Abstract
Introduction:
This is a report of a clinical trial on the therapeutic efficacy and safety of combined aliskiren and losartan (an angiotensin II receptor blocker (ARB)) versus aliskiren alone and ARB alone in non-diabetic chronic kidney disease (CKD) over a 3-year period.
Materials and methods:
This was a randomised trial in 155 patients with non-diabetic CKD comparing aliskiren (150 mg/day) (n=52) versus losartan (100 mg/day) (n=52) and the third group aliskiren (150 mg/day) combined with losartan (100 mg/day) (n=51). The trial utilised primary renal end points of eGFR <15 ml/min or end-stage renal failure.
Results:
All three groups had significant reduction of proteinuria (p<0.001 for all). The changes in eGFR, total urinary protein from baseline to each year were not significantly different between the three therapeutic groups.
Conclusion:
This study in non-diabetic CKD patients showed that combination therapy with aliskiren and ARB was as efficacious as aliskiren alone and ARB alone. There was one patient who developed a non-fatal stroke in the combined aliskiren and ARB group while the other two groups had none.
Introduction
One of the important strategies in the treatment of chronic kidney disease (CKD) is the use of angiotensin-converting enzyme inhibitors (ACEI) and angiotensin II receptor blockers (ARBs) to reduce proteinuria as well as to retard the progression to end-stage renal failure.1,2 ACEI and ARBs compete with the receptor for angiotensin and therefore inhibit the action of angiotensin. Aliskiren is a new renal protective agent that inhibits renin, the rate-limiting step in the renin–angiotensin–aldosterone system (RAAS). 3 In both healthy volunteers and disease states, aliskiren reduces angiotensin II levels and plasma renin activity (PRA), without stimulating compensatory increases in PRA, angiotensin I and angiotensin II as seen with ACEI and ARB. Aliskiren allows for total blockade of the renin–angiotensin system, and its beneficial effect is independent of blood pressure (BP) control. 4
One strategy which may prove to be even more effective would be to employ a combination of ARB (losartan) and aliskiren as shown in the AVOID Trial by Parving et al. 5 Such a strategy would achieve the dual purpose of ARB blockade of the RAAS system with the additional effect of aliskiren mopping up the excess renin resulting from ARB blockade which prevents the conversion of renin to angiotensin. Aliskiren has the effect of catalysing renin, hence reducing the high levels of PRA induced through the use of ARB.
The present study examines the effects of aliskiren (150 mg/day) combined with ARB (losartan 100 mg/day), aliskiren (150 mg/day) alone and ARB (losartan 100 mg/day) alone in patients with CKD (chronic glomerulonephritis). We compared the therapeutic efficacy between combined aliskiren (150 mg/day) with losartan (100 mg/day) versus aliskiren alone (150 mg/day) and the third group an ARB (losartan 100 mg/day) alone, to determine whether a combined dose of aliskiren and losartan is as effective as aliskiren alone or ARB (losartan) alone, and whether it confers additional renoprotective effects.
Materials and methods
For this study, 172 two patients with CKD due to chronic glomerulonephritis and not due to diabetic nephropathy, hypertensive nephrosclerosis, lupus nephritis or Henoch–Schönlein nephritis were recruited for the study. From September 2008 to July 2009, these patients entered a randomised study. Of these, 71% presented with asymptomatic haematuria and proteinuria, 29% with proteinuria alone, 34% had associated hypertension on presentation and 85% had Stage 3 CKD with estimated glomerular filtration rate (eGFR) <60 ml/min. Of the patients who had renal ultrasonography, 30% had normal sized or slightly shrunken kidneys but with increased echogenicity and no renal cysts. Patients with normal sized kidneys were offered renal biopsy examination, but most declined. Those with diminished sized kidneys were not offered renal biopsies. The biopsied patients were recruited into other trials and there were not many of them. In contrast, the non-biopsied CKD patients formed the bulk of our clinical practice and were more readily recruited. For purposes of standardisation of the study, we decided to recruit only non-biopsied patients into the study. Of the 172 patients, 58 patients were treated with combination therapy using aliskiren and ARB (losartan), 56 patients were treated with aliskiren alone and the remaining 58 patients were treated with ARB (losartan) alone. Informed consent was obtained for all patients. Entry criteria included proteinuria of 1 g or more and/or CKD Stage 3. Seven of the 58 patients on combination dose of aliskiren and ARB were withdrawn from the trial (two for hypotension, two for severe giddiness, one for diarrhoea and two defaulted follow-up), leaving 51 patients who completed the trial. Among the 56 patients treated with aliskiren alone, four were withdrawn (one for giddiness, one for drug rash and two defaulted follow-up), leaving 52 to complete the trial. For the remaining 58 patients who were on losartan alone, six were withdrawn (one for cough, two for hypotension and three defaulted follow-up), leaving 52 to complete the trial. The majority of patients who entered the trial had CKD Stage 3: 82% in the combined aliskiren and ARB group, 89% of the aliskiren alone group and 91% of the ARB (losartan) alone group.
