Abstract
Non-isothermal pyrolysis and gasification of
Introduction
Recently, large-scale
The process of waste-to-energy via gasification has been proposed as one of the crucial biomass utilization alternatives because gasification has high tolerance against the heterogeneous structures of biomass. 3 Moreover, it is suitable for producing gases that can be used to provide electrical power and heat, 4 and is compatible with the current infrastructures of the chemical industry. 5 Besides, it provides for limited formation of dioxins due to the oxygen-free environment. 6 Biomass gasification is a complex thermochemical process, which mainly consists of two stages: (1) pyrolysis or release of volatiles and (2) conversion of the pyrolytic char. 7 So far, considerable studies on the thermal–chemical processing of EP have been focused on pyrolysis under an inert atmosphere (i.e. N2 and Ar);8–11 note, however, that there is currently no investigation concerning the gasification of EP using other atmospheres like CO2 and steam. Recently, CO2 gasification has been regarded as a promising technology, because CO2 applied as a reaction medium is less corrosive than steam, 12 and it can be incorporated in a valorization cycle for the production of fuels, rather than simply being captured and stored. 13 Hence, CO2 gasification provides a long-term solution to mitigate directly the increasing concentration of greenhouse gases in the atmosphere. 14
In order to compensate for the shortcomings of previous studies, in this study, the pyrolysis and gasification of EP under a pure CO2 atmosphere was investigated, particularly by thermogravimetry–Fourier transform infrared spectroscopy (TG–FTIR). Weight loss data and evolution profiles of the major volatile species were obtained. Furthermore, based on the thermogravimetry (TG) data, the pyrolysis and gasification kinetic parameters were obtained by the Coats–Redfern method.
Experimental
Materials
