Abstract
Objective:
The Observational Study on Cognitive function And systolic blood pressure Reduction (OSCAR) was designed to evaluate the impact of eprosartan-based therapy on cognitive function in a cohort of 25,745 hypertensive subjects followed for six months.
Methods:
In this supplementary analysis, we studied the relationship between eprosartan-based therapy and cognitive function (assessed using the Mini-Mental State Examination (MMSE)) after 12-month follow-up of 3600 patients (the long-term follow-up on-treatment population).
Results:
Reduction in blood pressure was sustained over 12 months, with mean systolic blood pressure/diastolic blood pressure 130.9/79 mmHg at one year, compared with 164.3/92.8 mmHg at baseline (p<0.001). The overall mean MMSE score at completion of 12-month follow-up was significantly increased from baseline (27.8±2.7 vs. 26.3±3.5; p<0.001). The increase in MMSE score was observed when the population was stratified by age (p<0.001) and in a subgroup of patients with cerebrovascular disease at baseline (n=290) (p<0.001, 12 months vs. baseline).
Conclusions:
In this cohort of patients, use of eprosartan-based treatment for one year was associated with sustained reduction in blood pressure and stabilization or improvement of MMSE scores. These data are supportive of a role for blood pressure control in the prevention or delay of cognitive decline.
Introduction
Associations have been reported, primarily from longitudinal studies, between higher arterial blood pressure and a decline in cognitive function in adults. In particular, there are indications that sustained higher blood pressure may be associated with increased risk of cognitive decline.1-4
These observations have led to suggestions that intervention against high blood pressure for established clinical reasons such as the prevention of major cardiovascular events and strokes in the middle years of life might have a favourable impact on risk of cognitive decline in older age. 5
Relatively few large blood pressure-lowering trials have incorporated cognitive assessment and a diagnosis of dementia, and the results of individual studies have been variable. Meta-analyses are also not conclusive about the effect of antihypertensive treatment on risk of dementia.6,7 Considerable uncertainty therefore persists about the efficacy of antihypertensive drugs for lowering the risk of cognitive decline.
The Observational Study on Cognitive function And systolic blood pressure Reduction (OSCAR) was an observational study of planned six months’ duration that provided opportunities to explore the impact of once-daily therapy with eprosartan on trends in sitting systolic blood pressure (SBP) and cognitive performance (assessed by means of the Mini-Mental State Examination (MMSE)) in a very large community-dwelling population.
Primary findings from OSCAR indicated that a reduction in arterial blood pressure at six months was associated with a significant improvement in cognitive function, as represented by performance on the MMSE. 8 Cognitive decline was demonstrated in multiple linear regression to be independently associated with age, baseline MMSE score, baseline SBP and SBP reduction.
This report supplements these original findings, providing SBP and MMSE results from three countries in which the total duration of follow-up was extended to 12 months.
Methods
Methodological details and primary results from OSCAR have been published previously.8,9
In brief, this was an open-label, non-comparative, post-marketing survey conducted in 26 countries. Participating patients comprised adults aged ≥50 years with a sitting SBP of ≥140 mmHg (based on an average of two readings taken after 5 min of sitting) and who were eligible for eprosartan-based antihypertension therapy on the basis of newly diagnosed hypertension, inadequate response to existing therapy (comprising average sitting SBP ≥ 140 mmHg from two readings during treatment with pre-study drugs) or inability to tolerate existing therapy. Exclusion criteria were restricted to contraindications to eprosartan as specified in applicable local product labels.
Initial therapy comprised eprosartan 600 mg/day. Investigators were at discretion to supplement this with other drugs as necessary after the first month of treatment. There was an expressed preference, but not a requirement, for the first extra drug to be hydrochlorothiazide. No specific measure of compliance was applied.
The OSCAR protocol stipulated a follow-up of six months. In two countries – Greece and the Netherlands – follow-up was extended to 12 months and in Hungary it continued to 18 months. This extension of follow-up was anticipated in the study protocol. The long-term follow-up on-treatment (LTF) population so identified comprised 3600 patients who used eprosartan during the study.
Cognitive function was assessed using the MMSE 10 at baseline, at six months and after 12 months of follow-up. Official, validated and country-specific versions of the MMSE were used.
