Abstract
Introduction:
The aging-suppression gene α-klotho is potentially reno-protective. Animal studies suggest that angiotensin II may be a negative regulator of α-klotho expression. Therefore, we hypothesize that renin–angiotensin system antagonism may increase α-klotho secretion in type 2 diabetes (T2DM).
Subjects and methods:
In this post-hoc analysis of a randomized crossover study, 33 T2DM subjects with albuminuria received either 50 mg of losartan or 20 mg of quinapril (both 50% maximal dose) daily for 4 weeks with 4-week wash-out period in between.
Results:
Our data showed that losartan, but not quinapril, significantly increased circulating α-klotho level by an average of 23% (from 542 pg/ml to 668 pg/ml, p=0.001). Linear regression revealed that, besides different mode of treatment, increment in plasma α-klotho was associated with decrement in urine albumin/creatinine ratio (β=-0.263, p=0.029).
Conclusions:
The angiotensin receptor blocker losartan increases circulating α-klotho in T2DM with albuminuria. The clinical significance of this rise in α-klotho associated with losartan intervention deserves further investigation.
Introduction
α-klotho is a well-established aging-suppression gene. It is expressed as a type I transmembrane protein mainly in the distal tubules of kidney epithelial cells as well as in parathyroid cells, adipocytes and vascular endothelial cells.1–3 Membrane-bound α-klotho is shed from the cell surface by proteolytic cleavage and circulates in the blood as a hormone.4,5 Clinical studies have suggested that circulating α-klotho was a potential reno-protective factor in chronic kidney disease (CKD).6,7 In addition, circulating α-klotho may exert diverse vasculoprotective effects.7,8
Data from animal models showed that angiotensin II might be a negative regulator of α-klotho expression in the kidney. Infusion of angiotensin II in rats markedly down-regulated both α-klotho mRNA and protein expression in an angiotensin II receptor I-dependent, pressor-independent manner. In corollary, angiotensin II receptor blocker losartan reversed angiotensin II induced down-regulation of α-klotho in rats.9,10
Diabetic nephropathy (DN) is the leading cause of CKD globally. Increased activity of rennin–angiotensin system (RAS) contributes to the deterioration of DN.11–13 In corollary, clinical studies have demonstrated that RAS antagonism with angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers might retard the progression of DN. Our group previously reported that 4-week of losartan treatment resulted in amelioration of albuminuria in Asians with type 2 diabetes mellitus (T2DM). 13 It is, however, not known whether plasma α-klotho changes with losartan treatment. In the present post-hoc analysis, we aim to study whether 1) RAS antagonism by losartan or quinapril may increase circulating α-klotho, and 2) change in α-klotho, if any, is associated with anti-albuminuria effect of RAS inhibition.
Subjects and methods
Subjects
A total of 41 subjects with T2DM and albuminuria were recruited in our previous study. 13 Depending on sample availability, 33 subjects were included in the present post-hoc study (age 52.9 ± 9.5 years old, 63% male) and eight subjects were excluded (age 50.9 ± 12.8 years old, 70% male). There was no difference in demography between these 33 subjects and the eight excluded subjects. Type 2 diabetes was diagnosed according to American Diabetes Association Expert Committee recommendation in 1997. Albuminuria was defined by urinary spot albumin over creatinine ratio of ≥30 mg/g on two separate occasions without confounding reasons including urinary tract infection, congestive heart failure, febrile illness and uncontrolled blood glucose (HbA1c > 10%). Exclusion criteria included uncontrolled hypertension (systolic blood pressure (SBP) > 180 mmHg or diastolic blood pressure > 105 mmHg), uncontrolled dyslipidemia (triglycerides > 5 mM or total cholesterol > 8 mM), and major diabetic complications such as peripheral vascular disease or bypass surgery for coronary artery disease. Subjects with previous ACE inhibitor or angiotensin II receptor blocker treatment were also excluded.
