Abstract
Introduction:
Recently we established that pro-inflammatory actions of angiotensin (Ang) II in astrocytes involved Janus kinase 2 (JAK2), signal transducer and activator of transcription 3 (STAT3), and interleukin-6 (IL-6).
Materials and methods:
In our current study, we determined in brainstem and cerebellum whether Ang III also activates STAT3 leading to IL-6 mRNA expression and astrocyte proliferation.
Results:
Ang III induced STAT3 phosphorylation in a concentration- and time-dependent manner. Significant STAT3 phosphorylation was rapid and was maximal within 10 min, and with 100 nM Ang III. The Ang AT1 receptor was shown to mediate this action of Ang III. Ang III also significantly induced IL-6 mRNA expression within an hour, and maximal Ang III-mediated IL-6 mRNA expression occurred in the presence of 100 nM Ang III. Ang III-mediated IL-6 mRNA expression occurred by the interaction of the peptide with the Ang AT1 receptor and was mediated by STAT3. In addition, STAT3 was shown to mediate Ang III astrocyte proliferation.
Conclusions:
These findings suggest that Ang III, similar to Ang II, has pro-inflammatory effects since it induces STAT3 leading to an induction of IL-6 mRNA expression, outcomes that lend relevance to the physiological importance of central Ang III.
Keywords
Introduction
Angiotensin (Ang) II is a pleiotropic peptide that impacts the function of varied cells leading to the regulation of many organ systems. Centrally produced Ang II is well characterized for its ability to modulate blood pressure and cardiac hypertrophy by regulating sympathetic nerve activity, vasopressin release and fluid intake.1,2 The diverse actions of Ang II are mediated by its interactions with Ang AT1 and AT2 receptors, which are coupled to many signaling molecules, including small G proteins, phospholipases, mitogen-activated protein (MAP) kinases, phosphatases, tyrosine kinases, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, and transcription factors.3 –5 Recently, the heptapeptide Ang III, which is also known as Ang 2-8, has received attention due to similarities in its physiological and pathological actions as compared to Ang II. 6 Indeed, Ang III also couples with the Ang AT1 and AT2 receptors and binds with similar affinity as Ang II to these receptors and can activate these receptors with similar efficacy.2,7 Like Ang II, most of the physiological and pathological effects of the peptide are mediated by its interaction with the AT1 receptor with fewer actions mediated via the AT2 receptor. 6
As examples, Ang III induced protein and DNA synthesis as well as collagen synthesis and secretion in a concentration-dependent manner in neonatal cardiac fibroblasts. 8 Ang III via interaction with AT2 receptors induced aldosterone secretion from adrenal glomerulosa. 9 In the central nervous system (CNS), Ang III is shown to be physiologically important causing arginine vasopressin (AVP) release and blood pressure increases, similar to Ang II after intracerebroventricular injection of the peptides.10 –12
Other studies focused on the effects of Ang III on neurons in vivo and showed that the peptide is much more potent than Ang II in inducing responses from paraventricular nucleus receptors from both normal and hypertensive rats. 13 We have recently shown that Ang III interacts with Ang AT1 receptors to activate the extracellular signal-regulated kinase (ERK)1/2 and the c-Jun N-terminal kinase (JNK) MAP kinase pathways leading to astrocyte proliferation.14,15 The actions of Ang III to induce these MAP kinase pathways were very similar to those of Ang II suggesting that this peptide has equipotent potential as compared to Ang II. Huang et al., in a recent study in 2013, showed that inhibition of brain Ang III attenuated sympathetic hyperactivity and cardiac dysfunction in a myocardial infarction model. 16 Thus, these studies and others have established Ang III as a physiologically relevant peptide with a pivotal role in regulating blood pressure. 1
We have determined the pro-inflammatory potential of Ang II in astrocytes by studying its ability to induce the inflammatory mediator interleukin-6 (IL-6).17,18 IL-6 is a pleiotropic pro-inflammatory cytokine 19 whose expression in plasma is elevated through activation of the Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway. 19 JAK/STAT is one of several pathways that translate signals from cell membranes to the nucleus in response to many growth factors and cytokines. Studies have shown that JAK2 activates STAT3 in the presence of Ang II and induces various cell responses and releases many cytokines including IL-6.17,18,20,21 Our findings in astrocytes demonstrated that Ang II interacts with AT1 receptors to induce IL-6 production; the JAK2/STAT3 pathway was shown to mediate this action of Ang II.17,18
