Abstract
Introduction:
Our previous study of angiotensin II receptor blocker (ARB) administration in rats induced unusual proliferative changes of smooth muscle cells in renal arteriolar walls. The present study examined if the incidence of the changes depended on the rats’ age, and how long it would take to find changes.
Materials and methods:
Six-week-old (juvenile spontaneous hypertensive rats (SHRs)+ARB group, n=15) and 20-week-old (adult SHRs+ARB group, n=10) male SHRs were fed a standard diet (0.4% NaCl) containing valsartan (10 mg/kg/day; Novartis Co.). Fifteen age-matched SHRs were studied as controls. After 4, 8, and 12 weeks, the rat kidneys were examined under light and electron microscopes and through immunohistochemical studies.
Results:
Extremely concentric proliferative changes in afferent arteriolar walls were frequently observed in the juvenile SHR+ARB group compared to the adult SHR+ARB group (48.7±6.8% vs 19.3±6.9%; p=0.0307) at the 12th week. Increased renin expression and arteriolar changes were found from the 4th week in the juvenile SHR+ARB group.
Conclusion:
This study indicates that ARB administration induces unusual proliferative changes and a marked renin-producing cell increase in afferent arterioles more frequently in juveniles than adult rats. It is suggested that the treatment of ARB in juveniles might have a higher risk of changes in renal afferent arterioles.
Keywords
Introduction
It has been reported that renin-angiotensin-aldosterone system (RAAS) blockers, such as angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II type 1 receptor blockers (ARBs), introduce hypertrophy of the juxtaglomerular apparatus (JGA) and hyperplasia of renin-producing cells in renal tissue by activation of the RAAS feedback system.1 –5 We have reported that Zucker fatty rats (ZFRs) presented with unusual proliferative changes of smooth muscle cells (SMCs) in afferent arteriolar walls and an extreme increase in renin-producing cells following long-term oral administration of ARB. 6 ARBs are widely used in adult patients with hypertension and kidney diseases. Currently, ARBs are not an uncommon selection for the treatment of pediatric patients suffering from essential hypertension,7,8 glomerulonephritis 9 and primary nephrotic syndrome. 10 We planned to establish whether such lesions of afferent arterioles would be induced by other ARBs in other strains and in other ages of rats. We tried to histopathologically clarify the influences of ARB administration in juvenile and adult kidneys of SHRs, especially in renal afferent arterioles, because the SHRs are widely used as an animal model of human essential hypertension and they are a cross-bred strain, which is not as genetically exclusive as ZFRs. In this study, we compared the incidence of the unusual proliferative changes induced in juvenile and adult rats and studied how long it takes for the changes in afferent arteriolar walls to occur from ARB administration.
Materials and methods
Animals
Twenty-two six-week-old male SHRs and 16 20-week-old male SHRs were purchased from Charles River (Yokohama, Japan). The rats were divided into the following four groups: the juvenile SHR+ARB group (n=15) and the adult SHR+ARB group (n=10) which were fed a standard (0.4% NaCl) diet containing an ARB (0.08% valsartan, Novartis Co.). The juvenile SHR+ARB group was fed the diet for four weeks (n=4), eight weeks (n=4) and 12 weeks (n=7). The adult SHR+ARB group was fed the same diet for eight weeks (n=4) and 12 weeks (n=6). The two control groups (juvenile control group, n=7, adult control group, n=6) were fed the standard diet for 12 weeks. The body weight was 163.2±7.1 g in the juvenile groups and 356.8±24.8 g in the adult groups. The rats were housed in a room at a temperature of 23±1°C with a 12-h light/dark cycle and were allowed free access to diet and water. All experiments were carried out in accordance with the Animal Experimentation Guidelines of Toho University.
Histological analysis
Rats were anesthetized using thiobutabarbital (Inactin, Wako Pure Chemical Industries Ltd, Osaka, Japan; 100 mg/kg). The left kidneys were removed just after blood samples had been drawn. Each kidney was cut along the long axis, including the hilum. One half of each removed kidney was used for light microscopic studies and immunohistochemistry, and the other half was used for electron microscopic studies. Specimens for light microscopic examination were fixed in 10% neutral-buffered formalin solution and embedded in paraffin. Blocks from each of the four groups were sectioned at 2 µm and stained with hematoxylin and eosin (HE) and periodic acid-silver methenamine-HE (PASM-HE). The sections were examined independently by two pathologists.
