP was detected directly in the dog atria in our study. It is generally believed that ANP is produced predominantly in the atria. 22 Therefore, further research is needed to confirm the expression of ANP mRNA in the atria using a chronic AF model. Second, left ventricular hypertension induced by Ang-II infusion was considered the stimulus for enhancing ANP secretion in the former study, 21 while the main stimulus was considered atrial distension induced by atrial tachycardia in our study. Although the distension of atrial and ventricular cardiomyocytes is considered the main mechanical stimulus for ANP secretion, 23 some cytokines (i.e. endothelin-1, tumor necrosis factor-α, and interleukin-1) are also powerful stimuli that change according to different pathophysiologic statuses, such as hypertension and AF.24–26 Therefore, increased ANP secretion induced by Ang-(1-7) was complicated in both studies. The function of the cytokines related to inflammatory reaction should be analyzed using different animal pathology models in future studies.
It has been confirmed that Ang-(1-7) can selectively bind to cardiomyocytes and directly prevent cardiac remodeling in vitro and in vivo. However, in Mas receptor-deficient mice, Ang-(1-7) failed to bind to cardiomyocytes.5,27 Furthermore, Ang-(1-7) can activate NO synthase and NO generation mediated by the PI3K/Akt pathway in adult ventricular cardiomyocytes. 28 Therefore, the Mas/PI3K/Akt/NO axis was recognized as the main signaling pathway of Ang-(1-7). In isolated and pacing rat atria, the antagonists of the Mas/PI3K/Akt/NO axis were shown to decrease ANP secretion promoted by Ang-(1-7). 9 In this study, the Mas receptor antagonist (A-779), PI3K inhibitor (wortmannin), Akt/protein kinase B inhibitor (API-2), and NO synthase inhibitor (L-NAME) blocked the effects of Ang-(1-7) on both AF vulnerability and the secretion of atrial ANP after atrial pacing. According to our results, Ang-(1-7) prevented atrial electrical remodeling and enhanced ANP secretion in dogs with atrial tachycardia through the Ang-(1-7)/Mas/PI3K/Akt/NO signaling pathway in vivo. Furthermore, Ang-(1-7) is also proven to prevent cardiac remodeling through other intracellular signaling pathways associated with hypertrophic remodeling, including mitogen-activated protein kinase signaling and the activation of kinase cascades, which lead to the ultimate phosphorylation of terminal kinases (i.e. extracellular regulated protein kinase (ERK), p38, and c-jun N-terminal kinase). 29 Overexpression of Ang-(1-7) in the heart was found to significantly decrease the phosphorylation of c-Src and p38 kinase, but failed to affect ERK1/2 phosphorylation after Ang-II infusion. 21 In our previous study, Ang-(1-7) prevented the increase of ERK1/ERK2 mRNA expression induced by chronic atrial pacing for 14 days. Although Ang-(1-7) was proven to prevent cardiac remodeling through different pathways, we were able to observe that the potentiation of atrial ANP secretion by Ang-(1-7) was mediated by the Mas/PI3K/Akt/NO axis.
The antagonist of the ANP receptor (A-71915) blocked the preventional effects of Ang-(1-7) on atrial electrical remodeling, induced by rapid atrial pacing in our study. Correspondingly, ANP attenuated the shortening of the atrial ERPs and atrial monophasic action potential duration induced by rapid atrial pacing for 7 h. However, ANP was unable to change the expression of the mRNA encoding Kv4.3, L-Ca2+ channel, and Na+ channel. 30 In a previous report, ANP decreased both basal and cAMP pre-stimulated ICaL levels in fetal heart cells and increased ICaL levels in human atrial cells. 31 Miao et al. reported that ANP depressed action potentials and inhibited basal ICaL levels through the cGMP/PDE2/cAMP pathway in developmental cardiomyocytes. 32 According to our previous study, the effects of Ang-(1-7) on ion channels influenced the effects of Ang-(1-7) on atrial electrical remodeling. The densities of ITO and ICaL and the related ionic channel subunits gene expression were reduced in paced atria, while Ang-(1-7) prevented the decrease in ITO, ICaL, and Kv4.3 mRNA expression. 15 Therefore, the effects of Ang-(1-7) on atrial electrical remodeling could be mediated, at least partially, by the biological activities of ANP.
Study limitations
There were several limitations in this study. First, the ventricular rate in this study was not controlled by producing an atrioventricular node block and the ventricular function was not assessed with echocardiography. However, the ventricular rates and blood pressures were not significantly changed by atrial pacing in each group. Therefore, the left ventricular dysfunction induced by ventricular tachycardia might not have influenced atrial remodeling in this acute rapid atrial pacing model. However, the maintenance of physiological atrioventricular conduction is more similar to clinical cases of AF. Second, the electrophysiological study may have slightly affected the concentration of ANP in the left atrium; however, the same methods were consistently applied in all eight groups. Therefore, the electrophysiological parameters would not directly affect the final results of our study. Third, the protein and gene expression related to the ion channel subunits and the Mas/PI3K/Akt/NO pathway were not evaluated in the acute rapid atrial pacing canine model. Currently, researchers have failed to determine changes of the aforementioned protein and gene expressions in acute atrial remodeling.9,30 Therefore, a model with chronic atrial remodeling might be more suitable for evaluating the protein and gene expression related to ion channel subunits and the Mas/PI3K/Akt/NO pathway.
Conclusions
In the atrial tachycardia canine model, Ang-(1-7) prevented acute electrical remodeling and vulnerability of AF by activating the Mas/PI3K/Akt/NO signaling pathway. ANP played an important role in the effects of Ang-(1-7) on atrial electrical remodeling.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Program of Natural Science Foundation of China (grant number 81100131) and the Specialized Research Fund for the Doctoral Program of Higher Education of China (grant number 20111202120008).