The ability of Ang III to induce the STAT pathway leading to IL-6 gene expression is unknown in our system. However, it has been recently reported that Ang III also has pro-inflammatory properties. In rat mesangial and mononuclear cells, Ang III induced the transcription factors nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and activator protein-1 (AP-1), and increased the expression of related genes, such as monocyte chemoattractant protein-1 (MCP-1). 22 Thus, the focus of the present study was to use cultured rat brainstem and cerebellum astrocytes as model systems to determine whether Ang III induced STAT3 phosphorylation leading to an effect on the gene expression of IL-6. We chose to study STAT3 because this early response gene is well known to mediate pro-inflammatory effects and growth promoting effects of Ang II.18,23,24 In addition, astrocytes from brainstem and cerebellum will be investigated because we and others have established that these brain regions have well defined renin-angiotensin system (RAS) components.25 –27
Materials and methods
Materials
Tissue culture supplies such as Dulbecco’s modified Eagles Medium (DMEM)/F12 (1:1), fetal bovine serum (FBS), antibiotic solution, and trypsin/ethylenediamine tetraacetic acid (EDTA), were purchased from VWR (Grand Island, New York, USA) or Fisher Scientific (Milford, Massachusetts, USA). Ang III was obtained from Bachem (Torrance, California, USA). Losartan was a kind gift from Du Pont-Merck (Wilmington, Delaware, USA) while PD123319 was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Stattic, the STAT3 inhibitor, was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). 3 H-thymidine (2000 Ci/mmole) was purchased from MP Biomedicals (Solon, Ohio, USA). The phospho-STAT3 antibody and the STAT3 antibody were purchased from Cell Signaling Technology (Beverly, Massachusetts, USA). Protein measurement supplies, gel electrophoresis and western blotting supplies including bicinchoninic acid (BCA) protein reagents, acrylamide, electrochemiluminescence (ECL) reagents, and nitrocellulose membrane were purchased from either VWR (Piscataway, New Jersey, USA) or Biorad Laboratories (Hercules, California, USA). Supplies to measure IL-6 gene expression such as master mix, probes, cDNA kit and others were purchased from Applied Biosystems (Foster City, California, USA) or Life Technology (Carlsbad, California, USA). All other chemicals were purchased from either VWR international (Suwannee, Georgia, USA), Fisher Scientific (Milford Massachusetts, USA) or Sigma-Aldrich (St. Louis, Missouri, USA).
Preparation of astrocytes
Timed, pregnant Sprague-Dawley rats were obtained from Charles River Laboratories (Wilmington, Massachusetts, USA) and maintained in the Association for Assessment and Accreditation of Laboratory Animal Care (ALAAC) accredited animal facility of Nova Southeastern University. Primary cultures of astrocytes were prepared from the brainstem and cerebellum of 2–3 days old neonatal pups by physical dissociation as previously described.25 –27 Cells were maintained in DMEM/F12 with 10% FBS, 100 μg/ml penicillin, and 100 units/ml streptomycin at 37°C in a humidified CO2 incubator (5% CO2 and 95% air). Cultures were fed every 3–4 days until confluent. Confluent monolayers were placed in DMEM/F12 containing 10 mM HEPES, pH 7.5, 10% FBS and antibiotics and shaken overnight to remove oligodendrocytes and microglia. This method yields an astroglial enriched culture system. Astrocytes were detached with trypsin/EDTA (0.05% trypsin, 0.53 mM EDTA), replated at a ratio of 1:10, and grown to about 85% confluence prior to use. Isolated cells showed a positive immunoreactivity with an antibody against glial fibrillary acidic protein and negative immunoreactivity with markers for neurons, fibroblasts or oligodendrocytes.
Western blot analysis
Cultured brainstem and cerebellum astrocytes were made quiescent by a 48 h treatment with serum-free media. Cells were treated with increasing concentrations of Ang III (0.1 nM to 1 µM) or with 100 nM Ang III for 1–30 min. In other experiments, brainstem and cerebellum astrocytes were treated for 10 min with 100 nM Ang III in the presence and absence of the STAT3 inhibitor (5 µM Stattic) or the AT1 receptor inhibitor (10 µM losartan) or the AT2 receptor blocker (10 µM PD123319). Cells were also treated with the inhibitors alone. Basal and stimulated levels of the proteins were determined in the presence of dimethyl sulfoxide (DMSO) in experiments involving Stattic. Immediately following treatments, cell lysates were prepared by washing cells with phosphate-buffered saline containing 0.01 mM NaVO4 followed by solubilization in supplemented lysis buffer (100 mM NaCl, 50 mM NaF, 5 mM EDTA, 1% Triton X-100, 50 mM Tris-HCl, 0.01 mM NaVO4, 0.1 mM PMSF and 0.6 μM leupeptin, pH 7.4). The supernatant was clarified by centrifugation (12,000 × g for 10 min, 4ºC) and the protein concentration was measured based on the BCA method according to the manufacturer’s instructions (Pierce Biotechnology, Rockford, Illinois, USA).
