Abstract
Introduction:
The purpose of this study was to examine whether the insertion (I) and deletion (D) of angiotensin-converting enzyme (ACE) polymorphism confers susceptibility to psoriasis, vitiligo and rheumatoid arthritis (RA).
Materials and methods:
A meta-analysis was conducted on the association between the ACE I/D polymorphisms and psoriasis, vitiligo and RA.
Results:
Fifteen studies comprising five on psoriasis, five on vitiligo and five on RA were available for the meta-analysis consisting of 2094 cases and 2871 controls. Meta-analysis of the DD+ID genotype showed significant associations with psoriasis (odds ratio (OR) 0.753, 95% confidence interval (CI) 0.601–0.921, p = 0.006). Meta-analysis showed no association between vitiligo and the ACE I/D polymorphism. Meta-analysis of the DD+ID genotype showed an association with RA (OR 2.199, 95% CI 1.379–3.506, p = 0.001). Ethnicity-specific meta-analysis of the D allele showed no association with psoriasis in Europeans, and vitiligo in South Asians. However, subgroup analysis by ethnicity revealed a significant association between the D allele and RA in Arab populations (OR 2.697, 95% CI 1.803–4.034, p = 1.3 × 10−5).
Conclusions:
Our meta-analysis demonstrates that the ACE I/D polymorphism is associated with susceptibility to RA, especially in Arab populations.
Introduction
Psoriasis is a chronic inflammatory skin disorder characterized by keratinocyte hyperproliferation and increased blood flow induced by the stimulation of tissue-resident immune cells by markedly altered cutaneous cytokine profiles.1,2 Vitiligo is a progressive depigmenting disorder characterized by the loss of functional melanocytes from the epidermis and affects 1–2% of the world population. 3 Rheumatoid arthritis (RA) is a chronic inflammatory disease of predominantly synovial joints that causes significant morbidity and shortens life expectancy. 4 Although the etiology of these diseases is not fully understood, interactions between a susceptible genetic background and environmental factors have been suggested. 5
Angiotensin-converting enzyme (ACE) converts angiotensin I into angiotensin II and inactivates bradykinin via the kallikrein–kininogen system. 6 Angiotensin II is the main effector molecule of the renin–angiotensin–system (RAS), is pleiotropic, and is a mediator of the development and progression of diseases. 7 Furthermore, angiotensin II is a potent pro-inflammatory modulator that augments and perpetuates the immune response. 7 The ACE gene, located on chromosome 17q23, contains an insertion (I)/deletion (D) polymorphism within intron 16 that may contain or lack a 287 bp repeat sequence. 8 The DD genotype is associated with approximately a two-fold higher tissue and plasma concentration of ACE than the II genotype. 8 Thus, it seems possible that the D allele could play a role in the pathogenesis of autoimmune diseases.
The ACE I/D polymorphism has been studied in the context of autoimmune disease. However, published results on the genetic association for the ACE I/D polymorphism are controversial and inconclusive.9 –22 This may be due to small sample sizes, low statistical power, and/or clinical heterogeneity. In order to overcome the limitations of individual studies, resolve inconsistencies and reduce the likelihood that random errors are responsible for false-positive or false-negative associations, we employed meta-analysis to further characterize the association. In the present study,23 –25 we used meta-analysis to investigate whether the ACE I/D polymorphism contributes to susceptibility to psoriasis, vitiligo and RA.
Materials and methods
Identification of eligible studies and data extraction
A literature search was conducted for studies that examined the association between the ACE I/D polymorphism and psoriasis, vitiligo and RA. We utilized the MEDLINE and EMBASE citation indices to identify articles in which the ACE I/D polymorphism was determined in patients and controls (up to October 2012). In addition, all references mentioned in the identified articles were reviewed to identify studies not indexed by MEDLINE and EMBASE. The following keywords and subject terms were searched: ‘angiotensin-converting enzyme’, ‘ACE’, ‘psoriasis’, ‘vitiligo’, ‘rheumatoid arthritis’, and ‘RA’. Studies were included in the analysis if they: (1) were case control studies; (2) contained original data; and (3) contained sufficient data to calculate odds ratios (ORs). No language restriction was applied. We excluded the following: (1) studies containing overlapping data; (2) studies in which the number of null and wild genotypes or alleles could not be ascertained; and (3) studies in which family members had been studied because their analysis was based on linkage considerations. The following information was extracted from each identified study: author, year of publication, ethnicity of the study population, demographics, numbers of cases and controls and the frequencies of the genotypes and alleles of the ACE I/D polymorphism.
