Abstract
Neodymium (Nd)- or yttrium (Y)- modified bulk Ni2P catalysts (Nd-Ni2P or Y-Ni2P) have been successfully prepared and their catalytic performance in benzofuran hydrodeoxygenation have been investigated. The as-prepared catalysts were characterised by X-ray diffraction, N2 adsorption–desorption, CO uptake and X-ray photoelectron spectroscopy. The addition of Nd or Y, especially Nd, can increase the surface area of the catalysts and promote the formation of smaller and more highly dispersed Ni2P particles. The Nd-Ni2P catalyst showed the highest benzofuran hydrodeoxygenation activity of 95.3% and the O-free products yield of 74.6%, which gives an increase of 25.3% and 35.4% when compared with that found for Ni2P.
Introduction
Bio-oil is an abundant and inexpensive renewable resource, which has attracted much attention as an alternative to fossil fuels. 1 However, highly oxygenated compounds degrade the chemical and physical stability of bio-oil, so a hydrodeoxygenation (HDO) process is needed. 2 A number of catalysts have been developed and investigated for HDO, 3 among which, Ni2P was the most promising catalyst with unique chemical and physical properties in the HDO reaction. 4
The activity of Ni2P catalysts has been found to be influenced by promoters. Aluminium (Al), 5 citric acid (CA), 6 boron (B), 7 titanium (Ti) 8 and platinum (Pt) 9 have been reported as efficient promoters. The rare-earth metals with unfilled 4f electron shells exhibit outstanding optical, chemical, physical and electronic properties. 10 Hence, the rare-earth metals have been adopted and studied as promoters to enhance catalysts. 11 To our knowledge, the study of rare-earth metals, such as Nd and Y, to modify bulk Ni2P for the HDO reaction has not been reported. Therefore, it was meaningful to explore the effect of Nd and Y on the HDO performance over Ni2P catalysts.
In this article, Nd or Y were successfully incorporated into bulk Ni2P catalysts, and the effects of Nd or Y on the catalytic properties for benzofuran (BF) HDO were investigated.
Experimental
Preparation of bulk Ni2P, Nd-Ni2P and Y-Ni2P catalysts
A solution of Ni(NO3)2 together with (NH4)2·HPO4 was stirred and evaporated (initial P/Ni molar ratio of 1). The solid obtained was calcined at 500°C for 3 h to an oxidic Ni2P precursor.
Nd or Y were incorporated by impregnating the oxidic precursor with Nd(NO3)3 or Y(NO3)3 solution, respectively, and the molar fraction of Nd or Y to Ni2P was 0.1. The resulting solid was dried and calcined at 500°C for 3 h to obtain Nd-Ni2P or Y-Ni P catalyst precursors.
The bulk Ni2P, Nd-Ni2P and Y-Ni2P catalysts were prepared by reducing the precursors at 500°C for 2 h in flowing H2, then cooling naturally to room temperature and passivating in an O2/N2 mixture (0.5 vol% O2) for 1 h. The bulk catalysts obtained are denoted as Ni2P, Nd-Ni2P and Y-Ni2P, respectively.
Characterisation of catalysts
The crystal structures were determined by X-ray diffraction (XRD) analysis, which was carried out with a D/max-2200PC-X-ray diffractometer using CuKα radiation. The textural properties were analysed using Micromeritics adsorption equipment type NOVA2000e. Carbon monoxide (CO) uptakes were performed with a Micromeritics ASAP 2010 installation under static volumetric conditions. X-ray photoelectron spectroscopy (XPS) patterns were obtained with an ESCALAB MKII spectrometer with Mg radiation (E = 1253.6 eV) and a hemispherical analyzer.
Catalytic activities
The BF HDO was performed in a fixed-bed reactor using a feed consisting of a decalin solution of BF (2 wt%). The HDO conditions were 3.0 MPa, 300°C, weight hourly space velocity (WHSV) = 4.0 h–1 and an H2/Oil ratio of 500 (V/V). The feed and reaction products were analysed by flame ionisation detector (FID) – gas chromatography with a GC-14C-60 column.