EP was collected from the Yellow Sea coast in Qingdao, Shandong Province China. Prior to use, EP was first washed with tap water to remove solid particles and then dried at 65°C for 36 h. The dried EP was ground and sieved into particle sizes of 120–180 mesh.
TG–FTIR analysis
Pyrolysis and gasification of EP was performed using a TG–FTIR instrument that consists of a simultaneous thermal analyzer (STA 2500 Regulus; NETZSCH, Germany) and a Fourier transform infrared spectrometer (TENSOR II; Bruker, Germany). A sample mass of 13–15 mg was used in this study. The sample was heated from 35 to 1000°C at a pure CO2 flow rate of 70 mL min−1 and a heating rate of 20°C min−1. The volatiles released were detected online by FTIR spectroscopy, in which IR spectra were recorded at 4000–5000 cm−1 with a resolution of 4 cm–1. The transfer pipe and the gas cell in the FTIR were both heated at a constant temperature of 200°C. The experimental results of TG and FTIR were recorded automatically on a computer.
Kinetic methods
A kinetic study of EP is essential to achieve the production of fuel gas, chemicals, and energy. The information is also important for the design of large-scale gasification reactors. The Coats–Redfern method was used in this study to determine the kinetic parameters of pyrolysis and gasification of EP.15,16
The rate of conversion, dα/d

where α is the degree of conversion and

where
The reaction rate constant,

where
For a constant heating rate β during gasification, β = d
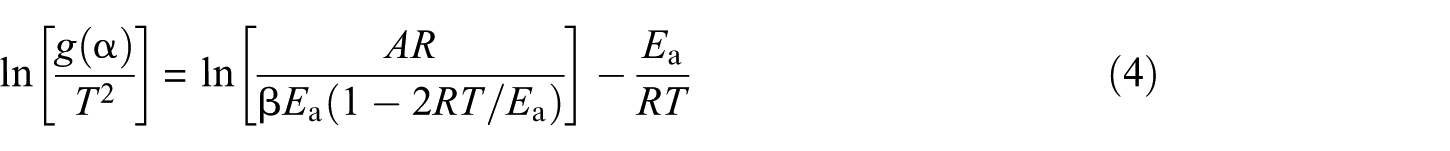
As the term 2

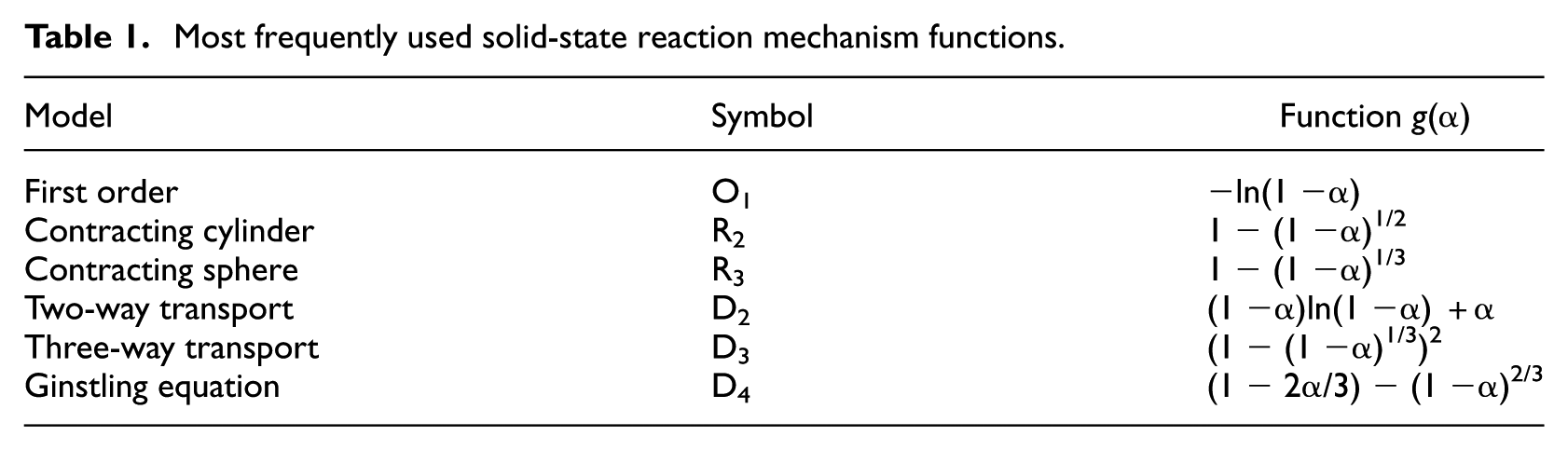
Various reaction models define
Most frequently used solid-state reaction mechanism functions.
Results and discussion
Thermal behavior
TG and derivative thermogravimetry (DTG) curves for EP are shown in Figure 1. The characteristic parameters obtained from the TG and DTG curves such as weight loss, the corresponding temperature ranges of the weight loss, the maximum weight loss rates (
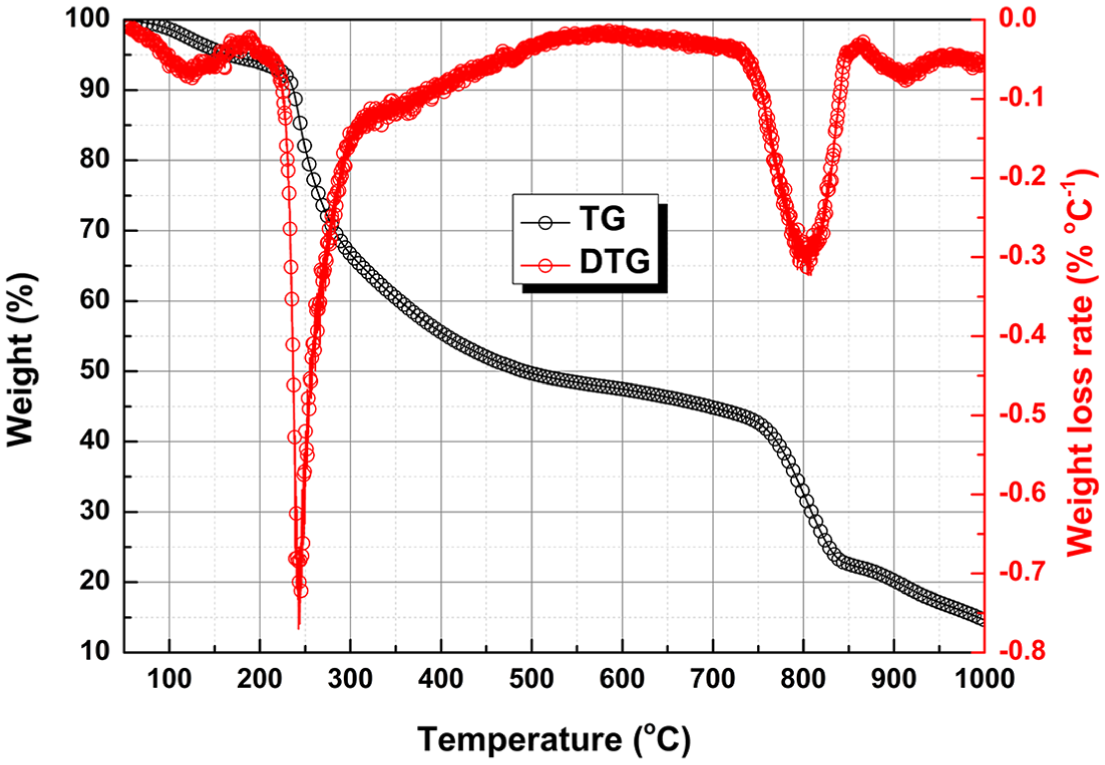
TG and DTG curves for EP at 20°C min−1.