Twenty microphotographs were taken using a digital microscopic camera (Olympus BX61, Olympus Co., Tokyo, Japan) at 100-fold magnification in each rat kidney, in random areas that did not overlap. Two pathologists counted the total number of glomeruli and estimated the mesangial expansion of glomeruli, including the mesangial matrix increase and/or mesangial cell proliferation. The degree of lesions was scored as follows: 0, not remarkable; 1, mild; 2, moderate; 3, marked. The mesangial scores were calculated by multiplying each of the affected glomeruli by its degree of mesangial expansion, adding these numbers together and finally dividing the sum by the total number of glomeruli. Tubular atrophy and interstitial fibrosis were estimated as the percentage of the affected area occupying each microphotograph. These were scored as follows: 0, none; 1, 0–30%; 2, 31–50%; 3, ≥51%. These scores were summed up in each group and analyzed statistically. The total numbers of the vertical and transverse cross-sections of arteries and arterioles were counted, and the numbers of arterioles with more than three layers of SMC walls in each microphotograph were enumerated. These were then summed and analyzed statistically. All scoring was performed in a blind manner.
Immunohistochemistry
The additional sections from all of the above paraffin blocks were used for immunohistochemistry. The sections underwent deparaffinization, rehydration, and treatment using antigen-retrieval techniques for each antigen. The primary antibodies used were goat anti-rat renin (gift from Tadashi Inagami, Vanderbilt University, Nashville, USA), anti-rat α-smooth muscle cell actin (SMA, Sigma, Missouri, USA) and monoclonal anti-smooth muscle myosin heavy chain isoform monoclonal antibody (SM-2, Yamasa, Japan).
Immunohistochemistry was performed using the ABC method (LSAB2 kit for use on rat specimens; Dako Japan, Tokyo, Japan).
To verify antibody specificity, sections from each paraffin block were used as negative controls by omitting the primary antibody and replacing it with normal goat immunoglobulin.
The positive stain of an anti-renin antibody was light microscopically examined by 200-fold, scored as follows: 0, negative; 1, locally positive; 2, strong positive in two parts; 3, wide and strong positive in more than two parts of the afferent arteriole in each half of the kidney, and these scores were summed up and divided by the numbers of glomeruli in each specimen. After that, the data was analyzed statistically in the four groups.
Electron microscopic studies
The specimens for electron microscopy were fixed in 5% glutaraldehyde solution, postfixed in osmium, embedded in Epon resin, sliced into ultrathin sections, and then examined under the electron microscope (JEM-1400; JOEL, Tokyo, Japan).
Blood pressure and biochemical measurements
Beginning at six or 20 weeks of age, and every three weeks thereafter, body weight was recorded and systolic blood pressure (SBP) was measured in all conscious rats using the indirect tail-cuff method (BP-98A; Softron, Tokyo, Japan) on a 37°C preheated cloth jacket for about 10 min. The mean of three such recordings was taken from the individual rat’s SBP and heart rate. Each rat was transferred to a metabolic cage for collection of a 24-h urine sample. All urine for each rat was collected for the measurement of protein and creatinine concentrations every three weeks and at the end of the each study. At the 4th, the 8th and the 12th week, the rats were anesthetized with thiobutabarbital (100 mg/kg), and blood samples were obtained from the inferior vena cava, and the animals were sacrificed. The blood samples were used for blood chemistry (Mitsubishi Chemical, Tokyo, Japan) and prorenin/renin concentration was measured by Enzyme-Linked Immunosorbent Assay (ELISA) (RPRENKT-1, Innovative Research, Florida, USA). Plasma aldosterone concentration was measured by liquid chromatography (Liquid chromatography-tandem mass spectrometry (LC-MS/MS) method; Asuka Medical, Kawasaki, Japan).
Statistical analysis
Values are presented as means±standard deviation (SD) except where otherwise stated. One-way repeated measures analysis of variance (ANOVA) was performed for each group by the Kruskal-Wallis test to assess the significance of results. Values of p<0.05 were considered to be significant.
Results
Histological findings
Large and small arteries
The segmental arteries showed over three SMC layers with the same size and regularly arranged SMCs in the four groups. However, the interlobar and arcuate arteries in the four groups sometimes showed irregularities of the SMCs in the arterial walls.
In all four groups, interlobular arteries with increased SMC layers and SMC irregularities were observed frequently. Percentages of morphologically changed interlobular arteries out of all observed interlobular arteries per kidney in the 12th week varied considerably and showed no statistical significances among the four groups (Table 1). Interlobular arteries with multiplication of SMC layers showed a decrease of the diameter of the lumen compared to the thickness of the arterial walls.
