Abstract
Hypothesis/introduction:
Preeclampsia is associated with alterations in the maternal renin–angiotensin–aldosterone system (RAAS), increased blood pressure (BP), and cardiovascular risk in the offspring. We hypothesized that preeclampsia is associated with alterations in the RAAS in the offspring that persist into adolescence.
Materials and methods:
We compared components of the circulating (n = 111) and renal (n = 160) RAAS in adolescents born prematurely with very low birth weight (VLBW) of preeclamptic (PreE) and normotensive (NoHTN) pregnancies. Multivariable linear regression was used to evaluate potential confounding and intermediate variables. Analyses were stratified by sex.
Results:
Adjusting for race and antenatal steroid exposure, male offspring of PreE mothers had higher circulating aldosterone than those of NoHTN mothers (adjusted mean difference = 109; 95% confidence limits: −9, 227 pmol/L). Further adjustment for current BMI attenuated this difference (adjusted mean difference: 93; 95% confidence limits: −30, 215 pmol/L).
Conclusion:
Among male preterm VLBW infants, maternal preeclampsia is associated with increased circulating aldosterone level in adolescence, which appears to be mediated in part by higher BMI.
Introduction
Preeclampsia, a pregnancy specific syndrome of hypertension and proteinuria, is a source of maternal and fetal morbidity and mortality, and a risk factor for future cardiovascular disease in both the mother and her offspring.1–3 While the pathophysiology of preeclampsia remains unclear, reported alterations in the renin–angiotensin–aldosterone system (RAAS) include decreased aldosterone levels, 4 enhanced vascular sensitivity to angiotensin II (Ang II), 5 increased Ang II content in the placental chorionic villi, 6 reduced circulating levels of angiotensin-(1-7) (Ang-(1-7)), 7 and circulating autoantibodies that stimulate the Ang II type I receptor. 8 The heightened Ang II sensitivity of preeclamptic pregnancies persists into the early postpartum period, even after the hypertension resolves, and might contribute to the increased risk of future vascular disease in these women. 9 In addition, Ang II and preeclampsia decrease placental activity of 11β-hydroxysteroid dehydrogenase type 2, the enzyme that protects the fetus from elevated maternal cortisol.10,11 In utero exposure of ovine fetuses to exogenous glucocorticoids leads to changes in the RAAS that predispose the offspring to elevated blood pressure as adults.12,13
Early-onset preeclampsia resulting in preterm delivery tends to be more severe than when onset is later and, thus, more likely to cause alterations in the developing RAAS.14,15 Additionally, early onset preeclampsia is associated with placental DNA hypomethylation compared to late onset preeclampsia. 16 These altered placental DNA methylation patterns have been linked to cortisol-signaling genes, providing a potential link between early onset preeclampsia and increased fetal exposure to glucocorticoids. 17
The present study evaluated the hypothesis that preeclampsia is associated with alterations in the RAAS in offspring that persist into adolescence, predisposing these individuals to cardiovascular disorders. To test our hypothesis, we evaluated adolescents with very low birth weight (VLBW; <1500 g), among whom early onset preeclampsia in the mother was a frequent antecedent of preterm delivery. Because sex differences have been described in models of fetal programming and the RAAS, 18 we also hypothesized that associations between preeclampsia and the RAAS components might differ between males and females.
Materials and methods
Study participants
The study sample was derived from a cohort of 479 consecutive singleton births with VLBW who were born between 1 January 1992 and 30 June 1996 at a regional perinatal center (Forsyth Medical Center), had no major congenital malformation, and returned to the medical center for evaluation at one year corrected age (Figure 1). Of these children, 193 (40%) were evaluated at 14 years of age. Thirty-three participants were excluded from the analysis (2 twins, 1 chromosomal abnormality, 1 polycystic kidney disease, 1 severe cerebral palsy, 16 born to hypertensive mothers who did not have preeclampsia, and 12 with no blood or urine samples). Of the 160 participants whose data are included in the study of the renal RAAS (i.e. participants who provided a urine specimen), 111 (69%) provided a blood sample to evaluate the circulating RAAS.
