Abstract
Introduction:
A low-sodium diet (LSD) was shown to increase both angiotensin II (AngII) and aldosterone levels, and to accelerate atherosclerosis in apolipoprotein E-deficient (E0) mice. The aim of the present study was to examine whether accelerated atherosclerosis in E0 mice fed a LSD is mediated by aldosterone, using the mineralocorticoid receptor blocker, eplerenone (Epl).
Methods and Results:
Mice were divided into three groups: normal diet (ND), LSD and LSD treated with Epl at 100 mg/kg per day (LSD+Epl) for 10 weeks. LSD significantly enhanced plasma renin and aldosterone levels, which were further increased in mice fed LSD+Epl. The aortic lesion area increased three-fold with LSD, while LSD+Epl significantly reduced the lesion area to values similar to ND. Serum and peritoneal macrophages obtained from LSD-fed mice exhibited pro-atherogenic properties including increased inflammation, oxidation and cholesterol accumulation, which were inhibited in mice fed LSD+Epl. In a J774A.1 macrophage-like cell line stimulated with lipopolysaccharide, Epl was shown to have a direct anti-inflammatory effect.
Conclusion:
In E0 mice, Epl inhibited LSD-accelerated atherosclerosis, despite the elevation of renin and aldosterone levels. It is therefore suggested that the atherogenic action of LSD could be mediated, at least in part, by activation of the mineralocorticoid receptor. In addition, eplerenone may have direct anti-inflammatory actions.
Keywords
Introduction
The physiological actions of aldosterone, primarily in epithelial cells, include the regulation of electrolytic balance, followed by water osmosis. In addition, aldosterone is considered to play an important role in the homeostasis of blood pressure. 1
A high dietary sodium intake is positively correlated with hypertension, which is a known risk factor for cardiovascular disease. 2 However, restricted dietary sodium intake, although it reduces blood pressure, is associated with activation of the renin–angiotensin–aldosterone system (RAAS), and with an increased risk for cardiovascular disease (CVD). 3
Dietary sodium restriction has previously been shown to accelerate atherosclerosis in apolipoprotein E-deficient (E0) mice, which was correlated with an increase in angiotensin II (AngII) and plasma cholesterol levels. 4 We have recently shown 5 that a low-salt diet (LSD) increased serum aldosterone levels, macrophages inflammation and lesion size in E0 mice. LDL (low density lipoprotein) receptor knockout mice subjected to a LSD were also shown to have increased plasma renin concentrations and renal expression. 6 Inhibition of angiotensin-converting enzyme (ACE) was recently shown to reduce serum aldosterone levels, and attenuate aortic inflammatory gene expression and atherosclerotic plaque accumulation in E0 mice fed a LSD. 7
High aldosterone levels have been reported to be associated with increased rates of CVD.8,9 The adverse effects of dietary sodium restriction on the cardiovascular function could be attributed to aldosterone, via the mineralocorticoid receptor (MR). The MR blockers spironolactone, and the more specific eplerenone, were shown to improve cardiovascular function and enhance survival in patients with CVD. 10,11 In animals with normal serum aldosterone levels, eplerenone has also been shown to inhibit atherosclerosis 12 and reduce oxidative stress. 13 However, the beneficial effects of MR blockers on oxidative stress in macrophages were only partial when mice were administered high levels of exogenous aldosterone. 14 As a sodium-restricted diet was shown to increase both AngII and aldosterone, the accelerated atherosclerosis induced by a LSD could result from the direct effect of AngII or could be due to the effect of aldosterone, which is downstream to AngII on the RAAS axis. Thus, the aim of the present study was to examine the role of aldosterone and/or MR activation in LSD-induced accelerated atherosclerosis by antagonizing aldosterone using the MR blocker eplerenone in E0 mice.