Evaluation of publication bias and study quality
Funnel plots are used to detect publication bias, but they require a range of studies of varying sizes and subjective judgments, and thus, we evaluated publication bias using Egger’s linear regression test. 26 The Egger’s linear regression test measures funnel plot asymmetry on a natural logarithmic scale of ORs. The chi-square test was used to determine whether observed genotype frequencies in controls conformed to Hardy–Weinberg (H-W) expectations.
Evaluation of statistical associations
We performed meta-analyses using: (1) the allelic contrast (D vs. I); (2) the recessive (DD vs. ID + II); (3) the dominant (DD + ID vs. II); and (4) the homozygote contrast (DD vs. II) models. Point estimates of risks, ORs and 95% confidence intervals (CIs) were estimated for each study. In addition, within- and between-study variations and heterogeneities were assessed using Cochran’s Q-statistic. Cochran’s Q-statistic test assesses the null hypothesis that all studies evaluated the same effect. The effect of heterogeneity was quantified using I2, with a range between 0 and 100%, and represents the proportion of between-study variability attributable to heterogeneity rather than chance. 27 I2 values of 25%, 50% and 75% were nominally assigned as low, moderate and high estimates. The fixed effects model assumes that a genetic factor has a similar effect on disease susceptibility across all studies investigated and that observed variations among studies are caused by chance alone. 28 The random effects model assumes that different studies show substantial diversity and assesses both within-study sampling error and between-study variance. 29 When study groups are homogeneous, the two models are similar. If the study groups lack homogeneity, the random effects model usually provides wider CIs than the fixed effects model. The random effects model is most appropriate in the presence of significant between-study heterogeneity. 29 Statistical manipulations were undertaken using a Comprehensive Meta-Analysis computer program (Biosta, Englewood, NJ, USA). The power of each study was computed as the probability of detecting an association between the ACE polymorphism and vasculitis using a significance level of 0.05 and assuming an OR of 1.5 (small effect size). Power analysis was performed using the statistical program G*Power (http://www.psycho.uni-duesseldorf.de/aap/projects/gpower).
Results
Studies included in the meta-analysis
Twenty studies were identified by electronic and manual searches and 15 were selected for a full-text review based on title and abstract details.9 –22,30 One study was excluded because it contained duplicate data. 30 One eligible study contained data on two different RA groups, and these were treated independently. 21 Thus, a total of 15 separate comparisons met our inclusion criteria.9 –22 These studies comprised five each on psoriasis, vitiligo and RA. They consisted of four European, three East Asian, three Arab, three Turkish and two South Asian studies. In total the studies included 2094 cases and 2871 controls. Selected details of the individual studies are summarized in Table 1. The statistical power of these 15 studies ranged from 16.4% to 86.1%. One of the studies had a statistical power exceeding 80%. 12
Details of the individual studies included in the meta-analysis.

RA: rheumatoid arthritis; OR: odds ratio; CI: confidence interval; H-W: Hardy–Weinberg.
Power calculations assume α = 0.05, OR = 1.5.
Meta-analysis of the association between the ACE I/D polymorphism and psoriasis, vitiligo and RA
Meta-analysis was performed on each patient with psoriasis, vitiligo or RA. A summary of the meta-analysis findings of the relation between the ACE I/D polymorphism and the diseases is provided in Table 2. Meta-analysis of the DD+ID genotype showed significant association with psoriasis (OR 0.753, 95% CI 0.601–0.921, p = 0.006), whereas no such relation was found for the D allele, the DD genotype or the DD vs. II genotype (Table 2; Figure 1).