The principal efficacy variables of the present analysis were the mean change in SBP and mean change in MMSE scores from baseline to 12 months. Trends in diastolic blood pressure (DBP) and pulse pressure (PP) were also monitored. Correlations between changes in MMSE and SBP/DBP were performed using Pearson analyses.
Within the overall MMSE assessment, a sub-analysis of the three-word recall was performed to evaluate the relationship between blood pressure reduction and episodic memory.
Safety was monitored through the identification of adverse drug reactions (ADRs) by active enquiry.
The design and conduct of the survey conformed to extant principles of medical ethics and good clinical practice.
Statistical methods
Changes in mean blood pressure between each follow-up visit and baseline were compared using a paired t-test. Tests were two-sided with significance defined as p<0.05 (i.e. the 5% probability level). MMSE scores and blood pressure values (SBP, DBP and PP) were compared between visits in patients receiving one, two or ≥3 antihypertensive drugs using analysis of covariance (ANCOVA) and adjustment of the initial value.
Categorical qualitative variables were compared using the Chi squared test or by direct calculation of the degree of significance using Fisher’s exact test. For ordinal qualitative variables the Wilcoxon or Kruskal–Wallis tests were used. Quantitative variables were compared by analysis of variance.
The same methods were applied to an exploratory analysis of outcomes in a subset of 290 patients identified as having a history of cerebrovascular disease (defined as ischaemic stroke, cerebral haemorrhage or transient ischaemic attack) at baseline, which was designed to investigate possible influences of cerebrovascular lesions on changes in blood pressure and MMSE.
Results
Demographics and patient profile
A initial total of 5918 patients were enrolled in Greece (n=4057), Hungary (n=1030) and the Netherlands (n=831). The derivation of a LTF population of 3600 patients and a safety population of 5918 patients is shown in Figure 1.

Flow chart of subject disposition.
Summary demographic and baseline blood pressure data appear in Table 1. This was predominantly a population with systo-diastolic hypertension. SBP and PP trended higher with age whereas DBP declined with age (data not shown).
Demographic and baseline blood pressure data of the long-term follow-up on-treatment population population (n=3600). Unless stated otherwise, values are presented in the form mean±SD.

BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; PP: pulse pressure.
After the first month of the survey, 1828 patients (50.8%) were recorded as receiving monotherapy, 1177 (32.7%) were receiving two drugs and 595 (16.5%) were receiving three drugs or more. No further changes to antihypertension regimen were recorded for 75% of patients (n=2643 of 3520) followed up to one year. Most of the changes recorded during that time (n=761) involved the prescription of at least one additional antihypertensive drug.
A total of 220 patients (6.1%) were recorded as receiving a total of 405 treatments that might, in the opinion of the investigator, impact cognitive function. The largest identified classes of such treatments (according to the World Health Organization Drug Classification) were ‘other psychostimulants and nootropics’ (n=55 (13.6%)) and ‘benzodiazepine derivatives’ (n=37 (9.1%)).
Blood pressure trends during 12-month follow-up
Overall trends in SBP, DBP and PP are illustrated in Figure 2. For all three indices there was a substantial and statistically significant (p<0.001) reduction during the first six months of the study, followed by a trend for continued reductions during extended follow-up (p<0.001 for comparisons of 12-month data versus baseline). Some statistically significant differences were noted in blood pressure responses in men and women but these were numerically small (<2 mmHg). The absolute scale of SBP reduction was similar in all age cohorts, but 12-month mean values were progressively higher with increased age, reflecting higher starting values. Mean SBP at 12 months had fallen by 4–5 mmHg more in patients receiving multi-drug therapy than in those receiving monotherapy (p<0.001) but achieved mean levels were similar (130–132 mmHg).