Methods
Mice
Three-month-old E0 mice were randomly divided into three groups (n=6 per group). One group received regular chow diet and served as control (ND). The other two groups received a low-salt chow diet (LSD) containing less than 0.01% sodium (TD90228, Harlan Teklad, Indianapolis, IN, USA); one of these two groups also received 100 mg/kg per day eplerenone in the drinking water (LSD+Epl). Mice were treated for 12 weeks. Three weeks before the end of the experiment, mice were put in metabolic cages for two consecutive days for urine collection. Blood, mouse peritoneal macrophages (MPM), kidneys, adrenal glands and aortas were harvested at the end of the experiment.
The animal study protocol was approved by the Committee for the Supervision of Animal Experiments of the Technion - Israel Institute of Technology and was conducted in accordance with the Israeli law regulating animal care.
Histopathology of aortic atherosclerotic lesions
The heart and entire aorta were rapidly dissected out and immersion-fixed in 3% glutaraldehyde in 0.1 mol/L sodium cacodylate buffer with 0.01% calcium chloride, pH 7.4 at room temperature. Preparation of the lesion sections and histomorphometric analysis have been described previously. 14
Serum and urine analyses
Serum electrolytes and lipids were determined using commercial kits (Sigma-Aldrich, St. Louis, MO, USA). Aldosterone levels were determined using a commercial radioimmunoassay (RIA) kit (Coat-a-count aldosterone; DPC, Los Angeles, CA, USA). Plasma renin activity (PRA) was determined using a commercial RIA kit (DiaSorin, Stillwater, MN, USA). Renal function was assessed by measuring urine and plasma creatinine and calculating the glomerular filtration rate (GFR) according to the following formula: urine concentration × urine volume × plasma concentration-1.
Blood pressure (BP) measurement
BP was measured in conscious mice with a tail-cuff (five measurements per mouse) linked to an IITC-229 NIBP System and computer software IITC-31 (Life Science Instruments, Woodland Hills, CA, USA).
Serum lipid peroxidation
To determine serum lipid peroxidation, serum samples were incubated for 2 hours at 37°C in the absence or presence of 100 mM 2,2′-azobis-2-amidinopropane hydrochloride (AAPH; Wako Chemical Industries, Osaka, Japan) for 2 hours. AAPH is a water-soluble azo compound that thermally decomposes to produce peroxyl radicals at a constant rate. Plasma lipid peroxide content was determined using the thiobarbituric reactive substance assay. 15
Macrophage cell culture
MPM isolation
Mice were injected with 3 ml phosphate-buffered saline (PBS) intraperitoneally. MPMs were harvested from the peritoneal fluid, washed three times with PBS and resuspended to 106/ml in Dulbecco’s modified Eagle medium supplemented with 5% heat-inactivated fetal calf serum, 100 U/ml penicillin, 100 µg/ml streptomycin, and 2 mM glutamine. The cells were plated on 1.5 mm coverslips pre-coated with fetal calf serum. Cells were incubated in a humidified incubator (5% CO2, 95% air) for 2 hours then washed once with Dulbecco’s modified Eagle medium to remove non-adherent cells. Monolayers were incubated for another 18 hours prior to assay.
Mouse macrophage-like cell line
Mouse macrophage-like cell line, J774A.1, purchased from American Type Culture Collection (Manassas, VA, USA), was plated on 1.5 mm coverslips pre-coated with fetal calf serum. Cells were either untreated (control) or activated with 1 µg/ml lipopolysaccharide (LPS; Sigma-Aldrich, St. Louis, MO, USA), either alone or with the addition of 50 nM eplerenone (LPS+Epl) for 24 hours. Cells were then processed for immunocytochemistry.
