Abstract
The mechanism of synthesis of 1,3,5-trisubstituted-1,2,4-triazoles from 3-thiopheneacetic acetic acid, 4-pyridine formamidine, and tri-fluoro ethyl hydrazine has been first investigated with B3LYP/6-311++G** method in this article. According to the potential energy profile, it can be predicted that the course of the reaction consists of six elementary reactions. The 3-thiopheneacetic acetic acid and 4-pyridine formamidine form first an intermediate product through a dehydration reaction; the intermediate product further combines with hydrogen ion to form a positive ions; the positive ion reacts with three fluorine ethyl hydrazines by a dehydration reaction to form another positive ions; then, it followed by two isomerization reactions, the final reaction with the acetate ion (Ac−) produces the final product. The research results reveal the laws of synthesis reaction of 1,3,5-trisubstituted-1,2,4-triazoles by the carboxylic acids, amidines, hydrazines, and their derivatives on theoretical level. These studies provide the systematic theoretical basis to design and synthesize new 1,3,5-trisubstituted-1,2,4-triazoles.
Introduction
1,2,4-Triazole is a five-membered heteroaromatic compound, containing three nitrogen atoms. It has been extensively used in the field of medicine and pesticide.1–3 At the same time, it presents the potential values of research and development in the field of chemistry, supramolecules, physics, material science, and life science.4–6 Keeping in view immense development value and the potential wide variety of applications of the 1,2,4-triazole compounds, their effective syntheses have received much interest of the chemists and great progress has been made.7–12 According to some exiting results reported in literature, we studied the mechanism of 1,3,5-trisubstituted-1,2,4-triazoles formed by the reaction between 3-thiopheneacetic acid, benamidine, and 2-(2,2,2-trifluoroethyl)hydrazine with the synthetic reaction equations as follows

The results show that the course of the reaction consists of five reactions containing six elementary reactions



The research findings theoretically revealed the mechanism of formation of 1,3,5-trisubstituted-1,2,4-triazoles by the carboxylic acids, amidines, hydrazine, and their derivatives, and it has provided systemic theoretical basis for the synthesis, develop, and application of the 1,3,5-trisubstituted-1,2,4-triazole compounds.
