Abstract
Introduction:
Various stress hormones are responsible for bringing out stress-related changes and are implicated in learning and memory processes. The extensive clinical experience of angiotensin receptor blockers (ARBs) and direct renin inhibitor as antihypertensive agents provides anecdotal evidence of improvements in cognition. The neurochemical basis underlying the anti-stress and nootropic effects are unclear. This study was aimed to determine the effects of aliskiren, valsartan and their combination on the neuromediators of the central nervous system (CNS) and periphery as well as on cognitive function.
Materials and methods:
Groups of rats were subjected to a forced swim stress for one hour after daily treatment with aliskiren, valsartan and their combination. The 24 h urinary excretion of vanillylmandellic acid (VMA), 5-hydroxyindoleacetic acid (5-HIAA), 6-β-hydroxycortisol (6-β-OH) cortisol and homovanillic acid (HVA) was determined in all groups under normal and stressed conditions. Nootropic activity was studied using cook’s pole climbing apparatus and acetylcholinesterase (AChE) inhibitory activity by Ellman’s method.
Results:
Administration of aliskiren (10 mg/kg), valsartan (20 mg/kg) and their combination at a dose of 5 and 10 mg/kg respectively reduced the urinary metabolite levels. Further, all drugs showed significant improvement in scopolamine-impaired performance and produced inhibition of the AChE enzyme.
Conclusions:
The present study provides scientific support for the anti-stress and nootropic activities of aliskiren, valsartan and their combination.
Keywords
Introduction
Stress is known to induce alterations in various physiological responses, leading to pathological states. Stress causes disturbance in the body’s normal physiological equilibrium and results in threatened homeostasis. 1 Every human today faces stressful situations in day-to-day life and overstress has been postulated to be involved in the pathogenesis of a variety of diseases, such as depression and anxiety, immunosuppression, endocrine disorders including diabetes mellitus, male sexual dysfunction, cognitive dysfunction, peptic ulcers, hypertension and ulcerative colitis. 2 There is increasing evidence that severe stress affects cognitive functions and leads to the pathogenesis of various neurodegenerative disorders such as Alzheimer’s disease (AD), Parkinson’s disease and aging.3,4
The stress response is often associated with high levels of stress hormones, such as catecholamines (effectors of the sympathetic adrenomedullary system), glucocorticoids (effectors of the hypothalamic pituitary adrenal axis), and their regulator, central angiotensin II (Ang II). 5 Both catecholamines6,7 and glucocorticoids8,9 are known to be linked to the pathogenesis of AD. 10 The renin-angiotensin system, besides blood pressure regulation, affects learning and memory as evidenced by improvement in cognition in hypertensive patients being treated with AT1 receptor blockers like candesartan. 11 The central cholinergic system is considered to be the most important neurotransmitter system involved in the regulation of cognitive functions: there is a tight correlation between cholinergic function, acetylcholinesterase (AChE) activity and cognition. Accordingly estimation of AChE activity provides an important correlation of cholinergic activity and cognitive function. 12 Angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have shown benefits in AD and prone-to-AD animals. However, the mechanisms responsible for their efficacy remain unknown, and no studies have directly addressed the role of central Ang II, a fundamental stress hormone, in the pathogenesis of AD. 10
Although pharmacologic manipulation of the renin-angiotensin system with ACEIs and ARBs improves outcomes in hypertension and cardiovascular and renal disease, it provides only partial protection from disease progression. 13 Possible mechanisms for the inadequacies include interruption of negative feedback and a compensatory increase in renin and angiotensin I levels, which can overcome ACE inhibition or result in the production of angiotensin II (Ang II) by non-ACE pathways (i.e. ACE escape). 14 Further, the inability of ARBs to occupy all Ang II type (AT1) receptors at any given time 15 and aldosterone breakthrough16,17 during ACE inhibition or ARB use are additional possible mechanisms for this lack of complete protection by ACE inhibition and angiotensin receptor blockade. For these reasons, inhibition of renin (the first and rate-limiting step in the renin-angiotensin system) has long been a pharmacologic target in the renin-angiotensin system. 13 By inhibiting the conversion of angiotensinogen to angiotensin I (Ang I) and by decreasing plasma renin activity, aliskiren, a novel direct renin inhibitor may provide a more complete blockade of the renin-angiotensin system and offers a new opportunity to explore multistep renin-angiotensin system blockade. 13 The combination of a direct renin inhibitor, aliskiren and an ARB, valsartan, is an effective approach in various cardiovascular, cerebrovascular and renal diseases, since the combination of valsartan-aliskiren targets the renin-angiotensin system in two ways. Valsartan blocks the action of angiotensin II at the receptor, an important endproduct of the renin-angiotensin system. Aliskiren reduces Ang II levels by directly inhibiting renin, an enzyme produced by the kidneys that starts a process that leads to the formation of Ang II. To our knowledge, no study has yet been conducted to find the influence of the said classes of drugs on the urinary metabolites of amines and cortisone. Encouraged by these observations the present study was performed to assess the effect of aliskiren, valsartan and their combination on urinary metabolites of amines and cortisone in normal and stress induced rats. The changes found in the levels of urinary metabolites of amines and cortisone were used to assesses the role of aliskiren, valsartan and their combination in learning and memory. The drugs were further evaluated for nootropic activity using the conditioned avoidance response (CAR) in rats.