Immunohistochemistry of macrophage inflammatory markers
Macrophages grown on coverslips were washed twice with PBS and fixed with ice-cold methanol for 10 min on ice. Following blocking of non-specific binding sites with T-TBS containing 5% BSA, coverslips were stained with the following primary antibodies diluted 1:100 in blocking solution: goat anti-mouse C-reactive protein (CRP; R&D Systems, Minneapolis, MN, USA), rat anti-mouse IL-6, Armenian hamster anti-mouse MCP-1, rat anti-mouse CD54 (ICAM1; Biolegend, San Diego, CA, USA) and rabbit anti-mouse Nf-Kb p65 (Santa Cruz Biotechnology, Santa Cruz, CA. USA). Following three washes with blocking solution, the respective fluorescein-conjugated secondary antibody (Jackson ImmunoResearch, West Grove, PA, USA) was applied. Negative controls consisted of slides in which only the second antibody was added. Nuclei were counterstained with propidium iodide (Sigma-Aldrich, St. Louis, MO, USA). Slides were examined in a Nikon E600 upright microscope with Plan Apo 60×/1.4 oil Dic objective using radiance 2100-confocal imaging system (Bio-Rad, Hercules, CA, USA), and visualized by a Green HeNe (543 nm) laser and red (637 nm) laser diode. Image Pro Plus® 5.0 software (Media Cybernetics, Bethesda, MD, USA) was used for intensity analysis.
LDL and oxidized LDL (oxLDL) uptake by MPM
LDL was separated from human serum by sequential ultracentrifugation. 16 oxLDL was prepared by incubation of 100 µg protein/ ml LDL with 5 µmol/l CuSO4 for 2 hours at 37°C. FITC-conjugated lipoproteins were prepared as described by Smythe et al. 17 MPMs were incubated at 37°C for 3 hours with FITC-conjugated native LDL or ox-LDL at a final concentration of 25 µg protein/ml. Cellular uptake of these lipoproteins was determined by fluorescence (excitation at 480 nm, emission at 520 nm) using BD FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA).
Gene expression
RNA was extracted from tissue or cells using MasterPure™ RNA purification kit (Epicentre Biotechnologies, Madison, WI, USA). cDNA was prepared using Verso™ cDNA kit (Thermo Scientific, Epsom, UK). Primers and probes for aldosterone synthase, renin and GAPDH were designed by Primer Design, Southampton, UK. Using ABsolute Blue QPCR ROX mix (Thermo Scientific), expression was determined by quantitative real-time polymerase chain reaction (PCR) with Rotor-Gene 6000 amplification detection system.
Statistics
Results are given as means ± SEM. The Student’s t-test was performed for all statistical analyses. A p<0.05 value is considered statistically significant.
Results
RAAS and metabolic changes induced by LSD and LSD+Epl in E0 mice
A LSD, as expected, resulted in RAAS activation. Serum aldosterone levels were 2.5-fold higher in LSD compared with ND mice. Treatment with eplerenone further doubled serum levels of aldosterone in mice fed a LSD (Figure 1A). Adrenal expression of aldosterone synthase was similarly enhanced (Figure 1B). Compared to ND, plasma renin levels were significantly increased, by 20% and 69% for LSD and LSD+Epl mice, respectively (Figure 1C). Expression of renin in the kidneys of LSD mice was 2-fold higher than that measured in ND mice, and LSD+Epl further enhanced expression of renin by 2.6-fold (Figure 1D). Both sodium and potassium secretion were significantly reduced in LSD mice, as expected. Urine volumes were not significantly different between ND and LSD mice. Although eplerenone significantly increased urine volume, the calculated GFR was not significantly different between all groups (Table 1). LSD mice had a significantly reduced systolic blood pressure (SBP) compared to ND or LSD+Epl mice (91.0±1.9, 99.5±2.0 and 96.3±3.0 mmHg, respectively).

A low-salt diet (LSD) increased renin and aldosterone levels, which were further increased by eplerenone treatment in E0 mice. E0 mice were treated for 10 weeks with normal diet (ND), LSD, or LSD+100 mg/kg per day eplerenone (LSD+Epl). Renin levels (A) and kidney expression (B), aldosterone circulating levels (C) and adrenal mRNA expression (D) were determined. Results are expressed as mean ± SEM (n = 6), *p < 0.05 vs ND, †p<0.05 vs LSD.