Calculation method
B3LYP/6-311++G** 13 implemented in the Gaussian 09 package is employed to locate all the stationary points along the reaction pathways. Full optimization and vibrational analysis are done for the stationary points on the reaction profile. Finally, the intrinsic reaction coordinate (IRC)14,15 is also calculated for all the transition states to determine the reaction paths and directions.
Results and discussions
Reaction mechanism
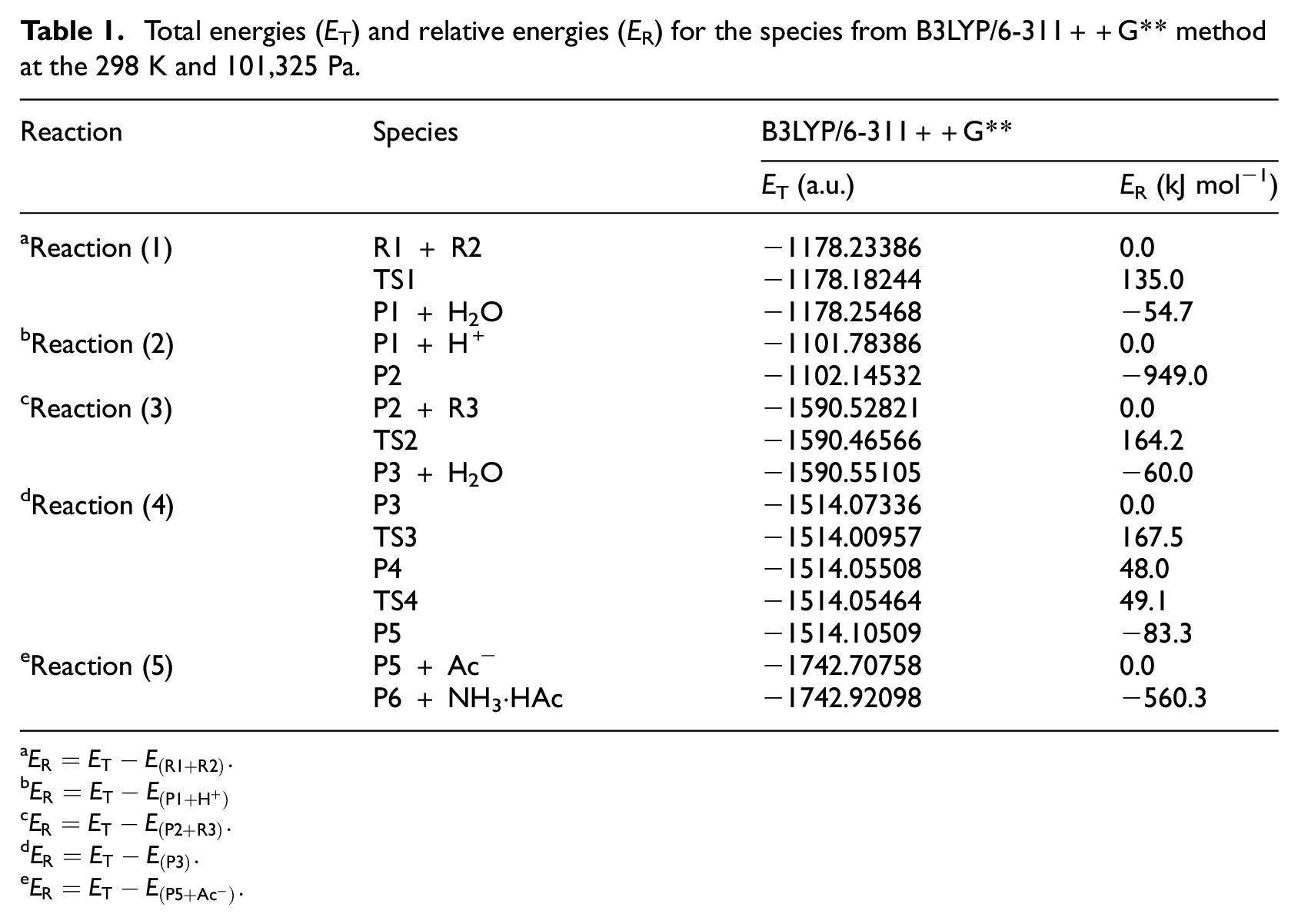
The theoretical calculations indicate that the ground state of 3-thiopheneacetic acetic acid (R1), 4-pyridine formamidine (R2), and three fluorine ethyl hydrazines (R3) are the singlet state. The mechanism of reaction and geometrical parameters of the reactants (R1, R2, R3), transition states (TS1, TS2, TS3, TS4), and products (P1 + H2O, P2, P3 + H2O, P4, P5, P6 + NH3·HAc) which appear in synthesis reaction of 1,3,5-trisubstituted-1,2,4-triazoles by 3-thiopheneacetic acetic acid, 4-pyridine formamidine, and tri-fluoro ethyl hydrazine are given in Figure 1. The energies are listed in Table 1, and molar heat of reaction (ΔrHm) and the molar Gibbs free energy of reaction (ΔrGm) are listed in Table 2. The potential energy profile of the reaction is shown in Figure 2.

The mechanism of reaction and optimized B3LYP/6-311++G** geometrical parameters and the atomic numbering for the species in the reaction. Bond lengths and bond angles are in angstrom and degrees, respectively.
Total energies (ET) and relative energies (ER) for the species from B3LYP/6-311++G** method at the 298 K and 101,325 Pa.
The molar heat of reaction (ΔrHm) and the molar Gibbs free energy of reaction (ΔrGm) from B3LYP/6-311++G** methods at the 298 K and 101,325 Pa.


The potential energy surface for the synthesis reaction of 1,3,5-trisubstituted-1,2,4-triazoles from 3-thiopheneacetic acetic acid, 4-pyridine formamidine, and tri-fluoro ethyl hydrazine with B3LYP/6-311++G**.
The unique imaginary frequencies of the transition states TS1, TS2, TS3, and TS4 are 606.5i, 770.6i, 1659.0i, and 20.2i. Therefore, the transition states can be affirmed as the real ones. The IRC (with the step-length of 0.1 amu−1/2bohr) analysis confirms that TS1 connects R1 + R2 and P1 + H2O, TS2 connects P2 + R3 and P3 + H2O, TS3 connects P3 and P4, and TS4 connects P4 and P5.