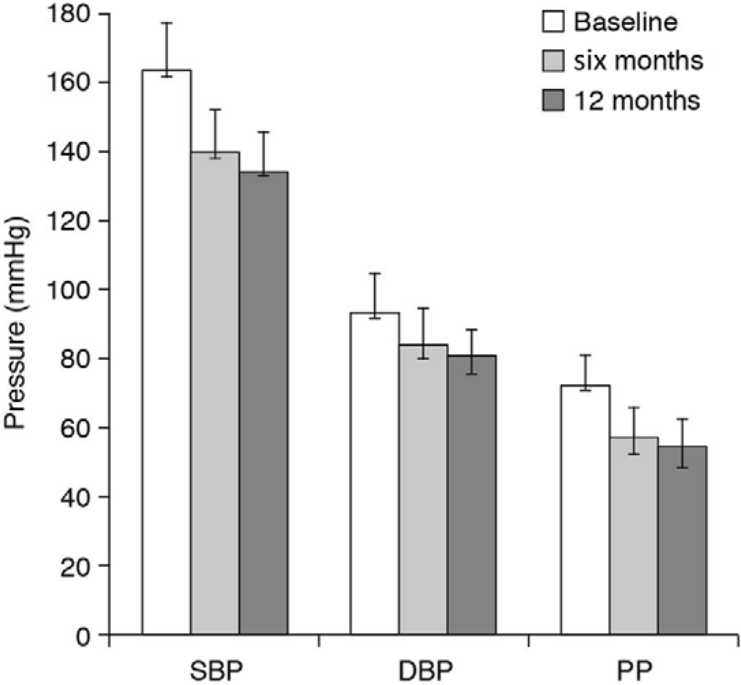
Evolution of systolic blood pressure (SBP), diastolic blood pressure (DBP) and pulse pressure (PP) in the OSCAR long-term follow-up on-treatment population (n=3600).
Changes in PP between baseline and 12 months of follow-up differed significantly according to age, being consistently larger in older patients than younger ones (p<0.001 for all comparisons between age decades).
Normalization of blood pressure (defined as SBP <140 mmHg and DBP <90 mmHg) was recorded in 81.1% of patients (n=2894) at 12 months. A response to therapy (defined as SBP <140 mmHg and/or €SBP ≥15 mmHg or DBP <90 mmHg and/or €DBP ≥10 mmHg) was recorded at 12 months in 97.0% of patients (n=3462). The 12-month normalization rate in men was 83.9%, compared with 78.9% in women. Twelve-month normalization and responder rates did not differ significantly by age, though some arithmetic variations were noted.
Rates of hypertension control were significantly higher in patients receiving monotherapy rather than any form of combination therapy (p<0.001) but rates of response were similar in all categories.
MMSE trends during 12-month follow-up
Table 2 shows mean MMSE scores at baseline and six and 12 months. The statistically significant increase in mean MMSE score reported at six months 8 was still evident at 12 months.
Overall MMSE trends up to 12 months in the OSCAR survey.

MMSE: Mini-Mental State Examination; LTF: long-term follow-up on-treatment.
A significant increase in MMSE score was observed between baseline and 12 months when the population was stratified by age (p<0.001 for overall test by ANCOVA adjusted on initial value). Thus, the mean change in MMSE score was 0.94±2.02 among individuals aged 50–59 years, 1.53±2.12 in those aged 60–69 years, 1.96±2.44 in those aged 70–79 years and 2.18±3.18 in those aged >80 years (Figure 3).

Mini-Mental State Examination (MMSE) trends over 12 months in the OSCAR long-term follow-up on-treatment population (n=3600), stratified by patients’ age at baseline; p<0.001 for overall test of MMSE change between baseline and 12 months, according to age.
The increase in mean MMSE score between baseline and 12 months was larger in subjects with a marked cognitive impairment at baseline (MMSE score <24) (3.87±2.76) than in subjects with an MMSE score at baseline ≥24 (0.94±1.74), although this difference was not significant (p≈0.14). The trend of improvement in MMSE score was similar in all categories of antihypertensive treatment (i.e. monotherapy, bitherapy, multi-drug therapy; p=0.563 by ANCOVA).
Three-word recall element of the MMSE
There was an improvement in performance of the three-word recall test (question 5 of the MMSE) (Figure 4). A very similar profile was discerned when this analysis was repeated with the omission of 220 patients recorded as having received concomitant treatments with potential to influence cognition (data not illustrated).

Change in distribution of performance of the three-word recall element of the Mini-Mental State Examination from baseline to 12 months of follow-up in the OSCAR long-term follow-up on-treatment population (n=3600).
