Abstract
Introduction:
Bahrain has a high prevalence of type 2 diabetes mellitus (T2DM). Previously, Angiotensin-converting enzyme (ACE) insertion/deletion (I/D) polymorphism was found to be associated with T2DM in Bahrainis. The relationship between the disease progression in Bahraini T2DM population and the genetic polymorphism of methylene-tetrahydrofolate-reductase (MTHFR) C677T is still under investigation.
Aim:
The current study investigated the distribution of MTHFR C677T gene polymorphism among Bahraini T2DM patients and examined the interaction between ACE I/D and MTHFR C677T polymorphisms on the risk of developing T2DM and its long-term complications.
Materials and methods:
Polymerase chain reaction (PCR) and PCR-restriction fragment length polymorphism (RFLP) were used to test for the presence of ACE I/D and MTHFR C677T polymorphisms in 171 patients with T2DM compared to 188 healthy (non-diabetic) age-matched control subjects from Bahrain.
Results:
The incidence of the DD genotype and D allele of the ACE gene was high among Bahraini T2DM patients. MTHFR allele and genotype frequencies did not differ between patients and controls. No significant relationship was identified between the combinations of ACE I/D and MTHFR C677T polymorphisms with T2DM.
Conclusions:
The results clearly showed an association of the ACE I/D polymorphism with the progression of T2DM, but when it interacts with MTHFR polymorphism no influence was detected on the increased risk of T2DM.
Keywords
Introduction
The Bahraini people consist of an Arabian Peninsula population with a high prevalence of type 2 diabetes mellitus (T2DM) which occurs among 20–30% of the adults. 1 This raises the involvement of genetic risk factors with the disease and the development of one or more of its long-term complications including cardiomyopathy, retinopathy, nephropathy and neuropathy. We have observed that some patients develop one or more of the diabetes-induced complications despite the short duration of their disease whereas others may have had the disease for 10 or more years without any of these complications. 2 These initial observations prompted us to investigate whether genetic risk factors may play a role in the development of T2DM or subsequently acquiring any related complications. Both of the angiotensin-converting enzyme (ACE) and methylene-tetrahydrofolate-reductase (MTHFR) enzymes have been previously suggested as potential candidate genes for the pathogenesis of T2DM and its complications in addition to the development of cardiovascular diseases.3–6
The insertion/deletion (I/D) polymorphism of the ACE gene was the first candidate gene of the renin–angiotensin system (RAS) for a predisposition to diabetic nephropathy. 7 The gene was discovered and described in 1990 by Rigat et al. 8 and was shown to account for about half of the phenotypic variance of serum ACE concentrations in the normal population. 8 The human ACE gene is located on the long arm of chromosome 17 (17q23) and includes 26 exons and 25 introns. The polymorphism is characterized by the presence (insertion) or absence (deletion) of a 287-bp Alu1 element inside intron 16 of the ACE gene, producing three genotypes (DD, II and ID heterozygote).8,9 The highest serum ACE activity was seen in the DD genotype while the lowest was seen in the II genotype. 8 In addition to its role in a predisposition to diabetic nephropathy, the ACE polymorphism has been suggested as associated with such diabetic complications 10 as coronary heart disease,11,12 stroke 13 and hypertension.14,15 Earlier studies have also demonstrated an ethnic variation in the frequency of I/D polymorphism in relation to diabetic nephropathy.7,10,16,17
Frosst et al. 18 identified a C-to-T substitution at nucleotide 677 that converts an alanine to a valine residue which is responsible for the synthesis of a thermo-labile form of MTHFR, a key enzyme in homocysteine metabolism. Individuals with homozygosity for the T allele are predisposed to the development of hyperhomocysteinemia, especially with a low level of plasma folate. 19 However, the relationship between MTHFR polymorphisms and hyperhomocysteinemia has not been well established yet, although C677T polymorphisms of MTHFR variants have been implicated in the pathogenesis of cardiovascular diseases including hypertension. 18
The association of the ACE I/D gene polymorphism with Bahraini T2DM was published in 2011 2 followed by another study 20 in 2012. Collectively, the initial results showed that the frequencies of D allele and DD genotypes were high among the general Bahraini population. 20 Moreover, there is a strong association of D allele and DD genotypes with the high prevalence of T2DM, 2 these initial observations are in total agreement with most reports from other populations, yet not shown to be a risk factor for the development of T2DM. 2 Meanwhile, the relationship between the disease progression in Bahraini T2DM population and the genetic polymorphism of MTHFR C677T is still under investigation.