There were 23 patients with hypertension in the combined aliskiren and ARB group, 20 in the aliskiren group and 29 in the ARB alone group. There were no significant differences in the various parameters between the three groups on entry into the trial. Additional BP control was achieved with atenolol, amlodipine and nifedipine. All patients were given advice on a low-salt diet.
Study design
This was an open-label study, where 172 patients were randomised to combination dose of aliskiren and ARB (losartan) (n=58), aliskiren alone (n=56) and ARB (losartan) alone (n=58). A list of random numbers generated via computer by simple randomisation allocation method was used. In total, 155 patients completed the 3-year duration of the trial. Seven patients were withdrawn from the combined aliskiren and ARB arm, four from the aliskiren alone arm and another six from the ARB alone arm. BP control was targeted at 130 mmHg systolic and 80 mmHg diastolic. There were no significant differences in either the systolic and diastolic BP between the ARB and the ACEI groups on entry into the trial. None of the patients in the study were prescribed hydrochlorothiazide.
Patients had the following investigations performed at 6-monthly intervals: serum creatinine, eGFR and total urinary protein (TUP). Serum creatinine was quantitated with alkaline picrate and TUP was quantitated by biuret agent. Glomerular filtration rate was estimated using the Cockcroft–Gault formula for eGFR. Decrease in eGFR was expressed as ml of eGFR loss per year over the 3-year duration from time of entry to exit of the trial. Improvement in eGFR was taken as the positive difference between the entry eGFR and the exit eGFR over the trial period. End-stage renal failure was equated with decline of eGFR to CKD Stage 5 with eGFR less than 15 ml/min/year. The primary end points were stage 5 CKD or end-stage renal failure. The secondary end points were reduction of proteinuria by 50% and change in eGFR.
Sample size
Sample size calculation was based on the proportion of patients achieving 30% decrease in TUP. A second sample size calculation was done to compare the rate of 30% TUP decrease between combined aliskiren and ARB and aliskiren alone. Assuming that the rate of TUP decrease to be 30% in the ARB alone group and aliskiren alone group, and 50% in the combined aliskiren and ARB group, the number of patients required in each group was 49 for a 2-sided test with alpha=0.05 and power of 80%. As we expected combined aliskiren and ARB to be even more efficacious, 50% reduction of TUP was chosen.
Statistical methods
SPSS 10.1 for Windows was used for all analysis. Results were expressed as mean ± SD or median (range) or count. For univariate analysis, Pearson’s chi-square test was used for comparing categorical data and ANOVA for comparing numeric data between the three treatment arms. ANOVA was followed by multiple comparison with Student–Newman–Keuls range test whenever statistical significance was found between the three arms. Next, a doubly multivariate ANOVA (MANOVA) with repeated measures was used to test the effect of drug treatment on both eGFR and TUP. The dependent variables were eGFR and TUP measured at four time points, namely baseline and thereafter every year of the 3 years of the study. The between-subject factor was treatment group with three levels corresponding to combination dose of aliskiren and ARB, aliskiren alone and ARB alone. Adjustment was made for the covariates of average systolic BP and average diastolic BP. Average BPs were calculated by taking the mean of all BPs while on medication (mean of BPs from year 1 to year 3). Within MANOVA, the effect of aliskiren and ARB on the outcomes of eGFR and TUP was compared with each of the other drug dosage groups by simple contrast comparison testing. Similarly, repeated contrast testing was done to obtain and compare the loss in eGFR in each year between the various drug groups. Plots of mean values of eGFR and TUP adjusted for covariates of systolic BP and diastolic BP were presented. So were the contrast estimates, their corresponding 95% confidence intervals and p-values for the comparison of eGFR and TUP between the levels of interest of the treatment group. Results of two types of comparison, change in eGFR and TUP from baseline to each time point compared between the three drug groups and loss in eGFR compared between the three drug groups were presented in a table with the F statistics and p-values.