Blood pressure correlations with changes in MMSE score
Exploratory Pearson analysis indicated a significant relation between lower SBP and preservation or enhancement of MMSE scores (correlation coefficient −0.116; p<0.0001). A similar relation was evident for PP (correlation coefficient −0.114; p<0.0001) but not for DBP (correlation coefficient −0.011; p≈0.5).
Trends in MMSE score in patients with cerebrovascular disease
Among the 290 patients identified as having cerebrovascular disease at baseline, mean initial MMSE score was 24.0±4.2 (men 24.5±4.3; women 23.5±4.1). This average value was strongly influenced by the very low mean score (21.6±4.4) registered in the 51 patients aged ≥80 years.
Mean MMSE score in this subset at 12 months of follow-up was 26.4±3.4, representing an average improvement of 2.39±2.75 (p<0.001 vs. baseline mean value). The average increase in MMSE score among patients aged 70–79 years or ≥80 years was 2.8, whereas among younger patients (<70 years) the mean increase was 1.7–1.8.
The 116 cerebrovascular disease patients with MMSE score <24 at baseline had an initial mean MMSE score of 19.7±2.9. This subgroup registered the largest increase in MMSE score of any analysed subset during 12 months of follow-up: MMSE score increased by 4.1±3 points to a group mean value of 23.8±3.4. The corresponding increase in those with initial MMSE score ≥24 was 1.3±1.9 (p<0.001 for comparison).
The scale of MMSE score improvement in the cerebrovascular subpopulation was similar (≈2.5 points) in all categories of treatment (i.e. monotherapy, bitherapy, multi-drug therapy).
Safety data
A total of 114 ADRs were recorded in 83 patients during 12 months of follow-up, of which 111 were treatment-emergent and 66 (in 44 patients) led to study drug termination. Most of these events (n=85) resolved without sequelae. Seventy-four ADRs were rated as mild and 31 as moderate; none were classified as severe.
No deaths were reported in this subset of OSCAR patients during follow-up to 12 months.
Discussion
The possibility of antihypertensive therapy – particularly agents that target the renin–angiotensin system – having beneficial effects on cognition has been a subject of research interest for some time. However, data, and the interpretation of data, from observational studies and controlled trials have frequently been at variance, as exemplified in recent reports from several groups of authors.11-13
Methodologically, OSCAR is an addition to the suite of observational studies that have examined this matter. As we report in this paper, follow-up was extended to 12 months in three countries (n=3600). The reduction in blood pressure (predominantly SBP) and the effect on mean MMSE score recorded at six months was maintained or enhanced at 12 months. After making allowance for the absence of a control group to compensate for the inherent variability of blood pressure, the antihypertensive effect recorded in this subset of the OSCAR population appears to be compatible with other recent reports.14-17
The continued reduction in blood pressure in our long-term cohort coincided with a 1.5-point increase in mean MMSE score from baseline. The clinical relevance of such an increase is open to question for a variety of reasons, some methodological, others arising from factors such as the variable trajectory of MMSE score regardless of baseline status,18,19 the extent to which changes in MMSE score are predictive for the development of recognized deficits of cognition 20 and the possibility that any adverse effects of high blood pressure on cognition may themselves be a function of cognitive status. 21 Moreover, care must be taken not to impute cause and effect to changes noted in a post-hoc analysis. We have no information on the effects (if any) of eprosartan-based therapy on indices such as cerebral blood flow distribution or extent of white matter hyperintensities (see, e.g., Debette et al., 22 Sierra et al. 23 and Sierra 24 ). We applied no techniques (e.g. magnetic resonance imaging/computed tomography scanning or neuropsychological assessment) to discriminate vascular cognitive impairment or degenerative disorders of primarily non-vascular origin. 25 Pearson analysis indicated a relation between lower SBP and PP and the preservation or enhancement of MMSE scores (p<0.0001) but the strength of the correlation was not great (correlation coefficients −0.116 and −0.114, respectively).
These findings are consistent with epidemiological data showing a relationship between PP 26 or arterial stiffness27,28 and cognitive impairment in elderly people.