Proteins previously solubilized in lysis buffer were separated in 10% polyacrylamide gels and transferred to nitrocellulose membranes. Nonspecific binding to the membranes was blocked by incubation with 5% Blotto (5% evaporated milk, 1% Tween-20 in Tris-buffered saline). Subsequently, membranes were sequentially probed with phospho-STAT3 antibody followed by the goat anti-rabbit secondary antibody as previously described.17,18 The immunoreactive bands were visualized using ECL reagents and the data quantified by densitometry. The non-phosphorylated form of STAT3 was used to visualize protein loading as described.17,18
Measurement of DNA synthesis
Subconfluent monolayers of astrocytes growing in 24-well culture dishes were made quiescent by 48 h treatment with serum-free media. Individual wells were then pretreated for 15 min with 5 µM Stattic, a selective inhibitor of STAT3. Subsequently, the cells were treated with 100 nM Ang III for 48 h. For comparative purposes, some wells did not receive any peptides or were treated with DMSO (the vehicle for Stattic) or were only treated with the inhibitor alone. 3 H-thymidine (0.25 Ci/ml culture medium) was added during the last 24 h of treatment. Newly synthesized DNA was precipitated with 5% TCA, dissolved in 0.25 N NaOH, and quantified by liquid scintillation spectrometry as previously described. 28
IL-6 mRNA expression
Quiescent astrocytes were treated with Ang III ranging in concentrations from 0.1 nM to 1 µM or with 100 nM Ang III for 30 min to 24 h. Total RNA was extracted from astrocytes using Tri reagent (Life Technology, Carlsbad, California, USA) according to the manufacturer’s instructions. DNA-free kit was used to eliminate any fractional genomic DNA contamination in the RNA. Total RNA concentrations and purity were determined using a Biorad SmartSpecTM spectrophotometer. Two micrograms of total RNA from each sample were reverse-transcribed into the complementary strand DNA using a high capacity reverse transcription reagent kit (Applied Biosystems, Foster City, California, USA).
Quantitative polymerase chain reaction (qPCR) was performed using TaqMan Universal master mix, and the Taq Man gene expression assay for rat IL-6 all supplied by Applied Biosystems (Foster City, California, USA). Samples were assayed in triplicates in 96 well plates using the StepOnePlus Real-Time PCR Systems from Applied Biosystems. The widely accepted comparative Ct (threshold cycle) method was used to perform relative quantification of qPCR results. 29 An arithmetic formula (fold difference = 2-ΔΔCt) was used to calculate the relative IL-6 mRNA expression in Ang III-stimulated astrocyte cultures as compared to the unstimulated controls, after normalization to levels of the housekeeping control gene, beta-actin. Data are thus expressed as fold change in IL-6 mRNA expression as compared to basal IL-6 mRNA expression in unstimulated cells.
Statistical analysis
All data are expressed as the mean±standard error of the mean (SEM) of six or more experiments, as indicated. T-tests or repeated measures of one-way analysis of variance (ANOVA) with Dunnett’s post-test were used to compare treatment groups with groups treated with no chemicals or those treated with the inhibitors, using PRISM (GraphPad). The criterion for statistical significance was set at p<0.05.
Results
STAT3 activation by Ang III
In previous studies we showed that Ang II rapidly induces STAT3 phosphorylation in brainstem and cerebellar astrocytes.17,18 To determine whether Ang III also activated STAT3 in cultured astrocytes from neonatal rat cerebellum and brainstem astrocytes, cells were incubated with Ang III ranging in concentrations from 0.1 nM to 1 µM. Ang III increased the phosphorylation of STAT3 protein in a concentration-dependent manner (Figure 1). Maximal phosphorylation was observed with 100 nM Ang III in astrocytes cultured from the brainstem (3.0±0.46 fold over basal, Figure 1(a)). Similarly, in astrocytes isolated from the cerebellum, maximal effects were observed with 100 nM Ang III (2.4±0.41 fold over basal, Figure 1(b)). Ang III also stimulated STAT3 phosphorylation in both brainstem and cerebellar astrocytes in a time-dependent manner (Figure 2). In both brainstem (Figure 2(a)) and cerebellum (Figure 2(b)) astrocytes, significant phosphorylation occurred as early as 1 min, with maximal effects observed by 10 min (Figure 2) suggesting that this Ang III effect was rapid and sustained. Comparison of effects in brainstem versus cerebellum astrocytes showed no significant differences in the abilities of cells from these two regions to respond to the peptide. Our findings suggest that brainstem and cerebellum astrocytes cultured under the same conditions do not differ in their responses to Ang III in terms of potency and time course of induction (See Figures 1 and 2).