Materials and methods
Animals
Female albino Wistar rats (180–220 gm) obtained from Sri Venkateshwara Enterprises, Bengaluru, India, were used in the study. They were housed three per cage at a temperature of 22±2°C with 12 h light/dark cycle under a controlled environment. Rats were fed a standard pellet diet and water ad libitum. Animals were kept for seven days in the laboratory for habituation. The experimental protocol was approved by the Institutional Animal Ethics Committee (IAEC), and care of the animals was carried out as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India.
Drugs and chemicals
5-Hydroxyindoleacetic acid (5-HIAA), 6-β-hydroxyl (6-β-OH ) cortisol, homovanillic acid (HVA) and iso-vanillylmandellic acid (VMA; internal standard) were purchased from Sigma, St Louis, Missouri, USA. Vanillylmandelic acid, scopolamine butyl bromide and acetylthiocholine iodide were procured from Himedia laboratories, Mumbai, India. Aliskiren (gift sample of Novartis, New Jersey, USA), valsartan (gift sample of Apotex Research Pvt Limited, Bengaluru, India). All other reagents used were of analytical grade.
Experimental protocol 18
Female albino Wistar rats were weight matched and divided into five groups (I, II, III IV and V), each containing six animals, and were housed individually in metabolic cages. Funnels of suitable size were arranged at the bottom of the metabolic cages for collection of urine. Perforated plastic discs were arranged in the funnels to retain fecal matter. The animals were maintained at room temperature and acclimatized to metabolic cages for seven days prior to drug administration. The 24 h urine samples from each group were collected into a beaker containing 0.5 ml of 6 M hydrochloric acid for determination of VMA, 5-HIAA, 6-β-OH cortisol and HVA. The volumes of the 24 h urine samples collected in the beakers were noted individually, and about 2 ml of urine (mixture) from each animal was taken into sample tubes and centrifuged at 3000 rpm for 10 min. The supernatants were transferred into another set of clean, dry tubes and stored at −20°C until analysis by high-performance liquid chromatography (HPLC).
The experimental protocol was divided into four phases. In the first phase of the experiment 24 h urine samples were collected in all five groups and subjected to analysis of VMA, 5-HIAA, 6-β-OH cortisol and HVA, and the normal values were recorded for six consecutive days. In the second phase the animals in each group were subjected to fresh-water swimming stress individually. In this method rats were forced to swim until they were exhausted (3–4 min) in a cylindrical vessel of height 60 cm and diameter 40 cm containing water at room temperature (28°C). Water depth was maintained at 30 cm. The 24 h urinary levels of VMA, 5-HIAA, 6-β-OH cortisol and HVA under stressed conditions were determined as described above daily for six consecutive days. The third phase of the experiment consisted of administration of drugs and vehicles to the same groups of animals after having recovered completely to normal condition. The treatment given to the groups of animals at the third phase for six consecutive days was as follows:
Group I: control animals administered with distilled water orally at a dose of 0.1 ml/100 gm body weight (b.w.).
Group II: animals administered orally with 0.5% carboxy methyl cellulose (CMC).
Group III: animals administered orally with aliskiren (dissolved in distilled water) at a dose of 10 mg/kg b.w.
Group IV: animals administered orally with valsartan (dissolved in distilled water) at a dose of 20 mg/kg b.w.
Group V: animals administered orally with combination of aliskiren (5 mg/kg b.w.) and valsartan (10 mg/kg b.w.) (dissolved in distilled water).
The 24 h urine samples were collected from each group of animals and the levels of VMA, 5-HIAA, 6-β-OH cortisol and HVA were determined.
The final phase of the experiment consisted of studying the influence of above-said drugs on stress-induced changes in the same groups of animals after a recovery period of one week. Groups III, IV and V were administered aliskiren (10 mg/kg b.w.), valsartan (20 mg/kg b.w.) and their combination (aliskiren (5 mg/kg b.w.) and valsartan (10 mg/kg b.w.)) by oral gavage respectively, one hour prior to the daily induction of stress for six consecutive days, where group I were administered with distilled water and group II with 0.5% CMC. The 24 h urine samples were collected and analyzed for VMA, 5-HIAA, 6-β-OH cortisol and HVA for six consecutive days to study the influence of the aliskiren, valsartan and their combination on the stress-induced biochemical changes. The urinary metabolites were determined by HPLC as described by Wako-chem Co. 19 with slight modifications. 18
Nootropic activity 20
The nootropic activity of aliskiren, valsartan and their combination was evaluated by using the (CAR) in rats as described by Cook and Weidley. 21 Rats were divided into five groups each containing six animals. Groups III, IV and V were administered orally with aliskiren (10 mg/kg), valsartan (20 mg/kg) and combination of aliskiren and valsartan (5 and 10 mg/kg) respectively. The animals in group I were administered with distilled water and group II with 0.5% CMC. After 60 min of drug administration, all the animals were subjected to a training schedule individually by placing them inside the perspex chamber of the pole climbing apparatus. After an accustomed period of 5 min to the chamber, a buzzer was given followed by a shock through the grid floor. The rat had to jump on the pole to avoid foot shock. Jumping on the pole functionally terminates the shock and this was classified as an escape while such jumping prior to the onset of the shock was considered as avoidance. The session was terminated after completion of 60 trials with an interval of 20–30 s given for each trial. This procedure was repeated at 24 h intervals until each group reached 95–99% avoidance. After attaining complete training of a particular group, the animals were treated with a single dose of scopolamine butyl bromide (1 mg/kg b.w., i.p. (intraperitoneal)), 30 min before the next day dosing. The training schedule was continued further with the daily doses of the drugs and vehicle until they returned to normal level from scopolamine-induced amnesia.