Effects of eprosartan-based therapy unrelated to blood pressure control or the preservation of vascular structure might be adduced to explain our observations. 29 Other observational studies have suggested that angiotensin receptor blockers (ARBs) may have superior effects on cognitive function compared with other antihypertensive agents. 11 Moreover, experimental studies have suggested that angiotensin II promotes production of amyloid centrally and that ARBs may decrease β amyloid deposition in the brain. 30 Recently, it has been observed that treatment with ARBs was associated with less Alzheimer’s disease-related pathology on autopsy evaluations in humans. 31 Here also, however, our data provide no direct evidence for the operation of specific ARB effects on cognition because of the lack of a control group.
Nevertheless, the MMSE data observed at 12 months are noteworthy and on a scale that might be indicative of meaningful change. 32 The tendency in our patients for the affirmative change in MMSE score to become greater with patients’ age is worthy of remark, as it is against the expected trend of cognitive trajectory. 33 We think it unlikely that these findings can be explained as the operation of a re-test or ‘training’ effect, given that six months elapsed between each administration of the MMSE. In addition, we noted a conspicuous association between time on treatment and performance in the three-word recall domain of the MMSE. This is believed to represent the functioning of episodic memory, which is one of the first domains of cognition to deteriorate in Alzheimer's disease. Performance in this test has been proposed as a simple measure of episodic memory. 34 Similar effects were reported in a substudy of the Study on Cognition and Prognosis in the Elderly, albeit in patients older than ours. 35 Nevertheless the possibility of a learning effect contribution to our findings cannot be dismissed.
The MMSE is an appropriate tool for screening in large populations and one of the most widely used. Nevertheless it is subject to a range of influences, several of which (e.g. ethnicity and education36-38) are likely to have been operative in our population. Recruitment of our patients from multiple centres in three countries may provide some assurance against systematic bias but cannot assure against local factors. Education level was not available in this study for adjustment of results and this is a limitation of our research. As regards the possible impact of stroke as a confounder, 39 it may be noted that the improvement in 12-month MMSE status in OSCAR patients with cerebrovascular disease at baseline echoes earlier findings in the Perindopril Protection Against Recurrent Stroke Study and the Heart Outcomes Prevention Evaluation study.40,41 Resource limitations precluded investigation of by-site effects but no such interplay was observed in the overall analysis of the OSCAR cohort. 8
A relationship between mid-life hypertension and the later incidence of dementia and Alzheimer’s disease has been adduced by some researchers42,43 but not others. 13 The population recruited to OSCAR is relevant to this question, with some 30% aged <60 years and ≈70% aged <70 years. Our one-year data confirm that the modest improvement in MMSE score seen after six months of eprosartan-based therapy was maintained. However, establishing the longer-term consequences of these effects would require at least several additional years of further follow-up and we do not have the resources to do this. In general, the need for extended follow-up (perhaps exceeding a decade) with the attendant implications for loss to follow-up is a major practical obstacle to conducting a decisive investigation of this matter. Initiatives such as www.Alzrisk.org may represent the best available way to resolve the question of a possible causal interplay between blood pressure control and the preservation of cognitive function.
Conclusions
Follow-up in 3600 patients participating in the OSCAR survey established that the reduction in SBP and improvement in MMSE score seen after six months in the entire study population was maintained when eprosartan-based hypertension treatment was extended up to one year. Similar results were recorded in subsets of patients with cerebrovascular disease at baseline.
Our interpretation of these new OSCAR data is that they support the possibility of a favourable influence of blood pressure therapy on cognitive function in middle-aged patients with hypertension at baseline but provide no conclusive proof of such an effect nor any proof that such an effect would be sustained or clinically relevant in later life.
Footnotes
Acknowledgements
Preparation of this report was assisted by Hughes associates, 3 Collins St, Oxford, OX4 1XS, UK.
Author contributions
All the named authors contributed to the formulation of the study, the development of the study protocol and the conduct of the study in their respective countries. All the named authors contributed to the analysis of the data and the development of the manuscript.
Conflict of interest
OH reports consulting and/or lecture fees from Abbott, Novartis, Boehringer Ingelheim, Sanofi-Aventis, Sankyo, Servier, Bayer, Schering Pharma and Bristol-Myers Squibb. Y-SL reports no relevant disclosures. AP reports receiving consulting fees and lecture fees from Abbott, Novartis, Sankyo, Servier, Pierre Fabre, Roche.
Funding
This work was supported by Abbott Products Operations AG, Hegenheimermattweg 127, 4123 Allschwil, Switzerland.