Effect of concentration on angiotensin (Ang) III-induced signal transducer and activator of transcription 3 (STAT3) protein phosphorylation. Quiescent monolayers of brainstem (1(a)) or cerebellum (1(b)) astrocytes were incubated for 10 min with Ang III ranging in concentration from 0.1 nM to 1 µM. STAT3 protein phosphorylation was measured by Western blot analysis using an antibody specific for the phosphorylated form of STAT3. Protein loading was visualized using a non-phosphorylated STAT3 antibody. The data were analyzed by densitometry and the amount of phosphorylation was calculated as the fold-increase over basal in the presence of vehicle after normalization with values obtained with the STAT3 antibody. Each value represents the mean± standard error of the mean (SEM) of preparations of brainstem and cerebellum astrocytes isolated from 6 or more litters of neonatal rat pups.
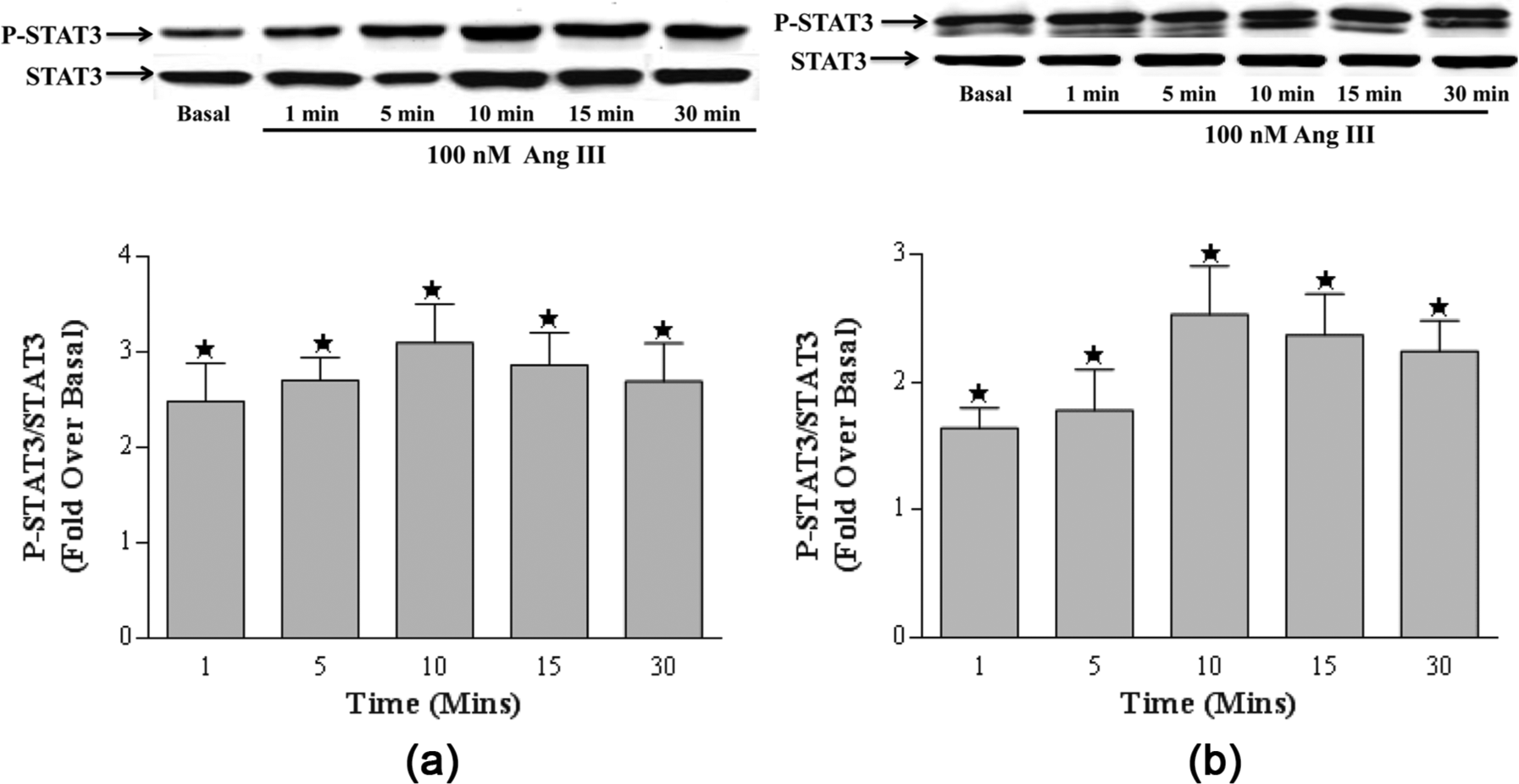
Effect of time on angiotensin (Ang) III-induced signal transducer and activator of transcription 3 (STAT3) protein phosphorylation. Quiescent brainstem (2(a)) or cerebellar (2(b)) astrocytes were incubated with 100 nM Ang III for 1 min to 30 min. STAT3 protein phosphorylation was measured by Western analysis using an antibody specific for the phosphorylated form of STAT3. Protein loading was visualized using a non-phosphorylated STAT3 antibody. The data were analyzed by densitometry and the amount of stimulation was calculated as the fold-increase over basal in the presence of vehicle after normalization with values obtained with the STAT3 antibody. Each value represents the mean±standard error of the mean (SEM) of preparations of brainstem or cerebellum astrocytes isolated from six or more litters of neonatal rat pups. *denotes p<0.05 as compared to basal levels for STAT3 phosphorylation in astrocytes prepared from the brainstem or cerebellum.
To determine the specificity in this Ang III effect, quiescent astrocytes were also pretreated for 15 min with the STAT3 inhibitor Stattic (5 µM), and then stimulated with 100 nM Ang III for 10 min. Pretreatment of astrocytes with Stattic attenuated Ang III-induced STAT3 phosphorylation by ~80% (brainstem) and 100% (cerebellum) suggesting that this was a direct and select effect of Ang III to activate STAT3 (Table 1).