Results
Table 1 compares the eGFR, proteinuria and decrease in eGFR between combination dose of aliskiren and ARB, aliskiren alone, and ARB alone, before and after the trial. The decrease in eGFR per year was not significantly different among the three arms. The decrease in eGFR was 1.0 ml/min/year in the combined aliskiren and ARB group compared with 1.5 and 1.6 ml/min/year in the aliskiren group and the group on ARB alone, respectively (p=0.117). There were 17 patients out of 51 (33%) who had improved eGFR at the end of the study in the combined aliskiren and ARB group compared with 15 out of 52 patients (29%) in the aliskiren alone group and 15 out of 52 patients (29%) in the group on ARB alone (NS, p=0.850 ). There were no patients with end-stage renal failure at the end of the study in all the three groups (Table 1). Though TUP was lower at the end of the study for the combined aliskiren and ARB group compared with the other two groups, its value at baseline was also lower. So, we will more meaningfully compare the change in TUP among the three groups in the next section on MANOVA results.
Comparing demographic and clinical profile of patients treated with combined dose aliskiren and ARB, aliskiren alone and ARB alone.
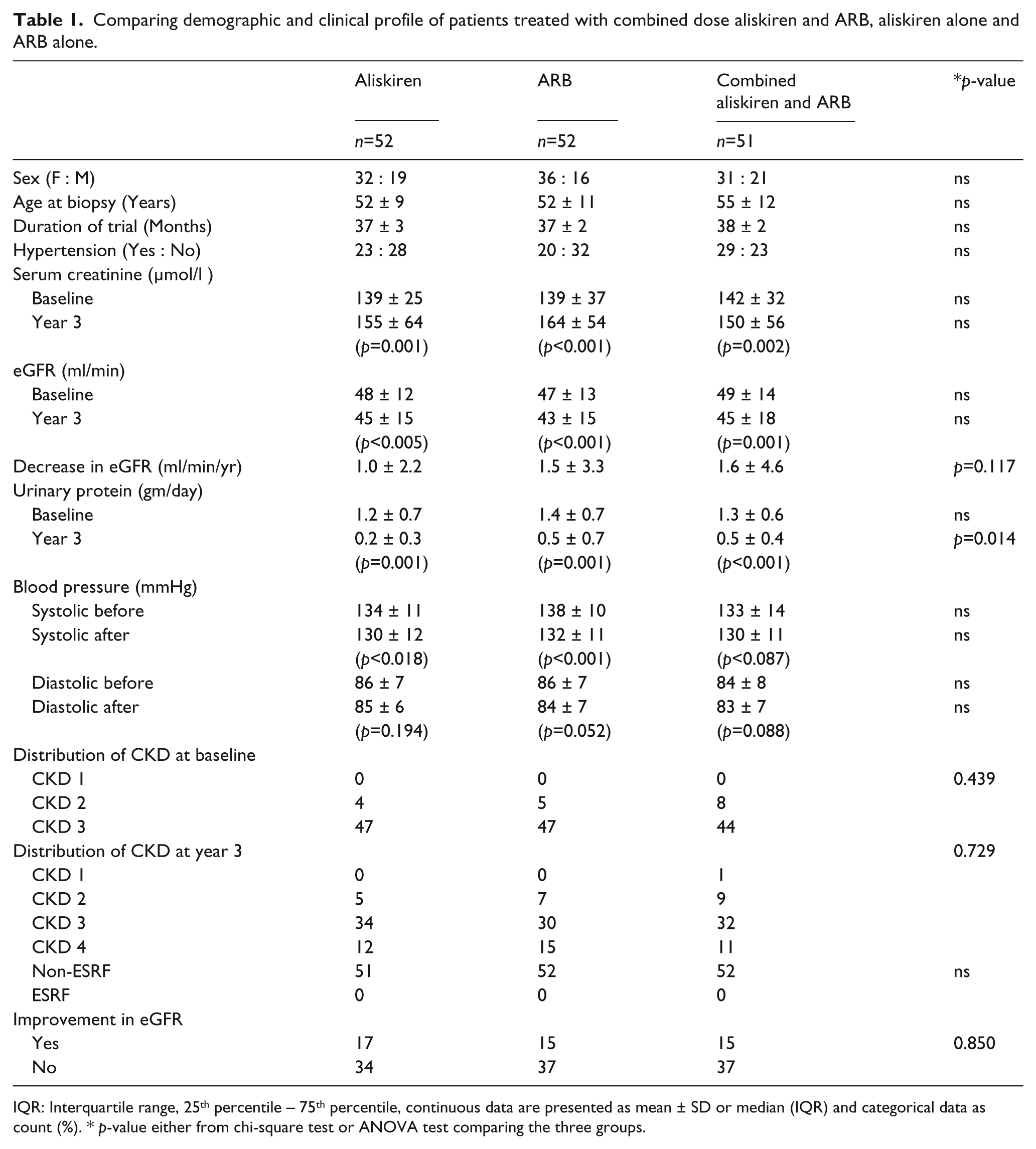
IQR: Interquartile range, 25th percentile − 75th percentile, continuous data are presented as mean ± SD or median (IQR) and categorical data as count (%). * p-value either from chi-square test or ANOVA test comparing the three groups.
