Abstract
The sorption kinetics and thermodynamic parameters of phosphate removal from aqueous solution using quaternary ammonium–modified bottle gourd biomass as a sorbent were studied in a batch reactor. The cationic sorbent, containing trimethylammonium and hydroxypropyl groups, was obtained through the chemical reactions of the lignocellulosic Lagenaria vulgaris shell with (3-chloro-2-hydroxypropyl)trimethylammonium chloride. Experimental data of phosphate sorption from aqueous solutions of different initial concentrations (5–140 mg P L−1) have been analysed by reaction kinetics and diffusion models. The characteristic rate constants calculated by linear and non-linear regression analyses of the experimental results are presented. The phosphate sorption reaches equilibrium in 20–30 min, depending on the initial phosphate concentration. The maximum sorption capacity of quaternary ammonium–modified bottle gourd (QABG) sorbent was 18 mg P g−1 at 20 oC. The sorption system is best described by a non-linear equation of the pseudo first-order model (R2 > 0.996). The Weber–Morris model indicated that the sorption process took place in three steps, whereby the intra-particle diffusion is not the only rate-controlling step. In addition, the effect of temperature (20 oC–50 oC) on sorption kinetics was also investigated. The various thermodynamic parameters suggest that phosphate sorption is favoured and is an exothermic process. The activation energy and the sticking probability confirmed that anion exchange is the dominant mechanism. These results provide valuable information for the potential use of agricultural residues in the treatment of wastewaters.
Introduction
Wastewater rich in phosphates is not eco-friendly, as phosphates are nutrients in aquatic systems and lead to eutrophication. 1 In order to remove phosphate from wastewater, before discharging into natural watercourses, appropriate treatment methods should be applied. For this purpose, many wastewater treatment technologies have been developed, based on the application of various chemical, biological, and physical methods, separately or in combination.2–4 However, these technologies are not sustainable due to many weaknesses, such as very strict control of specific conditions, high capital investment, potentially new water pollutants, disposal of the generated sludge and waste management costs. 4 Great effort is devoted to the ecological and economic issues of the sorption process using cost-effective and easily available materials. Further development of phosphate sorption has been focused on the investigation of biodegradable sorbents based on agricultural by-products and residues.5,6 Lignocellulosic material, such as bottle gourd biomass, can be used for this purpose. 7
The biosorbent (QABG) discussed in this paper is an improvement on that in reference 7, as demonstrated down by the results of elemental analysis, and then the effects on phosphate sorption. Although the same lignocellulosic biomass was used, this improvement resulted from a different synthesis mechanism. Namely, for the synthesis of biosorbent (labeled QALVS) in the previous experiment, some hazardous or carcinogenic chemical modification agents (epichlorohydrin, dimethylformamide, trimethylamine and pyridine) were used. The cationic biosorbent QABG discussed in this paper was obtained by modifying the same lignocellulosic biomass with an environmentally friendly and less harmful reagent, such as the original cationic monomer, N-(3-chloro-2-hydroxypropyl) trimethylammonium chloride (CHMAC reagent). In this case, the synthesized QABG biosorbent is structurally identical to the previously modified cationic biosorbents (QALVS), since they all contain trimethylammonium and hydroxypropyl groups as active phosphate sorption centers on the same lignocellulosic biomass. However, the significant advantages of this QABG biosorbent (such as better reaction efficiency, higher yield, better sorption capacity, etc.) are highlighted in relation to the previous QALVS biosorbent, taking into account the advantage of a simpler synthetic reaction, which justifies a new process of synthesis from the ecological and safety aspect and the application of more efficient sorbent for the removal of anionic pollutants from water.
Bottle gourd shell (BGS) is a widespread and highly available plant material that represents a valuable remnant after using a nutrient pulp. Due to the exceptional characteristics of strength and porosity, this material has become of interest for testing due to its industrial potential.8,9 Similar to the chemical composition of other lignocellulosic materials (35%–40% of cellulose, 15%–20% of hemicelluloses, 40%–45% of lignin), native BGS biomass has found potential application as a sorbent of cationic pollutants.10,11 However, the development and application of a cationic sorbent based on the modified BGS biomass is of great importance for the removal of anionic species from wastewater. The chemical modification of BGS biomass is possible due to the high content of readily available OH groups of its constituents (especially cellulose), which are subject to various reactions such as esterification, copolymerisation, condensation, grafting of surfactants etc.12,13
This article illustrates the application of BGS biomass as a sorbent for anionic species, primarily phosphate anions. Main objectives of this research are to study the kinetics and thermodynamics of the phosphate sorption process using the chemically modified biomass based on the Lagenaria vulgaris shell (LVS). The typical Lagergren and Ho models, in linear and non-linear form, were applied to define the sorption kinetic rates. The typical Weber–Morris and Chrastil diffusion models were used to determine a rate-controlling step. The effect of temperature on sorption kinetics was investigated in the range of 20 °C–50 °C. In order to clarify the sorption mechanism, the activation energy and the sticking probability were determined. These results, obtained by kinetic and thermodynamic testing, may provide important data for the industrial application of the sorbent, as well as the design of a potential plant for purifying wastewaters rich in anionic pollutants. In addition to the scientific contribution, the present study is of practical importance in the technological application of phosphate removal from aqueous solutions of different initial concentrations.
Sorption kinetic models
The kinetics of phosphate sorption were analysed using the Lagergren model (pseudo first-order equation), Ho model (pseudo second-order equation), Chrastil diffusion model and Weber–Morris intra-particle diffusion model. The pseudo first-order kinetic model, using Lagergren, 14 can be written as follows
where k1 (min−1) is the rate constant of phosphate sorption, Qe (mg P g−1) is the amount of phosphate sorbed at equilibrium and Qt (mg P g−1) is the amount of phosphate sorbed at time t (min). This pseudo first-order model can be rearranged for linearised data plotting as follows
The pseudo second-order kinetic model, using Ho and McKay’s, 15 can be written as follows
where k2 (g mg−1 min−1) is the rate constant of phosphate sorption, Qe (mg P g−1) is the amount of phosphate sorbed at equilibrium and Qt (mg P g−1) is the amount of phosphate sorbed at time t (min). This pseudo second-order model can also be linearised 16 as four different types
The diffusion model using Chrastil’s 17 can be written as follows
where kC (L g−1 min−1) is the rate constant, A0 (g L−1) is the dose of sorbent, n is the heterogeneous structural diffusion resistance constant (i.e. the order of reaction, 0 < n < 1), Qe (mg P g−1) is the amount of phosphate sorbed at equilibrium and Qt (mg P g−1) is the amount of phosphate sorbed at time t (min).
The Weber–Morris intra-particle diffusion model, developed to describe the diffusion mechanism and rate-controlling steps that affect the sorption process, 18 can be written as
where kid (mg g−1 min−0.5) is the intra-particle diffusion rate constant, Cid (mg g−1) is the thickness of the boundary layer (the intercept) and Qt (mg P g−1) is the amount of phosphate sorbed at time t (min).
Experimental
Reagents and materials
All reagents used in this study were of analytical grade (Sigma Aldrich Chemie GmbH). The solutions for modification and synthesis processes, as well as those used to rinse obtained products, were prepared with deionised water (18 MΩ). Stock solution of phosphate (1000 mg P L−1) was obtained by dissolving 4.392 g of pure KH2PO4 in 1 L of deionised water. Working standard solutions (5–140 mg P L−1) were prepared by suitable dilution of the stock solution. Mature fruit L. vulgaris from the southern part of Serbia (near Leskovac), grown at an altitude of 250 m, was used as the initial lignocellulosic material in this experiment. The naturally dried shell of L. vulgaris was manually cleaned and crushed to small pieces of 1 to 2 cm (irregular shape, diverse morphology and highly porous structure). The raw material was soaked and washed with deionised water to remove dust and soluble impurities. The washed material was dried in an oven for 24 h at 60 °C, milled in a crusher mill (Waring 8010 ES, Germany) and sieved (Oct-Digital 4527) to obtain particles of size 400–800 µm (irregular shape, diverse morphology and highly porous structure).
Characterisation methods
Elemental analysis (CHNS/O) of unmodified LVS biomass and quaternary QABG sorbent was performed using an elemental analyser (Vario EL III CHNS/O systeme GmbH). The infrared spectra of the investigated samples were recorded on a Fourier transform infrared (FTIR) spectrometer (BOMEM MB-100, Hartmann and Braun, Canada), in the range of 400–4000 cm−1, with 12 scans at a resolution of 2 cm−1. Win Bomem Easy software was used to analyse the obtained FTIR spectra.