Effects of Stattic, losartan and PD123319 on angiotensin (Ang) III-induced signal transducer and activator of transcription 3 (STAT3).

Denotes p<0.05 as compared to Ang III stimulation of STAT3 phosphorylation. Data was quantified by densitometric analysis and are the results of individual experiments of six or more preparation of astrocytes.
Effects of losartan and PD123319 on Ang III-induced STAT3 phosphorylation
The Ang receptor involved in this effect was determined by treating quiescent brainstem and cerebellar astrocytes with 100 nM Ang III in the presence and absence of the selective AT1 receptor antagonist (losartan, 10 µM) or the selective AT2 receptor antagonist (PD123319, 10 µM). Losartan attenuated by more than 80% and 90% Ang III-induced STAT3 phosphorylation in brainstem and cerebellum astrocytes, respectively (Table 1). The AT2 receptor antagonist was ineffective (Table 1). These findings suggest that Ang III interacts with the Ang AT1 receptor to activate STAT3 in these cells.
Effects of Stattic on Ang III-induced astrocyte DNA synthesis
Previous studies showed that Ang III significantly induced astrocyte DNA synthesis. 14 To determine whether Ang III-induced astrocyte growth involved STAT3, quiescent brainstem and cerebellum astrocytes were pretreated with 5 µM Stattic (STAT3 inhibitor) followed by 100 nM Ang III for 48 h. 3 H-thymidine was added during the last 24 h of treatment to measure astrocyte DNA synthesis, as an index of growth. The results showed that Ang III significantly stimulated brainstem and cerebellum astrocyte growth to a similar extent as we have previously observed (Table 2). We also observed that pretreatment of quiescent astrocytes with 5 µM Stattic completely prevented Ang III-mediated astrocyte growth in both brainstem and cerebellum astrocytes (Table 2). These findings suggest that Ang III activates STAT3 to induced astrocyte DNA synthesis, a measure of growth.
Effects of Stattic on angiotensin (Ang) III-induced DNA synthesis.

Denotes p<0.05 as compared to Ang III-mediated DNA synthesis. Values are calculated based on individual experiments of four or more preparation of astrocytes.