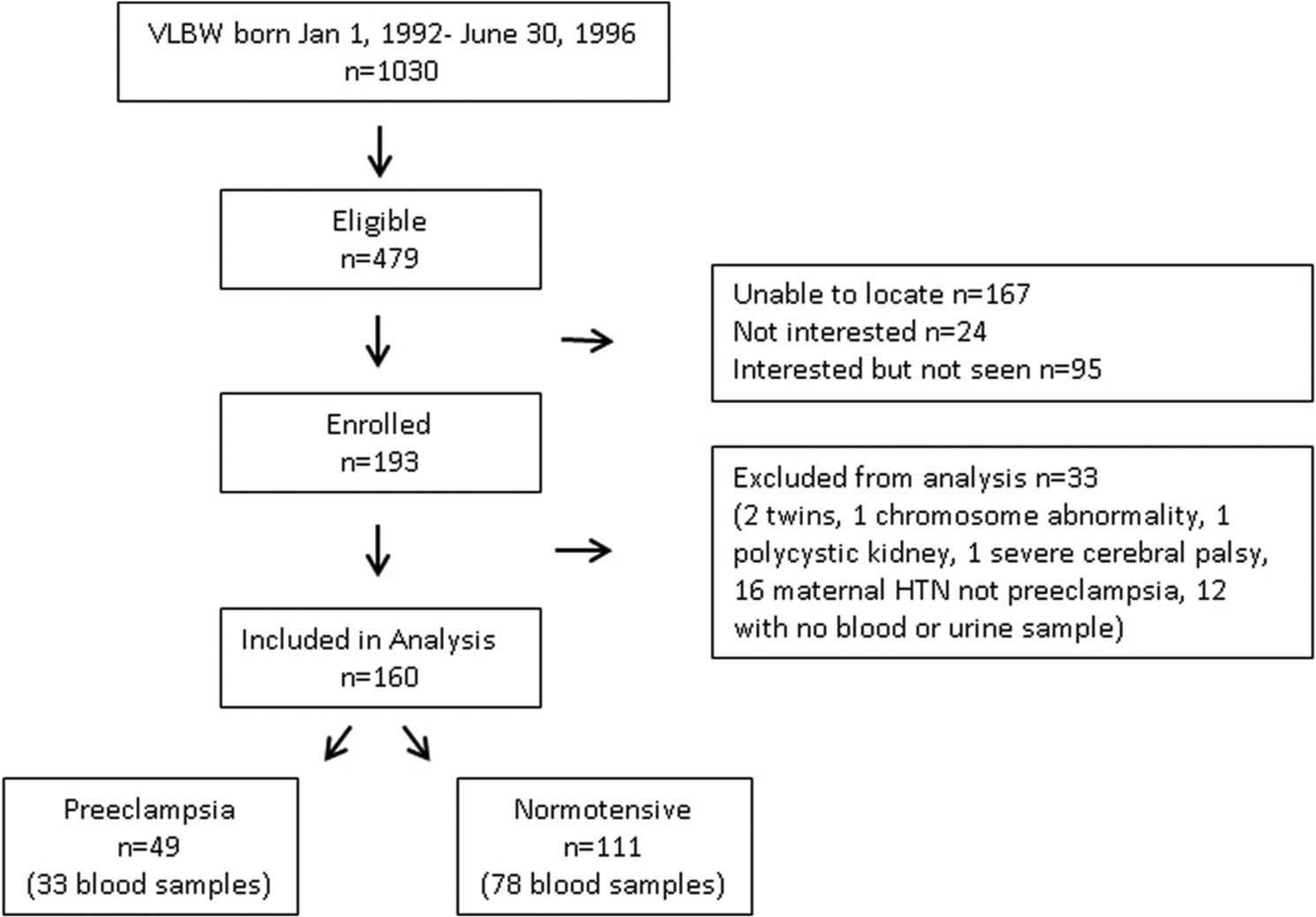
Progress of the participants through the observational follow-up study.