Sorbent synthesis
The dried and milled LVS (3 g) was first treated with an alkaline solution (20 mmol of NaOH per gram of biomass) at room temperature (23 °C ± 0.2 °C) with stirring (150 rpm), to activate primary and secondary –OH groups of glucopyranose units and translate cellulose into reactive alkali–cellulose with characteristic –ONa groups. After 60 min, the suspension was transferred to a three-neck round bottomed flask (250 mL). In the flask, an original 65% aqueous (3-chloro-2-hydroxypropyl)trimethylammonium chloride (CHTAC) solution (20 mmol CHTAC per gram of biomass) was added slowly, dropwise, with an increase in temperature to about 80 °C. The reaction mixture was further treated at 85 °C ± 0.2 °C for the next 10 h with constant stirring (150 rpm). Finally, the reaction product was washed with deionised water at 40 °C and vacuum dried at 60 °C for 4 h (to a moisture content < 3%). The resulting QABG product was then used in all the sorption experiments.
Sorption experiments
Phosphate sorption experiments were conducted in a batch reactor (pH 6 ± 0.1, 60 min, 150 rpm), using 50 mL of the phosphate working solution (5–140 mg P L−1). The solution was stirred at 150 rpm in order to ensure a good mixing without vortex effect. Heating was done using a water bath to the desired temperature (20 °C, 30 °C, 40 °C, 50 °C ± 1 °C). The defined dose of QABG sorbent (2 g L−1) was added to the working solution. Aliquots of the working solutions (3 mL) were withdrawn at appropriate time intervals (up to 120 min), filtered through a 0.45 μm micro-filtration membrane (Agilent Technologies, Germany), diluted in 4% HNO3 (1 mL) and preserved with CCl4 for measurement. Filtrates were analysed for residual phosphate concentration (expressed over phosphorus), axially at 213.618 nm using an inductively coupled plasma optical emission spectrometer (Spectro ARCOS FHE12, Germany), according to the manufacturer’s instructions. The equilibrium phosphate concentration was calculated as follows
where Qe (mg P g−1) is the amount of phosphate sorption per gram QABG at equilibrium, C0 and Ce (mg P L−1) are the original and equilibrium concentrations of phosphate, respectively, V (L) is the volume of solution and m (g) is the dry mass of QABG sorbent.
Data presentation and statistical analysis
Sorption phosphate tests were replicated 3 times and only mean values are presented. The kinetic model parameters were evaluated by linear and non-linear (Levenberg–Marquardt method) regression analyses using OriginPro 8.0 software (OriginLab Corporation, USA). Significance levels were set at p = 0.05. Statistical analysis was performed using the determination coefficient (R2) and chi-square distribution (χ2). The best matching of the applied models was verified using the lowest χ2 and highest R2 values.
Results and discussion
Elemental composition analysis
The chemical compositions of raw LVS biomass and QABG sorbent were characterised using CHNS/O elemental analysis. In addition to elemental analysis, reaction efficiency (RE) analysis was performed to assess added functional groups in the sorbent during the modification process. 7 Table 1 shows the elemental compositions and the amount of nitrogen added (Nadd). It was found that the H/C ratio of 0.13 was retained in the QABG sorbent. The presence of sulphur has not been identified. Attachment of quaternary ammonium groups from the CHTAC reagent was evaluated by increasing the nitrogen content. It was determined that 19.40 mg N g−1 was incorporated onto the QABG surface as a cationic R4N+ group, indicating a theoretical exchange capacity of 1.39 m Eq g−1. Based on the RE value, it is evident that the reaction between the LVS biomass and the quaternising reagent was not particularly efficient. This fact is supported by the small value of the degree of substitution (DS), indicating that the substitution takes place on each third glucopyranose unit in the cellulose chain of LVS. However, one should keep in mind that the amount of quaternising reagent was not the limiting factor of the RE. A barrier to the reactivity of biomass with the reactant can be the variation of bulk density as a function of biomass composition.7,19
Elemental composition analysis of lignocellulosic LVS biomass and QABG sorbent.

LVS: Lagenaria vulgaris shell; QABG: quaternary ammonium–modified bottle gourd; RE: reaction efficiency; DS: degree of substitution.
Amount of nitrogen added to LVS biomass.
Reaction efficiency.
Degree of substitution.
Spectroscopic FTIR analysis
The successful modification of LVS biomass to a functional QABG sorbent by the quaternisation process is confirmed using spectroscopic FTIR analysis. As shown in Figure 1(a), the spectrum displays a number of absorption bands, indicating the complex nature of lignocellulosic LVS biomass. Bands identified at 1036, 1106 and 1161 cm−1 are typical for O–H, C–OH, C–O and CH2 glycoside groups from cellulose. 20 The main peaks at higher wavenumbers (1260, 1324, 1376, 1424, 1462, 1508, 1601 and 1738 cm−1) arise from absorption of C–OH, C–H, CH2, CH3, C–O, C=C and C=O groups from lignin and hemicelluloses, respectively. These peaks are used for the detection of structural changes during chemical modification of raw LVS biomass. 7 The decrease in band intensity and shifting of their positions typical for C=O and C–OH groups may be due to the breakdown of the lignocellulosic structure and the reaction of glucopyranose units with CHTAC. 21 Most importantly, the FTIR spectrum of QABG (Figure 1(b)) shows the emergence of a new weak band at 1490 cm−1, corresponding to the C–H bending vibration from quaternary –N+–(CH3)3 groups. 20 These observations clearly indicate the incorporation of a cationic R4N+ group onto the QABG sorbent surface.
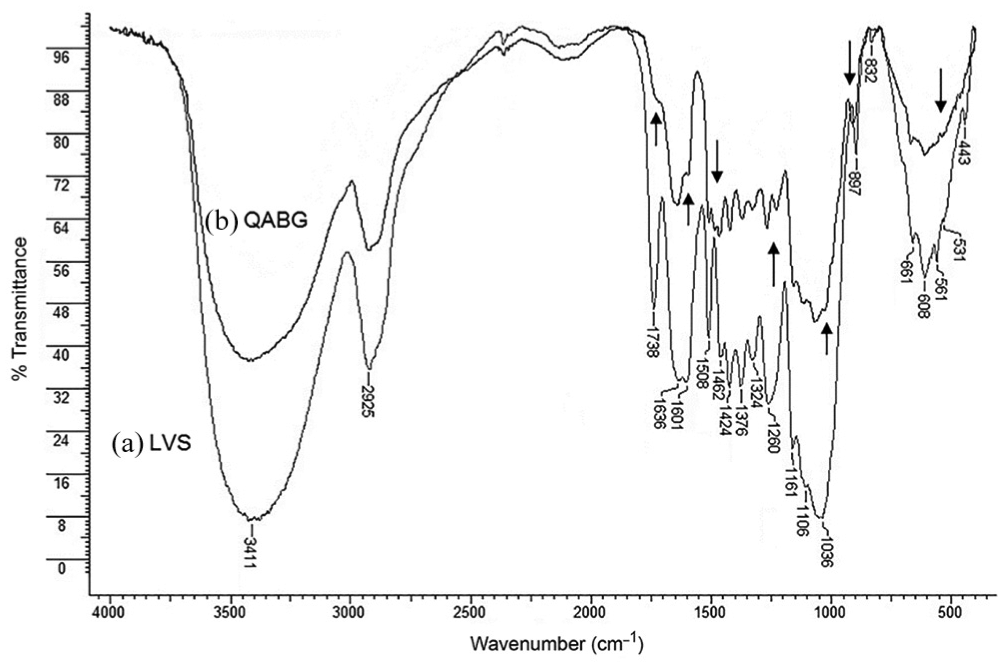
FTIR spectra of (a) raw LVS biomass and (b) QABG sorbent.
Effect of solution concentration on phosphate sorption
The effect of solution concentration (in the range 5–140 mg P L−1) on the removal efficiency of phosphate from water was investigated in a batch reactor with rapid stirring (150 rpm), under optimal conditions (temperature: 20.0 °C ± 0.2 °C, pH value: 6.0 ± 0.1) for 30 min. Compared with QABG sorbent, the raw LVS biomass has very low potential for phosphate removal from the solution (2.23% or 0.56 mg P g−1), indicating that the phosphate sorption of raw biomass is only based on surface sorption. 7 This fact confirms the importance of chemical modification to introduce some functional groups into the LVS structure and its application as an effective sorbent for removing phosphate from aqueous solution. The effect of the initial concentration of phosphate solutions on the efficiency of phosphate removal using QABG sorbent and its sorption capacity are shown in Figure 2. Maximum phosphate removal (≈ 99%) is achieved in a 5 mg P L−1 concentration solution for 30 min. The efficiency of phosphate removal decreases by increasing the initial concentration of phosphate from 5 to 140 mg P L−1. On the contrary, the sorption capacity of QABG sorbent for phosphate increases with the initial concentration of phosphate. Maximum sorption capacity of QABG (≈ 18 mg P g−1) is achieved in a 140 mg P L−1 concentration solution.