Histopathological data in the four groups.

ARB: valsartan; glo: glomerulus; SHR: spontaneous hypertensive rat; SMC: smooth muscle cell.
Data are mean±standard deviation (SD). p<0.05:statistically significant.
Juvenile SHR+ARB; bJuvenile SHR control; cAdult SHR+ARB; dAdult SHR control; ab: a versus b.
Afferent and efferent arterioles
Unusual proliferative changes of afferent arterioles were more frequently observed in the 12th week juvenile SHR+ARB group (Table 1, Figure 1(a)) compared to those in the 12th week adult SHR+ARB group (Table 1, Figure 1(c)). The changes were essentially similar between the juvenile and the adult groups. With PASM-HE staining, the changes of arteriolar wall SMCs very frequently showed an extremely marked increase in layers showing multiple concentric circles in vertical cross-sections. SMCs proliferated and showed marked irregularities in size, shape, and arrangement. In some arterioles, unusual proliferative changes extended to interlobular arteries beyond the bifurcation (Figure 1(a)). Those arteriolar changes showed marked thickness in the arteriolar walls without hyaline deposition which is commonly observed in the arteriolosclerotic arterioles.
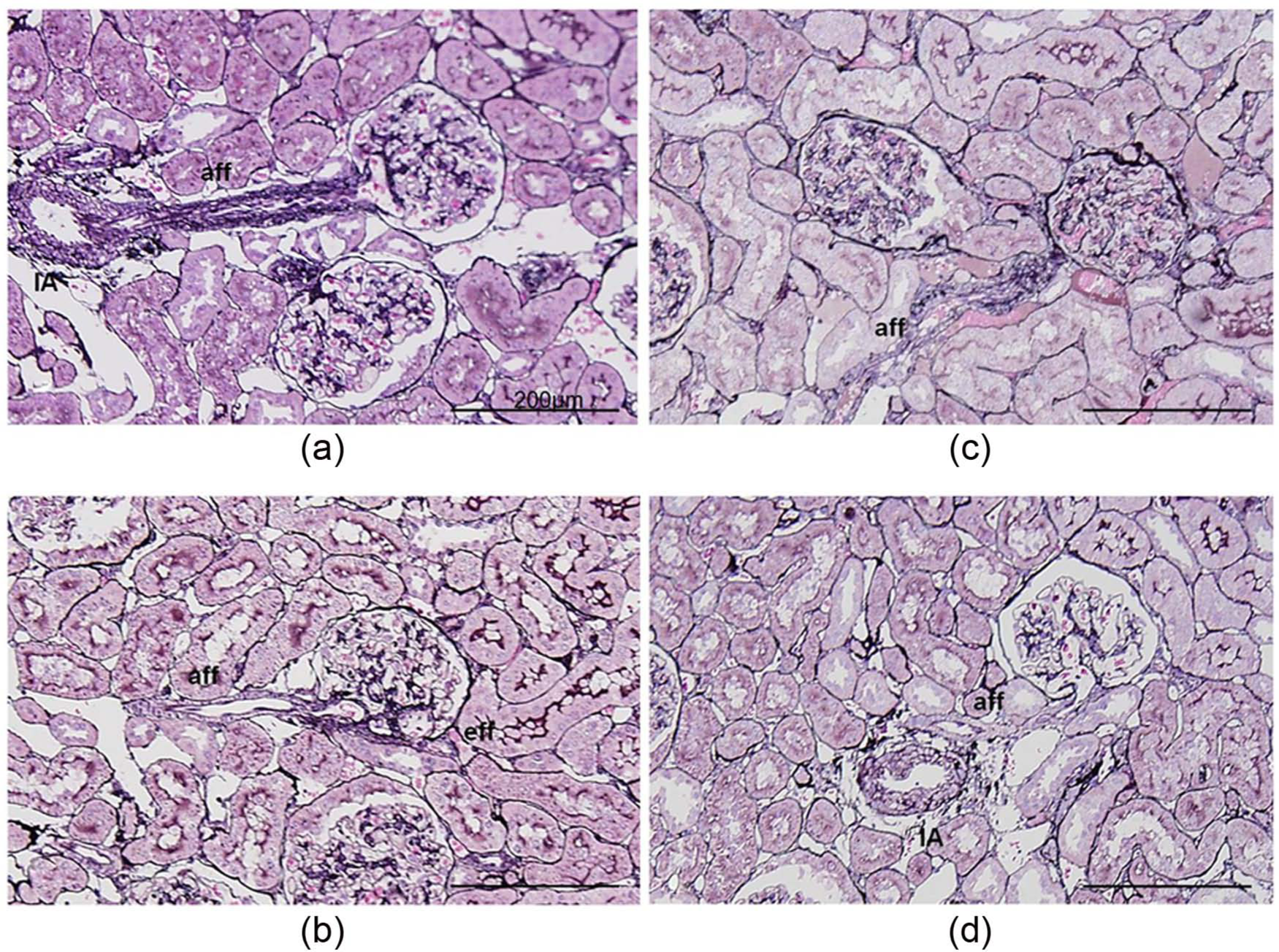
Microphotographs of afferent arterioles in the four groups (periodic acid-silver methenamine- hematoxylin and eosin stain, ×200).