Results and discussion
X-Ray Diffraction
The XRD patterns of the catalysts are exhibited in Figure 1. For all catalysts, diffraction peaks at 2θ = 40.6°, 44.5°, 47.1° and 54.1° (powder diffraction file: 03-0953) were assigned to the Ni2P phase. For bulk Ni2P catalyst, diffraction peaks at 2θ = 28.8°, 30.2°, 31.6°, 43.9°, 47.8° and 53.0° (PDF: 18-0883) can be ascribed to the Ni5P4 phase. No diffraction peaks could be seen at the corresponding positions for Nd-Ni2P and Y-Ni2P, which indicated that the addition of Nd or Y can restrain the formation of the Ni5P4 phase. The average sizes of Ni2P crystallites were calculated by the Scherrer equation (column 5 of Table 1). For Ni2P catalyst, the average size of Ni2P crystallites was 54 nm. Compared with Ni2P catalyst, the average Ni2P crystallite sizes for Nd-Ni2P (39 nm) and Y-Ni2P (21 nm) were smaller. This result may be caused by the strong interaction between nickel metal and certain reducible oxides, which constrain the growth of the nickel particle size. 12
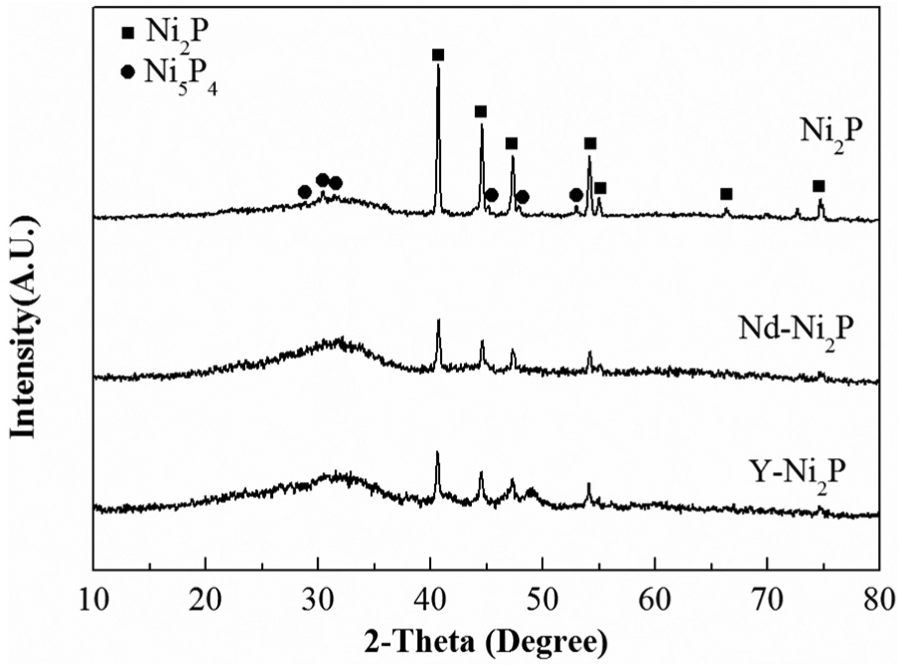
XRD patterns of the Ni2P, Nd-Ni2P and Y-Ni2P catalysts.
Textural characterisation of Ni2P, Nd-Ni2P and Y-Ni2P catalysts.

Textural properties
Table 1 shows the textural properties of the catalysts. The surface area of bulk Ni2P was 11.2 m2 g−1 with a pore volume of 0.060 cm3 g−1. Upon addition of Nd and Y, the surface area and pore volume were increased. What is worth noting is that the Nd-Ni2P catalyst displayed twofold higher surface area (25.6 m2 g−1) and pore volume (0.129 cm3 g−1) compared with bulk Ni2P catalyst. The result can be caused by the diminution in enrichment of phosphorus on the surface of the catalyst. This is discussed in X-ray photoelectron spectroscopy analysis.