Characteristic parameters obtained from the TG and DTG data for EP at 20°C min–1.
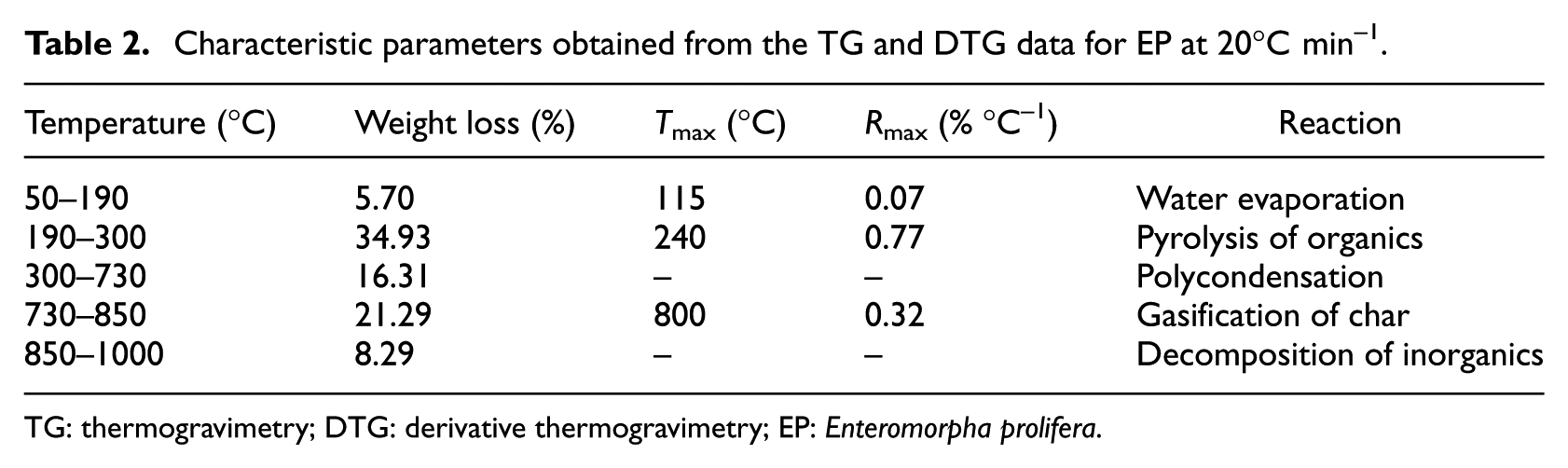
TG: thermogravimetry; DTG: derivative thermogravimetry; EP:
FTIR analysis
The volatiles evolved from the pyrolysis and gasification of EP were analyzed by FTIR in real time. As shown in Figure 2, the Gram–Schmidt curve shows the variation of the yields of the volatile compounds with temperature. It has two peaks centered at 240 and 800°C, respectively, which matched with the DTG peaks very well. Note that the second peak is much stronger than the first one, indicating that more gaseous products were generated during the gasification stage. The three-dimensional (3D) spectrogram (absorbance–wavenumber–temperature) of the volatiles is shown in the inset of Figure 2. When the temperature is fixed, absorbance information at different wave numbers can be obtained to study the volatile components released at that moment. Figure 3 shows the FTIR spectra at the typical temperatures for pyrolysis and gasification of EP. As a result of the cracking of the organics in EP, the complex gaseous products composed of a variety of molecules were evolved at 240°C, i.e.,

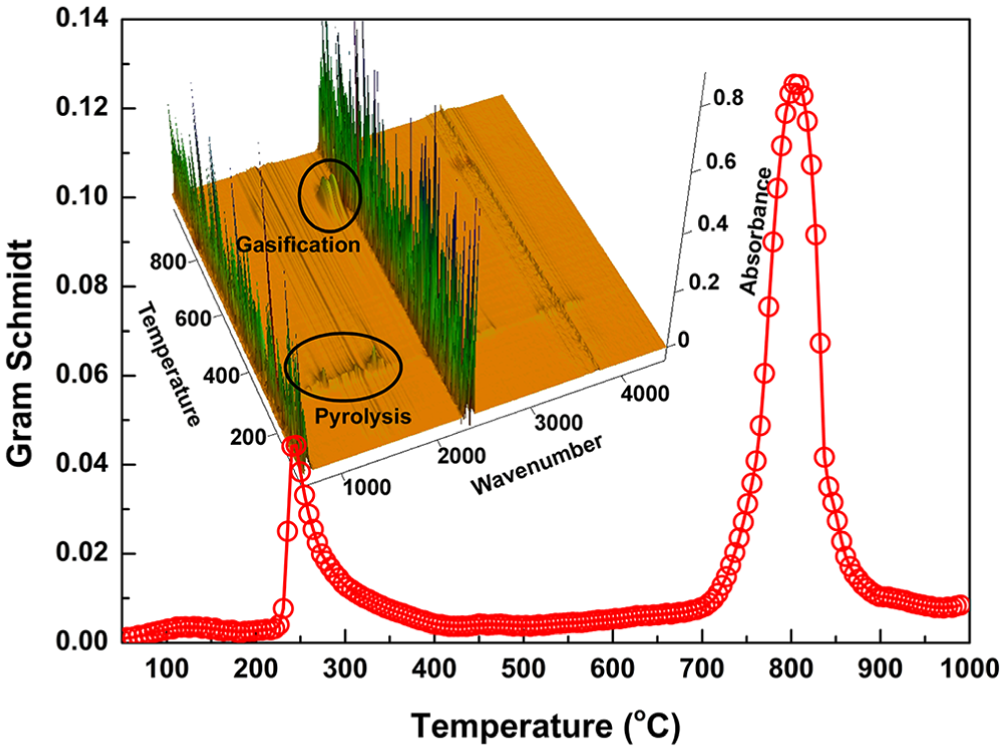
Gram–Schmidt curve for EP at a heating rate of 20°C min−1; the inset shows the 3D infrared spectrum of pyrolysis products.
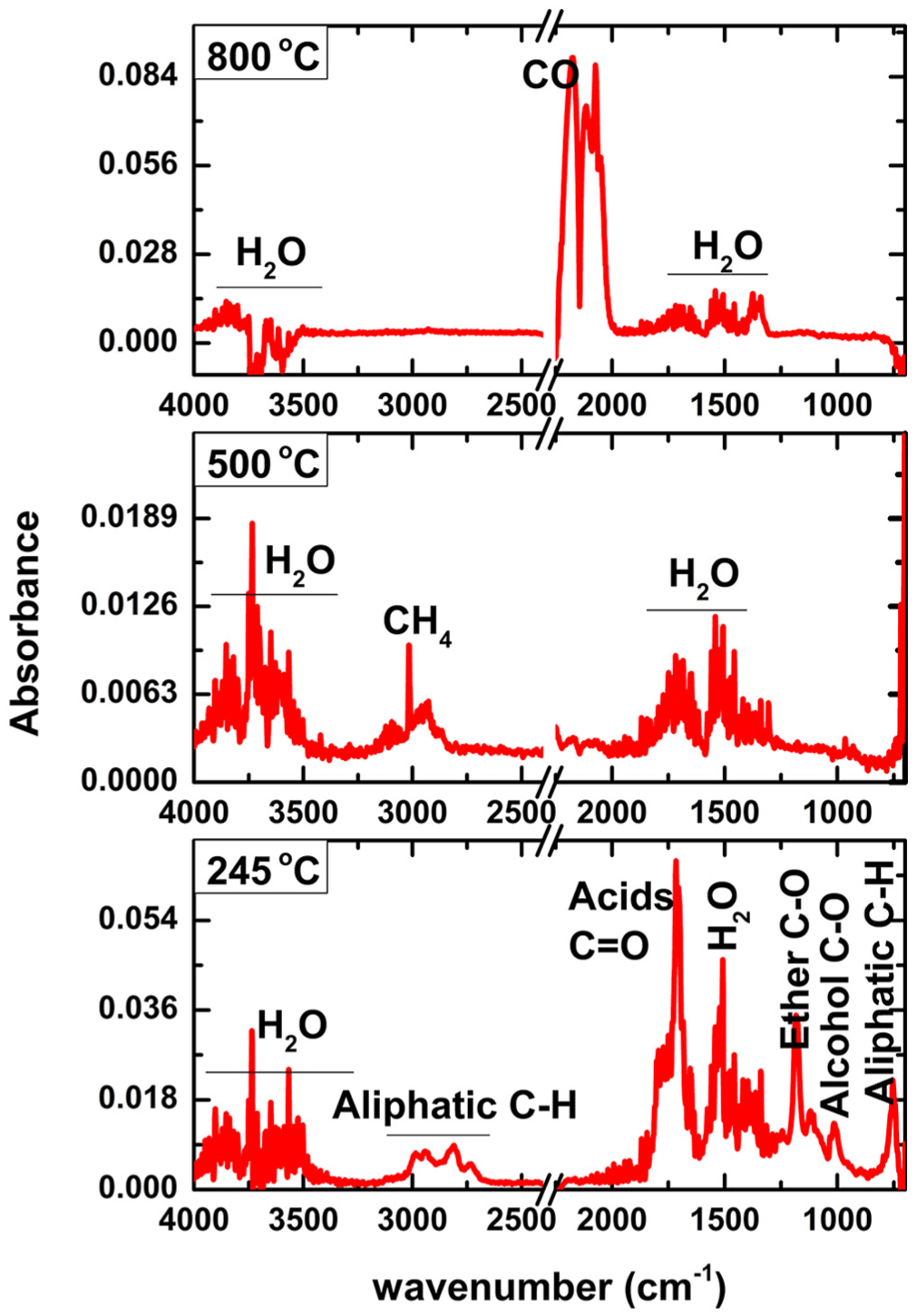
FTIR spectra for pyrolysis products corresponding to different temperatures.