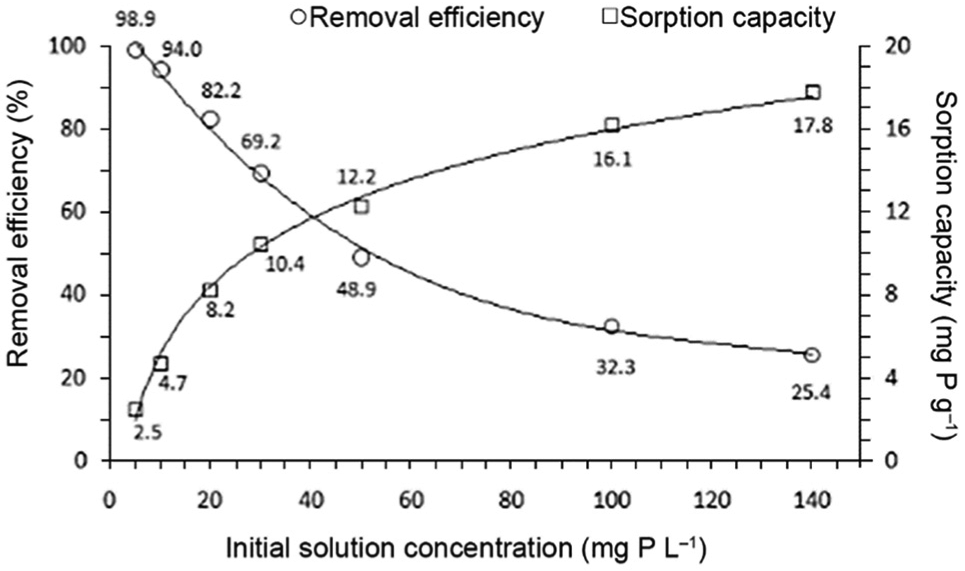
Effect of initial solution concentration on phosphate sorption by QABG for 30 min (sorbent dosage: 2 g L−1; pH: 6 ± 0.1; temperature: 20 °C ± 0.2 °C; mixing at 150 rpm).
Effect of contact time on phosphate sorption
The removal efficiency of phosphate from aqueous solutions of different initial concentrations (in the range 5–140 mg P L−1) as a function of sorbate–sorbent contact time was tested under optimum conditions (temperature: 20.0 °C ± 0.2 °C, pH value: 6.0 ± 0.1, stirring at 150 rpm). The extent of phosphate removal by QABG sorbent first increases rapidly with time (up to 10 min) and then becomes constant (after 30 min) for all the initial concentrations when the equilibrium state is established (Figure 3). It is obvious that the sorption rates for each initial concentration are different. Thus, the reaction and diffusion kinetic models were applied with the aim of defining the sorption kinetics and clarifying the possible sorption mechanism.
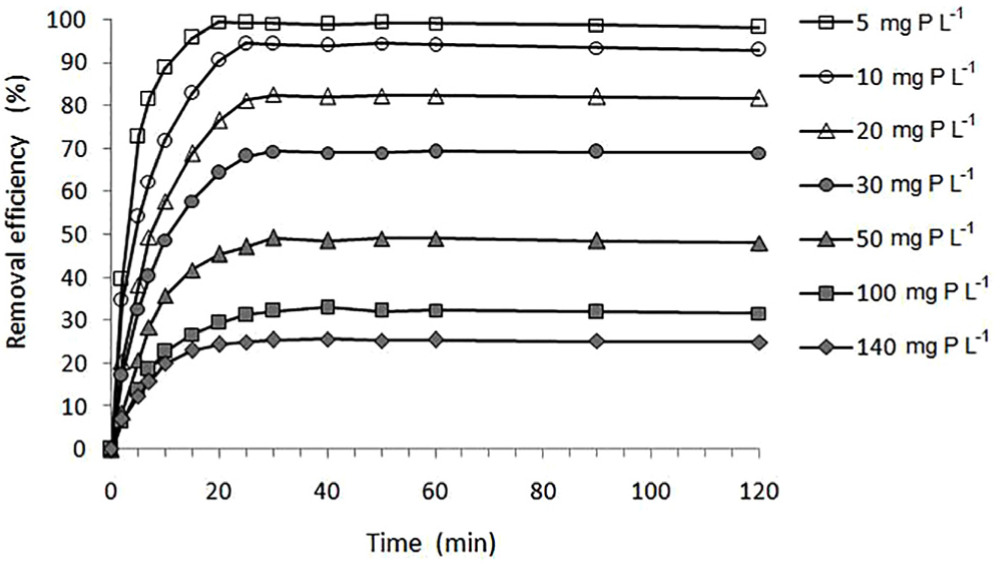
Effect of contact time on the removal efficiency of phosphate by QABG from aqueous solutions of different initial concentrations (sorbent dosage: 2 mg P L−1; pH: 6 ± 0.1; temperature: 20 °C ± 0.2 °C; mixing at 150 rpm).
Reaction kinetics
The analysis of sorption kinetics is very important considering that it clarifies the pathway of the reaction and the control mechanism of the sorption process. 22 In the case of anionic pollutants, the sorption process can be described by surface sorption analysis, that is, type of interaction (pseudo first-order and pseudo second-order) and diffusion models (Chrastil and Weber–Morris models).23–25 The selected models are typical for defining the sorption kinetics in the fluid–solid system and have been used in most studies dealing with this problem.4–6,26,27 In order to analyse the phosphate sorption on solid QABG sorbent, methods of linear and non-linear regression analyses of experimental results have been applied. The experimental data of the phosphate sorption obtained under optimum conditions (20 °C ± 0.2 °C, pH 6 ± 0.1, mixing at 150 rpm for 120 min) were analysed by the described kinetic equations (equations (2)–(9)).
Linear regression analysis of kinetics
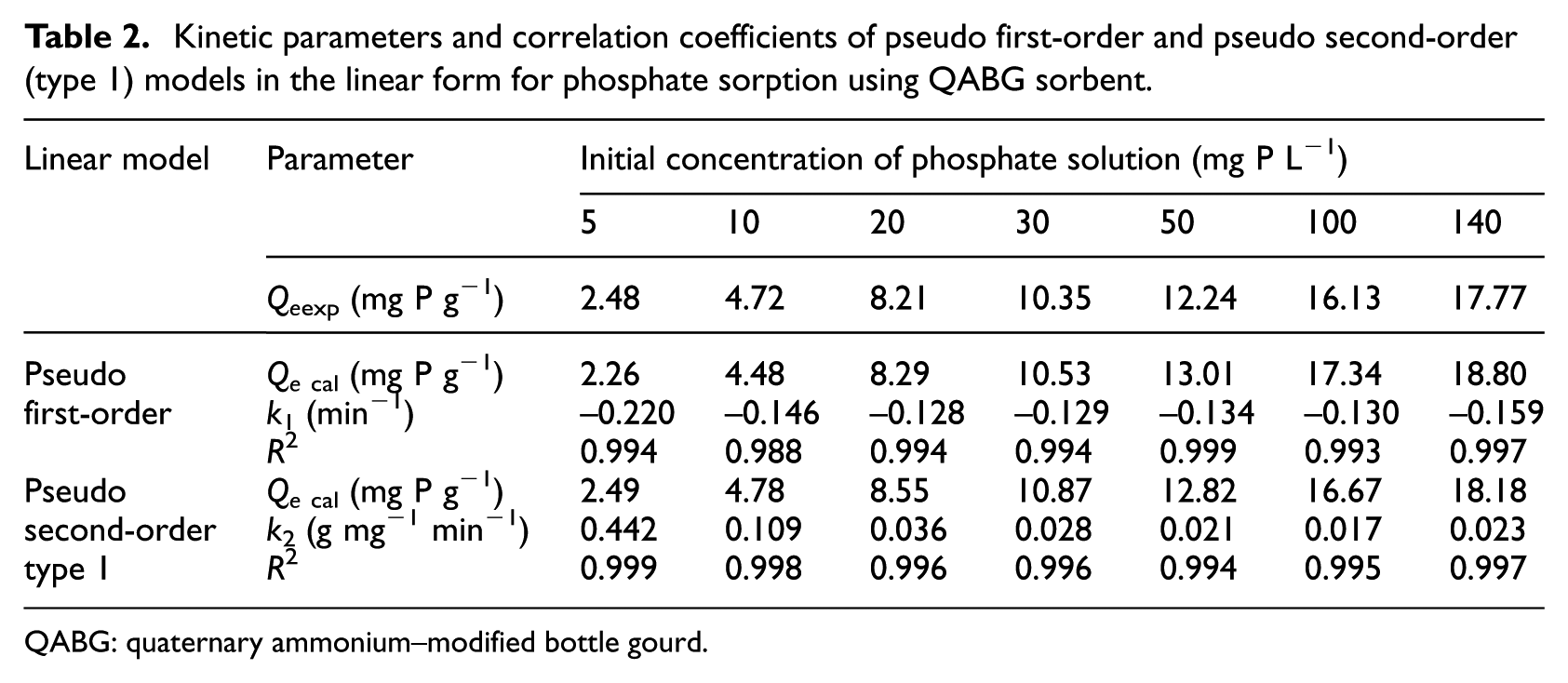
Figure 4 shows that the pseudo first-order equation in the linear form can only describe the sorption process before reaching equilibrium, from 0 to 25 min depending on the initial concentration of the phosphate solution (in the range 5–140 mg P L−1). Kinetic parameters for the pseudo first-order model and corresponding correlation coefficients are evaluated as shown in Table 2. The values of R2 for the pseudo first-order model for phosphate sorption are satisfactory (≥ 0.988). It was found that the values of k1 increase with increasing initial phosphate concentration. This fact suggests that phosphate sorption is faster in a solution of higher initial concentration, probably due to a larger driving force.