Effects of Ang III on IL-6 mRNA expression
To investigate whether Ang III stimulated IL-6 mRNA expression, astrocytes were stimulated for 45 min with concentrations of Ang III ranging from 0.1 nM to 1 µM with 10-fold increments in each concentration. As shown in Figure 3, Ang III induced IL-6 mRNA expression in a concentration-dependent manner. Maximal effects were observed with 100 nM Ang III (2.0± 0.09 fold over basal in brainstem astrocytes, and 1.9 ±0.3 fold over basal in cerebellum astrocytes). These findings suggest that the peptide had a similar potency in brainstem and cerebellum astrocytes to induce IL-6 mRNA expression.

Effect of concentration on angiotensin (Ang) III-induced interleukin-6 (IL-6) mRNA expression. Quiescent monolayers of brainstem (3(a)) or cerebellar (3(b)) astrocytes were incubated with increasing concentrations of Ang III (0.1 nM to 1 µM) for 45 min. IL-6 mRNA expression was analyzed by quantitative PCR. The amount of Ang III-stimulated IL-6 mRNA expression was calculated as the fold-increase over basal. Each value represents the mean±standard error of the mean (SEM) of preparations of brainstem or cerebellar astrocytes isolated from six or more litters of neonatal rat pups. *denotes p<0.05 as compared to basal levels for IL-6 mRNA expression in astrocytes prepared from the brainstem or cerebellum.
The time course of Ang III-mediated IL-6 mRNA expression was also investigated over a time period ranging from 30 min to 24 h. As shown in Figure 4, Ang III enhanced the expression of IL-6 mRNA in a time-dependent manner. In brainstem astrocytes (Figure 4(a)), there was a gradual increase in IL-6 mRNA expression with significant increases at all-time points up to 6 h. Maximal mRNA expression occurred after 3 h of treatment with 100 nM Ang III (1.7±0.2 fold over basal). Similarly in cerebellar astrocytes (Figure 4(b)), there was significant upregulation in IL-6 mRNA expression as early as 30 min and up to 3 h. The maximal Ang III-induced expression of IL-6 mRNA was 2.6±0.5 over basal observed at the 3 h time point, with effects tapering off thereafter. 100 nM Ang III treatments did not induce IL-6 mRNA expression after 24 h of treatment in both brainstem and cerebellum astrocytes (Figure 4).

Effect of time on angiotensin (Ang) III–induced interleukin-6 (IL-6) mRNA expression. Quiescent monolayers of brainstem (4(a)) or cerebellar (4(b)) astrocytes were incubated with increasing concentrations of Ang III for 30 min to 24 h. IL-6 mRNA expression was analyzed by quantitative PCR. The amount of Ang III-stimulated IL-6 mRNA expression was calculated as the fold-increase over basal. Each value represents the mean±standard error of the mean (SEM) of preparations of brainstem or cerebellar astrocytes isolated from six or more litters of neonatal rat pups. *denotes p<0.05 as compared to basal levels for IL-6 mRNA expression in astrocytes prepared from the brainstem or cerebellum.
Effects of Stattic, losartan and PD123319 on Ang III-mediated IL-6 mRNA induction
To investigate whether STAT3 mediates Ang III-induced IL-6 mRNA expression, quiescent brainstem and cerebellar astrocytes were pretreated for 15 min with the STAT3 inhibitor (Stattic, 5 µM) followed by stimulation with 100 nM Ang III. Our results demonstrated that Stattic significantly downregulated Ang III-mediated IL-6 mRNA expression by 80% and 60% in brainstem and cerebellum astrocytes, respectively (Table 3), suggesting that STAT3 mediates Ang III-mediated IL-6 mRNA induction.
Effects of Stattic, losartan and PD123319 on angiotensin (Ang) III-induced interleukin-6 (IL-6) mRNA.

Denotes p<0.05 as compared to Ang III stimulation of signal transducer and activator of transcription 3 (STAT3) phosphorylation. Data are the results of individual experiments of six or more preparation of astrocytes.
Cells were also treated with 100 nM Ang III in the presence and absence of the selective AT1 receptor antagonist (losartan, 10 uM) or the selective AT2 receptor blocker (PD123319, 10 uM) to determine whether Ang III stimulation of IL-6 mRNA occurred through interaction at one of the Ang receptors. Pretreatment with losartan attenuated the expression of IL-6 mRNA by about 70% and 100% in brainstem and cerebellar astrocytes, respectively. PD123319, the AT2 receptor antagonist was ineffective in preventing Ang III-mediated increases in IL-6 mRNA expression in both brainstem and cerebellum astrocytes (Table 3). These findings suggest that Ang III interacts with the Ang AT1 receptor leading to STAT3 induction and IL-6 mRNA expression.