The changes in eGFR and TUP from baseline to each year were not significantly different between the three therapeutic groups (Table 2, Figures 1 and 2). The changes in eGFR and TUP from each year to the next were also not significantly different between the three therapeutic groups (Table 3, Figures 1 and 2). The BP levels, systolic and diastolic, over the 3 years for the three drug groups are displayed graphically in Figures 3 and 4, respectively. There were no significant differences between the three drug groups throughout the 3 years.
Test of within-subject contrast (change from baseline) for eGFR and total urinary protein.

Each row of the table tests the change from baseline to that time between the three groups.
eGFR: estimated glomerular filtration rate; TUP: total urinary protein.

Mean eGFR with its standard error (SE) for the three treatment groups.

Mean total urinary protein with its standard error (SE) for the three treatment groups.
Test of within-subject contrast (change from previous year) for eGFR and total urinary protein.

Each row of the table tests the yearly change in eGFR between the four groups.
eGFR: estimated glomerular filtration rate; TUP: total urinary protein.

Mean systolic blood pressure with its standard error (SE) for the three treatment groups.

Mean diastolic blood pressure with its standard error (SE) for the three treatment groups.
Hyperkalaemia
The incidence of hyperkalaemia (>5.5 mmol/l) was 37.2% (19/51) in the combined aliskiren and ARB group, 13.3% (8/52) in the aliskiren alone group and 19.2% (10/52) in the ARB alone group (p<0.039). Hyperkalaemia was a major problem in the combined aliskiren and ARB group compared with the aliskiren alone and the ARB alone group.
Pre-trial comorbidities
Since a significant portion of the data analyses would involve events like ischaemic heart disease (IHD) and strokes, in accordance with the results of the ALTITUDE study, 6 it would be relevant to assess pre-trial status and certain factors like hypertension and hypercholesterolaemia which could predispose to these comorbidities.
The incidence of hypertension in the three groups combined aliskiren plus ARB, aliskiren alone and ARB alone was 45.1% (23/51), 38.5% (20/52) and 55.8% (29/52), respectively, no significant difference. For hypercholesterolaemia the incidence tended to be higher in the ARB alone group at 65.4% (34/52) compared with aliskiren alone 42.3% (22/52) and combined aliskiren and ARB group 49.0% (25/51), (p=0.053). The incidence of IHD in the combined aliskiren plus ARB group, aliskiren alone group and ARB alone group was 19.6% (10/51), 11.5% (6/52) and 15.4% (8/52) (p=0.527). Two patients had cerebrovascular accidents (lacunar infarction) among the ARB alone group but none in the other two groups. The difference was not significant. It must be emphasised that the events of IHD and strokes occurred prior to 2008 before entry of the patients into the trial.
Comorbidities during trial
During the duration of the trial, in each of the three groups there was one patient who developed IHD. All three patients had this as a new comorbidity. Only one patient developed a transient ischaemic attack (TIA) or non-fatal stroke in the combined aliskiren and losartan group, with none in the other two groups. This would have been considered a primary end point event as well as a serious adverse event in the ALTITUDE study. 6 In this present trial this was considered an associated event.
Discussion
The above data showed that among patients with non-diabetic CKD, treatment with combined aliskiren and ARB was as effective when compared with the other two treatment groups on aliskiren alone and ARB alone. The changes in eGFR and TUP from baseline to each year were not significantly differently between the three therapeutic groups. The BP of all three drug groups showed no significant differences and did not influence any of the above data. This study was concluded before the release of the report of the early termination of the ALTITUDE study 6 as the results of the ALTITUDE study showed that there was no benefit with aliskiren and that there were more cases of stroke, renal complications, hyperkalaemia and hypotension in patients who received aliskiren compared with patients who received a placebo.