Therefore, the aim of the current study was to test the distribution of MTHFR C677T gene polymorphism among Bahraini T2DM patients and to examine the interaction between ACE I/D and MTHFR C677T polymorphisms on the risk of development of T2DM and its long-term complications.
Materials and methods
Subjects
This case-control association study recruited a total of 171 unrelated adults amongst Bahraini T2DM patients and 188 age-matched, healthy control non-diabetic Bahraini adults. The project had the relevant ethnical clearance from the Ethics Committee of Arabian Gulf University and other related institutions to undertake the study.
DNA extraction
Venous blood was collected by venipuncture both from overnight fasting T2DM patients and healthy controls in vacutainer tubes with ethylene diamine tetraacetic acid (EDTA) as anticoagulant. Peripheral blood mononuclear cells were used as a source of genomic DNA. Extraction of DNA was accomplished according to the protocol of the QIAamp DNA Blood Mini Spin Column (QiageGmbH, D-40724 Hilden, Germany).
ACE I/D polymorphism genotyping
Genotyping was performed on extracted DNA by the olymerase chain reaction (PCR) using a pair of oligonucleotide primers: 50-CTGGAGACCACTCCCATCCTTTCT-30 and 50-GATGTGGCCATCACGTCAGAT-30 (Biobasic, Markham, Canada). The reactions were performed with 5 pmol of each primer in combination with Taq PCR Master Mix (Qiagen, Valencia, California, USA). PCR was used to detect two alleles of the 190 bp deletion (D allele) and 490 bp insertion (I allele) fragments. DNA was amplified using primer and PCR cycling conditions as described earlier. 21 Each sample that had the DD genotype was re-amplified with an insertion-specific primer pair, which recognized an insertion-specific sequence.22,23 The PCR products was analyzed on 2% agarose gels after staining with TMVistra Green.
MTHFR C677T genotyping
The single point mutation in the gene that codes for MTHFR C677T had been analyzed by PCR–restriction fragment length polymorphism (RFLP) assay as described by Frosst et al. 18 The regions containing the 677 base of MTHFR were amplified by PCR using a sense primer (5′-TGAAGGAGAAGGTGTCTGCGG GA-3′) and an antisense primer (5′-AGGACGGTGCGGTGAGAGTC-3′). The presence or absence of mutation depended on the restriction enzymes to recognize the restriction sequence and then cut it into two bands which were analyzed on 2% agarose gels after staining with TMVistra Green.
Statistical analysis
The distribution of allele and genotype frequencies between patients and controls was determined by allele counting and the statistical analysis was performed using SPSS version 20 statistical package for Windows, which also computed the p value, odds ratio (OR) and 95% confidence interval (CI). The distribution of alleles and genotypes between patients and controls was determined using the goodness-of-fit test, and statistical significance was determined at p<0.05.
Results
Study groups
Table 1 shows the general characteristics of the study participants including number, gender, age, duration of diabetes, body mass index and diabetes-induced long-term complications in both T2DM and control groups. Data are represented as mean±standard deviation (SD). In this study, patients were all diagnosed as T2DM and this was based on the World Health Organization diagnostic and classification criteria of 1998. 24 The criteria for inclusion of T2DM patients included insulin resistance (obesity, hyper- insulinemia and hyperglycemia). None of the patients had ever had keto-acidosis. The unrelated adult Bahraini T2DM patients (n=171) comprised of 85 (52.1%) males and 78 (47.9%) females with a mean age (±SD) of 53±10.00 years (Table 1). These patients also had a high incidence of diabetes-induced long-term complications. The control group (n=188) comprised of 72 (58.1%) males and 52 (41.1%) females with a mean age (±SD) of 55±10.00 years. Moreover, the diabetic patients had the disease for over 11 years. Both groups were obese.
General characteristics of study participants: Type 2 diabetes mellitus (T2DM) vs age-matched control.

p value in comparing T2DM vs controls.
Total number of individuals per group.
Mean±standard deviation (SD).