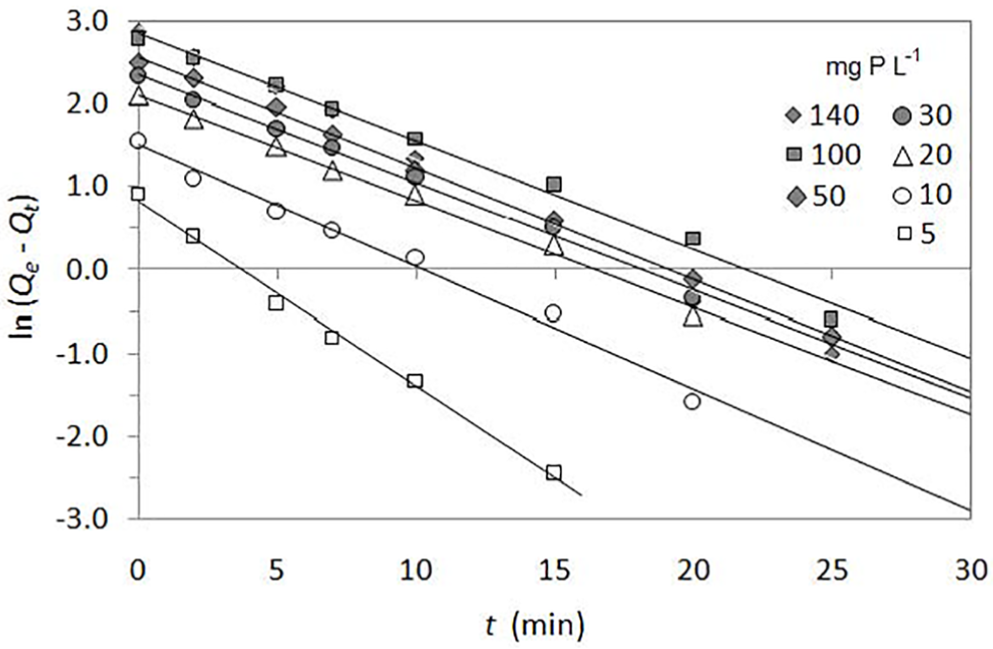
Kinetic model of pseudo first-order in the linear form for phosphate sorption from solutions of different initial concentrations (5–140 mg P L−1) using QABG sorbent (dosage: 2 mg P L−1, pH: 6 ± 0.1, temperature: 20 °C ± 0.2 °C, mixing at 150 rpm).
Kinetic parameters and correlation coefficients of pseudo first-order and pseudo second-order (type 1) models in the linear form for phosphate sorption using QABG sorbent.
QABG: quaternary ammonium–modified bottle gourd.
The linear pseudo second-order kinetic plots for the sorption of phosphate on QABG sorbent are shown in Figure 5. Based on the appearance of all four types of pseudo second-order kinetic plots, it is clear that the calculated values of the rate constant and the amount of phosphate sorbed at equilibrium, as well as the determination coefficient, will be significantly different. The best agreement with the experimental results is for the pseudo second-order model, which uses the linear equation of type 1 (Figure 5(a)). The corresponding kinetic and statistical parameters of this model are presented in Table 2. The linear equation of type 1 for the pseudo second-order kinetic model has been used in most studies to describe the kinetics of sorption processes.5,22,25
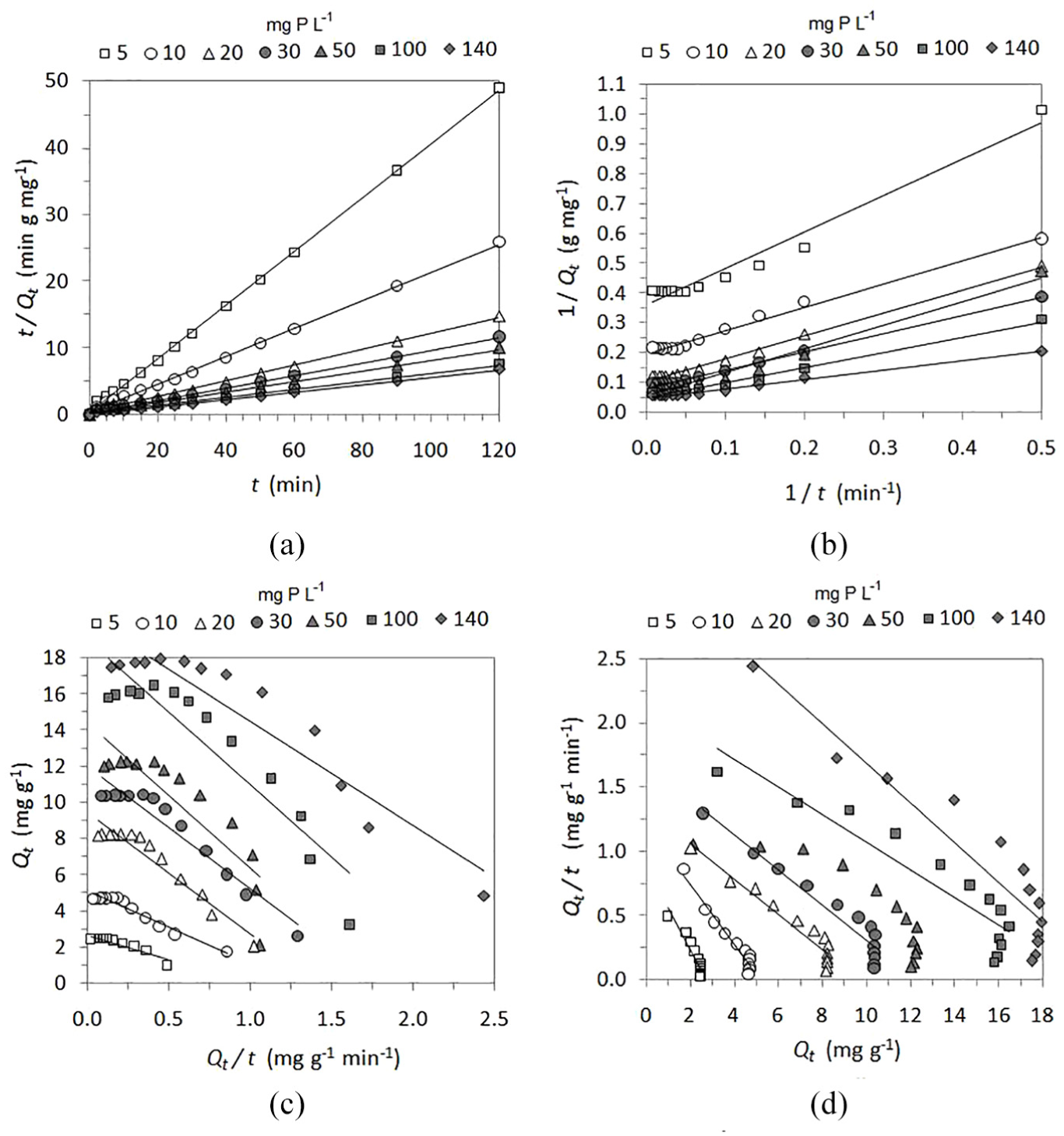
Pseudo second-order kinetic plots for four types of linear equations (a–d) for phosphate sorption from solutions of different initial concentrations (5–140 mg P L−1) using QABG sorbent (dosage: 2 mg P L−1, pH: 6 ± 0.1, temperature: 20 °C ± 0.2 °C, mixing: 150 rpm): (a) pseudo second-order type 1. (b) Pseudo second-order type 2. (c) Pseudo second-order type 3. (d) Pseudo second-order type 4.
Unlike the pseudo first-order model, the pseudo second-order model (type 1) can be applied for the entire sorption process (0–120 min) and for all the tested initial concentrations of phosphate solutions (5–140 mg P L−1), as shown in Figure 5(a). The values of R2 (Table 2) of the pseudo second-order model for phosphate sorption are satisfactory (> 0.994), indicating that the phosphate sorption on QABG can be well represented by this kinetic model. As shown in Table 2, the calculated values of the sorption capacity (Qecal) are relatively close to the experimental results (Qeexp) in both applied models and significantly better agreement of the data is obtained using the pseudo second-order equation. However, this is characteristic only for solutions of lower phosphate concentration (up to 30 mg P L−1). Therefore, non-linear regression analysis of kinetics was performed.