Definitions
We reviewed medical records and delivery log books to identify participants whose mothers were diagnosed as having preeclampsia/eclampsia, pregnancy-induced hypertension, or chronic hypertension. Maternal pre-pregnancy body mass index (BMI) was calculated from pre-pregnancy weight recorded in the medical record and height reported by the mother at the follow up visit. We defined exposure of the offspring to antenatal steroids as maternal treatment with betamethasone or dexamethasone. We estimated gestational age based on the mother’s last menstrual period, when available and confirmed with an early ultrasound. Otherwise, we used an estimate based on an early prenatal ultrasound, or, if no prenatal estimate was available, a clinical assessment of the infant was used. As an index of fetal growth, the birth weight z-score was calculated using US natality data. 19 Race of the participant was categorized as black or non-black, based on parents’ report. Pubertal maturation status was assessed using self-completed questionnaires based on Tanner staging for breast development and pubic hair in females and genital development and pubic hair in males. 20
Measurements
Participants were seen for three visits while 14 years of age as part of a larger study of cardiovascular and metabolic outcomes among VLBW adolescents. Measurements made during the third visit are reported here. After the child removed his or her shoes, height was measured in triplicate using a wall-mounted stadiometer. With the child in light clothing, weight was measured in triplicate using a digital platform scale. A random non-fasting venous sample was obtained by venipuncture, sitting upright, 30 minutes after application of 4% lidocaine for markers of serum/plasma RAAS. A random clean catch urine sample was obtained for the assessment of angiotensin peptides and creatinine. Blood pressure (BP) was measured in the right arm with a mercury sphygmomanometer by nursing staff certified in BP measurement. Guidelines established by the National High Blood Pressure Education Program Working Group on Hypertension Control in Children and Adolescents were followed. 21 The average of three measurements was used to estimate the auscultatory systolic and diastolic BP.
Laboratory
Serum aldosterone was measured by radioimmunoassay (RIA) using antibody-coated tubes (Diagnostic Products, Los Angeles, CA). The assay sensitivity is 27.74 pmol/L. The intra-assay and inter-assay coefficients of variation are 5% and 6.6%, respectively.
Plasma renin activity, defined as the rate of angiotensin I (Ang I) generation from the endogenous substrate angiotensinogen, was measured in plasma treated with EDTA and PMSF to prevent the degradation of the generated peptide. The renin product Ang I was quantified by RIA using a clinical human renin kit (DiaSorin, Stillwater MN). The minimal detectable activity is 77.2 pmol/L/h of incubation at 37°C. The intra-assay and inter-assay coefficients of variation are 10% and 12%, respectively.
For determination of angiotensin peptides, blood was collected in an EDTA tube containing a cocktail of protease inhibitors including 0.44 mM 1,20-o-phenanthroline monohydrate (Sigma, St. Louis, MO), 0.12 mM pepstatin (Peninsula Labs, Belmont CA), and 1 mM Na p-hydroxymercuribenzoate (Sigma, St. Louis, MO), centrifuged, and plasma removed. Urine was collected in acid and immediately frozen at −80°C. Plasma and urine were extracted using Sep-Pak columns activated with 5 mL sequential washes of a mixture of ethanol:water:4% acetic acid (83:13:4), methanol, ultra-pure water, and 4% acetic acid. After the sample was applied to the column, it was washed with ultrapure water and acetone and eluted with 3.3 ml washes of ethanol:water:4% acetic acid. The eluent was completely evaporated, reconstituted in assay buffer, and measured for Ang II and Ang-(1-7) by separate RIAs as previously described. 22 The minimum detectable levels of the assays were 0.8 pmol/L for Ang II and 2.8 pmol/L for Ang-(1-7). The intra-assay and inter-assay coefficients of variation are 12% and 22% for Ang II, and 8% and 20% for Ang-(1-7). Urinary concentrations of both peptides were normalized to urinary creatinine levels. Creatinine was determined by a modified Jaffe reaction. One value for urine Ang II and Ang-(1-7), 37 for plasma Ang-(1-7), two values for plasma renin activity, and four values for serum aldosterone were below the minimum detectable level of the assay.
Statistical analysis
We compared children born to mothers with preeclampsia (PreE) and children born to mothers who did not have preeclampsia or hypertension during pregnancy (NoHTN) in unadjusted analyses using the two-sample t-test or chi-square test. We evaluated the relationship of maternal preeclampsia to RAAS-related outcomes in the offspring using multiple linear regression models. Laboratory assay values below the minimum detectable levels were assigned one-half those levels; logarithmic transformation was then applied to all values to improve distributional characteristics for the t-test and multiple regression analyses for a subset of assays (urinary Ang II, Ang-(1-7), their ratio, and plasma renin activity, renin–aldosterone ratio, Ang-(1-7) and the ratio of Ang II/Ang-(1-7)). To assist in the assessment of this approach to using detection limit information and log transformation on our study data, we compared the t-test results to those using the Wilcoxon rank-sum test, which is preferred among nonparametric approaches to singly-censored data. 23 Potential confounding factors were assessed in relation to maternal preeclampsia and RAAS-related outcomes – birth weight, gestational age, race, antenatal steroid treatment of the mother, and maternal pre-pregnancy body mass index – using the two-sample t-test, chi-square test, or Spearman correlation. Potential confounders that were associated with both maternal preeclampsia and at least one RAAS-related outcome at a significance level of p < 0.2 (two-sided) were included in multiple regression modeling. Maternal pre-pregnancy BMI was not included in further modeling as limited data were available for this variable (24 males and 59 females). For evaluation of the maternal preeclampsia effect as potentially mediated through intermediate variables (cesarean delivery, birth weight z-score, and BMI at age 14 years) we entered these variables separately into models that contained the confounding variables, race, and antenatal steroids exposure. Primary effects are reported with and without adjustment. As we hypothesized sex differences, analyses were stratified by sex; p ≤ 0.05 (two-sided) was considered statistically significant in hypothesis tests.