Discussion
In recent years it has become clear that the central RAS is important in many pathophysiological conditions including hypertension, congestive heart failure, myocardial infarction, and diabetic nephropathy.1,2 The importance of neurons in the brain is well established, but in recent years the role of other cells such as astrocytes are now appreciated. Specifically for the functions of the central RAS, astrocytes have been shown to be important in the production of angiotensinogen, the precursor proteins for Ang II, Ang III and other physiologically relevant peptides produced by this system. 30 Thus, it is important that we understand how peptides produced by this system are regulated and the intracellular signaling pathways that govern the actions of these peptides in astrocytes. The central effects of Ang II are well known.1,2 Ang III has recently received attention as an important peptide produced by RAS, and even though some controversy still exists in terms of the effects of Ang III compared to Ang II, it is clear that the peptide is physiologically important particularly in the control of blood pressure, fluid homeostasis, and AVP release. 6
Most studies delineating the effects of Ang III have focused on the binding affinity of the peptide to AT1 receptors or measured the effects of the peptide to modulate blood pressure11,12,31 with few studies delineating the intracellular mediators of these Ang III effects. The earlier signaling pathways governing the effects of Ang III have been the recent focus of our laboratory. In cultured rat astrocytes, we showed that Ang III stimulated Ang AT1 receptor to induce the phosphorylation of ERK1/2 and JNK MAP kinase pathways with the same potency as Ang II.14,15 These pathways were also shown to mediate Ang III-induced astrocyte proliferation suggesting, at least in astrocytes, that these MAP kinase pathways mediate Ang III effects. In neonatal cardiac fibroblasts Ang III induced protein synthesis, DNA synthesis and collagen synthesis/secretion in a concentration-dependent manner. 8 Blume et al. 32 demonstrated in conscious rats that Ang III induced the expression of the transcription factors c-Fos, c-Jun and Krox-24, effects mediated by the AT1 receptor.
In the current study we explored the ability of Ang III to activate STAT3, a member of the JAK/STAT family of proteins. This pathway is thought to be important in development and homeostasis 33 and is a characteristic pathway via which several cytokines elicit their responses. JAK/STAT signaling mediates several Ang II-induced physiological and pathological responses,20, 34,35 effects that apparently involve an increase in angiotensinogen and IL-6 levels.20,23,36 IL-6 exhibits a variety of physiological actions including induction of cell growth, cellular development, and inflammation; it is also involved in several disease processes as well.19,37 The role of Ang III in inducing IL-6 levels is unknown. However, a link between Ang III and inflammation has been proposed. As previously mentioned, it has been shown in rat mesangial and mononuclear cells that Ang III has pro-inflammatory actions. 22
Here we show for the first time in astrocytes that Ang III induces STAT3 phosphorylation leading to an increase in IL-6 mRNA expression. This effect is mediated by the interaction of Ang III with the Ang AT1 receptor. Most importantly, our results showed that the effects of Ang II and Ang III to induce STAT3 and IL-6 mRNA levels were equipotent since the magnitude of the Ang III effects observed in our current samples were comparable to our previously published Ang II results for Ang II effects on IL-6 mRNA expression.17,18 Future studies will be done to determine whether Ang III induces IL-6 protein levels and IL-6 protein secretion in a similar manner and pattern as Ang II. Further, astrocytes isolated from the brainstem and cerebellum, were equally responsive to Ang III exhibiting similar sensitivity to the effects of the peptide to induce both STAT3 protein and IL-6 mRNA expression.