Non-linear regression analysis of kinetics
Non-linear regression analysis was carried out in this study using OriginPro 8.0 software (Levenberg–Marquardt method). The non-linear fitness examination of the plots, illustrated in Figure 6, confirmed that the sorption kinetics did not fit well with the pseudo second-order model. The examination indicates that only the pseudo first-order model in the non-linear form can be applied for the entire sorption process (0–120 min) and all the tested phosphate solutions. For all the initial phosphate concentrations (5–140 mg P L−1), an increase in sorption capacity during the initial period of the process (0–10 min) can be observed. The sorption capacity becomes constant after 30 min when the sorption reaches equilibrium.
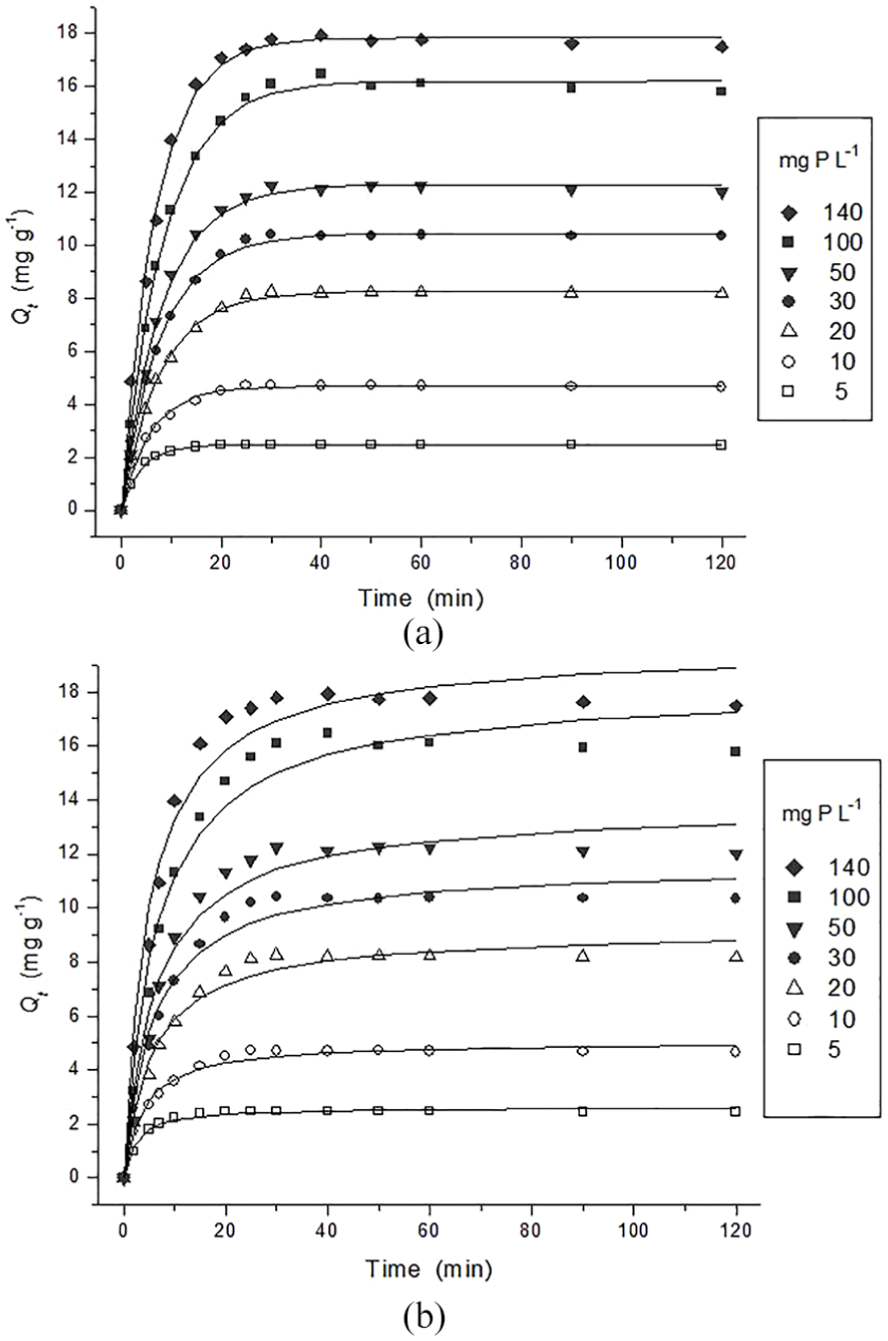
Kinetic model of (a) pseudo first-order and (b) pseudo second-order in the non-linear form (Levenberg–Marquardt method) for phosphate sorption from solutions of different initial concentrations (5–140 mg P L−1) using QABG sorbent.
Kinetic parameters for the selected models in the non-linear form are evaluated as shown in Table 3. The corresponding correlation coefficients were used for estimating the agreement of the models with the experimental results. The values of R2 of the pseudo first-order model for phosphate sorption are satisfactory (> 0.991) and are followed by those of the pseudo second-order model (< 0.985). Good fitting of the experimental data by non-linear regression is likely due to the nature of the pseudo first-order model, according to which the sorption kinetics are a function of the binding sites on the sorbent surface, regardless of the sorbate concentration. In this way, only the number of free active centres on the sorbent surface is significant, which is in accordance with the theoretical principles of the Lagergren kinetic model. In addition, the calculated values of the sorption capacity (Qecal) show a much better agreement with the experimental results than in the case of linear regression analysis (Table 2). This result indicates that the sorption of phosphate onto QABG can be well represented by the non-linear form of the pseudo first-order kinetic model. On the other hand, it is noticeable that the value of k1 is higher in the case of a lower initial phosphate concentration (Table 3). With increase in phosphate concentration the equilibrium rate constant decreases, so the total sorption rate is significantly lower and fairly balanced in solutions of higher initial concentration. This can be due to the overload of the sorbent surface with phosphates, which makes intra-particle diffusion difficult as an additional process. Therefore, it can be assumed that this is the slowest stage, which significantly affects the reduction in the total sorption rate in all cases of higher initial concentration.
Kinetic parameters and correlation coefficients of pseudo first-order and pseudo second-order models in the non-linear form for phosphate sorption using QABG sorbent.
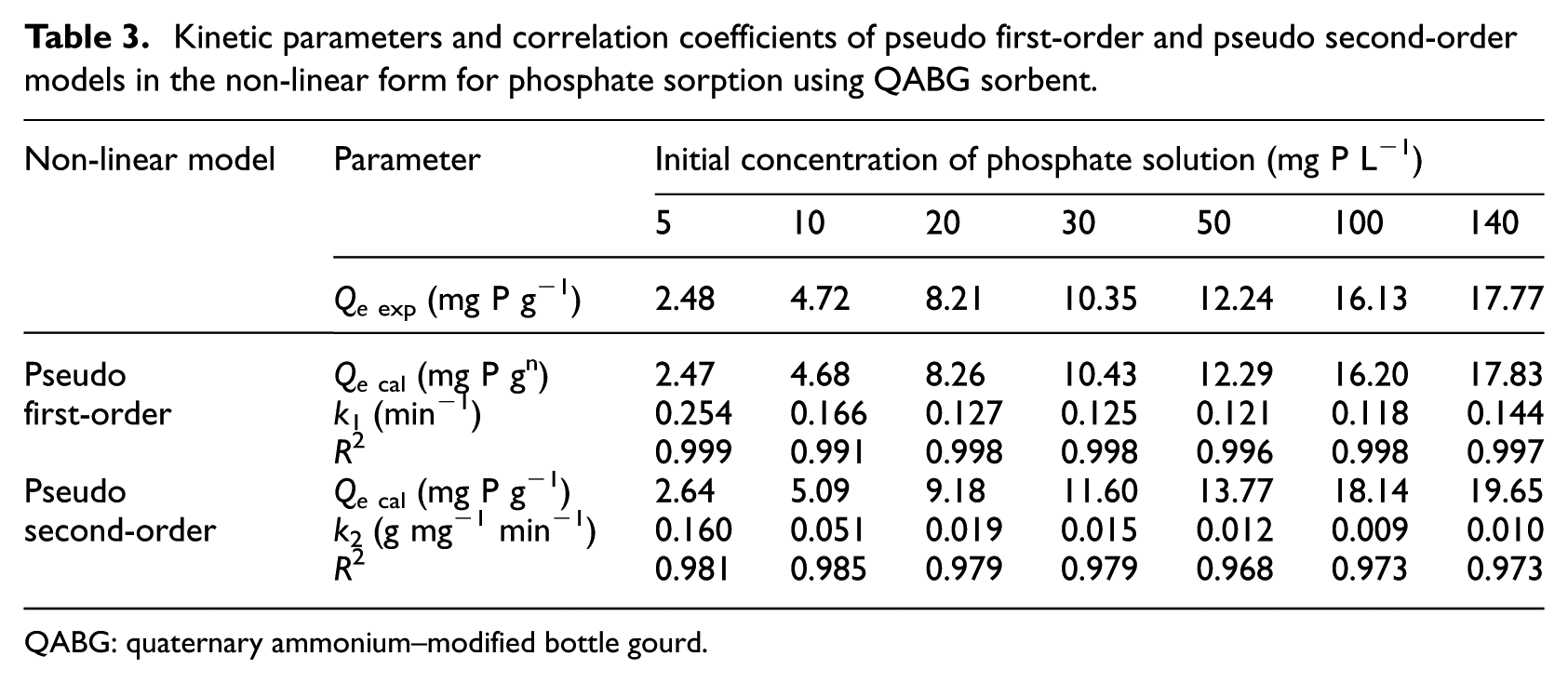
QABG: quaternary ammonium–modified bottle gourd.