In the juvenile and the adult control groups, SMCs of the arteriolar wall commonly formed one layer, or, infrequently two layers and rarely three layers (Figure 1(b),(d)). We calculated the percentage of arteriolar walls with more than three SMC layers out of all observed arterioles in each rat. It was alarmingly greater in the 12th week juvenile SHR+ARB group compared to that in the juvenile control group (48.7±6.20% vs 5.2±2.7%, p=0.00313: Table 1). The percentage in the juvenile SHR+ARB group was also significantly greater compared to the percentage in the 12th week adult SHR+ARB group (19.3±6.9%, p=0.0307).
Within the juvenile+ARB groups, the occurrence of the arteriolar lesions increased in accordance with the duration of ARB administration. The percentages were significantly increased in the 12th week compared to those in the 4th, and in the 8th week after administration of ARB. (Figure 2, 4W: 31.6±4.2% and 8W: 38.9±4.0% vs 12W: 48.7±6.20%, p=0.0281, respectively).

The change in the percentage of arterioles with more than three smooth muscle cell (SMC) wall layers within all the observed arterioles in the three valsartan treated juvenile spontaneous hypertensive rat (SHR) groups.
In the adult SHR+ARB group, the lesions were not so frequently seen in the 8th week (17.6±3.6%, p=0.0002) and in the 12th week (Table 1) compared to those in the juvenile SHR+ARB groups. At any period of the ARB administration, the lesions were more frequently observed in the deep cortex in both the juvenile and the adult SHR+ARB groups (Figure 3, p=0.0123, p=0.0384, respectively).

The localization of arterioles with more than three smooth muscle cell (SMC) wall layers in the kidney of juvenile and adult angiotensin II receptor blocker (ARB)-treated spontaneous hypertensive rat (SHR) groups.
In the arteriolar SMC lesions, mitosis was rarely identified, and there was no necrosis or inflammation in the arteriolar walls. Granular cells in the afferent arterioles were markedly increased, containing abundant renin granules stained as brownish-black in the outer layers of the afferent arterioles in both the juvenile and the adult SHR+ARB groups. The number of extramesangial cells in the JGA was sometimes increased: however, the macula densa showed no abnormal changes.
Under electron microscopy, affected afferent arterioles showed an increased outer diameter and a decreased inner luminal diameter, with extreme thickening and multiplication of layers in the arteriolar walls. The wall layers consisted of SMC cytoplasm with caveolae and spindle-shaped dense bodies. These were frequently divided into small compartments by irregularly undulating, and thickened basement membranes. The number of nuclei was also increased, but increased cytoplasm volume was more evident, and numerous cytoplasmic processes were arranged irregularly. SMCs in the outer layers of the afferent arteriole frequently contained abundant dense granular structures (arrows), considered to be prorenin/renin (Figure 4(a)). In the control groups, afferent arteriolar SMCs were arranged regularly in one to two layers and sometimes three layers. The SMCs frequently showed a regular cuboidal shape in both control groups (Figure 4(b)).
