Abstract
Introduction:
AGT is the first gene to be linked to essential hypertension (EHT). It harbors several variants of which only few polymorphisms are found to exhibit positive and negative associations with hypertension. In the present study, the AGT gene was screened to detect already reported and novel variations contributing to the development of hypertension.
Method:
In total, 215 hypertensives and 230 normotensives were screened for variations in all the five exons and a part of promoter of AGT gene using single strand conformation polymorphism analysis followed by sequencing of samples showing mobility shifts on polyacrylamide gels.
Results:
Five novel variants, namely c.-61G>A in promoter, c.-4+17C>T in intron1, c.24T>C and c.28A>T in Exon2, and c.*90 T>C in 3’ untranslated region were detected in the AGT gene. c.-61G>A lies in the promoter region that plays a critical role in its expression. Variation c.-4+17C>T created a new enhancer site. c.24T>C (TCT-TCC) is a silent mutation while c.28A>T (p. M10L) has a possible damaging effect on the AGT protein. c.*90T>C, detected in the 3’ untranslated region is thought to play an important role in the translation and stability of the mRNA.
Conclusion:
Studies on the functional role of these novel variants are warranted to understand the mechanism underlying the development of EHT.
Introduction
Essential hypertension is a common complex condition and a substantial public health problem. It occurs with a prevalence of about 30–50% 1 in several developed and developing countries, and its prevalence in India is estimated to be 25% in urban and 10% in rural populations. 2 It stands as a major risk factor for cardiac and renovascular diseases, 1 adding to the mortality rate when patients are not treated promptly and managed with proper surveillance. It is a multifactorial disease caused by interactions between genetic and environmental factors. The contribution of genes to the pathogenesis of human hypertension is estimated to be 30%. 3 The genome-wide scans that identified several independent loci for blood pressure revealed the polygenic nature of hypertension. 4 Little is known, however, about the number of genes involved or about the mode of interaction between genes and the environment.
To date, more than 300 genes have been implicated in the development of hypertension which are limited to proteins that are related to blood pressure regulatory systems and other pathways. Progress in identifying these genetic determinants has thus far been slow, probably because the individual genetic effects present are small. Among the most studied genes is angiotensinogen (AGT), the substrate of renin, playing the role of a key molecule in the renin–angiotensin system. Angiotensinogen, a polypeptide produced in the liver, is cleaved by the renin enzyme to angiotensin (Ang) I which is then converted to Ang II by another component, angiotensin-converting enzyme (ACE). This product produces vasoconstriction and sodium retention, thus regulating blood pressure. Also, levels of AGT in blood were found to be correlated with hypertension. Mice inserted with additional copy of the AGT gene became hypertensive, indicating its importance in blood pressure regulation. 5
The AGT gene is located on the long arm of chromosome 1(1q42-43) and contains five exons and four introns. It is the first gene reported to be linked to essential hypertension, by Jeunemaitre et al. 6 Although more than 23 variants of AGT have been cloned and sequenced,6,7 only a few polymorphisms in the promoter (g.-217G>A, g.-20A>C, g.-6G>A) and exon 2 (p.T174M and p.M235T) are reported to be exhibiting strong positive6,8–15 and negative associations with essential hypertension and cardiovascular diseases.16–29 Contradictions in these findings were attributed to the ethnicity of the populations studied. A functional relationship of the variant p.M235T with the development of hypertension was suggested which is also associated with increased levels of circulating angiotensinogen in the patients. The AGT gene is also reported to interact with other loci such as ACE and environmental factors, providing more insight into the mechanism that governs the relationship of the AGT gene with essential hypertension.
Studies delineating the importance of other AGT gene variants are limited, and lacking in the Indian population. Hence, the present study was conducted to screen the AGT gene promoter and all the five exons to find known and novel variants, contributing if any contribute to the development of essential hypertension. Here, we report the presence of five novel variants in patients with essential hypertension, which are registered in the NCBI database.
Materials and methods
A total of 215 hypertensives with confirmed diagnosis (with systolic blood pressure (SBP) ≥ 140 mm Hg and diastolic blood pressure (DBP) ≥ 90 mm Hg) and those who were already on antihypertensive drugs at the time of investigations were recruited from the outpatient clinics of (i) Gandhi Medical College and Hospital, Hyderabad, India, (ii) Nizam’s Institute of Medical Sciences, Hyderabad, India, and (iii) by personal contacts. For comparison with the patient group, 230 age- and sex-matched healthy controls (with SBP/DBP <130/90 mmHg) were selected at random by personal contacts, by house visits, and among the employees of Government and private organizations with the provision for annual health checkup. The purpose and outcome of the study were explained to the patients and controls, and only those who gave their consent to participate in the investigations by providing the blood samples and demographic history were considered. The study was approved by the Institutional Ethical Committee.