AChE activity 22
Adult female Wistar rats weighing 200–250 gm were divided into six groups of six each. Group I animals served as control, administered orally with distilled water at a dose of 0.1 ml/100 gm b.w.; Group II animals were administered orally with 0.5% CMC; Group III, IV, V and VI animals were administered with piracetam (200 mg/kg, i.p.); aliskiren (10 mg/kg b.w. (b.w.), p.o.); valsartan (20 mg/kg b.w., p.o.) and combination of aliskiren and valsartan (5 mg/kg b.w., p.o. + 10 mg/kg b.w., p.o.) respectively for eight days. On the ninth day the animals were sacrificed by cervical decapitation under light anesthesia, 90 min after administration of last dose of respective treatment/vehicle. The whole brain was carefully removed weighed and transferred to a glass homogenizer. Homogenization of the brain was carried out in an ice bath with 0.1 M phosphate buffer (pH:8). The homogenate was centrifuged at 3000 rpm for 10 min and the resultant cloudy supernatant was used for AChE activity following Ellman’s method. 22
Statistical analysis
Results are expressed as mean±standard error of mean (SEM). The data for anti-stress activity were subjected to the Student’s paired t test. The data for AChE inhibitory activity was subjected to one way ANOVA followed by Dunnett’s test. In all tests, the criterion for statistical significance was p<0.05.
Results
Anti-stress activity
The urinary data of VMA, 5-HIAA, 6-β-OH cortisol and HVA observed in various phases of the experiment are shown in Figures 1, 2, 3 and 4 respectively. Induction of the forced swim stress to the animals produced significant (p<0.05) increases in the excretion of VMA, 5-HIAA and 6-β-OH cortisol in groups I–V; HVA in group III and V compared to their respective basal values in normal condition. All the metabolite levels were found to return to their normal levels in six to seven days after withdrawal of stress. Daily administration of aliskiren, valsartan and their combination administered to the animals under normal conditions produced a decrease in the excretion of all the metabolites compared to normal basal levels (Table 1). Aliskiren significantly (p<0.05) decreased urinary levels of HVA, valsartan significantly (p<0.05) decreased urinary levels of VMA, 6-β-OH cortisol and