Meta-analysis of the association between the ACE I/D polymorphism and psoriasis, vitiligo and RA in overall group.

ACE I/D: angiotensin-converting enzyme insertion and deletion; RA: rheumatoid arthritis; OR: odds ratio; CI: confidence interval; F: fixed effects model; I: insertion; D: deletion; R: random effects model.
Studies in Hardy–Weinberg equilibrium.
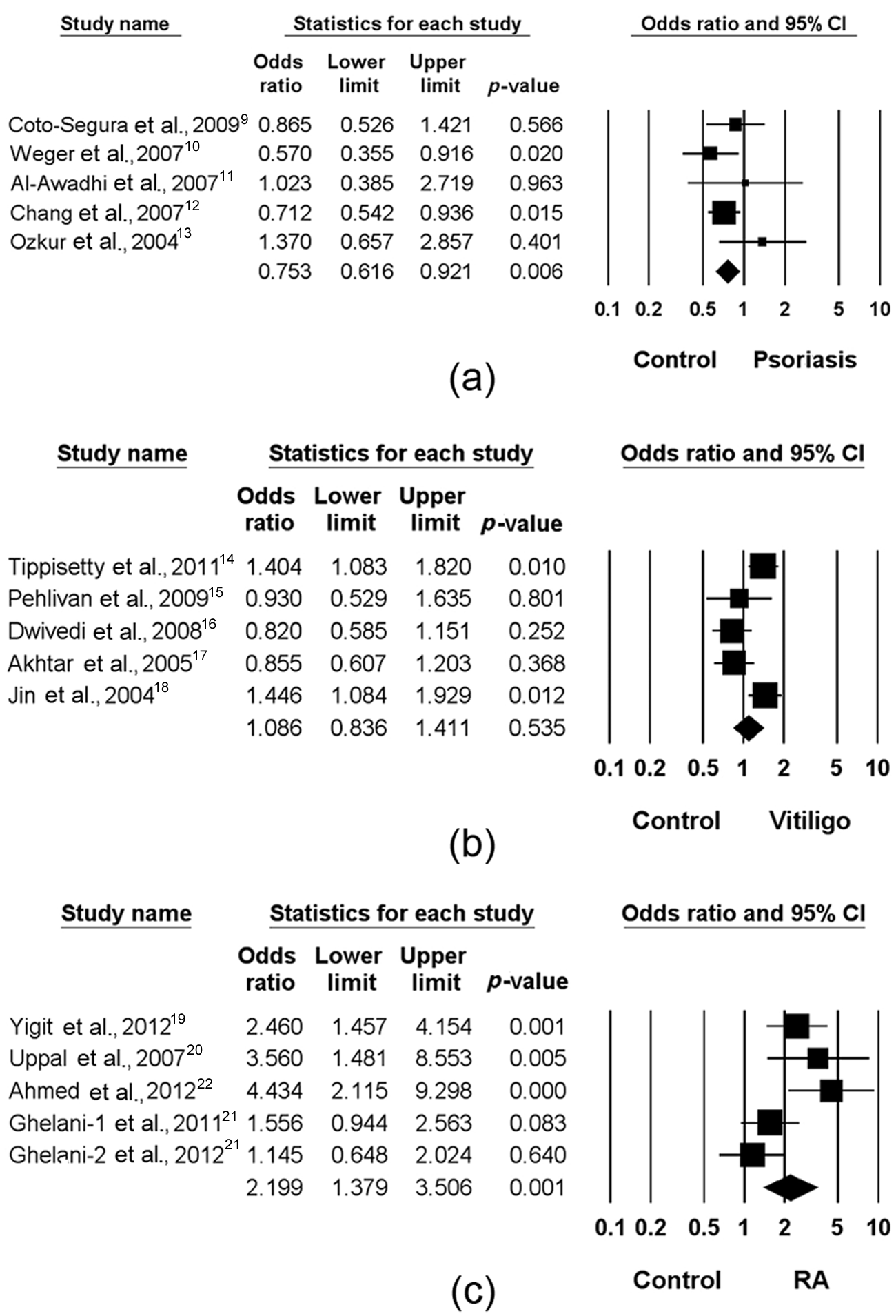
Odds ratios and 95% confidence intervals (CIs) of individual studies and pooled data for the association between the DD + ID genotype and psoriasis (a), between the D allele and vitiligo (b), and between the DD genotype and rheumatoid arthritis (RA) (c).