The genotype frequencies of I/D polymorphism of the ACE gene in T2DM patients compared to control subjects
The genotype and allele distribution of the ACE I/D polymorphism in T2DM patients (n=171) and control subjects (n=188) are shown in Table 2. The frequencies of the II, ID, and DD genotypes of the ACE gene in control subjects were 18.1%, 47.3% and 34.6%, respectively, and in T2DM patients were 12.8%, 39.2% and 48%, respectively. Both groups were tested for the Hardy Weinberg Equilibrium (HWE) at the ACE I/D locus by Chi square (χ2) test with one degree of freedom. The frequency of DD genotypes was significantly higher in T2DM patients than in control subjects (p=0.01, OR=1.7, 95% CI=1.14–2.67). In addition, the frequency of the D allele was significantly higher among patients than control subjects (p=0.01, OR=1.49, 95% CI=1.01–2.25).
Genotype frequencies of angiotensin-converting enzyme (ACE) insertion/deletion (I/D) polymorphism in type 2 diabetes mellitus (T2DM) patients compared to control subjects.

CI: confidence interval; OR: odds ratio.
Comparing T2DM vs controls.
p values compared subjects carrying ID genotype with subjects carrying II genotype.
p values compared subjects carrying DD genotype with subjects carrying II genotype.
The genotype frequencies of MTHFR C677T gene polymorphism in T2DM patients compared to control subjects
Both genotype and allele distributions of the MTHFR C677T gene polymorphism in both groups were tested, and the results are shown in Table 3. The data show no significant association between the MTHFR C677T alleles and genotypes detected within the studied groups.
Genotype frequencies of methylene-tetrahydrofolate-reductase (MTHFR) C677T polymorphism in type 2 diabetes mellitus (T2DM) patients compared to control subjects.
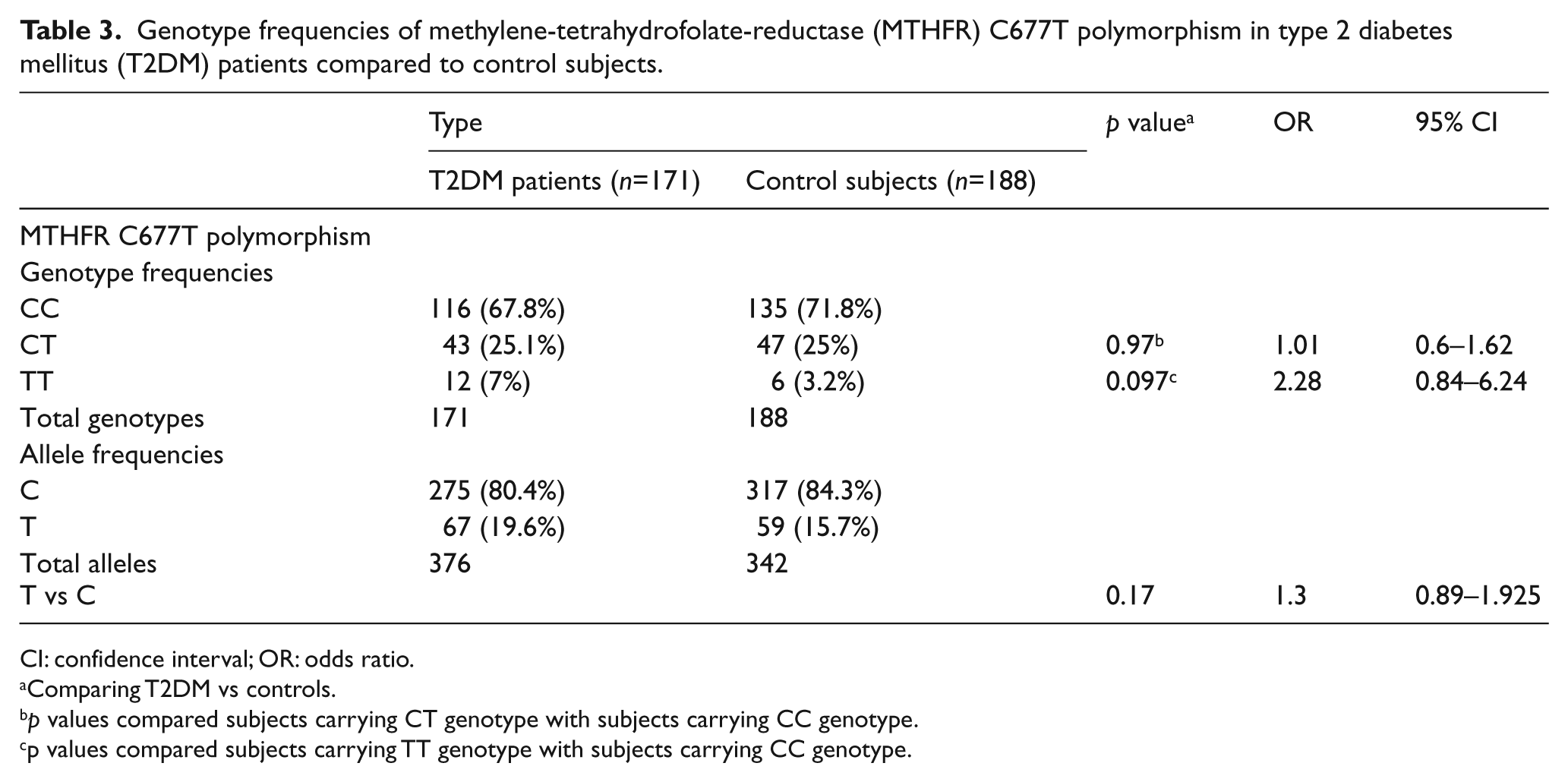
CI: confidence interval; OR: odds ratio.