HVA and the combination of aliskiren and valsartan significantly (p<0.05) decreased urinary levels of VMA and HVA. Daily administration of aliskiren, valsartan and their combination one hour prior to the induction of stress inhibited the increase in VMA, 5-HIAA, 6-β-OH cortisol and HVA which was manifested during stress alone. The inhibition was found to statistically significant (Table 1). Hence the combination of aliskiren and valsartan was found to be more effective than aliskiren and valsartan in reducing the increased urinary metabolite levels caused by forced swim stress. These findings indicate that the combination of aliskiren and valsartan possesses maximum anti-stress activity followed by aliskiren and valsartan in experimental rat model.

Influence of aliskiren, valsartan and combination of aliskiren and valsartan on 24 h urinary levels of vanillylmandellic acid (VMA) in normal and stress-induced rats. Each bar indicates the mean excretion of six animals. *p<0.05 significant difference from normal conditions of the corresponding groups; #p <0.05 significant difference from normal conditions of the corresponding groups; $p <0.05 significant difference from stress conditions of the corresponding groups. CMC: carboxy methyl cellulose.
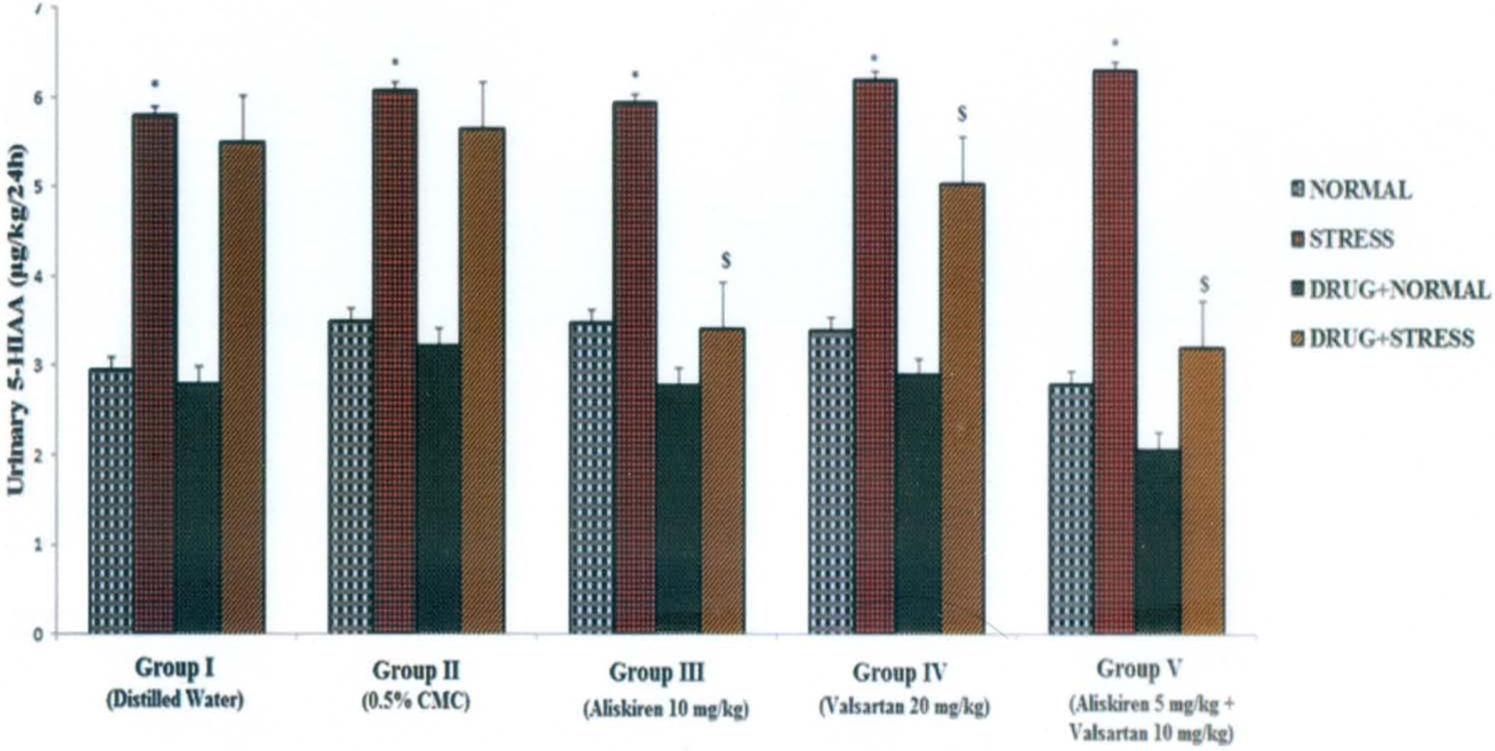
Influence of aliskiren, valsartan and combination of aliskiren and valsartan on 24 h urinary levels of 5-hydroxyindoleacetic acid (5-HIAA) in normal and stress-induced rats. Each bar indicates the mean excretion of six animals. *p<0.05 significant difference from normal conditions of the corresponding groups; $p <0.05 significant difference from stress conditions of the corresponding groups. CMC: carboxy methyl cellulose.
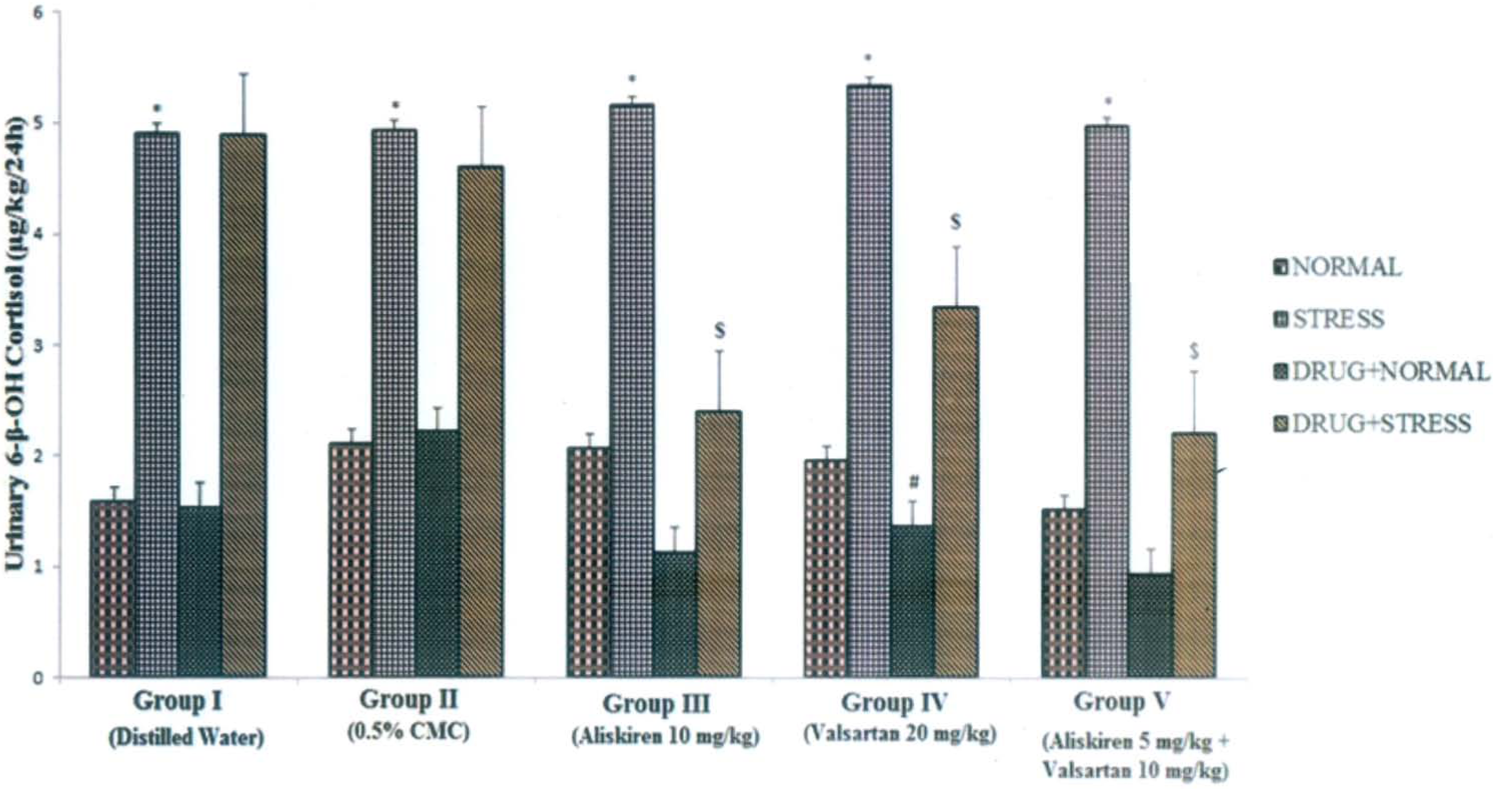
Influence of aliskiren, valsartan and combination of aliskiren and valsartan on 24 h urinary levels of 6-β-hydroxycortisol (6-β-OH cortisol) in normal and stress-induced rats. Each bar indicates the mean excretion of six animals. *p<0.05 significant difference from normal conditions of the corresponding groups; #p <0.05 significant difference from normal conditions of the corresponding groups; $p <0.05 significant difference from stress conditions of the corresponding groups. CMC: carboxy methyl cellulose.