Meta-analysis showed that the DD+ID genotype was associated with susceptibility to vitiligo (OR 1.277, 95% CI 1.019–1.600, p = 0.034) (Table 2). However, when one study not in H-W equilibrium was excluded from the meta-analysis, the association did not remain significant (OR 1.149, 95% CI 0.867–1.521, p = 0.334) (Table 2). Meta-analysis showed no association between vitiligo and the I/D polymorphism using the allele contrast, recessive model or homozygote contrast model (Table 2; Figure 1).
Meta-analysis showed that the D allele was associated with susceptibility to RA (OR 1.610, 95% CI 1.059–2.450, p = 0.026) (Table 2). However, when one study not in H-W equilibrium was excluded from the meta-analysis, the association did not remain significant (OR 1.558, 95% CI 0.925–2.627, p = 0.096) (Table 2). Meta-analysis of the DD+ID genotype showed an association with RA (OR 2.199, 95% CI 1.379–3.506, p = 0.001) (Table 2). In addition, excluding the study that deviated from the H-W equilibrium among controls did not affect the result (OR 2.169, 95% CI 1.174–4.007, p = 0.003) (Table 2; Figure 1). However, meta-analysis showed no association between the RA and I/D polymorphism using the dominant model or homozygote contrast model (Table 2).
Meta-analysis of the association between the ACE I/D polymorphism and psoriasis, vitiligo and RA in each ethnic group