Comparing T2DM vs controls.
p values compared subjects carrying CT genotype with subjects carrying CC genotype.
p values compared subjects carrying TT genotype with subjects carrying CC genotype.
The genotype frequencies of ACE I/D and MTHFR C677T polymorphisms in T2DM patients compared to controls
Table 4 summarizes the results obtained when combining the ACE I/D and MTHFR C677T polymorphisms and comparing their effect on both case and control groups. The results did not show any significant differences for genotype frequencies between the studied groups for this combination.
Genotype frequencies of angiotensin-converting enzyme (ACE) insertion/deletion (I/D) and methylene-tetrahydrofolate-reductase (MTHFR) C677T polymorphisms in type 2 diabetes mellitus (T2DM) patients compared to controls.
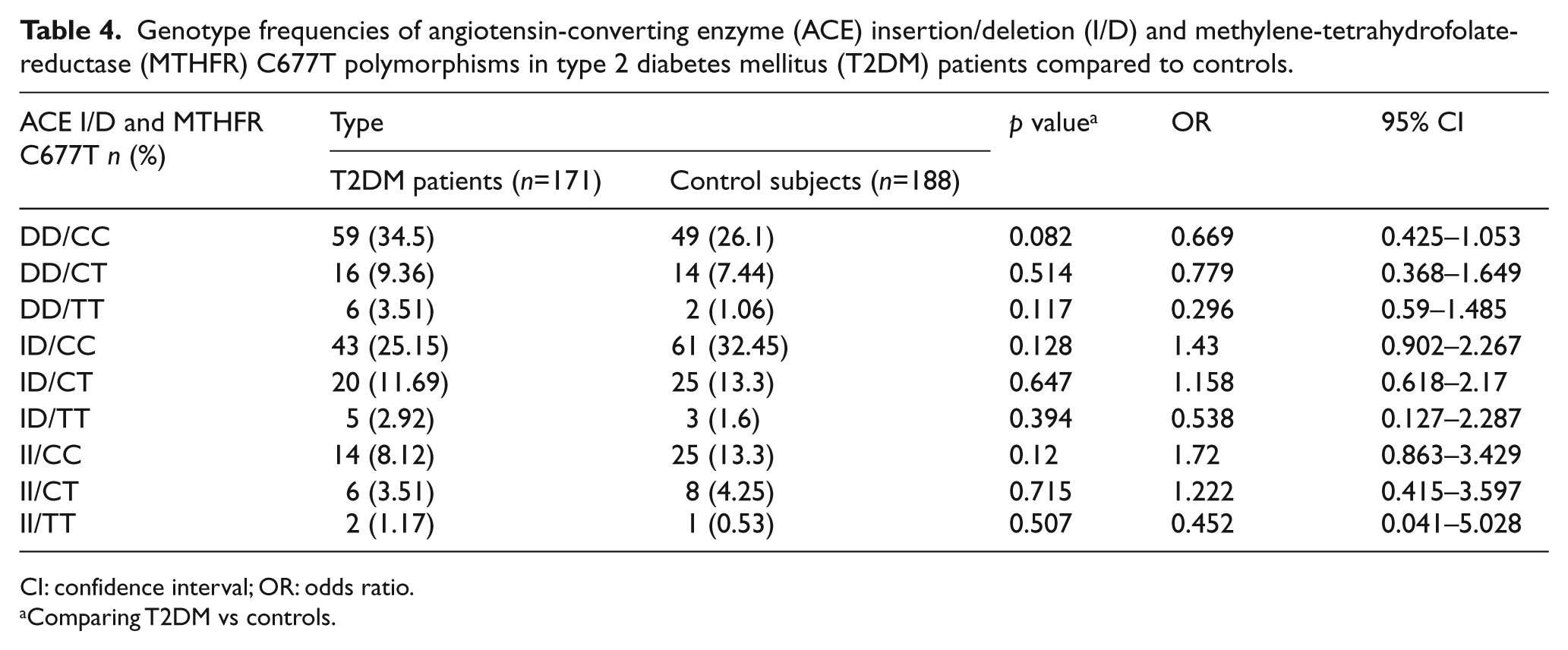
CI: confidence interval; OR: odds ratio.