Each subject was given either 50 mg of losartan or 20 mg of quinapril (50% of maximum dose) for 4 weeks with a 4-week wash-out period in between, in a crossover design. The order of intervention was randomized. Pill counting was used as a means to verify adherence to assigned intervention. This study design has been explained in detail elsewhere. 13 Briefly, we took into careful consideration the reported onset of action of RAS intervention and anticipated adverse event-related dropout rate.
The study was conducted in accordance with the ethical standards of Declaration of Helsinki. It was approved by our Institution Review board and written informed consent was obtained from each participant.
Biochemical measurement
Plasma glucose was measured by glucose oxidase method and HbA1c was measured using the immunoturbidimetric method (Cobas Integra 800 Chemistry Analyzer, Roche, Basel, Switzerland). An early morning urine sample was collected for measurement of urinary albumin. Albumin and creatinine were measured by commercial assay (Immulite, DPC, Gwynedd, UK). Human soluble α-klotho in plasma was assayed by a solid phase sandwich ELISA using two highly specific antibodies (Immono-Biological Laboratories, Japan) with an intra-assay CV of 3.1% and inter-assay CV of 6.9%.14,15
Statistical analysis
Analysis was performed using SPSS for windows version 19.0. Variables with skewed distribution (e.g. urine albumin/creatinine ratio (ACR)) were log transformed before data analysis. Student’s two-tailed paired t-test was used to compare normally distributed continuous variables before and after each intervention. A general linear model was used to compare post-treatment α-klotho levels between losartan and quinapril after correcting for baseline α-klotho variations. Linear regression was employed to explore the contribution of treatment-induced changes towards changes in ACR. Therefore, δ log-transformed ACR was the dependent variable while the following were considered as independent variables: mode of treatment (losartan or quinapril), δ SBP and δ α-klotho. p<0.05 was considered as statistically significant.
Results