Subgroup analysis by ethnicity revealed a non-significant association between the D allele and psoriasis in Europeans (OR 0.912, 95% CI 0.644–1.291, p = 0.602) (Table 3). Ethnicity-specific meta-analysis showed that the D allele was not associated with susceptibility to vitiligo in South Asians (OR 1.086, 95% CI 0.642–1.837, p = 0.759) (Table 3). However, subgroup analysis by ethnicity revealed a significant association between the D allele and RA in Arab populations (OR 2.697, 95% CI 1.803–4.034, p = 1.3 × 10−5) (Table 3).
Meta-analysis of the association between the ACE I/D polymorphism and psoriasis, vitiligo and RA in overall group.
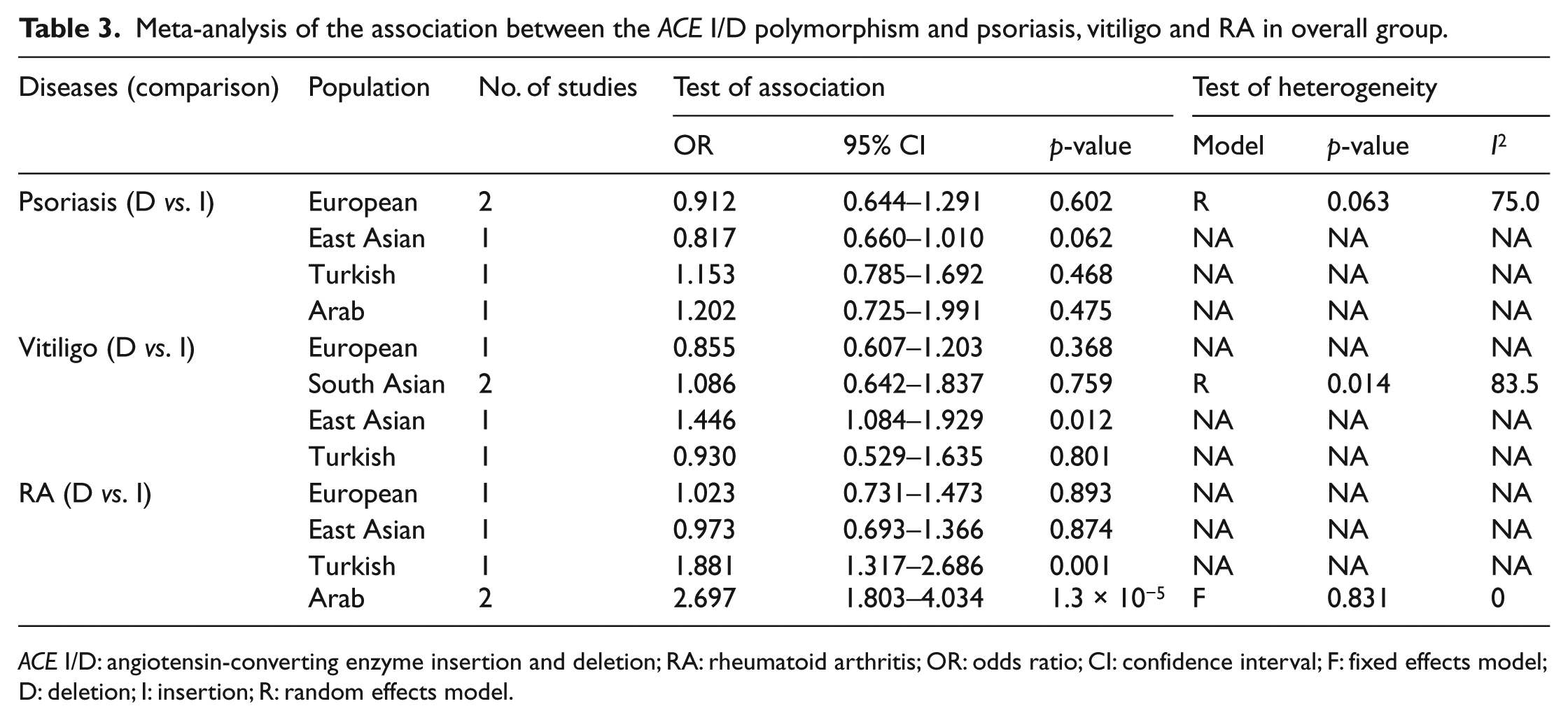
ACE I/D: angiotensin-converting enzyme insertion and deletion; RA: rheumatoid arthritis; OR: odds ratio; CI: confidence interval; F: fixed effects model; D: deletion; I: insertion; R: random effects model.
Heterogeneity and publication bias
The distribution of genotypes of the ACE I/D polymorphism in control groups was consistent with H-W equilibrium in 13 of the 15 studies. Deviation from H-W equilibrium among controls suggests the possibility of bias during control selection or genotyping errors. When we excluded these studies,14,19 the overall results of the DD+ID genotype in RA were not substantially affected (Table 2). Between-study heterogeneity was found during meta- analyses of all diseases, vitiligo and RA, but no evidence of heterogeneity was found for meta-analyses in psoriasis (Tables 2 and 3). Publication bias causes a disproportionate number of positive studies, and poses a problem for meta-analyses. Egger’s regression test showed no evidence of publication bias in this meta-analysis of the ACE I/D polymorphism in any of the diseases studied (Egger’s regression test p-values > 0.1).