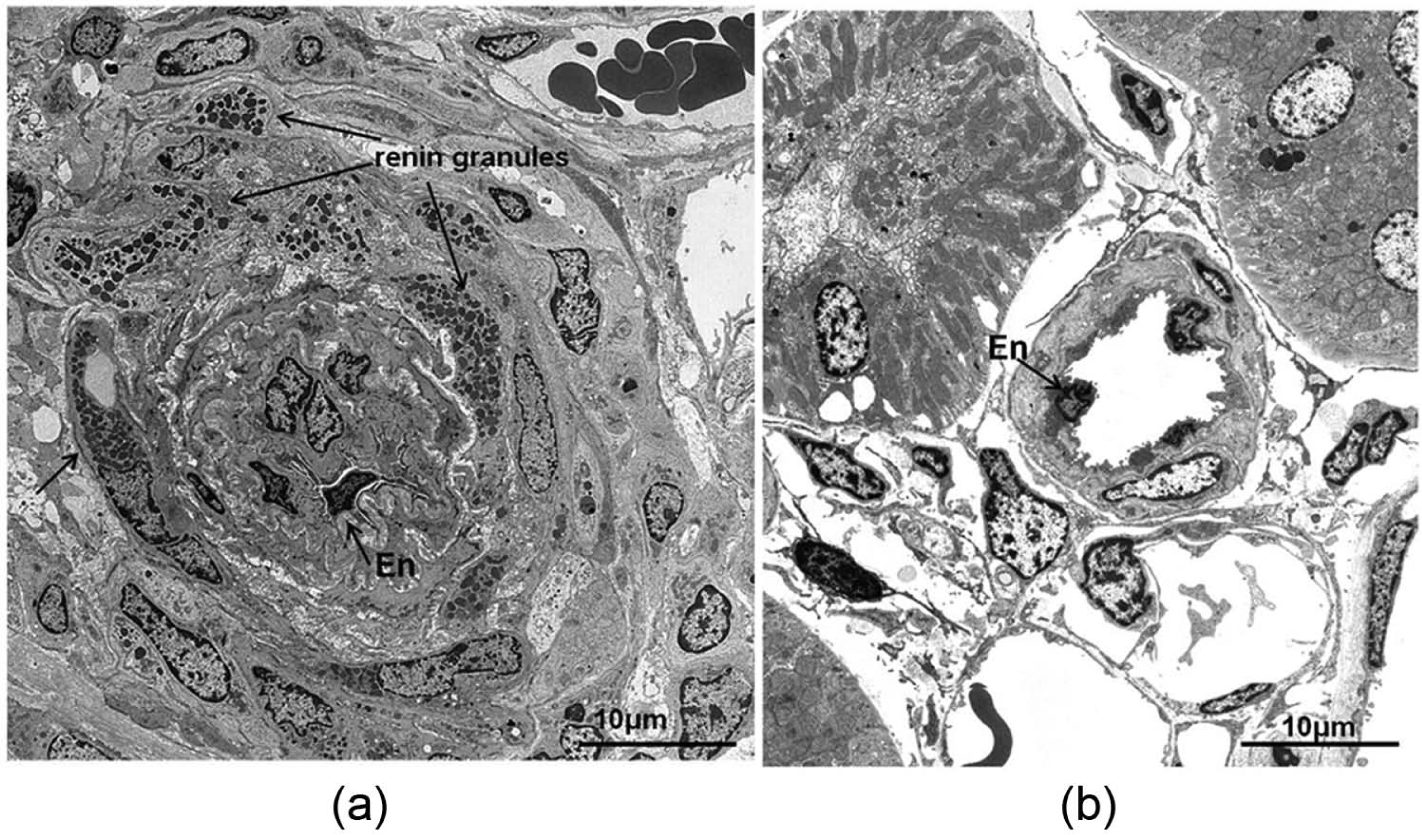
Electron micrographs of afferent arterioles in the juvenile spontaneous hypertensive rat (SHR) groups.
Endothelial cells comprised one layer along the inner lumen, with no significant changes. Changes in efferent arterioles were not as evident as in afferent arterioles, and SMC layers were not increased in both the juvenile and the adult SHR+ARB groups. However, irregularities in the size and shape of SMCs were occasionally seen in each group with or without ARB administration. Lumens of efferent arterioles were not narrowed.
Immunohistochemistry
In the juvenile and the adult SHR+ARB groups, the expression of renin in the afferent arterioles was markedly increased in width, length, and quantity. Renin expression was increased mostly along the outer layer of afferent arterioles, sometimes reaching up close to the bifurcation from interlobular arteries (Figure 5(a)). The positive expression of SMA in the multiplying afferent arteriolar SMCs was distinctly observed; however, smooth muscle cell myosin-2 (SM-2) expression in the affected arteriolar walls was markedly decreased in the juvenile (Figure 5(c),(e)) and the adult SHR+ARB group. The similar positive expression of SMA and SM-2 in the afferent arteriolar SMCs was observed in the both juvenile (Figure 5(d),(f)) and the adult control groups.