The duration of diabetes in the 33 subjects was 6.4 ± 6.6 years. 13 As shown in Table 1, the baseline clinical profiles of these patients before each drug intervention were similar. As stated in our early reports, losartan treatment lowered SBP slightly better than quinapril (from 135 ± 15 before treatment to 130 ± 13 mmHg after treatment, p=0.045 for losartan; from 135 ± 15 before treatment to 134 ± 15 mmHg after treatment for quinapril). In addition, losartan significantly reduced urine albumin secretion while quinapril treatment showed a non-significant trend towards urine albumin reduction.
Clinical and biochemical parameters before and after 4-week of losartan and quinapril intervention (mean±SD).
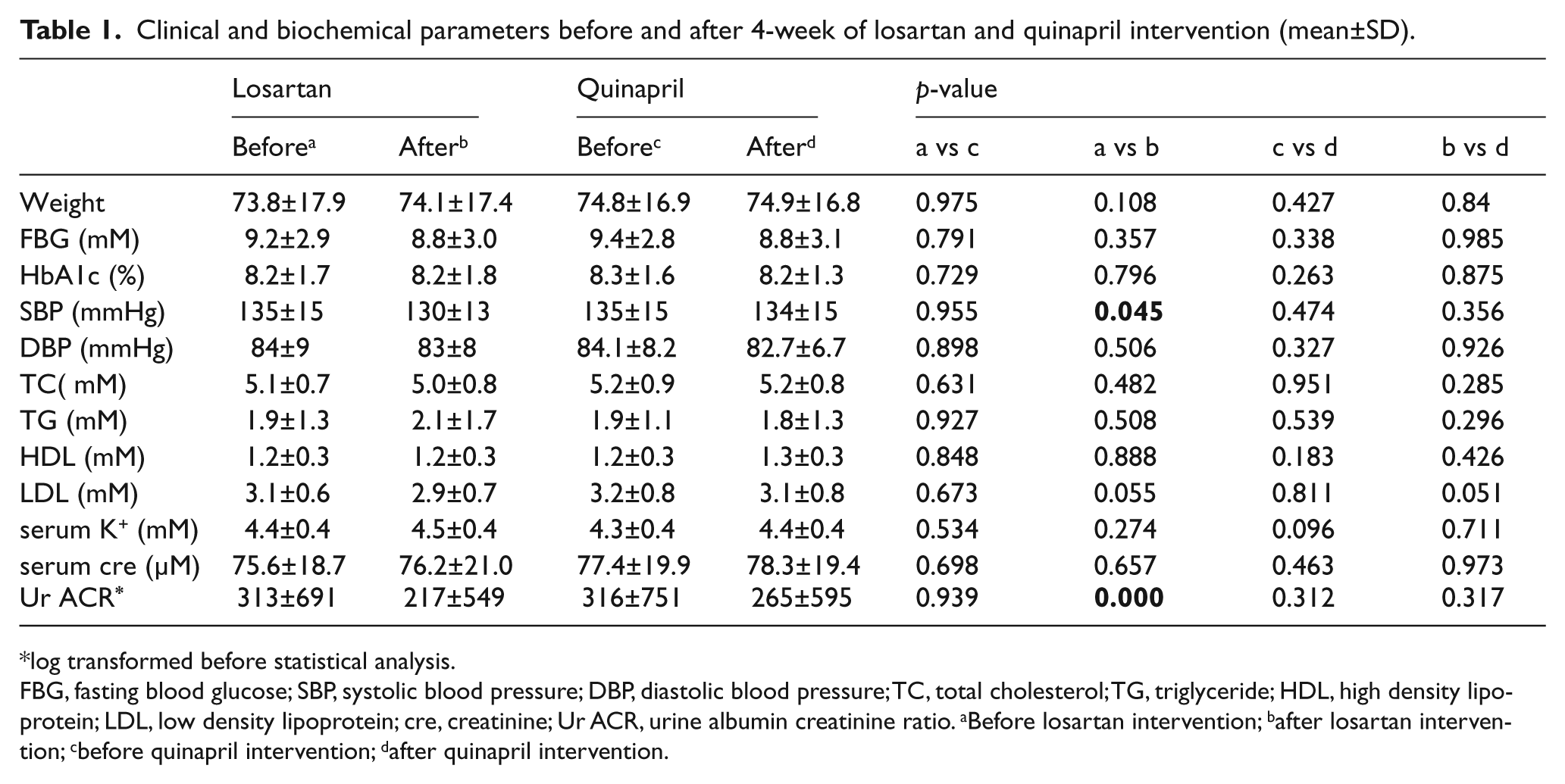
log transformed before statistical analysis.
FBG, fasting blood glucose; SBP, systolic blood pressure; DBP, diastolic blood pressure; TC, total cholesterol; TG, triglyceride; HDL, high density lipoprotein; LDL, low density lipoprotein; cre, creatinine; Ur ACR, urine albumin creatinine ratio. aBefore losartan intervention; bafter losartan intervention; cbefore quinapril intervention; dafter quinapril intervention.
Interestingly, 4 weeks’ intervention of losartan significantly increased the circulating α-klotho level by 23% (from 542 ± 34 pg/ml before treatment to 668 ± 38 pg/ml after treatment, p=0.001). However, the same period of quinapril intervention did not affect the level of circulating α-klotho (from 497 ± 25 pg/ml before treatment to 495 ± 26 pg/ml after treatment, p>0.05, Figure 1). Analysis by general linear model suggested that the α-klotho level after losartan intervention was significantly higher than that of post-quinapril intervention after correcting for baseline α-klotho variations (p<0.0001).

Changes of circulating α-klotho after losartan and quinapril treatment (mean±SEM).
To examine whether treatment-induced changes in SBP and α-klotho contribute to amelioration of ACR, multivariate linear regression analysis was conducted. This revealed that besides mode of treatment (losartan or quinapril), changes in α-klotho was the important predictor of variation in ACR (β=-0.263, p=0.029) while change in SBP showed no association with ACR. Collectively, the model explained approximately 29% of variation in ACR.