Physiological parameters of E0 mice fed a normal diet (ND), low-salt diet (LSD) or LSD+eplerenone (LSD+Epl).

Data show mean ± SEM, p<0.05 a vs ND, b vs LSD
Effects of LSD and LSD+Epl on aortic atherosclerotic lesions
In this model, where RAAS is activated by a LSD, atherosclerosis is indeed accelerated, with aortic lesion size 3-fold larger in LSD compared with ND mice. Eplerenone significantly inhibited development of atherosclerotic lesion size in LSD mice, and the size of the aortic lesions did not significantly differ from those in ND mice (Figure 2).

Eplerenone reduced LSD-accelerated atherosclerosis in E0 mice. The aortic arches from E0 mice treated for 10 weeks with a normal diet (ND), low-salt diet (LSD), or LSD+100 mg/kg per day eplerenone (LSD+Epl), were analyzed for their lesion size using histomorphometric techniques. A, B, and C, are photomicrographs of typical atherosclerotic lesions of the aortic arches of E0 mice in ND, LSD and LSD+Epl groups, respectively. D. Mean of atherosclerotic lesion size ± SEM (n = 6), *p < 0.05 vs ND, †p<0.05 vs LSD.
Effects of LSD and LSD+Epl on serum and MPM atherogenic properties
The anti-atherosclerotic effect of eplerenone could be mediated by its action on one or more of the major determinants of atherosclerotic plaque development: oxidation, inflammation and cholesterol accumulation. Serum total cholesterol did not significantly differ between the groups (data not shown). However, high-density lipoprotein (HDL) cholesterol was significantly reduced in LSD mice, to 50% of that measured in ND mice. Eplerenone attenuated the reduction in HDL in LSD mice, such that serum HDL was 28% higher in LSD+Epl mice than LSD mice (Figure 3A). As HDL is known to protect serum from oxidation, we measured the oxidation of sera obtained from these mice. Serum oxidation mirrored the changes in serum HDL with increases of 77% serum peroxidation in LSD mice compared with only 23% in LSD+Epl mice (Figure 3B). The hallmark of atherosclerotic lesion development is the accumulation of oxidized LDL in macrophages. Eplerenone significantly reduced oxidized LDL uptake by peritoneal macrophages as compared to LSD alone (Figure 3C). However, peritoneal macrophages obtained from all treatment groups did not significantly differ in their ability to uptake LDL (data not shown).

Eplerenone reduced LSD-induced pro-atherogenic markers in serum and peritoneal macrophages from E0 mice. E0 mice were fed for 10 weeks with a normal diet (ND), low-salt diet (LSD), or LSD+100 mg/kg per day eplerenone (LSD+Epl). Serum obtained from these mice was analyzed for HDL cholesterol (A) and serum lipid peroxidation (B). Results are expressed as mean ± SEM (n = 6), *p < 0.05 vs ND, †p<0.05 vs LSD. Peritoneal macrophages obtained from these mice were processed for uptake of fluorescent oxidized-LDL using flow cytometry (C) and CRP immunohistochemistry analyzed for fluorescence intensity using confocal laser scanning microscopy (D). Results are expressed as mean ± SEM (n = 3), *p < 0.05 vs ND, †p<0.05 vs LSD.
Effects of eplerenone on macrophage inflammation
Eplerenone also had an anti-inflammatory effect as determined by CRP expression in peritoneal macrophages obtained from treated mice. LSD alone induced 9-fold increase in CRP expression in MPMs compared with ND. Eplerenone treatment in LSD mice reduced CRP in MPM by 69% compared with LSD alone (Figure 3D). The anti-inflammatory effect of eplerenone was also observed in vitro. The mouse macrophage-like cell line J774A.1, activated by LPS, exhibited a marked increase in expression of inflammatory markers, which was significantly attenuated by 50 nM eplerenone (Figure 4). These results suggest that the anti-inflammatory effects of eplerenone might be aldosterone-independent.