The following factors were used to convert conventional units to SI units (pmol/L): aldosterone (molecular weight, mw = 360), ng/dL × 27.74; Ang I (mw = 1295), pg/mL × 0.772; Ang II (mw = 1045), pg/mL × 0.957; Ang-(1-7) (mw = 899), pg/mL × 1.112; creatinine (mw = 113), mg/dL × 88.4 (µmol/L).
This study was approved by the Wake Forest School of Medicine Institutional Review Board. Informed consent was obtained from parents or legal guardians and participants gave assent.
Results
The participants whose data are included in the study had lower gestational age and greater birth weight z-score than those eligible but not included (Table 1). Participants from PreE mothers were more likely to have been born by cesarean delivery (p < 0.001) and had lower birth weight z-scores (p < 0.001). Females from PreE mothers were more likely to be exposed to antenatal steroids (p=0.002) and be non-black (p = 0.011) compared to females from mothers with NoHTN. Male offspring of PreE mothers had higher gestational age (p<0.001), and their mothers had higher pre-pregnancy BMI (p = 0.042) (Table 2).
Comparison of neonatal characteristics of adolescents included with those not included, expressed as mean ± SD or N (%).

A subset of 111 participants gave a blood sample.
Characteristics of participants at birth expressed as mean ± SD or N (%).

Provided a blood and or urine sample: entire group: No HTN n = 70, PreE n = 29; Males: No HTN n = 49, PreE n = 20; Females: No HTN n = 41, PreE n = 18.
At age 14, male participants in the PreE group had higher systolic BP (t-test p = 0.047), BMI (p = 0.012), renin activity (p = 0.053), level of circulating aldosterone (p = 0.041, Figure 2) and a higher percentage of Tanner stage 5 in pubic hair (p = 0.018) (Table 3). Female participants in the PreE group had higher levels of urinary Ang II (p = 0.052). Findings were similar using a nonparametric approach to significance testing (results not shown).

Serum aldosterone levels in very low birth weight adolescent male offspring of preeclamptic and normotensive pregnancies. Horizontal lines denote medians.
Comparison of offspring of normotensive and preeclamptic pregnancies at age 14 years expressed as mean ± SD.

Data missing for 1 male in No HTN group.
Log-transformed for analysis.
Table 4 summarizes the results of multivariate analyses of RAAS-related outcomes that reached or nearly reached statistical significance in univariate analyses. Adjustment for race or exposure to antenatal steroids had little effect on group differences in renin activity and aldosterone in males while adjustment for antenatal steroid exposure attenuated group differences in urinary Ang II (females). Adjustment for maternal pre-pregnancy BMI, (available for only 24 males and 59 females) attenuated the group difference in aldosterone levels in males (PreE-NoHTN adjusted mean difference (95% CI); 79 (−127, 286) pmol/L, p = 0.43). Adjustment for both race and exposure to antenatal steroids attenuated group differences in aldosterone (males) and urinary Ang II (females). We tested the influence of the variables cesarean delivery, birth weight z-score, and participant BMI at age 14 years using models that included the confounding variables (antenatal steroid exposure and race). Adjustment for cesarean delivery attenuated group differences in log of plasma renin activity (0.24 (−0.38, 0.86) pmol Ang I/L/h; p = 0.44), aldosterone (86 (−54, 227) pmol/L; p = 0.22) and log of urinary Ang II (0.35 (−0.15, 0.86) pmol/g creatinine; p = 0.17). Adjustment for birth weight z-score attenuated renin activity and urinary Ang II but not aldosterone levels. Adjustment for participant BMI at age 14 years attenuated differences in aldosterone but not renin activity or urinary Ang II.