Influence of aliskiren, valsartan and combination of aliskiren and valsartan on 24 h urinary levels of homovanillic acid (HVA) in normal and stress-induced rats. Each bar indicates the mean excretion of six animals. *p<0.05 significant difference from normal conditions of the corresponding groups; #p <0.05 significant difference from normal conditions of the corresponding groups; $p <0.05 significant difference from stress conditions of the corresponding groups. CMC: carboxy methyl cellulose.
Average values of metabolites (μg) excreted in 24 h rat urine per kg body weight (b.w.).
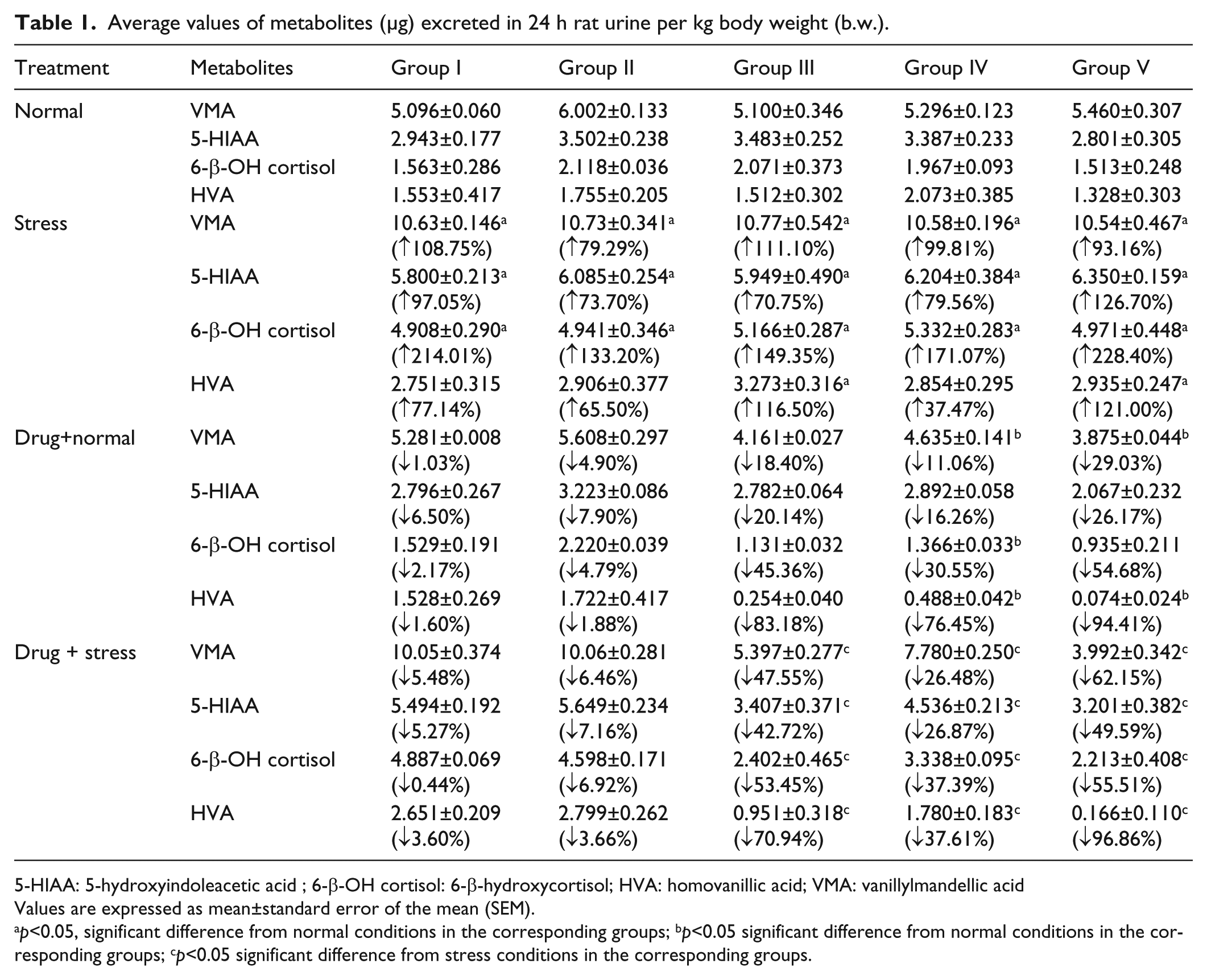
5-HIAA: 5-hydroxyindoleacetic acid ; 6-β-OH cortisol: 6-β-hydroxycortisol; HVA: homovanillic acid; VMA: vanillylmandellic acid
Values are expressed as mean±standard error of the mean (SEM).
p<0.05, significant difference from normal conditions in the corresponding groups; bp<0.05 significant difference from normal conditions in the corresponding groups; cp<0.05 significant difference from stress conditions in the corresponding groups.