The evolution histories of the main volatile products as a function of temperature are presented in Figure 4. The release of CH4 and aliphatic C–H exhibits two peaks, those in the low-temperature region (200–300°C) were mainly generated from the rupture of oxygen-containing aliphatic chains, such as methoxy (–O–CH3) and ethoxy (–O–CH2–CH3) groups, while the peaks at higher temperatures (>450°C) were caused by the rupture of the side chains of aromatic rings. Both the curves of CH4 and aliphatic C–H had long tails beyond 600°C, indicating the carbonization of EP to a greater extent. Carboxylic acids, ethers, and alcohols were the dominating condensable products; they were mainly released within a narrow temperature range (230–300°C), suggesting that the violent pyrolysis reactions were finished in a short time. CO was the main non-condensable product, and it could be detected from 700°C to as high as 900°C, indicating the temperature range of the occurrence of the Boudouard reaction. Based on the evolution histories of the main volatile products, the thermal conversion process of EP under a CO2 atmosphere can be illustrated by the schematic diagram in Figure 5.
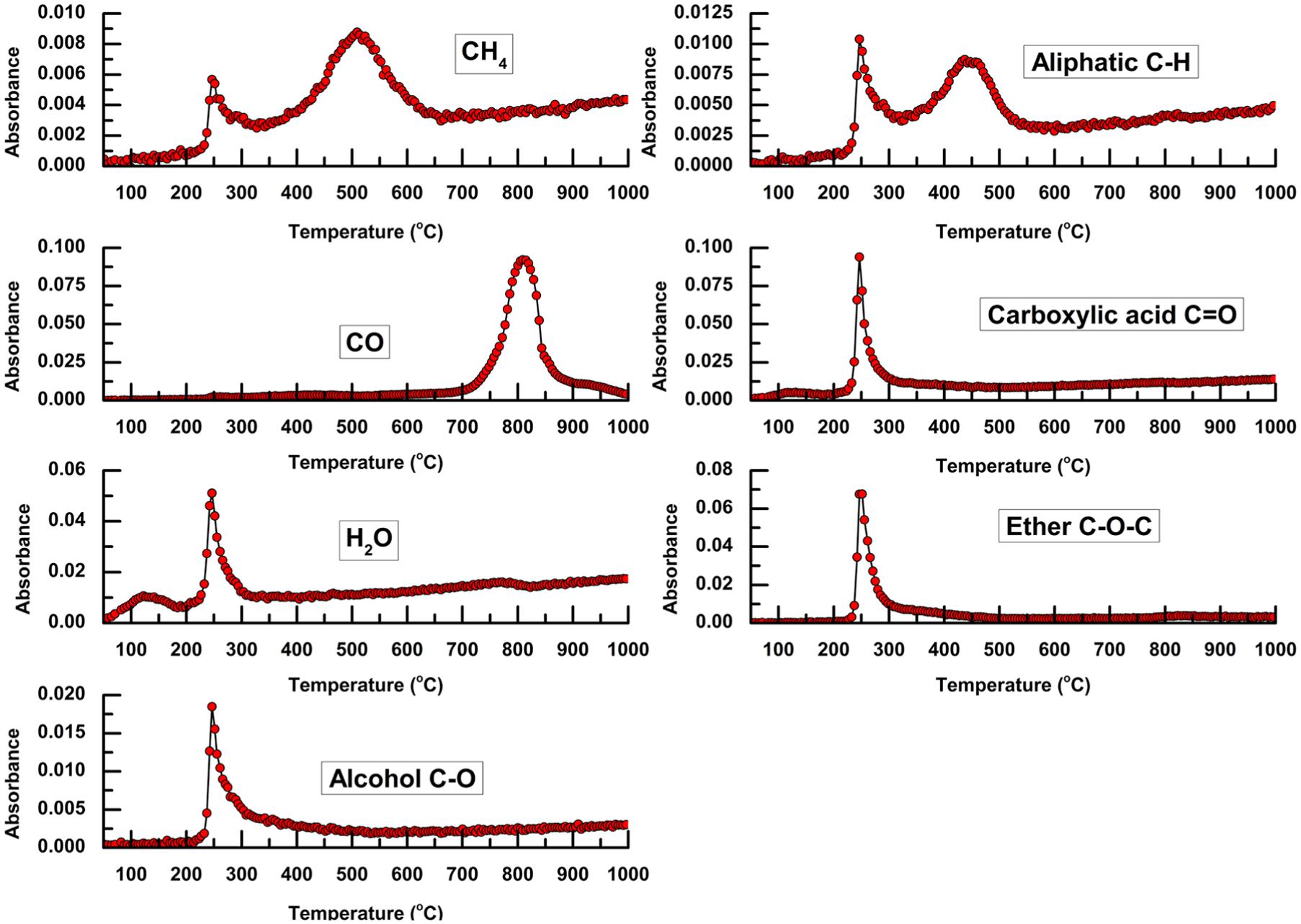
Evolution histories of main volatile products as a function of temperature.
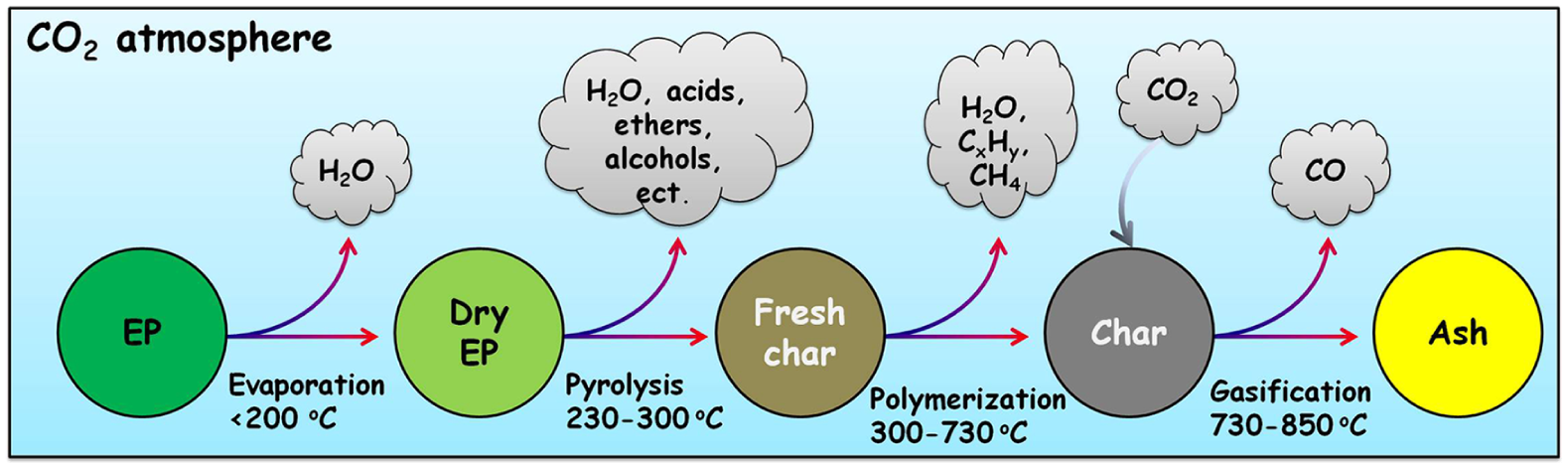
Schematic diagram of the thermal conversion process of EP under a CO2 atmosphere.