Multiple members of the STAT family of transcription factors are known to mediate Ang II effects23,35 but the ability of Ang III to induce STAT family of transcription factors is unknown and the reason for our studies. We selected STAT3 for our studies with Ang III because it protects against apoptosis and enhances cell proliferation. 38 As we have shown previously, 14 and as shown in Table 3, 100 nM Ang III induces cell proliferation in cerebellar and brainstem astrocytes. Moreover, inhibition of STAT3 prevented Ang III-mediated astrocyte proliferation. We have previously shown that Ang III stimulated the proliferation of rat astrocytes through the Ang AT1 receptor, an effect mediated by the ERK1/2 and JNK MAP kinase pathways.14,15 These findings suggest that, along with the other intracellular signals mentioned, STAT3 is also involved in Ang III growth-promoting effects as well as Ang III inflammatory actions. Further, these studies also show that both Ang II17,18 and Ang III can cause similar effects in astrocytes (increase DNA synthesis), and may use similar pathways, here the JAK2/STAT3 pathway, to achieve these effects. The mechanism responsible for astrocyte proliferation caused by Ang III-induced STAT3 activation is unknown. However, STAT3-mediated, IL-6-dependent T cell proliferation occurred through prevention of apoptosis suggesting a possible pathway by which STAT3 may promote astrocyte proliferation. 39 Future studies will be geared at determining the mechanism(s) by which this peptide induces STAT3 leading to an increase in DNA synthesis.
It is apparent that activated astrocytes contribute to central inflammatory effects and tissue repair as a participant in the local innate immune response triggered by a variety of insults.40,41 Astrocytes display an array of receptors involved in innate immunity, and astrocyte activation leads to cellular proliferation and secretion of a number of soluble mediators, including IL-6, which impacts immune responses.42 –44 Thus, it is important to study the pathways that lead to local immune responses (here, IL-6 production and astrocyte proliferation) in the brain. Moreover, it has been shown that STAT3 is a key regulator of reactive astrocytes in the healing process after spinal cord injury also suggesting a role for this mediator in central inflammatory processes involving astrocytes. 45 In this study, we are using an astrocyte-enriched culture system that to some extent is contaminated with microglia. Since cultured microglia express components of the RAS, and STAT3 is present in these cells, it is possible that the astrocyte response we detect in this study is influenced by the small numbers of microglia.46,47
Over the last two decades, the RAS has become much more complex with the identification of novel peptides such as Ang III that exhibit biological activity. Ang III interacts at the same receptor as Ang II and is produced from the same precursor molecule angiotensinogen by a series of reactions which included conversion of Ang II to Ang III by the enzyme aminopeptidase A. 6 This has led to the discovery of a number of physiological effects that are similar between Ang II and Ang III. Actually, it has been theorized that in the central nervous system it is Ang III that is the active peptide not Ang II.12,48,49 However, this theory has been invalidated due to a number of studies that have shown otherwise.50 –52
Ang II role in inflammation has been investigated in a number of systems;53 –56 however, the role of Ang III in inflammatory processes is unknown. Thus, it is important that we delineate whether Ang III also plays a key role in inflammation since a dysregulated central RAS is thought to be involved in inflammatory processes due to the significant increase in the production of brain angiotensinogen during an inflammatory response. 57 In addition, during cold injury angiotensinogen-deficient mice exhibits an attenuated glial fibrillary acidic protein (GFAP) expression and decreased laminin levels in astrocytes; these deficiencies result in an incomplete reconstitution of the blood brain barrier. 58 These findings suggest that during inflammation or physical trauma to the brain, there is dysregulation in the RAS which may lead to astrocyte proliferation and activation. Activated astrocytes, as previously mentioned, are important in the body’s response to inflammation. Most importantly, this dysregulated RAS may involve effects of Ang II and Ang III as well.
Indeed in our previous studies, we have shown that Ang II increases mRNA expression and release of the pro-inflammatory cytokine IL-6 from astrocytes, effects that were mediated by the JAK/STAT pathway. 17 Here we show for the first time that Ang III also acts in a similar manner inducing STAT3 phosphorylation and IL-6 mRNA expression. STAT3 and the AT1 receptor were shown to mediate this Ang III effect. It is significant that Ang III induces IL-6 mRNA levels since this cytokine is a known mediator of inflammation.18,19,59 Since Ang III induces IL-6 expression in a similar manner as Ang II, and these two peptides other physiological effects are mostly similar, it is plausible that both peptides may promote inflammatory processes in the brain via this inflammatory cytokine.
Conclusion
Our studies are important in delineating RAS pathways that may be important in inflammatory responses in areas of the central nervous system known to express Ang receptors and associated signaling pathways. It is important to establish an association between inflammatory pathways and the RAS since these systems on their own are associated with diseases such as stroke, Alzheimer’s disease, heart failure, and hypertension. Deleterious interactions between these two systems could make these conditions even more prevalent and validate the importance of studying connections/crosstalk between these systems in the brain in particular in glial cells.
Footnotes
Acknowledgements
The authors are grateful to David Gazze for his review of the manuscript.
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by a President’s Faculty Research and Development Grant from Nova Southeastern University (grant number 335465).