Nootropic activity
The CAR of rats administered with the vehicle, aliskiren, valsartan and combination of aliskiren and valsartan increased gradually to 95% over seven days. The percentage avoidance was found to be higher in groups administered the combination of aliskiren and valsartan followed by separate administration of aliskiren and valsartan compared to the control (distilled water) and 0.5% CMC treated groups. Animals receiving the combination of aliskiren (5 mg/kg) and valsartan (10 mg/kg) took three days to show 95–99% avoidance while the group administered with 10 mg/kg of aliskiren took four days and the group administered with 20 mg/kg of valsartan took five days respectively, whereas control (distilled water) and 0.5% CMC treated groups took seven days (Figure 5).

Effect of aliskiren, valsartan and combination of aliskiren and valsartan on mean percentage conditioned avoidance response (CAR) after oral administration in rats. Scopolamine butylbromide (SBB) was administered 30 min before next day dosing with aliskiren, valsartan and the combination of aliskiren and valsartan after attaining complete acquisition. CMC: carboxy methyl cellulose.
Administration of scopolamine-produced amnesia as seen from the reduction in the observed CAR. The amnesia was found to be greater in 0.5% CMC and control groups than in drug-treated groups. However, continued administration with aliskiren, valsartan and combination of aliskiren and valsartan produced higher retention and recovery than in the control and 0.5% CMC groups. Recovery from scopolamine-induced amnesia in groups administered with the combination of aliskiren (5 mg/kg) and valsartan (10 mg/kg) took three days, the group administered with aliskiren (10 mg/kg) took four days and the group administered with valsartan (20 mg/kg) took six days and the control group took nine days and the 0.5% CMC group took 12 days respectively. From the results of our study we found that combination of aliskiren and valsartan had better retention and recovery from scopolamine-induced amnesia compared with aliskiren and valsartan groups.
AChE activity
In the present study, the increased AChE activity seen in control groups was significantly (p<0.05) reduced in animals receiving the combination of aliskiren and valsartan (0.411±0.003 μmol/min/g tissue); aliskiren (0.458±0.033 μmol/min/g tissue); valsartan (0.530±0.007 μmol/min/g tissue) when compared with their respective control groups. However the combination of aliskiren and valsartan (5 mg/kg b.w. + 10 mg/kg b.w.) was observed to be more effective than aliskiren (10 mg/kg b.w.) and valsartan (20 mg/kg b.w.) in reducing the increased AChE activity. Piracetam (0.395±0.007 μmol/min/g tissue) at a dose of 200 mg/kg b.w. was observed to be more effective than aliskiren, valsartan and their combination in reducing the increased AChE activity (Table 2). These findings indicated that the evaluated drugs possess AChE inhibitory activity in rats.
Effect of aliskiren, valsartan and their combination on acetylcholinesterase activity in rat’s brain.

AChE: acetylcholinesterase; b.w.: body weight; CMC: carboxy methyl cellulose.
Values are expressed as mean±standard error of the mean (SEM), one-way analysis of variance (ANOVA) followed by Dunnett’s test. Significant at ap<0.05 when compared to control group; bp<0.05 when compared to 0.5% CMC.
Discussion
In the present investigation, a non-invasive biochemical approach was followed to determine the 24 h urinary excretion of VMA, 5-HIAA, 6-β-OH cortisol and HVA in rats treated with aliskiren, valsartan and their combination in normal and stress-induced conditions. VMA, the peripheral metabolite of noradrenaline (NA); 5-HIAA, the main metabolite of serotonin (5-HT); 6-β-OH cortisol, metabolite of cortisone and HVA, the predominant metabolite of dopamine (DA) were selected as the biomarkers for evaluation. Estimation of these metabolites in rat urine can indirectly reflect the change in the synthesis and metabolism of their precursor molecules. Hence all these parameters were determined in 24 h urine samples from the rats. The result of our present study indicated that VMA, 5-HIAA, 6-β-OH cortisol and HVA were excreted in urine daily at certain levels (basal values) as metabolites of NA, 5-HT, cortisone and DA respectively.
Stress had a definite influence on the neurotransmitter levels. It increased VMA, 5-HIAA, 6-β-OH cortisol and HVA excretion. It is well established that the application of stress influences the hypothalamus-pituitary-adrenal (HPA) axis leading to an increase in 6-β-OH cortisol. 23 Stress also increases central and peripheral sympathetic activity, leading to an increase in VMA.24–26 Literature reports indicate that stress also influences serotonin and the limbic system and such activity might be responsible for increases in 5-HIAA and HVA. 27
Aliskiren, valsartan and their combination when administered to normal animals decreased the levels of VMA, 5-HIAA, 6-β-OH cortisol and HVA in comparison with basal values. The renin-angiotensin system system exists centrally as well as peripherally and the agonist Ang II acts on AT1 and AT2 receptors. Since ARBs act by blocking Ang II action at the receptor level, rather than by inhibiting its synthesis, they ought to antagonize the AT1-mediated effects of Ang II: hence it was reasonable to expect that the influence of renin-angiotensin system activity would decrease on the sympathetic nervous system (SNS) and HPA axis and the corresponding metabolites would also decrease because of the reduction of the corresponding precursors. The direct renin release inhibitor, aliskiren, effectively suppresses the rise in renin activity which was considered as most logical target for inhibition of the renin-angiotensin system, thereby decreasing the levels of VMA, 5-HIAA, 6-β-OH cortisol and HVA in comparison with basal values. The renin inhibitor (aliskiren) and ARB (valsartan) combination provides a synergistic effect on renin-angiotensin system hormone levels and hence the metabolites levels of the mediators were decreased.