Microphotographs of immunohistochemistry in the juvenile hypertensive rat (SHR) groups, (a)–(f) ×100.
The extremely increased expression of renin was seen from the 4th week in the juvenile SHR+ARB group, and the renin expression in the 8th and the 12th week were more markedly observed in the juvenile SHR+ARB group compared to those in the adult SHR+ARB group (Figure 6).

Scores of renin expression by immunohistochemistry.
Laboratory findings
The laboratory data from the four groups is shown in Table 2. Systolic blood pressure (SBP) in the juvenile and the adult SHR+ARB groups showed significant decreases compared to those of the SHR controls (p=0.0205, p=0.0313, respectively). The levels of serum creatinine and blood urea nitrogen were significantly higher in the adult SHR+ARB group compared to those in the adult control group (p=0.0296, p=0.0216): however, the creatinine clearance showed no significant differences.
Laboratory data in the four groups.
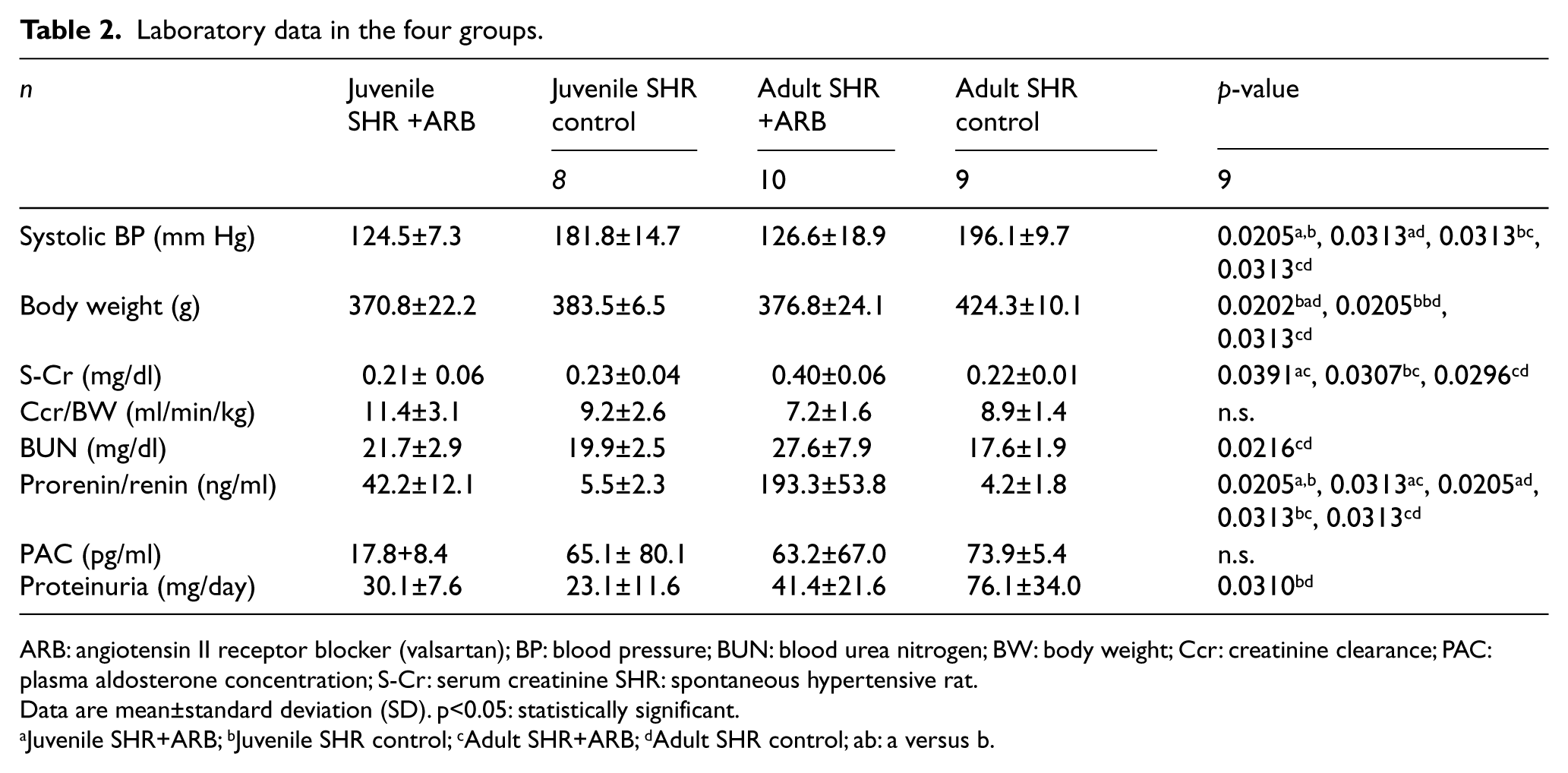
ARB: angiotensin II receptor blocker (valsartan); BP: blood pressure; BUN: blood urea nitrogen; BW: body weight; Ccr: creatinine clearance; PAC: plasma aldosterone concentration; S-Cr: serum creatinine SHR: spontaneous hypertensive rat.