All the subjects were examined clinically and through biochemical and other investigations for thyroid disorders, diabetes mellitus, stroke, cardiac diseases, renal problems, and so on, and were excluded from the study if any of such conditions was present. Detailed information relating to sex, age, body mass index (BMI, determined by considering weight in kg and height in m2), diet, family history, consanguinity, and habits such as smoking and alcohol consumption were collected from both patients and controls. Family information relating to three to four generations was also obtained and pedigrees were constructed.
DNA was isolated from whole blood collected in ethylenediaminetetraacetic acid (EDTA) vacutainers from patients and controls using a rapid non-enzymatic method as described by Lahiri and Nurnberger. 30 Fourteen commercially designed overlapping primer sets (Table 1) were used to amplify a part of promoter and all the five exons of AGT gene by polymerase chain reaction (PCR). PCRs were performed using 10 µl PCR mix containing 1× PCR buffer, 200 µM dNTPs, 0.25 units of Taq polymerase and 2.5 pmol of forward and reverse primers. PCR products were denatured with low ionic strength (LIS) buffer (10% sucrose, 0.01% bromophenol blue and 0.01% xylene cyanol) at 95°C for 10 min followed by snap cooling in ice. Single strand conformation polymorphism (SSCP) analysis was carried out by running the samples in 10–12% denaturing gels prepared by using 40% acrylamide solution (37.5 g acrylamide, 1 g bisacrylamide), and the run was carried out at 100 V for 18 h. Bands were visualized by silver staining and recorded using Systronics gel documentation. Samples showing mobility variations were sequenced on ABI 3100 DNA sequence analyzer to identify the nucleotide changes.
Primers used for amplification of promoter and different exons of AGT gene.

Sequencing results were analyzed using NCBI blast. PolyPhen (Polymorphism Phenotyping; (http://genetics.bwh.harvard.edu/pph2/)) is an automatic tool employed for prediction of the possible impact of an amino acid substitution on the structural and functional properties of a protein. The SIFT (Sorting Intolerant From Tolerant) tool (http://sift.jcvi.org/) was also used to predict the effect of sequence changes on the protein function based on homology search and the physical properties of amino acids. Human splice site finder (HSF) was used to predict the effects of mutations on splicing signals or to identify splicing motifs if any in human sequence.
Results and discussion
Screening of the AGT gene for a part of promoter and all the five exons by SSCP analysis showed various mobility patterns which, on sequencing, revealed eight known, three rare and five novel variants in the AGT gene. Table 2 describes the distribution of genotypic and allelic frequencies of known variants detected in the present study. The three rare variants, namely p.T137M in three hypertensives, c.*90T>C in one hypertensive and c.*158C>G in six hypertensives and two normotensives were all observed in heterozygous state. The five novel variants, c.- 61G>A (g.-23G>A) in promoter, c.-4+17C>T in intron1, c.24T>C and c.28A>T in Exon2, and c.*90 T>C in 3’ untranslated region of the AGT gene have been registered in the NCBI database with reference numbers rs312262681, rs312262682, rs207482231, rs199476082 and rs207482232, respectively. All the novel variants were detected only in hypertensive samples, except for c.-4+17C>T which was observed in both hypertensives and normotensives.
Distribution of genotypes and allele frequencies of AGT known variants in hypertensives and normotensives.
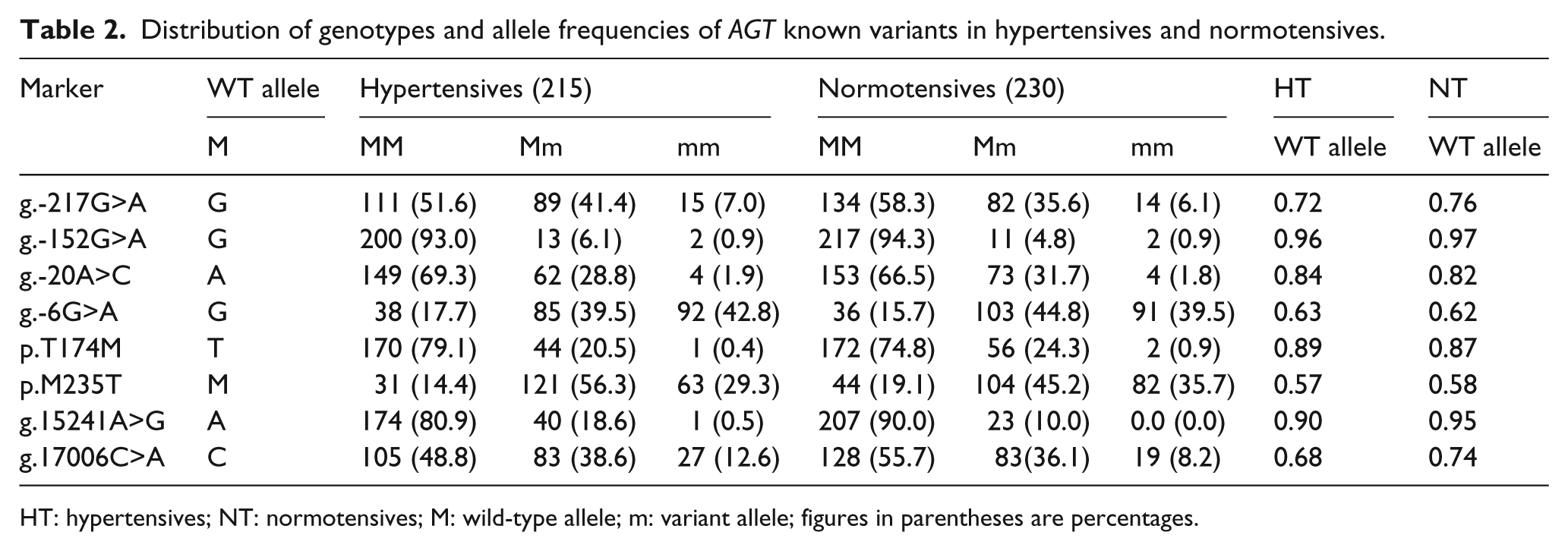
HT: hypertensives; NT: normotensives; M: wild-type allele; m: variant allele; figures in parentheses are percentages.