Mean differences (preeclampsia-normotensive, 95% confidence intervals and p values) for multivariate analyses relating preeclampsia to outcome measures of the renin–angiotensin–aldosterone system (RAAS) in adolescent offspring who were born prematurely.

Discussion
This is the first study to compare elements of the circulating and renal RAAS in VLBW adolescent offspring of PreE pregnancies and offspring of normotensive pregnancies. Our results suggest increased aldosterone levels in male adolescents born to PreE mothers. We also observed trends for increased circulating renin activity in males and urinary Ang II in females. The group differences in aldosterone levels in males persist when adjusted for intrauterine growth but are attenuated by adjustment for the adolescent’s current BMI, consistent with the possibility that increased adiposity among males born to PreE mothers contributes to increased aldosterone. This finding of higher aldosterone levels during adolescence may predict the development of hypertension, 24 and would be consistent with the higher blood pressure in offspring of PreE mothers as seen in our study and others. 15
Race, antenatal steroid exposure, cesarean delivery, and RAAS
When examining the effects of potential confounding variables, we found adjustment for race slightly attenuated group differences among males whereas adjustment for antenatal steroid exposure attenuated group differences in females. This finding in females is consistent with the possibility that fetal exposure to glucocorticoids has a programming effect on the RAAS. Adjustment for cesarean delivery attenuated group differences in aldosterone levels. One possible explanation is that in our cohort cesarean delivery is a marker for more severe preeclampsia. It is less likely that cesarean delivery leads to higher aldosterone levels, since cesarean birth has been associated with lower blood pressure in preterm children, 25 and lower renin concentration and plasma renin activity in umbilical cord blood compared to vaginal birth.26–29
Fetal growth and the RAAS
Aldosterone, which is elevated in normal pregnancies and reduced in pregnancies complicated by preeclampsia, contributes to placental growth through stimulation of placental trophoblasts. 27 Recently, Gennari-Moser et al. found that vascular endothelial growth factor (VEGF) stimulates aldosterone synthesis independently and in synergy with Ang II; and soluble fms-like tyrosine kinase-1 (sFLT-1), a VEGF inhibitor that is elevated in preeclampsia, reduces aldosterone levels. 28 They hypothesized that the elevated levels of sFLT-1 associated with preeclampsia inhibit the VEGF-induced increase in aldosterone that normally occurs in pregnancy. Low maternal aldosterone levels also are associated with decreased plasma volume expansion and idiopathic fetal growth restriction in normotensive women. 29
Franco et al. described activation of the RAAS in children who were born small for gestational age at term compared to those born appropriate for gestational age and Martinez-Aguayo et al. reported an inverse association of aldosterone to birth weight standard deviation score in a cohort of mostly full term children.30,31 We found that adjustment for fetal growth restriction attenuates group differences in circulating renin activity among males and urinary Ang II levels among females. However, poor intrauterine growth did not explain the preeclampsia–increased aldosterone relationship in males.
Obesity and the RAAS
We and others have described increased BMI in offspring of PreE women.3,32 In adults, obesity is associated with increased aldosterone levels and renin activity which are reduced with weight loss. 33 A local RAAS exists in adipose tissue and contributes to circulating angiotensinogen levels, and weight loss is associated with a decrease in RAAS activity in adipose tissue.34,35 Additionally, aldosterone stimulates the differentiation of mouse 3T3-L1 cells into mature adipocytes. 36 Thus, there is biological plausibility for the possibility that differences in BMI explain the differences in aldosterone that we observed. However, we have no explanation as to why adjustment for current BMI had little effect on group differences in circulating renin activity in males.
Sex differences
Fetal responses to the stressful intrauterine environment of preeclampsia may be sex specific. 37 Studies in animal models suggest that the hormonal status of offspring might alter the phenotype associated with adverse intrauterine exposures. In rats, exposure to reduced uterine artery perfusion, a model of intrauterine growth restriction, results in hypertension in adult males. Gonadectomy in males reduces blood pressure while gonadectomy in females increases blood pressure.38,39 Furthermore, these sex-specific differences appear to be influenced by the RAAS. 18 Additionally, Lu et al., using a mouse model of preeclampsia induced with infusion of sFLT-1, describe sex-specific programming of elevated BP in male offspring. 40 Our finding suggests that alterations of the RAAS programmed by the intrauterine environment of early preeclampsia are sex specific.