Data are mean±standard deviation (SD). p<0.05: statistically significant.
Juvenile SHR+ARB; bJuvenile SHR control; cAdult SHR+ARB; dAdult SHR control; ab: a versus b.
The prorenin/renin concentrations were significantly elevated in the juvenile and the adult SHR+ARB groups compared to those in the juvenile and the adult control groups (p=0.0205 and p=0.0310, respectively; Table 2). The mean levels of plasma aldosterone concentration in the ARB treated juvenile and adult groups were lower compared to those of each control group; however, there were no significant differences among the four groups. The amount of daily urinary protein excretion increased in the adult control group compared to that in the juvenile control group (p=0.0310).
Discussion
In the 1990s, investigators reported that treatment with ACEIs and /or ARBs caused JGA hyperplasia in renal tissue.1–5 In those reports, toxicopathologists sometimes described thickening of afferent arteriolar walls.1–3 However, this attracted little attention because JGA hyperplasia was a self-evident result induced by the RAAS feedback system. In 2001, Smit-van Oosten reported that, in experimental kidney transplantation, a chronic RAAS blockade preserved glomerular morphology in the absence of proteinuria but induced graft vasculopathy and reduced renal function. 11 In 2004, Racasan et al. described marked SMC hyperplasia and disruption of the wall structure of renal arteries and arterioles that were induced by ACEI or ARB administration in pregnant SHRs starting from the third trimester. 12
In the previous study, we had reported that the extreme proliferation of afferent arteriolar walls were observed in ZFRs given an ARB for 12 weeks. 6 In this study, we found that the same unusual proliferative changes were induced by administration of another ARB in the rats of a different strain from the previous study. We consider that these pathological lesions induced in the intrarenal arteriolar walls are common changes occurring from the treatment of any ARB.
In the unusual proliferative arteriolar lesions, the mural SMCs increased in numbers and layers. The SMCs showed extreme irregularities in size, shape, arrangement and the cytoplasm of the SMCs was markedly enlarged in each cell, with the extension of many cytoplasmic processes observed in the electron microscopy. The expression of SMA in the affected arteriolar walls was preserved: however, the expression of SM-2, a marker of mature myosin heavy chain, 13 disappeared in immunohistochemistry. The findings indicate the increased SMCs are considered to represent activated and dedifferentiated SMCs. The cause of SMC activation or dedifferentiation in hypertensive situations has been reported as endothelial damage from high blood pressure, 14 because the endothelial cells are a defensive wall against many trigger factors for dedifferentiation, such as Platelet-Delived Growth Factor (PDGF), 15 including blood flow. In contrast to this, dedifferentiation in the SMCs of afferent arterioles in our study is not considered to be induced by endothelial damage. A great deal of evidence has suggested that ARBs have protective effects on endothelial cells in both humans and animals,16,17and we found almost no morphological abnormalities in endothelial cells in the juvenile and the adult SHR+ARB groups under electron microscopy. None of the rats with these arteriolar lesions had hypertension.
In this study, the unusual proliferative lesions were observed more frequently in the juvenile SHR+ARB group compared with the adult SHR+ARB group. The results indicate that the occurrence of the proliferative changes on afferent arterioles induced by ARB administration depends on the age of the rat. It is suggested that the SMCs of arteriolar walls in the juvenile rats might be more immature and dedifferentiation of the SMCs would happen more easily compare with the adult rats.