c.-61G>A, detected in the heterozygous state in three hypertensive samples, lies in the promoter region located between the TATA box and transcriptional initiation site. It has been shown by Yanai et al. 31 and Zhao et al. 32 that the nucleotide sequence located between the TATA box and transcriptional initiation site of the human AGT gene plays a critical role in the expression of the gene, and referred it as angiotensin core promoter element 1. It is possible that this variation in the promoter may affect the binding of transcription factors to this region of the promoter and alter the transcriptional regulation of the gene; this has to be further elucidated.
c.-4+17C>T variation in intron1 of AGT gene when analyzed using HSF was predicted to alter enhancer motifs with respect to wild-type sequence. The PESE octamers and EIEs predicted the creation of a new enhancer site. Besides this a silencer motif identified by Sironi et al. 33 was predicted to be broken, with a variation score of 7.12 between wild-type and the mutant motif. This variation was detected in the heterozygous state in both hypertensives and normotensives.
SSCP analysis of region 1 of Exon 2 of the AGT gene showed two different hetero patterns in two different hypertensive samples, which on sequencing revealed T-C and A-T substitutions at position 24 and 28 of the cDNA sequence, denoted as c.24T>C and c.28A>T, respectively, both of which are found to be novel variants. c.24T>C (TCT-TCC) is a silent mutation while c.28A>T (p. M10L) is found to have a possible damaging effect on the AGT protein through Polyphen analysis. Both the variations are located in the region that codes for the signal peptide. The signal peptide plays an important role in translation by ribosomes; the nascent protein is bound via the signal peptide to the signal recognition particle, which guides the complex to the endoplasmic reticulum (ER). 6 Translocation of the protein into the ER is followed by post-translational modifications such as glycosylation, signal peptide cleavage, disulfide bond formation, and folding. 34 The sequences of signal peptides are heterogeneous, but typically three conserved regions have been recognized and shown to be essential for protein export as follows: (i) an amino-terminal positively charged region; (ii) a central hydrophobic region; and (iii) a more polar carboxylterminal region. The amino-terminal amino acid sequence deduced from the cDNA of human angiotensinogen revealed the presence of a signal peptide of 33 amino acids containing all three consensus regions.35,36 The 5’ segment of human angiotensinogen mRNA contains two AUG codons in the same reading frame. The c.28A>T variation results in the conversion of a second initiation codon located at the 5’ segment of human angiotensinogen mRNA to leucine. It was shown by Nakajima et al. 37 that alteration of either the first or second AUG codons abolished the synthesis of the longer and the shorter form, respectively, of native angiotensinogen. Furthermore, the rate of secretion of the shorter form was lower than that of the longer form. By transplanting angiotensinogen signal peptide onto green fluorescence protein, however, they found that both forms of the signal peptide could target green fluorescence protein, normally localized in the cytoplasm, to the secretory pathway.
c.*90T>C, a novel variant, was detected in one hypertensive sample in the 3’ untranslated region of the AGT gene. The 3’-UTR plays an important role in the translation, localization and maintenance of stability of the mRNA. Research on the pathophysiology of diseases and mutations affecting the functionality of the 3’-UTR is still sparse.
In conclusion, the study revealed five novel variants whose functional roles have to be further elucidated through in vivo studies correlating AGT levels and onset and progression of essential hypertension. Also, screening of larger samples for these variations might throw more light on their contribution to the development of essential hypertension.
Footnotes
Acknowledgements
The authors are thankful to the patients and controls who voluntarily extended their cooperation in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Department of Science and Technology (DST) [SAN.F.No: SR/WOS-A/LS-134/2009], New Delhi, India.