Kinetic study
The kinetic parameters were determined assuming single separate reactions for a particular stage of thermal conversion. By considering the DTG curve for EP (Figure 1), two temperature ranges for pyrolysis and gasification reactions were selected, equation (5) was applied separately to each of them, and a series of straight lines can be obtained by plotting ln(
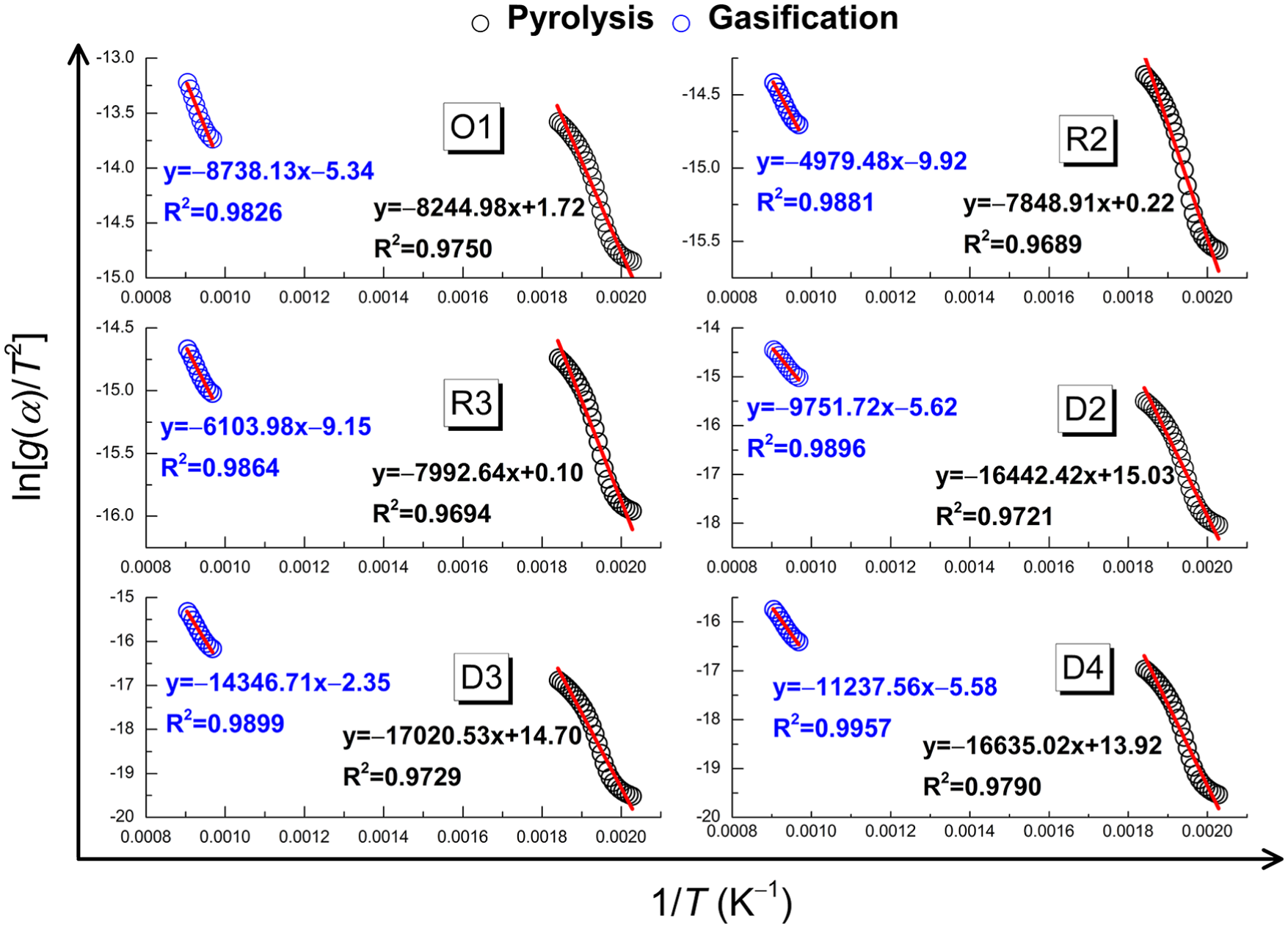
Plots of ln(
The D4 model, which is also known as the Ginstling equation, assumes that the chemical reactions occur on the entire surface of the spherical solid particles, leading to the formation of a layer with a certain thickness increasing with the reaction time. The reaction rate is controlled by the diffusive resistance of the layer to the gaseous reactants or products. Based on the above assumption, kinetic models of the pyrolysis and gasification stages of EP are illustrated in Figure 7.