The marked increase of renin expression in the juvenile SHR+ARB group has been shown from the 4th week after administration of ARB (Figure 6). Although renin expression in the 8th and the 12th week adult SHR+ARB group showed an increase compared to that of the adult control group, the levels of expression were less than those shown in the 12th week juvenile SHR+ARB groups. The activation of the RAAS feedback system by ARB administration induces a more extreme increase of renin-producing cells in juvenile rats within a shorter period compared to adult rats. The renin-producing cells arise from the SMCs of afferent arteriolar walls: 18 therefore, we suspect that the SMCs of afferent arterioles in juvenile rats could change phenotype more easily compared to those in adult rats. These results indicate that tissue renin expression shows the same tendencies as the unusual proliferative changes of SMCs in juvenile rats which occur more seriously within a shorter duration compared to those in adult rats.
The extreme arteriolar thickening observed in the juvenile and the adult SHR group narrowed the arteriolar lumens. After 12 weeks of ARB administration, mild renal dysfunction was detected in the adult SHR+ARB group compared to the adult control group in our study (Table 2). Many reports have recently been published about the beneficial effects of ARB treatment for atherosclerosis in the coronary arteries.19,20 Small arteries or arterioles, such as in mesenteric resistant vessels in rats 21 and gluteal subcutaneous arteriolar biopsies in humans, 22 showed a decreased media/lumen ratio with ARB treatment. However, most of these reports on arteriolar media thickness (media/lumen ratio) examined extra renal vessels.23–25 It is suggested that the proliferative changes in arteriolar walls induced by ARB treatment arise only in the renal vessels accompanied by the extreme increase of renin-producing cells, because no reports have described thickened arteriolar walls caused by ARBs in any other organ.
Sakoda et al. reported human prorenin/renin-induced proliferation of cultured human vascular SMCs without an angiotensin II signaling pathway. 26 Liu et al. reported that rat recombinant prorenin directly stimulates rat vascular SMCs, inducing proliferative and hypertrophic changes dependent on the (pro)renin receptors ((P)RRs), independent of angiotensin II. This pathway was blocked by neither ARBs nor ACEIs. 27 Nguyen et al. described how prorenin/renin binds to (P)RR and directly activates Mitogen-activated Protein Kinase (MAPK) in SMCs independent of angiotensin II. 28 We consider that the proliferative afferent arteriolar changes of SMCs were caused by extremely increased tissue prorenin/renin from a feedback reaction of the RAAS by the ARB.
We consider that the involvement of ARB treatment on proliferative changes in renal arteriolar walls has no relation to lowering blood pressure because both of the adult and the juvenile groups showed lowered blood pressure, and the levels were similar (124.5±7.3 mm Hg in the ARB-treated juvenile and 126.6±18.9 mm Hg in the ARB-treated adult rats: there were no significant differences). In a preliminary study, the blood lowering effect of ARB on the arteriolar changes was investigated using very low concentration of ARB (fed by 0.4% NaCl with 0.003% Olmesartan diet) treatment without lowered blood pressure in ZFRs. However, since we found the same unusual proliferative changes in renal afferent arterioles after 12 weeks (unpublished data), we consider that the blood lowering effect itself does not play any role in these proliferative changes.
The proliferation via the direct blocking effect of AT1 receptors only is deniable because we found the same unusual proliferative changes in afferent arteriolar walls in rats treated by angiotensin II converting enzyme inhibitor which blocks both AT1 and AT2 receptors (unpublished data: abstract presented at the 54th Annual Meeting of the Japanese Society of Nephrology 2011, June, Tokyo, Japan). In 2004, Raccasan et al. also described marked SMC hyperplasia in renal arterioles by ACEI or ARB administration. 12
Conclusions
This study indicates that ARB administration induces unusual proliferative changes in SMCs on afferent arteriolar walls and an extreme increase of renin-producing cells in rats. These two changes are more frequent and more serious in juvenile rats compared with adult rats. We consider that the excessive activation of the RAAS feedback system plays the key role of the induction of these changes: therefore, these changes could be induced by any ARB, and could be accelerated by combination therapy with ACEIs or diuretics. These arteriolar changes narrow the lumens of afferent arterioles and protect glomeruli, reducing transglomerular pressure: however, these changes could ultimately induce renal dysfunction. Close attention must be given when using ARBs for juvenile human subjects because of the risks of such serious renal afferent arteriolar changes, especially when using high-dose ARBs or in combination with ACEIs or diuretics which also activate the RAAS feedback system.
Footnotes
Acknowledgements
The authors are grateful to Toshie Shimozeki, Toho University, for her excellent technical assistance. They would also like to thank Mitsuko Sagawa for the measurement of blood pressure, and Mitsuko Sato for her secretarial assistance.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Funding
This research received no specific grants from any funding agency in the public, commercial, or not-for-profit sectors.