Discussion
In this post-hoc analysis of a randomized crossover study, we found that losartan increased circulating α-klotho in type 2 diabetes with albuminuria. To the best of our knowledge, this is the first human study showing that angiotensin II receptor I antagonism increased circulating α-klotho, even though similar findings have been reported in animal models.
Plasma α-klotho was shed from cell surface after cleavage by proteases. The exact source of circulating α-klotho is not well elucidated, since membrane-bound α-klotho is expressed in several types of tissues and cells.1-3 Earlier studies showed that kidney injury significantly lowered plasma α-klotho in rodents. 16 It is therefore tempting to speculate that the kidney may be the main source of circulating α-klotho.
Our observation in humans was certainly compatible with earlier studies which demonstrated that restoration of α-klotho abundance in the kidney by gene transfer could ameliorate angiotensin II-induced proteinuria. It is thus tempting to speculate that amelioration of albuminuria with losartan treatment was partly secondary to an increase in α-klotho.17,18 However, it is also conceivable that tubular injury alleviation secondary to amelioration of proteinuria may increase α-klotho. Although we observed a statistically significant association between increased α-klotho and reduced proteinuria in our subjects, we would like to point out that data provided in this preliminary report alone were insufficient to establish a causal relationship between α-klotho and ACR reduction. Therefore, our novel observation would require replication in another independent cohort.
How losartan caused an increase in α-klotho expression and secretion is unknown. Data from animal models showed that angiotensin II directly repressed expression of α-klotho.9,10 It is thus tempting to hypothesize that antagonism of angiotensin action by losartan may explain the increase of α-klotho in the current study. Lack of change in α-klotho by quinapril might appear to contradict this hypothesis. We postulated that the null of effect by quinapril could be in part dose dependent. Quinapril used this study was only half maximal dose over a short duration of only 4 weeks. Given that ACE inhibition by quinapril can be circumvented by other endopeptidases (e.g. chymase endopeptidase), we speculated that this might explain the null effect of quinapril on α-klotho. 19 This hypothesis is indirectly substantiated by the unexpected non-significant trend towards albuminuria amelioration by quinapril. On the other hand, losartan has several AT1 receptor-independent actions, including its anti-inflammatory and antioxidant properties.20,21 An animal study suggested that free radicals and oxidative stress may involve in angiotensin II-induced α-klotho down-regulation. 9 Hence, it is possible that increased α-klotho by losartan might also be partially attributable to its antioxidant effect.
The strength of our studies included a randomized crossover design. Nearly all of our subjects were still in the early stage of CKD and thus there are fewer confounding factors (e.g. co-morbidities and polypharmacy) affecting the regulation of α-klotho. Our study had several limitations. Firstly, small sample-size studies tend to over-estimate effect size, thereby justifying the need for a larger, similarly well-designed study to more accurately estimate the efficacy of losartan treatment. Secondly, the study was based on post-hoc analysis using bio-banked samples. Therefore, the observation generated could only serve as hypothesis generating. Thirdly, it is known that many factors, such as parathyroid hormone (PTH), vitamin D and serum phosphate etc, may involve in regulation of circulating α-klotho levels. Although the crossover randomization design may reduce the confounding effect of these factors, the relationship between hormones such as PTH and vitamin D, anti-RAS treatment and changes of circulating α-klotho cannot be revealed in our current study.
In conclusion, short-term half-maximal dose angiotensin II receptor 1 blockade with losartan in type 2 diabetes was associated with amelioration of albuminuria and a rise in plasma α-klotho concentration. The clinical significance of this rise in α-klotho will require further investigation.
Footnotes
Conflict of interest
None declared.
Funding
This work was partially supported by Singapore National Medical Research Council Grant [PPG/AH(KTPH)/2011] and KTPH enabling grant [AHEF2012/028].