Discussion
ACE is expressed in a wide range of tissues including those of the kidneys, heart, lungs, vascular endothelium, skin, joints and testes. ACE plays an important role in RAS, and angiotensin II conversion, directly increases vascular smooth muscle cell contraction and affects smooth muscle proliferation, monocyte adhesion, platelet adhesion, and aggregation. Angiotensin II also acts as a potent pro-inflammatory modulator.7,31 The ACE I/D polymorphism is located on an intron of the ACE gene, and is in strong linkage disequilibrium with genetic factors that influence serum ACE levels. 8 In fact, the ACE I/D polymorphism accounts for approximately one-half of the variance in ACE plasma levels in humans. Furthermore, the polymorphism has been studied in several diseases, including Alzheimer’s disease, myocardial infarction, cerebral infarction, hypertension and diabetic nephropathy. These studies also reported associations between susceptibilities to these diseases and the DD genotype and D allele.9 –22
In this meta-analysis, we combined evidence on the associations between the ACE I/D polymorphism and susceptibility to psoriasis, vitiligo and RA. Meta-analysis of the DD+ID genotype showed significant associations with psoriasis (OR 0.753, 95% CI 0.601–0.921, p = 0.006), whereas no such relation was found for vitiligo. In addition, meta-analysis of the DD+ID genotype showed an association with RA (OR 2.199, 95% CI 1.379–3.506, p = 0.001). Subgroup analysis by ethnicity revealed a significant association between the D allele and RA in Arab populations (OR 2.697, 95% CI 1.803–4.034, p = 1.3 × 10−5).
ACE is expressed by skin components such as vascular endothelial cells, fibroblasts, and keratinocytes.32,33 It has been known that administration of ACE inhibitors can induce or exacerbate psoriasis in clinical practice. 34 Tissue ACE can control the cutaneous inflammatory response by degrading bradykinin and substance P, which are strong mediators of inflammation. 35 Activation of the kallikrein–kininogen system in plasma and tissue has been associated with psoriasis. 36 Moreover, in view of the fact that the ACE I/D polymorphism accounts for approximately one-half of the variance in ACE plasma levels, it is likely that the ACE I/D polymorphism plays a role in susceptibility to psoriasis. 8 This finding suggests that the ACE II genotype reduces ACE activity in skin and may augment the activation of the kallikrein– kininogen system and increase the risk of psoriasis. 12
Although the etiology of vitiligo has not been determined, autoimmune destruction of melanocytes has been suggested. Genetic studies have established that vitiligo susceptibility has a genetic component.37,38 In the present study, we found no association between vitiligo and the ACE I/D polymorphism. The lack of association between vitiligo and psoriasis is unclear. The difference may be due to small sample sizes or differences in the type of skin disease. These two disorders may follow different pathways at the cellular and molecular levels. Relatively few studies reported the association between vitiligo and the ACE I/D polymorphism and their results were heterogeneous. Further studies are required on this topic.
ACE, as a key regulator in inflammatory signal transduction pathway, has been considered to be involved in the pathogenesis of RA. 39 ACE levels are increased in RA synovial fluid suggesting that locally produced ACE may contribute to the joint destruction of RA. 40 ACE converts angiotensin I to angiotensin II, which has a role as a growth factor and as a proinflammatory modulator. Angiotensin II modulates the expression of many cytokines, chemokines, adhesion molecules, endothelin-1, prostaglandins, platelet-activating factor, nitric oxide and cyclooxygenase-2. 39
Our results should be interpreted with caution due to the limited number of studies included in this meta-analysis, which restricted further sub-group analyses. The relative importance of the ACE I/D polymorphism during the development of autoimmune diseases may be dependent on ethnicity. However, we were unable to perform ethnicity-specific meta-analysis due to limited data. Thus, further studies are required to allow meta-analysis by ethnic group to clarify the role of the ACE I/D polymorphism in various ethnic populations.
Our study has several limitations including potential publication bias, heterogeneity and confounding factors that may have distorted the meta-analysis. Secondly, there are varying levels of severity for each disease and the activity level in each disease was unclear. Further research is required to examine whether an association exists between the ACE I/D polymorphism and the activity or clinical features of the diseases. This was not possible in the present study due to limited data.
In conclusion, this meta-analysis of the ACE I/D polymorphism based on a total sample of 2094 patients and 2871 controls demonstrates that the ACE I/D polymorphism is associated with susceptibility to RA, especially in Arab populations. Accordingly, our findings support the notion that the ACE I/D polymorphism plays a role in the pathogenesis of RA. Larger scale studies in populations with different ethnicities are required to explore relationships between polymorphisms of the ACE gene and the pathogenesis of psoriasis and RA.
Footnotes
Conflict of interest
None declared.
Funding
This work was supported by the Korean Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (grant no. A102065).