The values of the sorption capacities (Qecal) calculated by the linear and non-linear regression methods for the pseudo first-order and pseudo second-order equations were compared with the experimentally determined capacities (Qeexp). The main criterion for determining the applicability of the tested kinetic equations was the chi-square (χ2) statistical parameter

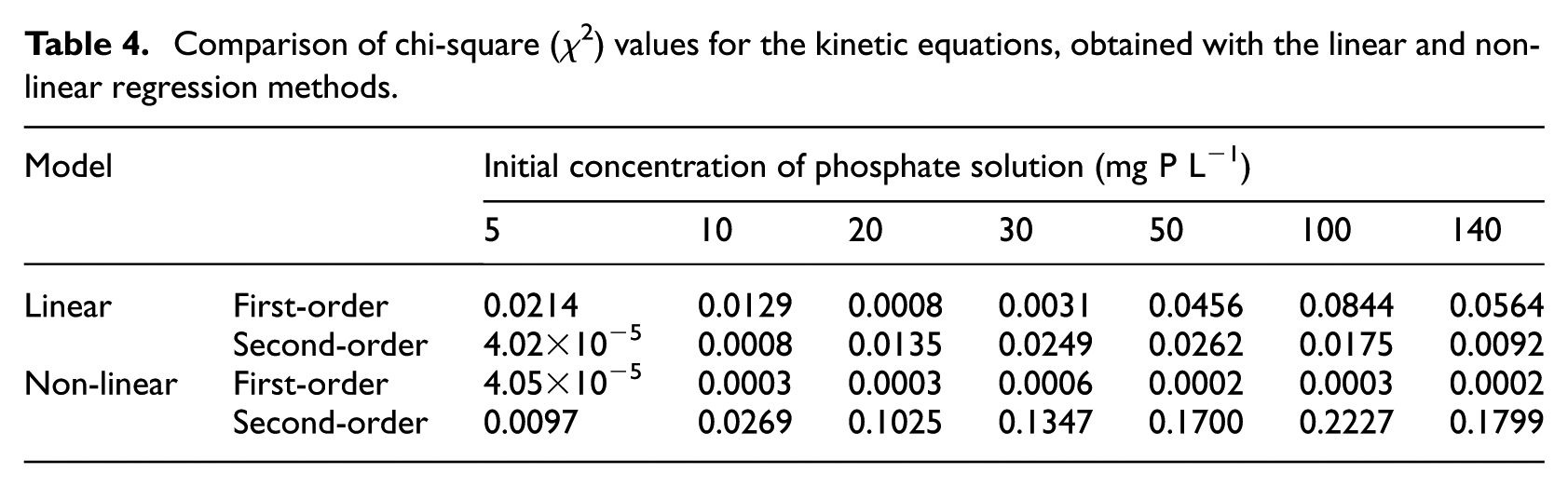
The lowest χ2 values (Table 4), as well as the highest R2 values (Tables 2–3), obtained using non-linear regression for all the initial phosphate concentrations (5–140 mg P L−1), suggest that the pseudo first-order model is best for describing the kinetic nature of phosphate sorption on QABG sorbent.
Comparison of chi-square (χ2) values for the kinetic equations, obtained with the linear and non-linear regression methods.
Diffusion kinetic models
It was found that the process of sorption by a porous sorbent can generally be represented by four stages: (1) diffusion of sorbate in the solution, (2) boundary layer diffusion or external binding, (3) intra-particle diffusion and (4) sorption–desorption of sorbate on the outer surface and within the particles of sorbent.28–30 One or more of these stages can be responsible for controlling the total rate of the sorption process. Therefore, the diffusion effect in the bulk of the solution and the rate-controlling stages that affect the sorption process were examined with the Chrastil and Weber–Morris diffusion models.
Chrastil’s diffusion model
The sorption process in a diffusion limited system can be described using the Chrastil model, 17 which is based on equation (8). This model was applied in this study to the entire sorption process (0–120 min) and all of the tested initial phosphate concentrations (5–140 mg P L−1). The parameters of the model (Qe, n and kC) were determined by non-linear regression analysis using OriginPro 8.0 software (Levenberg–Marquardt method). The values of these parameters are shown in Table 5.
Chrastil’s diffusion kinetic and statistical parameters for phosphate sorption onto QABG.
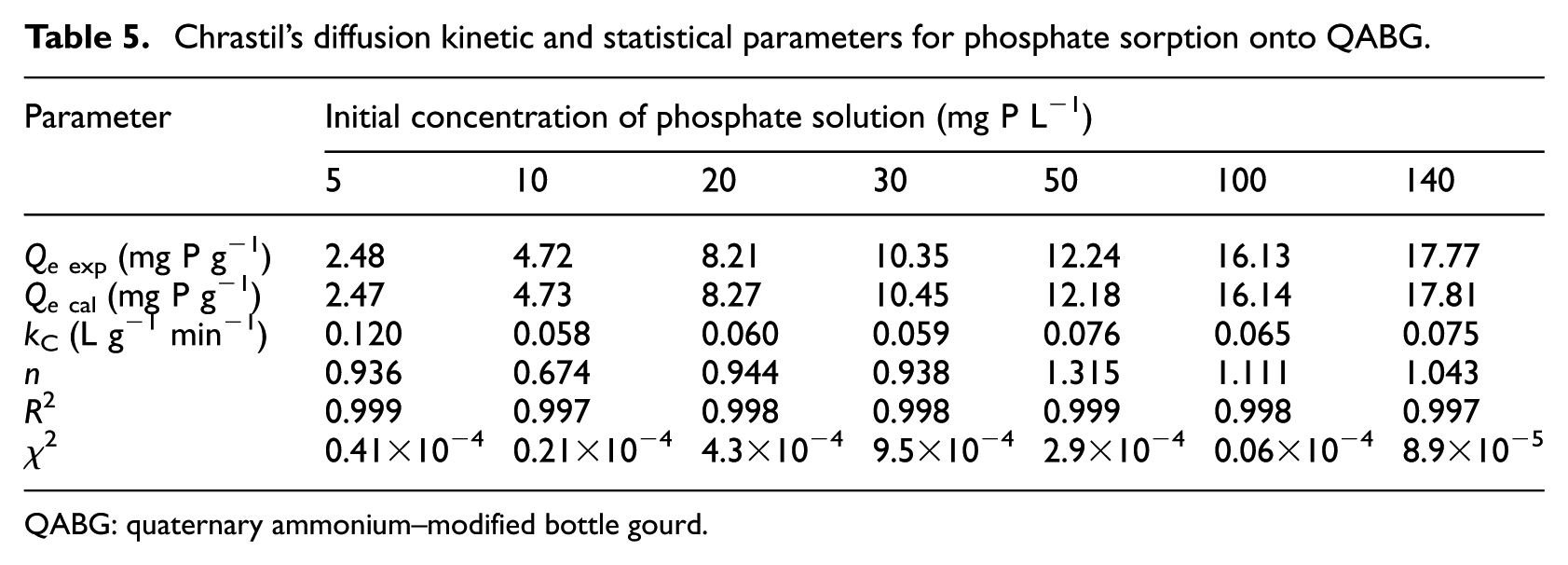
QABG: quaternary ammonium–modified bottle gourd.