In stress-induced conditions with prior treatment with renin inhibitor (aliskiren), ARB (valsartan) and their combination, similar changes were seen in the estimated metabolites. Stress is known to increase renin-angiotensin system activity and also influence the SNS and HPA systems. The role of Ang-II in restraint stress and isolated stress was studied previously. In isolation stress, increased adrenal corticosterone, aldosterone (peripheral), catecholamine and tyrosine hydroxylase mRNA (TH mRNA) (central) were found in a manner consistent with the classical stress response. The renin inhibitor, aliskiren, an ARB, valsartan, and their combination significantly prevented the increments in all estimated metabolite levels induced by chronic stress when given before the stress regimen. Immobilization stress has been shown to cause increases in plasma norepinephrine (NE) and epinephrine (E) levels.28,29 Intracerebroventricular application of ARBs inhibits the increases in plasma NE and E levels during stress exposure, indicating that the central Ang II system has an excitatory role in sympathetic responses to stress. 29 Armando et al. 30 found that pre-treatment with candesartan, an ARB, eliminated the increase in adrenal NE and E concentrations induced by isolation stress. On the other hand, it has been shown that acute and chronic stress stimulates the renin-angiotensin system to increase the levels of Ang II, both in the plasma and the brain. 31 Armando et al. also found that isolation stress enhanced Ang II receptor expression to a similar extent as occurs during repeated immobilization stress.32–34 Pretreatment with candesartan almost completely abolished the hormonal and sympatho-adrenal responses to stress which further supports our results. 35 The increase in 6-β-OH cortisol in stress conditions indicates the increased release of cortisone is due to stimulation of the HPA axis. In the presence of valsartan there was a decreased effect of the SNS both centrally and peripherally resulting in decreased VMA. Further the AT1 receptor blockade causes inhibition in the HPA axis stimulation resulting in decreases in 6-β-OH cortisol levels. The role of Ang II on the release of serotonin is known in stress conditions. The ARB valsartan significantly decreased the levels of 5-HIAA. It has been reported that aliskiren, a novel drug, has some advantages over existing renin-angiotensin system blockers such as: it does not have angiotensin-converting enzyme (ACE)-escape like activity, 36 it prevents the formation of both Ang I and Ang II and it produces an effective blockade of the renin-angiotensin system without the compensatory increase in Plasma renin activity (PRA). 37 Hence when the renin release inhibitor, aliskiren, was administered, renin release was blocked which subsequently leads to reduced SNS activity as well as HPA axis activity, explaining the reduction in the related amine and cortisone metabolites. Earlier investigators36,38,39 noted that a renin inhibitor and ARB combination could provide synergistic effects on renin-angiotensin system hormone levels, and also stated that the presence of a renin inhibitor effectively suppressed the rise in renin activity that would normally be stimulated by an ARB. Such observations are scientifically supported by our current biochemical study.36,38,39
The concept and definition of nootropic was first proposed in 1972 by Guirgea who coined the term ‘nootropic’ from the Greek words ‘noos’ (mind) and ‘tropein’ (to turn toward) to mean enhancement of learning and memory. 40 The nootropic drugs represent a class of agents that facilitates the integrative functions of the CNS, particularly the intellectual performance, learning and memory. They are thought to work by increasing the brain’s supply of neurochemicals (neurotransmitters, enzymes, hormones), improving the brain’s oxygen supply or by stimulating nerve growth. Learning and memory are closely associated with the functional status of the central cholinergic system. 41
The cholinergic muscarinic antagonist scopolamine is the drug most widely used to induce amnesia in experimental animals. 42 AChE inhibitors, which enhance the availability of acetylcholine (ACh) in the synaptic cleft, were able to reverse the scopolamine-induced deficit, indicating that the cognitive deficit is cholinergic in nature. The characteristics of the scopolamine model suggest that the cognitive deficits that can be observed after scopolamine treatment are directly related to a decrease in central cholinergic functions. 43 The basal brain provides the major source of cholinergic inputs to the neocortex and hippocampus. 41
The anti-stress activity was correlated with the nootropic activity of drugs, since the role of stress have been implicated in the loss of memory, concentration and also in AD.44,45 The process of nootropic activity involves acquisition, retention and retrieval and is measured using the CAR. Acquisition was quicker in groups of treated rats administered with aliskiren, valsartan and their combination in comparison to control groups, indicating the effect of the anti-stress activity of the drugs. When challenged with scopolamine butyl bromide (1 mg/kg b.w. i.p), the amnesia was less in the treated group showing better retention and recovery than in the control, aliskiren, valsartan and their combination treated groups. Hence we have shown a decrease in memory loss which could be due to central cholinomimetic activity. The role of stress has been emphasized in diseases ranging from psychiatric and endocrine disorders to cognitive function. 46 The decrease observed during the forced swim stress induced an increase in amine and cortisone levels which was correlated to the improved learning and memory process in drug-treated rats. Nalini et al. reported that peripheral administration as well as central administration of epinephrine, norephinephrine, and their agonists were found to suppress avoidance behavior but were also facilitatory in some experiments.47,48 Nootropics have also been demonstrated to interfere with serotonergic transmission and also have an inhibitory effect on norepinephrine function. 49 These reports support the inhibitory role for norepinephrine in learning and memory. The role of 5-HT in learning and memory is found to be inhibitory in various experimental studies. Treatment with a 5-HT antagonist 50 and depletion of brain 5-HT, facilitate learning and enhance retention process in avoidance task. 51 Fenfluramaine, a serotonin reuptake inhibitor and releasing agent, effectively increases serotonin levels and appeared to impair the working memory in human subjects. Evidence suggests that serotonergic transmission inhibits the performance of working memory and it also suggests that blocking serotonergic transmission in the brain is a possible mechanism to enhance working performance.52,53 Elevation of circulating corticosterone concentrations by systemic administration of corticosterone or a glucocorticoid receptor agonist exerts an inhibitory influence on learning and memory, 54 and stress exposure or glucocorticoid administration profoundly impairs working memory. 55 Hence the decrease in cortisone is responsible for improved learning and working memory. Use of a DA agonist as well as electrical stimulation suggested that heightened activity in the dopaminergic system during the learning and memory process might lead to retention failure. 47 Hence decreased dopaminergic activity is favorable for an improved learning and memory process. Based on these reports, we can hypothesize that the anti-stress activity of aliskiren, valsartan and their combination is correlated with nootropic activity.