Genetic polymorphisms, epigenetics, and preeclampsia
Genetics of the mother, father, and fetus are known to influence risk of preeclampsia. In a Swedish population-based study, 55% of the liability of preeclampsia was explained by genetic factors (35% maternal); however, 32% of the liability was unexplained by genetics or shared environment. 41 Many candidate genes and polymorphisms have been studied in their relationship to preeclampsia. Polymorphisms for the aldosterone synthase gene (CYP11B2), which influences aldosterone levels, are of importance in preeclampsia as gain-of-function variants reduce the risk of preeclampsia. 42 Epigenetic changes or changes in DNA methylation patterns have also been described in the pathogenesis of preeclampsia. MicroRNAs play a role in the development of preeclampsia and may influence gene expression in an epigenetic fashion via DNA methyltransferase activity. 43 Additionally, epigenetic mechanisms have been described for many animal models in which intrauterine exposures result in effects on the offspring. 44
Comparison of RAAS values
The values we obtained in our study of the circulating RAAS are comparable to those described by others. The levels of serum aldosterone, plasma renin activity and aldosterone to renin ratio that we report are in the normal range for normotensive adults. 45 Compared to normotensive children (ages 4–16 years) our cohort’s plasma renin activity was similar while serum aldosterone and aldosterone-to-renin ratio were higher. 46 Simoes e Silva et al. assessed the RAAS profiles in childhood hypertension (essential and renovascular) and the plasma renin activity reported for children with renovascular disease is comparable to our findings. 47 The plasma levels of Ang II in the present study are also comparable to controls and patients with essential hypertension, but lower than those with renovascular disease. 47 Additionally, plasma Ang-(1-7) levels are lower in the subjects in our study than all groups of children in the report by Simoes e Silva et al., 47 those of normotensive adults, 48 and a control group of young women. 7 While there are differences in study populations and laboratory methods, when compared to these previous reports, the VLBW adolescents we studied had increased serum aldosterone, aldosterone-to-renin ratio, and plasma renin activity, and lower plasma Ang-(1-7).
There are fewer clinical studies that assess urinary levels of the RAAS components. Yamamoto et al. reported a positive correlation of urinary Ang II to angiotensinogen and to Ang II immune-staining in kidney tissue suggesting that urinary peptide levels reflect intrarenal Ang II concentrations. 49 The concentrations of both urinary Ang II and Ang-(1-7) measured in a spot urine sample of preterm participants in our study were higher than that of adult subjects who underwent renal transplants; 50 however, the latter study did not block peptidase activity in the collected urine which may result in lower levels of both peptides. Moreover, urinary levels of Ang-(1-7) were higher than that in an adult population of normotensive and hypertensive patients which may reflect the enhanced formation of the peptide in younger subjects. 48 The urinary Ang II concentration in our VLBW adolescents was lower than those reported in a cohort of adults with chronic kidney disease. 49
Limitations
A limitation of our study is that we lack data on additional RAAS enzymes including angiotensin converting enzyme, angiotensin converting enzyme 2, and neprilysin that contribute to the synthesis and degradation of Ang II and Ang-(1-7), as well as the angiotensin receptor subtypes within the kidney which contribute to the functional balance of the RAAS. The enzyme components are excreted in the urine along with significant levels of the precursor protein angiotensinogen.13,51 We also lack complete data on maternal pre-pregnancy BMI, a variable that influences cardiovascular risk in the offspring. 52 Additionally, all subjects in this cohort were born prematurely, thus the normotensive pregnancies were not normal. Despite these limitations the present finding of elevated aldosterone levels in adolescent male offspring of pregnancies complicated by severe preeclampsia provides one potential explanation for the later increased cardiovascular risk that has been described in this population.
Conclusion
In summary, our findings suggest that among adolescent males born prematurely with VLBW, maternal preeclampsia is associated with elevated aldosterone levels. This elevation is partially explained by increased adiposity and might predispose to the development of hypertension in adulthood. Further study of the RAAS in offspring of pregnancies complicated by severe early-onset preeclampsia might provide insight as to strategies for decreasing the risk of later cardiovascular disease.
Footnotes
Acknowledgements
We thank Alice Scott RN, study coordinator, and the participants and their families.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Child Health and Human Development (grant number PO1 HD047584), the Clinical Research Unit of Wake Forest Baptist Medical Center (grant number MCRR/NIH MO1-RR07122), Forsyth Medical Center, and the Wake Forest School of Medicine Department of Pediatrics Research Fund.