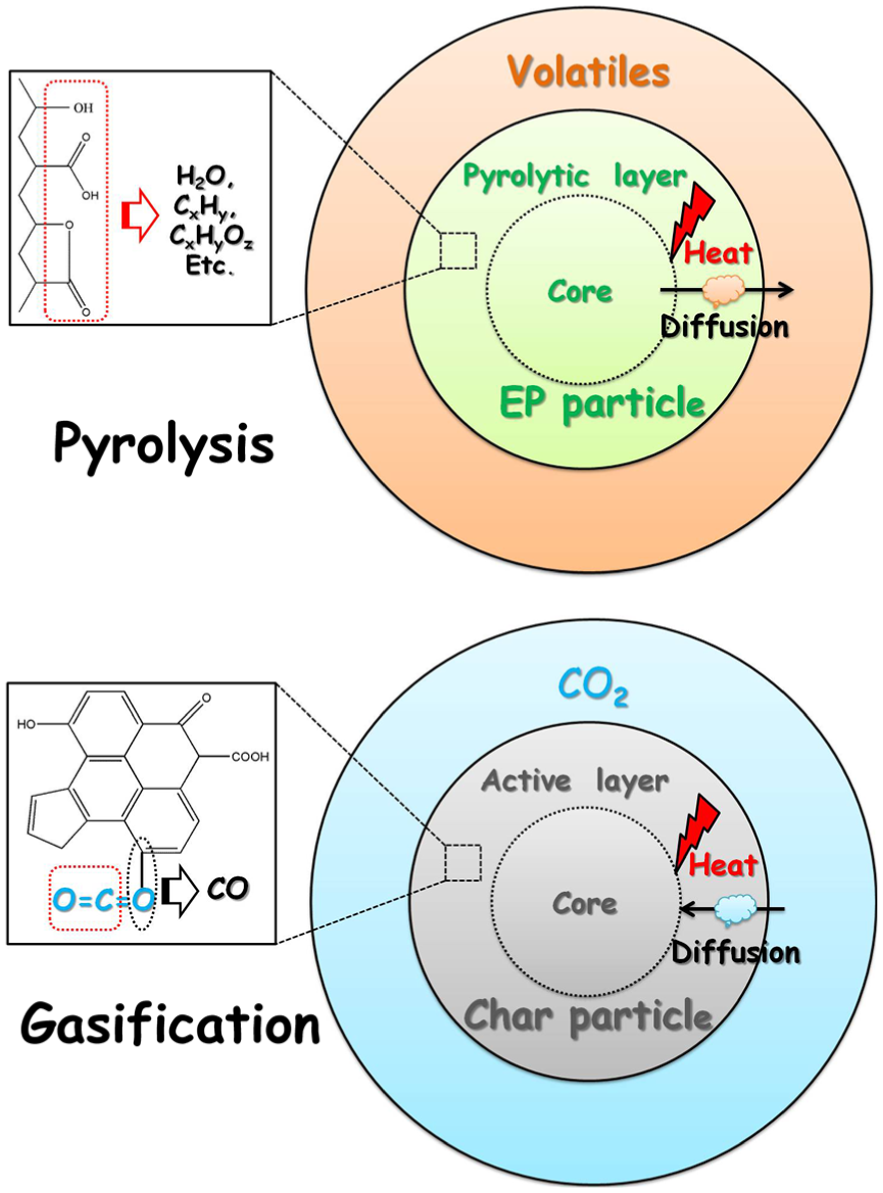
Schematic diagram of the D4 model for pyrolysis and gasification of EP.
Conclusions
Pyrolysis of EP occurred between 200 and 300°C, and gasification of pyrolytic char occurred between 700 and 900°C. Carboxylic acids, ethers, and alcohols were the dominating condensable products generated from pyrolysis. H2O, CH4, and aliphatic hydrocarbons were also formed during pyrolysis, and their continuous release at higher temperatures suggested the polymerization of the freshly generated pyrolytic char. The yield of the gasification product CO was much higher than that of the pyrolytic gaseous products. The activation energies of the pyrolysis and gasification stages were 138.30 and 93.43 kJ mol−1, respectively, and their kinetics can be described by the D4 model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Fundamental Research Funds for Central Public Welfare Scientific Research Institutes (No. K-JBYWF-2017-T09).