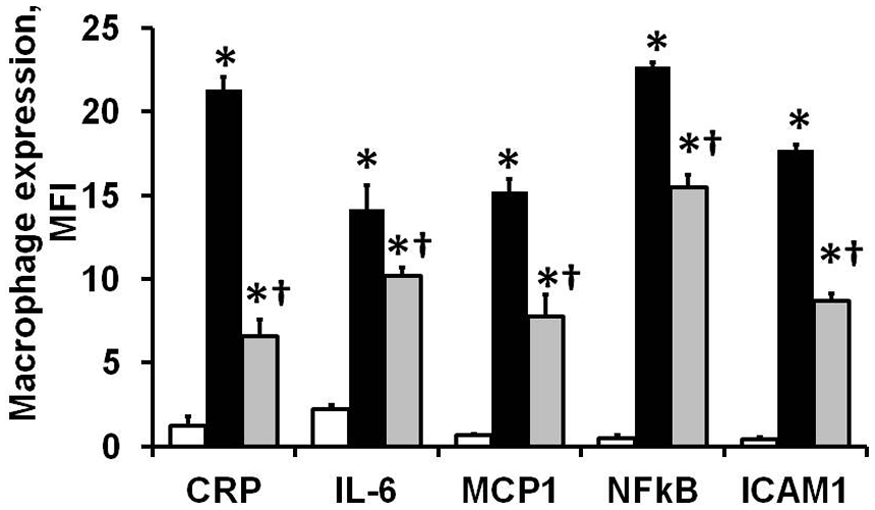
Eplerenone reduced LPS-induced inflammation in a mouse macrophage-like cell line in vitro. The mouse macrophage-like cell line grown in culture was treated for 24 hours with vehicle (control, white bars), 1 µg/ml lipopolysaccharide (LPS; black bars) or LPS with 50 nmol/l eplerenone (LPS+Epl, grey bars). Cells were processed for staining of various inflammation markers and confocal laser scanning micrographs were analyzed for the fluorescence intensity normalized to nuclei. Results are expressed as mean ± SEM (n = 3), *p < 0.05 vs ND, †p<0.05 vs LPS.
Discussion
The results of the present study demonstrate that a sodium-restricted diet activates the RAAS and increases experimental atherosclerosis in E0 mice. The mineralocorticoid blocker eplerenone was shown to counteract the LSD-induced atherosclerosis through effects on reducing oxidation, cholesterol accumulation, and inflammation.
High dietary salt intake is known to be correlated with high BP and an increased risk for CVD. 2 However, the present study and previous reports6,7 demonstrate that, although LSD is associated with lower SBP, LSD is paradoxically linked with an increase in atherosclerosis. Hence, factors other than BP per se could be related to the development of atherosclerosis. The results of the present study, showing that eplerenone reduced LSD-induced atherosclerosis, suggest that aldosterone may mediate the acceleration of atherosclerosis, independent of its effects on BP. This is supported by a recently published study, showing that the elevation of plasma aldosterone, but not other ‘classical’ variables such as blood pressure, is significantly associated with the progression of atherosclerosis in human. 18 The results of the present study are also in line with those of a study showing that the effect of eplerenone on the development of atherosclerosis in monkeys fed a high-cholesterol diet is independent of its blood-pressure lowering effects. 19
Plasma AngII was previously reported to be significantly correlated with aortic atherosclerotic lesion area in E0 mice fed a LSD. 4 ACE inhibition was shown to prevent the effect of LSD on atherosclerosis, 7 while reducing circulating aldosterone levels. In genetically modified mice, where RAAS is constitutively activated, atherosclerosis is accelerated to a similar extent in LSD and ND mice. 7 Consistent with these reports, our results demonstrate activation of RAAS and accelerated atherosclerosis in E0 mice fed a LSD. Our results also demonstrate that blockade of the RAAS downstream to AngII in the RAAS axis, using specific mineralocorticoid receptor blocker, prevented the effects of a LSD on atherosclerosis. Moreover, eplerenone successfully reduced lesion size in LSD-fed mice, despite additional increases in renin and aldosterone.