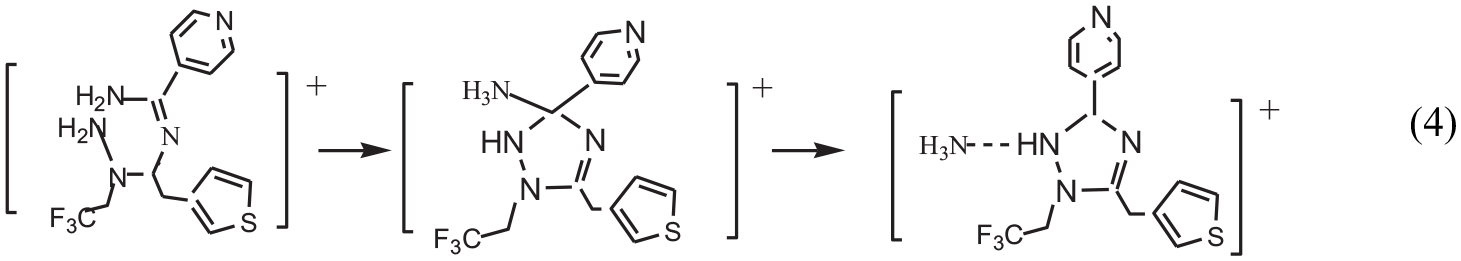
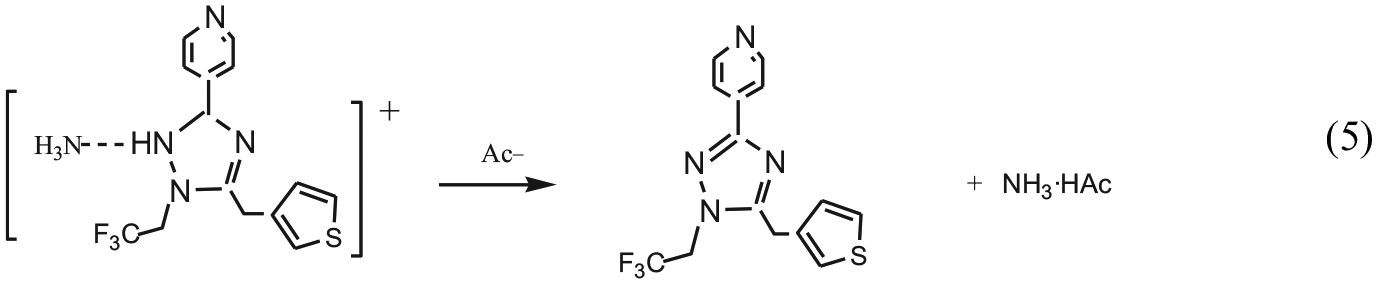
According to Figures 1 and 2, it can be seen that the course of the reaction consists of five reactions containing six elementary reactions: (1) 3-thiopheneacetic acetic acid (R1) and 4-pyridine formamidine (R2) form an intermediate product (P1) through a dehydration reaction with an energy barrier of 135.7 kJ mol−1. (2) In acidic conditions provided by acetic acid (HAc), the P1 further combines with hydrogen ion to form a positive ion (P2), and the reaction is a barrier-free exothermic reaction. (3) P2 and R3 form another positive ion (P3) through a dehydration reaction with an energy barrier of 164.2 kJ mol−1. (4) P3 isomerizes to a positive ion (P4) via transition state (TS3) with an energy barrier of 167.5 kJ mol−1. (5) P4 is further isomerized to the P5 cation via the transition state (TS4) with an energy barrier of 1.1 kJ mol−1. (6) P5 further reacts with Ac− to form 1,3,5-trisubstituted-1,2,4-triazole and ammonium acetate (NH3·HAc), and the reaction is a barrier-free exothermic reaction.
According to Table 2, it can be known that except for the P3 → P4 reaction, the ΔrHm and ΔrGm of the other five reactions are all less than zero. Therefore, at normal temperature and pressure (298 K, 101,325 Pa), these five reactions are spontaneous exothermic reactions. According to Figure 2, it can be known that P4 is a metastable state, P3 → P4 → P5 is a continuous reaction, and the ΔrHm and ΔrGm of P3 → P5 are −88.6 and −96.3 kJ mol−1, respectively. Therefore, although the ΔrGm of P3 → P4 is greater than zero, the continuous reaction of P3 → P4 → P5 is not hindered.