In all the cases, a good agreement of Chrastil’s model with the experimental data (R2 > 0.997 and χ2 < 0.00095) was obtained. Accordingly, the diffusion characteristics of the phosphate system can be analysed by estimating the parameters kC and n. The rate constant kC is independent of the parameter A0, which is associated with the sorbent concentration, but it depends on the sorption capacity of the sorbent and the diffusion coefficient. In this study, the rate constants have relatively low values, which are slightly changed with the change in the initial phosphate concentration, as shown in Table 5. On the other hand, the parameter n characterises the overall heterogeneous diffusion structure of the sorbent. This constant is independent of the phosphate concentration, the sorbent concentration and temperature. In a system with pronounced diffusion resistance the constant is usually small (n < 0.5), which is not the case here. According to the applied diffusion model, the constants n are approximately 1 in all the cases (Table 5), indicating that diffusion resistance is small and the kinetics of phosphate sorption are of first-order. This can be understood if it is taken into account that the sorption process occurs in the system with rapid stirring (150 rpm) where the transport of phosphate (from the bulk of the solution to the sorbent surface) could be neglected.31,32
Weber–Morris model
The rapid stirring systems are most commonly characterised by intra-particle diffusion as a rate-controlling step. 28 According to the Weber–Morris model, there should be a linear dependence between Qt and t0.5, which confirms that intra-particle diffusion is involved in the sorption process. In the case where the plot of Qt versus t0.5 starts from the origin, then intra-particle diffusion is the only rate-limiting step. These plots for phosphate sorption from aqueous solutions of various initial concentrations are shown in Figure 7.
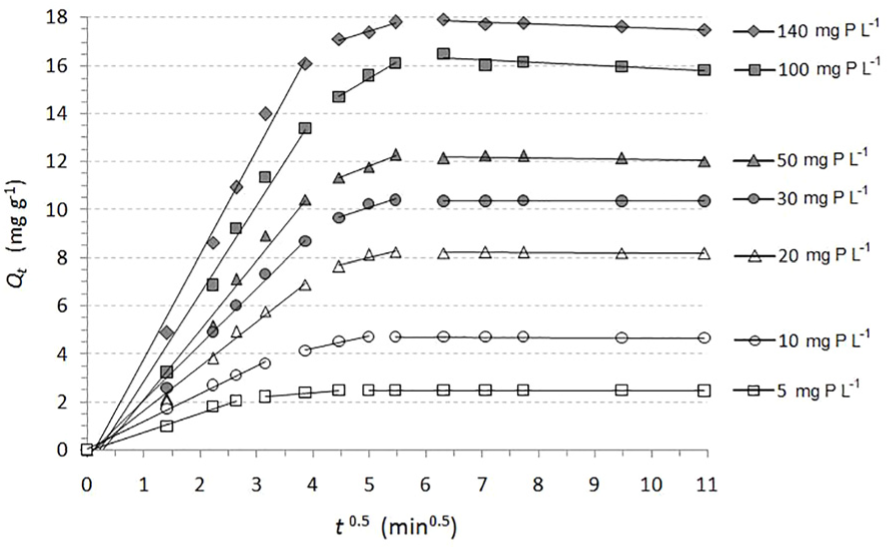
Intra-particle diffusion sorption kinetics of phosphate onto QABG sorbent (dosage: 2 g L−1, pH: 6 ± 0.1, temperature: 20 °C ± 0.2 °C, mixing at 150 rpm, initial concentrations: 5–140 mg P L−1).
The applied diffusion model suggests a characteristic multilinearity for all the initial phosphate concentrations (5–140 mg P L−1). It is obvious that all the plots show three separate stages. For the initial period of phosphate sorption (0–15 min), satisfactory linearity was established (Table 6), but the plots do not pass through the origin (the intercept Cid≠ 0). These deviations of linear plots near the origin, which relate to changes in the thickness of the boundary layer, suggest that another sorption mechanism is involved in addition to intra-particle diffusion. It is evident that the effect of the boundary layer is characterised by the first stage. In accordance with the relatively high values of R2, a rate-limiting step in the initial period of phosphate sorption is the external mass transfer. As shown in Table 6, the rate constants increase with increasing initial phosphate concentration, probably due to the larger driving force of the process. The second linear parts of the plots (15–30 min) are characteristic of intra-particle diffusion (Figure 7). The values of kid, which were calculated from the slopes of the plots in the second stage, suggest a slower process compared with the initial period, probably due to the characteristic porosity of the sorbent. 11 The third part of the plots start after 30 min and represent the final stage of equilibrium, when the intra-particle diffusion and the surface sorption of phosphate begin to slow down. According to the applied model, it can be assumed that the sorption mechanism of phosphate on QABG is complex. In addition, the sorption process is not solely rate-limited by the intra-particle diffusion, but also by ion exchange as the dominant mechanism. Similar observations have also been described in other sorption processes, where the lignocellulosic materials were used as sorbents of anionic pollutants, primarily phosphate.5,6,22,27
Rate constants and intercept values of intra-particle diffusion model for the sorption of phosphate onto QABG sorbent.
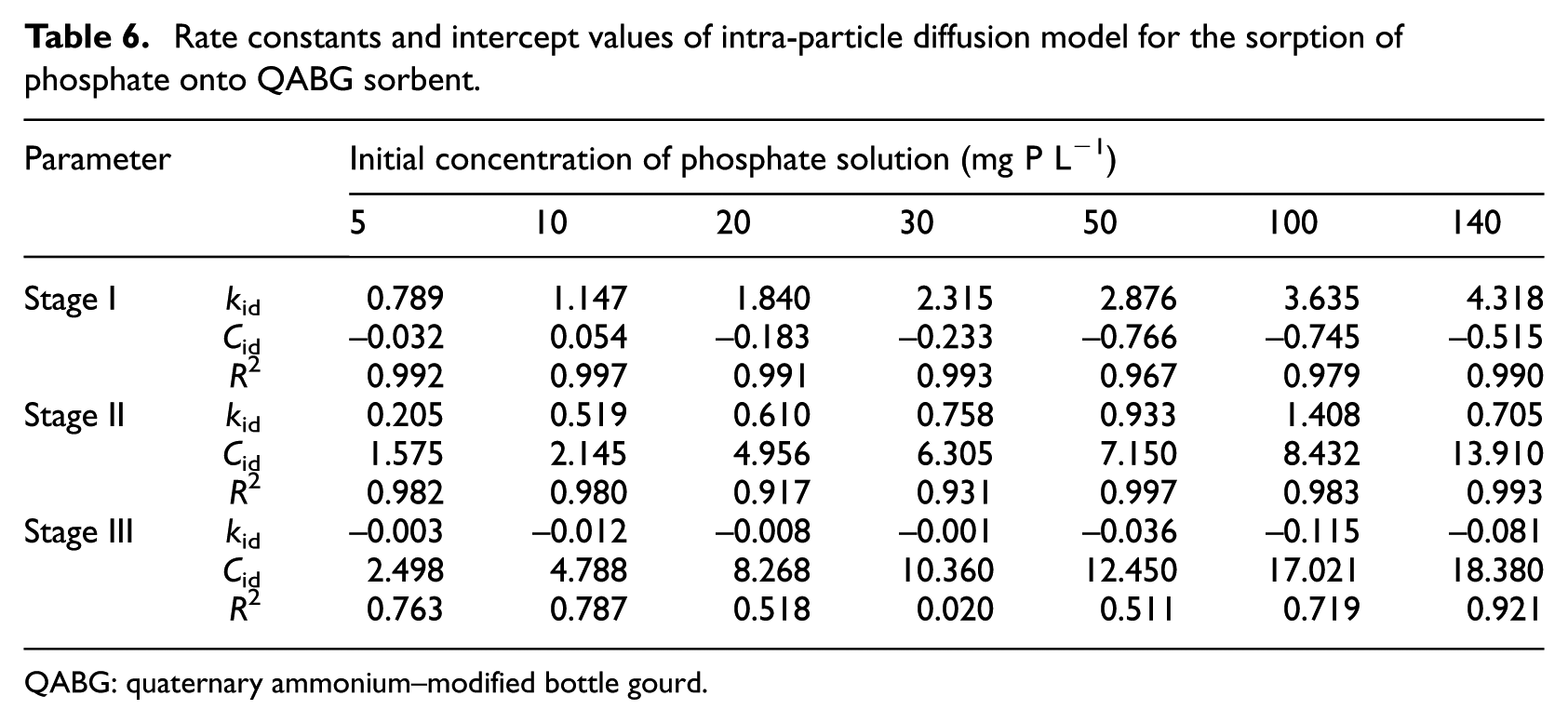
QABG: quaternary ammonium–modified bottle gourd.
Sorption thermodynamics
The effect of temperature on phosphate sorption from aqueous solution by QABG was studied by varying the temperature between 20 °C and 50 °C. This study showed that sorption of phosphate decreased with increase in temperature, indicating that lower temperatures favour the removal of phosphate by QABG. Accordingly, phosphate desorption from the solid surface occurs due to the increase in the temperature of the solution. 7 These observations suggest weak sorption interaction between sorbent surface and phosphate, which supports physisorption. A similar effect was reported for phosphate sorption by some other modified biomasses.1,6,33
In order to determine which sorption process actually occurs, different thermodynamic parameters (primarily energy and entropy factors) should be considered. The typical thermodynamic parameters of enthalpy change (ΔHo) and Gibbs free energy change (ΔGo), as well as the entropy change (ΔSo), for the phosphate sorption are calculated using the following equations


where KD is the coefficient of distribution (KD = Csorb/Ce), Csorb is the concentration of sorbed phosphate in equilibrium (mg P L−1), Ce is the equilibrium phosphate concentration in the solution (mg P L−1), T is the absolute temperature (K) and R is the universal gas constant (8.314 J mol−1 K−1). The corresponding functional dependence of ln KD on 1/T was found to be linear (R2 = 0.999). The values of ΔHo and ΔSo, respectively, were determined from the slope and intercept of the plot. These thermodynamic parameters are shown in Table 7.