The protective effects of eplerenone shown in the present study could contribute to our understanding of LSD-induced atherosclerosis in a number of different ways. First, as MR is blocked by eplerenone, and as the rate-limiting enzyme for the formation of RAAS up-stream to aldosterone, i.e. renin, is further elevated in LSD+Epl mice, we would have expected that the extent of atherosclerosis would have been at least similar to that observed in mice fed LSD alone if AngII is the mediator of the adverse effects of a LSD. However, as feeding LSD+Epl resulted in a complete prevention of atherosclerosis, it is less likely that AngII is the principal mediator of the adverse effects of LSD in accelerating atherosclerosis. Second, activation of MR was previously suggested, in in vivo and in vitro studies, to mediate both aldosterone and AngII-induced gene transcription related to cardiovascular injury, fibrosis and inflammation. 20 , 21 Thus, even in the presence of elevated AngII, the results of the present study may point to a possible key role for aldosterone and MR activation, rather than AngII, in the adverse effects of LSD. Third, the ‘classical’ effects of aldosterone are mediated by the MR, but aldosterone also has non-MR effects. 22 As we have shown protective effects of eplerenone despite the elevated circulating aldosterone levels in LSD+Epl, it is suggested that MR-mediated, rather non-MR actions of aldosterone, contribute to the effects of LSD on atherosclerosis.
An excess of aldosterone was previously shown to be associated with increased inflammatory status 23 and eplerenone attenuated the expression of pro-inflammatory markers. 24 In the present study where endogenous aldosterone levels are increased by a LSD, CRP expression is significantly enhanced in MPMs obtained from LSD-fed mice. As the expression of this LSD-induced marker of inflammation was attenuated by eplerenone in these mice, we could conclude that the anti-inflammatory action of eplerenone is related to aldosterone antagonism. In this regard, it is interesting to note that deletion of MR from macrophages has previously been shown to be protective against macrophage activation and inflammatory response.25,26 However, we have also shown that eplerenone reduced expression of inflammatory markers in macrophage-like cells, which were activated by LPS, independent of aldosterone. This result is in line with previously reported independent anti-inflammatory effects of another MR blocker, spironolactone, in mononuclear cells activated by LPS in vitro 27 . It is important to note that, although the affinity of spironolactone for the receptor is 10–20-fold higher than that of eplerenone, the direct anti-inflammatory effects in vitro observed in our study were achieved at concentrations three orders of magnitude lower than those of spironolactone in previous studies. 28 Given the high selectivity and the low concentration required for the direct anti-inflammatory effects of eplerenone in our study, it is suggested that the MR blockade is involved. However, the inflammation signaling pathways induced by LPS and inhibited by MR antagonism needs to be elucidated.
In addition to the anti-inflammatory effects of eplerenone in LSD-fed mice, we have observed other serum and macrophage anti-atherogenic effects, such as reduced oxLDL uptake by macrophages, which was also observed at normal RAAS activity in our previous study. 14 In agreement with other studies,6,7,14,29 LSD did not affect serum total cholesterol levels in E0 mice. However, LSD reduced serum HDL cholesterol in the present study, and eplerenone restored HDL levels in LSD-fed mice. The effect of eplerenone on serum HDL levels, which is considered to be anti-atherogenic, 30 together with its effects on oxidation and oxidized cholesterol uptake by macrophages, make this drug efficient in limiting plaque accumulation under conditions of activated RAAS.
In conclusion, eplerenone, a selective MR blocker, inhibits the acceleration of atherosclerosis associated with AngII /aldosterone activation in LSD-fed mice. Our study suggests a key role for MR in mediating the pro-atherogenic effects of a LSD through regulation of macrophage inflammation, cholesterol accumulation and oxidation.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