In the CNS, ACh has a variety of effects as a neuromodulator upon plasticity, arousal and reward. ACh has an important role in the enhancement of sensory perceptions when we wake up and in sustaining attention. Damage to the cholinergic (ACh-producing) system in the brain has been shown to be plausibly associated with the memory deficits associated with AD. 56 The most commonly recognized symptom of AD is memory loss. Memory impairment in the patients results from a deficiency in cholinergic function in the brain. Approaches to enhance cholinergic function in AD have included stimulation of cholinergic receptors or prolonging the availability of ACh released into the neuronal synaptic cleft by inhibiting ACh hydrolysis by AChE. 57 Hammond and Brimijoin had given evidence on the role of AChE in cognitive function studies on biopsy tissues of AD patients, which showed decreased AChE with a concurrent loss of cognitive function. 58 Several studies have demonstrated that chronic stress leads to cognitive dysfunction and results in disorders like depression, anxiety, and impairment of learning and memory. Hence it can be speculated that there is a tight correlation between stress, cholinergic function, AChE activity and cognition. Based on these reports the AChE inhibitory activity of aliskiren, valsartan and their combination was also explored in the brain homogenates of rats using Ellman’s method. Aliskiren, valsartan and their combination significantly decreases the AChE activity, indicating a normal level of ACh was maintained in the brain, which is responsible for memory. The use of piracetam is expected to compensate cholinergic deficits indirectly by inhibiting the destruction of ACh and directly by increasing the expression of choline transferase. In our study piracetam, aliskiren, valsartan and their combination significantly lowered AChE activity indicating the stimulating action of these drugs on the cholinergic system. Hence the memory enhancement effect of evaluated drugs can be attributed to anticholinesterase activity.
It is believed that the renin-angiotensin system of the brain is involved in the modulation of multiple functions in the brain, including processes of sensory information, learning and memory and regulation of emotional responses. 59 Elevated brain Ang II may interfere with ACh release that in turn interferes with cognitive function. 60 It also interferes with potassium-mediated release of ACh from rat entorhinal cortex slices. 61 Centrally administered Ang II was reported to disrupt learning and memory in various working and spatial memory tasks in rodents.62–64 Raghavendra et al. 65 concluded that a decrease in endogenous Ang II activity in the brain might result in improved cognitive performance by enhancing cyclic GMP (cGMP) pathways. Fogari et al., demonstrated that valsartan improves the quality of life scores and has beneficial effects on cognition in older hypertensive patients.66,67 Raghavendra et al. 68 reported that losartan was able to enhance memory in mice, as assessed by a passive avoidance task and by the elevated plus-maze. Furthermore, losartan reversed the cognitive defects induced by scopolamine and itself was potentiated by co-administration of cholinesterase inhibitor. These results support the initial hypothesis of Barnes et al., 69 suggesting the cognitive enhancing effect of Ang II suppression and indicating a mediating role of ACh.
In the present study, the scopolamine reversal of the memory-enhancing effect of valsartan, aliskiren and their combination suggests the possible role of ACh in the memory-enhancing action of these drugs. The neurochemical basis behind Ang II and ACh interaction supports the memory-enhancing action of Ang II inhibitors. Binding sites for Ang II have been identified in the brain regions involved in the processing of sensory information, such as the cortex, hippocampus, mid brain and lateral septum.70,71 Ang II was shown to inhibit the in vitro release of ACh from entorhinal cortex which was reversed by peptide Ang II receptor antagonists.61,69 These findings suggest the importance of apparent control via Ang II of cholinergic processes which have long been associated with cognitive performance.
Conclusion
These experiments indicate the possible novel therapeutic effect of an Ang II AT1 receptor blocker, a direct renin inhibitor and their combination in antagonizing the effect of renin-angiotensin system stimulation on the HPA axis and SNS activities. In this way, the AT1 receptor antagonist, valsartan, and the direct renin inhibitor, aliskiren, and their combination decreases vulnerability and sensitivity to stress, preventing stress-related disorders. The decrease in the levels of VMA, 5-HIAA, 6-β-OH cortisol and HVA in the urine of treated rats indicates that these drugs acting on the renin-angiotensin system cause an overall decrease in the turnover of monoamines and cortisone. Hence the above findings together suggest the involvement of noradrenergic, serotonergic and dopaminergic systems in learning and memory, mainly as inhibitory neurotransmitters.
Footnotes
Conflicts of interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