Thermodynamic parameters, activation energy and the sticking probability for phosphate sorption onto QABG sorbent.
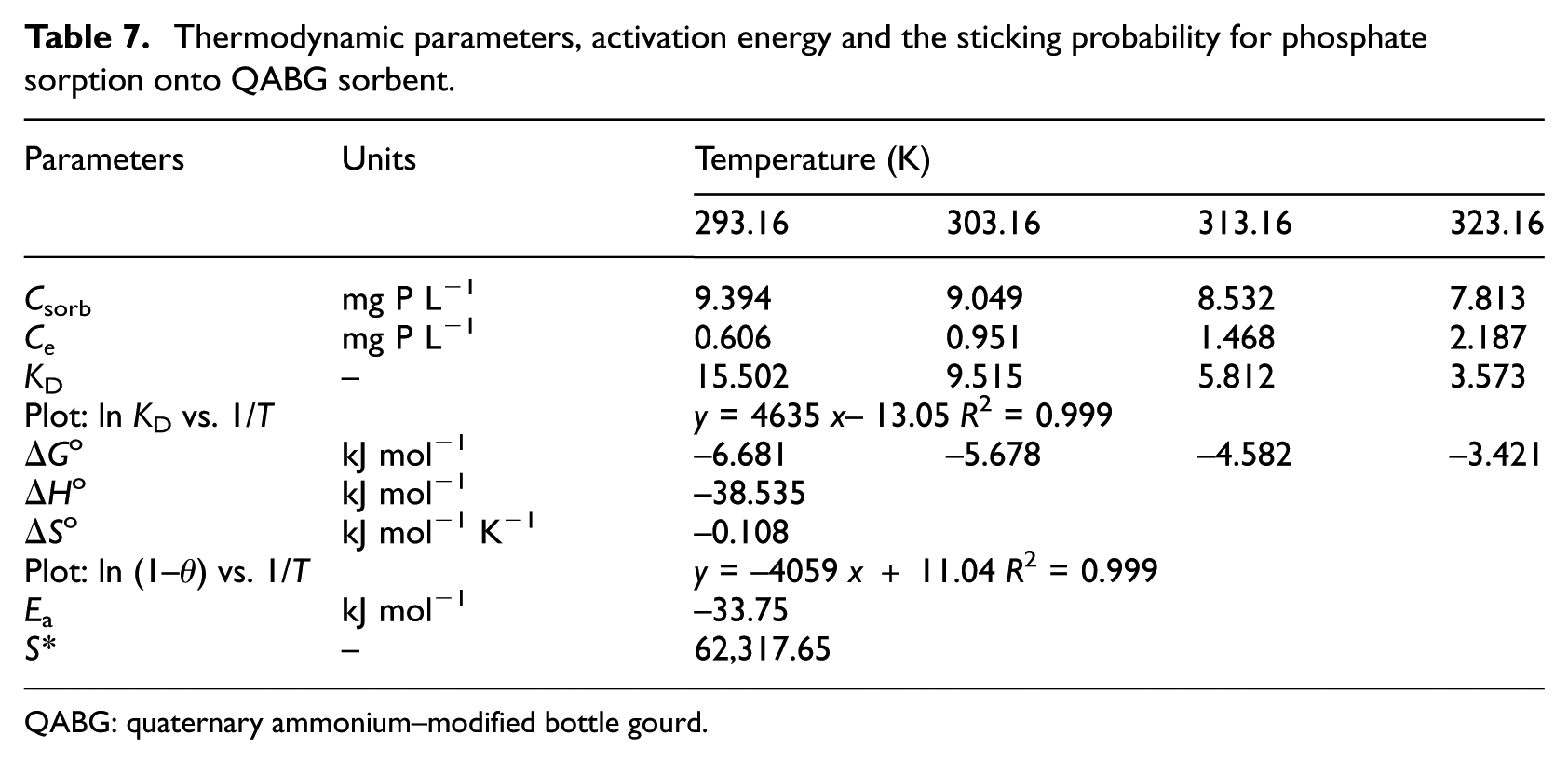
QABG: quaternary ammonium–modified bottle gourd.
The negative values of ΔGo at all temperatures confirm the feasibility of the process, 33 as well as the spontaneous nature of phosphate sorption by QABG sorbent. It should be noted that values of ΔGo up to −20 kJ mol−1 are consistent with electrostatic interaction between sorption sites and the phosphate, as is the case here. The values of ΔSo and ΔHo for the phosphate sorption process were calculated to be −0.11 kJ mol−1 K−1 and −38.54 kJ mol−1, respectively. The negative value of ΔSo shows the stability of the process with no structural change at the solid/liquid interface and suggests that the system exhibits random behaviour. 34 The negative value of ΔHo suggests that the sorption reaction is exothermic. It is established that the value of ΔHo is in the range −4 to −20 kJ mol−1 for multilayer physisorption, in the range −40 to −80 kJ mol−1 for ion exchange (electrostatic interactions between the solid charged surface and the ions from the solution) and in the range −80 to −800 kJ mol−1 (the same order of magnitude as for the average chemical reaction) for unilayer chemisorption. 7 In this case, based on the calculated ΔHo value, the ion exchange mechanism for the phosphate sorption is implied as the main one.
The assumption that ion exchange is the predominant mechanism can be confirmed by estimating the values of activation energy (Ea) and sticking probability (S*). Based on the experimental data, the surface coverage (θ) was calculated from the relation θ = 1 –C/C0, where C0 and C are the initial and residual concentrations of phosphate in the aqueous solution (mg P L−1), respectively. The corresponding plot of ln(1–θ) versus 1/T gave linear functional dependence (R2 = 0.999) with an intercept of ln S* and slope of Ea/R. The activation energy, estimated from the plot with good fitting, was found to be −33.75 kJ mol−1, as shown in Table 7. This negative value of Ea suggests that a lower temperature favours phosphate removal by QABG and the sorption process is exothermic. Further, a relatively low value of Ea indicates that phosphate sorption is a diffusion-controlled process.
The sticking probability indicates the measure of the potential of a sorbate to remain on the sorbent indefinitely. The parameter S* is a function of the sorbate/sorbent system being considered. If S* > 1, then there is no strong sorption (sorbate unsticking to sorbent), when S* = 1 a linear relation of sticking exists between sorbate and sorbent (both chemisorption and physisorption mechanisms are included) and S* = 0 is characteristic of indefinite sticking of sorbate to sorbent (mechanism of chemisorption is dominant). For 0 < S* < 1, there is favourable sticking of sorbate to sorbent (mechanism of physisorption is dominant). The sticking probability value is calculated using a modified Arrhenius-type equation related to surface coverage

It is known that the parameter S* depends on the temperature of the system. Therefore, the sticking probability value was estimated over the entire temperature range (from 20 °C to 50 °C), based on the surface coverage at different temperatures (Table 7). This result suggests that the probability of the phosphate to stick to the sorbent surface is very low (as S* >> 1), confirming that the sorption process is ion exchange.
Conclusion
This research confirmed that a cationic sorbent, prepared from BGS as a lignocellulosic agricultural by-product, can be used successfully to remove phosphate anions from aqueous solution. The sorption of phosphate on QABG was evaluated by surface sorption analysis and diffusion models using linear and non-linear regression analyses of experimental results. The kinetics of phosphate sorption were extraordinarily fast, reaching ≈99% of the total sorption at equilibrium in 30 min. The best model that described the kinetic data was the non-linear pseudo first-order kinetic model. According to the applied Weber–Morris model, which shows a characteristic multilinearity for all the initial phosphate concentrations, it has been established that the sorption mechanism is complex and that the sorption process is not solely rate-controlled by the intra-particle diffusion step. Sorption thermodynamics indicated that the sorption process was exothermic and spontaneous. The anion exchange mechanism is the dominant sorption process. The activation energy and the sticking probability further supports lower solution temperatures and the proposed mechanism. It may be concluded that QABG sorbent has the potential to be used as an efficient and cost-effective material in ecological treatment and restoration systems for the removal and recovery of anionic pollutants from wastewater, as an alternative to more costly sorbents.
Footnotes
Acknowledgements
These researches were supported by the Serbian Ministry of Education, Science and Technological Development through Project TR-34012.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.