To date, the Health Science Authority (HSA) of Singapore 7 has received 14 suspected adverse reaction reports associated with the use of aliskiren, of which four involved cardiovascular events (one case of hypotension, myocardial infarction and stroke and three cases of hypotension). HSA has recommended that aliskiren or aliskiren combination with ACEI or ARBs should not be used in diabetics or in patients with severe renal failure (eGFR < 30 ml/min). 7
The initial trials of aliskiren involved patients with hypertension, the drug being first developed as an antihypertensive agent. They were short trials in hypertensive patients,8-14 many lasting only 8 weeks, and did not address the question of renoprotection. Later trials by Parving et al.5,8 and Tang et al. 9 were conducted in patients with kidney disease, diabetes and IgA nephropathy, and these trials lasted longer − 6 months and 12 months – and both showed a reduction of proteinuria, an indicator of renoprotection. In all the trials so far, cardiac and neurological adverse events such as cardiovascular and central nervous system morbidity and mortality were not an issue until the interim results of the ALTITUDE study were made known. 6
The ALTITUDE study was able to unmask serious adverse events such as IHD and strokes because it had included cardio-renal endpoints among its primary end points. It may be advisable to require future trials on drugs which could impact on the kidneys, heart and brain to have similar cardio-renal endpoints or cardio-neuro-renal endpoints to further ensure therapeutic safety of the trial drug.
Our present study in 155 patients with CKD over 3 years showed that the use of combination therapy of aliskiren with ARB was efficacious as an antiproteinuric drug when compared with aliskiren alone or ARB alone. However, it should be noted that aliskiren was prescribed in a dose of 150 mg a day in our patients, compared with the dose of 300 mg a day in the European patients because of the lower body weight of our Asian patients in the trial. Perhaps aliskiren at a dose of 300 mg a day would prove to be even more efficacious. All the three drug groups in our study did not show significant differences in their BP at the end point. Neither aliskiren nor losartan appear to be very potent hypotensive agents. This was the reason for the use of additional calcium channel and beta blockers for BP control for patients with inadequate BP control, to ensure adequate BP control throughout the study.
Like Parving et al., 5 our study showed that the incidence of hyperkalaemia (>5.5 mmol/l) was 37.2% (19/51) in the combined aliskiren and ARB group, 13.3% (8/52) in the aliskiren alone group and 19.2% (10/52) in the ARB alone group (p<0.039).
The problem of hyperkalaemia was 36.9% in the aliskiren group versus 27.1% in the placebo group in the ALTITUDE study. Parving et al.’s AVOID study 5 showed that patients on combined aliskiren and ARB totalled 13.7% with hyperkalaemia >5.5 mmol/l compared with 10.8% for placebo group and 4.7% and 1.7%, respectively, for serum K+ ≥6 mmol/l (p<0.113). Like Parving et al., 5 all our patients had Stage 3 CKD, but we were only using 150 mg/day of aliskiren compared with 300 mg/day in Parving et al.’s study.
Our study, modest compared with the magnitude of the ALTITUDE study, still managed to detect the problem of hyperkalaemia in the group treated with combination therapy with aliskiren and ARB, like those of Parving et al. 5 and Tang et al. 9 We did not measure the levels of aldosterone and PRA in the patients with hyperkalaemia and were therefore unable to ascertain if the hyperkalaemia was due to reduction of serum levels of aldosterone. In the study by Tang et al. 9 all the patients on aliskiren and losartan had reduction of PRA, but only 52% had reduction of serum aldosterone. There was no mention of correlation of hyperkalaemia with reduction of aldosterone levels in Tang et al.’s study.
The solitary patient with TIA in the combination therapy group represents 2.0% or 1/51 patient, with no occurrences in the other two therapeutic groups. This is almost the same incidence of 2.6% reported in the ALTITUDE study. Based on our study it would appear that the findings of the ALTITUDE study would also apply to non-diabetic CKD patients. Recently, Shavit et al. 15 reported that over the past decade some investigators have advocated the use of a mineralocorticoid receptor antagonist, such as eplerenone, in combination with ACEI/ARB, focussing on proteinuria reduction and stabilising renal function in patients with CKD, and the results have been promising.16-18 This may be an alternative to the combined use of aliskiren with ACEI/ARB. 19
Footnotes
Acknowledgements
We would like to acknowledge M/s Irene Ow, M/s Tan Hwee Boon and M/s Chin Yok Moi for Administrative and other support.
Conflict of interest
None declared.
Funding
This is an Academic Trial supported by Singhealth Cluster with IRB approval, CIRB Ref: 208E.