The analysis and explanation of the reaction mechanism
According to Figure 1, when R1 starts to react with R2, as the length of C(1)–N(1) bond (R1 + R2: ∞; TS1: 1.484 Å; P1 + H2O: 1.394 Å) decreases, the lengths of C(1)–O(2) and N(1)–H(7) bond (R1 + R2: 1.365 Å, 1.017 Å; TS1: 2.134 Å, 1.078 Å; P1 + H2O: 3.676 Å, 1.948 Å) elongate gradually. Before the transition state (TS1), the C(1)–O(2) bond was broken, N(1) and C(1) to form a covalent bond. After the transition state (TS1), the N(1)–H(7) bond was broken, H(7)+ ion and [O(2)H(1)]− hydroxyl combine to form a stable water molecule. Thus, the first step reaction has completed (R1 + R2 → P1 + H2O). In the second step reaction, under acidic condition provided by acetic acid (HAc), the O(1) atom in P1 combines with H(14)+ ion to form a P2 positive ion. In the third step reaction, as the length of C(1)–N(4) bond (P2 + R3: ∞; TS2: 1.579 Å; P3 + H2O: 1.364 Å) decreases, the lengths of O(1)–C(1) and H(15)–N(4) bond (P2 + R3: 1.323 Å, 1.017 Å; TS2: 1.998 Å, 1.135 Å; P3 + H2O: 6.222 Å, 7.112 Å) elongate gradually. Before the transition state (TS2), the O(1)–C(1) bond and H(15)–N(4) bond were broken. After the transition state (TS2), H(15)+ ion and [O(1)H(14)]− hydroxyl combine to form a stable water molecule. Thus, the third step reaction has completed (P2 + R3→P3 + H2O). In the fourth step reaction, as the length of C(7)–N(5) bond (P3: 2.769 Å; TS3: 1.569 Å; P4: 1.446 Å) gradually decreases, the lengths of H(17)–N(5) and C(7)–N(2) bond (P3: 1.016 Å, 1.330 Å; TS3: 1.309 Å, 1.516 Å; P4: 2.543 Å, 1.639 Å) elongate gradually. Before the transition state (TS3), H(17)–N(5) bond was broken, C(7) and N(5) to form a covalent bond. After the transition state (TS3), H(17) undergoes hydrogen transfer from N(5) to N(2), the C(7)–N(2) bond was broken. Thus, P3 isomerizes to P4 via transition state (TS3). In the fifth step reaction, as the length of C(7)–N(2) bond (P4:1.639 Å; TS4: 1.653 Å; P5: 3.706 Å) elongates gradually, it forms hydrogen bonds between N(2) and H(16); thus, P4 isomerizes to P5 via transition state (TS4). In the sixth step reaction, because of the interaction between [N(2)H(8)H(9)H(17)] in P5 and Ac−, so that the N(5)–H(16) bond fracture occurred, [N(2)H(8)H(9)H(17)H(16)]+ in P5 and Ac− combine to form NH3·HAc, which results in the formation of P6 + NH3·HAc.
Conclusion
On the basis of the potential energy profile obtained with the B3LYP/6-311++G** method for the synthesis of 1,3,5-trisubstituted-1,2,4-triazoles by the singlet 3-thiopheneacetic acetic acid, 4-pyridine formamidine, and tri-fluoro ethyl hydrazine, it can be predicted that the course of the reaction consists of five reactions containing six elementary reactions. (1) 3-thiopheneacetic acetic acid (R1) and 4-pyridine formamidine (R2) form an intermediate product (P1) through a dehydration reaction with an energy barrier of 135.7 kJ mol−1. (2) Under acidic condition provided by acetic acid (HAc), the P1 further combines with hydrogen ion to form a positive ions (P2). This is a barrier-free exothermic reaction. (3) P2 and R3 form a positive ion (P3) through a dehydration reaction with an energy barrier of 164.2 kJ mol−1. (4) P3 isomerizes to a positive ion (P4) via a transition state (TS3) with an energy barrier of 167.5 kJ mol−1. (5) P4 further isomerizes to a positive ion (P5) via a transition state (TS4) with an energy barrier of 1.1 kJ mol−1. (6) P5 further reacts with Ac− to form 1,3,5-substituted-1,2,4-triazole compound and ammonium acetate, and this is a barrier-free exothermic reaction. The synthesis of 1,3,5-trisubstituted-1,2,4-triazoles by the singlet 3-thiopheneacetic acetic acid, 4-pyridine formamidine, and tri-fluoro ethyl hydrazine is a spontaneous exothermic reaction at ordinary pressure and room temperature (101,325 Pa, 298 K).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (NSFC) (grant number: 31370090) and Project of Key R&D of Shandong Province in China (grant number: 2015GSF121006).