Comparing T2DM vs controls.
Discussion
In view of the high incidence of T2DM among Bahrainis, 1 the present case–control association study was undertaken for the first time to investigate the distribution of ACE I/D gene polymorphism together with MTHFR C677T gene polymorphism in this population. At the beginning, this study compared the allele and genotype frequencies of both polymorphisms separately in Bahrain’s T2DM patients compared with age-matched non diabetic controls. The results of the ACE I/D gene polymorphism showed high frequencies of the D allele and DD genotypes among T2DM patients. In contrast, MTHFR C677T gene polymorphism was negatively associated with T2DM. In addition, the data for gene combination with the ACE I/D polymorphism did not show any significant correlations with the development of T2DM among Bahraini patients.
The Bahraini population is an Arabian Peninsula population. The characteristics of Arabic populations make them ideal for the study of complex, polygenic, multifactorial disorders such as T2DM. The Arabic populations are characterized by genetic homogeneity due to the tribal structure of their society, large family size and extensive consanguinity1,2 which indicate
In relation to ACE gene polymorphism and its association with T2DM and its long-term complications in the Bahraini people, we reported earlier that there was a high level of the D allele and DD genotypes of the ACE I/D gene among the general Bahraini population. 20 In turn, this may explain the highest prevalence of T2DM and its complications in addition to hypertension in Bahraini population. 1 The mode of action of the ACE I/D gene was not known until now, however, earlier reports showed that D allele is associated with higher plasma ACE concentrations.9,21 Similarly, other studies have now shown that patients with the DD genotype can develop cellular hypertrophy, proliferation and disruption of the extracellular matrix and secrete cytokine and growth factors. Collectively, these initial data suggest that ACE I/D polymorphism is a candidate gene for increasing the incidence, pathogenesis and progression of several diseases including cardiomyopathy, 25 hypertension 26 and T2DM with its long-term macro and micro vascular complications 27 including diabetic nephropathy. 28 This is further supported by data in this and earlier studies confirming the association of D allele and DD genotypes with the risk of development T2DM. 2 Moreover, these observations are also in close agreement with other population studies confirming the association of ACE I/D polymorphism and increased risk of developing T2DM.3,29,30
Although the current study did not show any effect of the MTHFR gene and its combination with ACE I/D gene in the progression of T2DM in Bahrainis, further investigation is required to support the current study. Yet, the relationship between the MTHFR C677T polymorphism and its exact action with T2DM worldwide is under question. Several previous studies have shown no association between the MTHFR C677T polymorphism and T2DM among Taiwanese, 31 Tunisian,3,32 Brazilian, 33 German 34 and Czech populations. 35 In contrast, other data have indicated a significant association with T2DM among Moroccan, 36 Chinese 37 and Polish 38 populations with the possibility of increased risks of cardiovascular disease and hypertension. 39 In addition, ACE I/D and MTHFR C677T polymorphisms in combination have been reported to increase the risk of developing T2DM in the Tunisian populations 3 who suffer from ischemic stroke 6 and pregnancy-induced hypertension. 40 However, neither the ACE I/D nor the C677T MTHFR polymorphism was reported to be associated with risk of myocardial infarction in the Polish population. 38
In conclusion, the present data confirmed the significant association of T2DM with ACE I/D polymorphism but no significant association with MTHFR C677T. Furthermore, no influence was detected on the progression of diabetes when both polymorphisms were combined.
Footnotes
Acknowledgements
The authors would like to thank Khalid Matar, Chief of Medical Technologists, and nursing staff in the Bahrain Defence Force (BDF) for providing the opportunity to collect blood samples for the current study from the BDF Hospital, Kingdom of Bahrain.
Conflicts of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