CO uptake
CO uptakes were used to quantify the nickel atoms on the catalyst surface. 13 The measured CO uptakes are listed in column 6 of Table 1. The CO uptake of bulk Ni2P was 171 μmol g−1, which is very low. This may be caused by the lower surface area and the accumulation of phosphate. Upon addition of Nd or Y, the CO uptakes were 326 μmol g−1 for Nd-Ni2P and 314 μmol g−1 for Y-Ni2P. The CO uptake of Nd-Ni2P was larger than that of Y-Ni2P, which is possibly because Nd-Ni2P possesses a large surface area (Table 1, BET results) and lower surface accumulation of phosphate (see discussion of the XPS results) than those of Y-Ni2P.
X-Ray Photoelectron Spectroscopy
The XPS spectra are shown in Figure 2. Table 2 gives the binding energies and the Ni/P atomic ratios. As depicted in Figure 2(a), for all catalysts, the bands centred at 852.5–852.7 eV and 856.4–856.7 eV belong to the Niδ+ species in the Ni2P phase and Ni2+ species interacting with phosphate as a consequence of superficial oxidation, respectively. 14 In Figure 2(b), the peaks centred at 128.8–130.3 eV can be assigned to a Pδ− species in the Ni2P phase 15 and that at 134.6–134.8 eV can be attributed to phosphate (P5+) due to superficial oxidation. 16
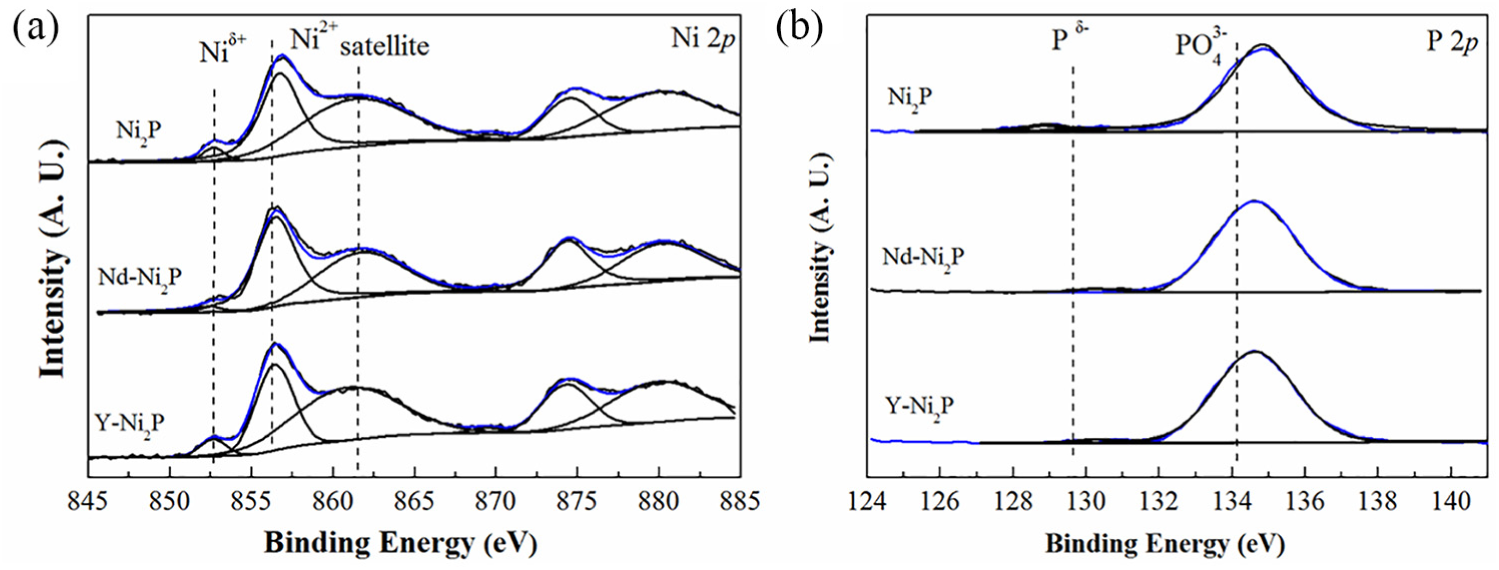
XPS spectra of the Ni2P, Nd-Ni2P and Y-Ni2P catalysts. (a) Ni 2p core level spectra, (b) P 2p core level spectra.
Spectral parameters obtained by XPS analysis.
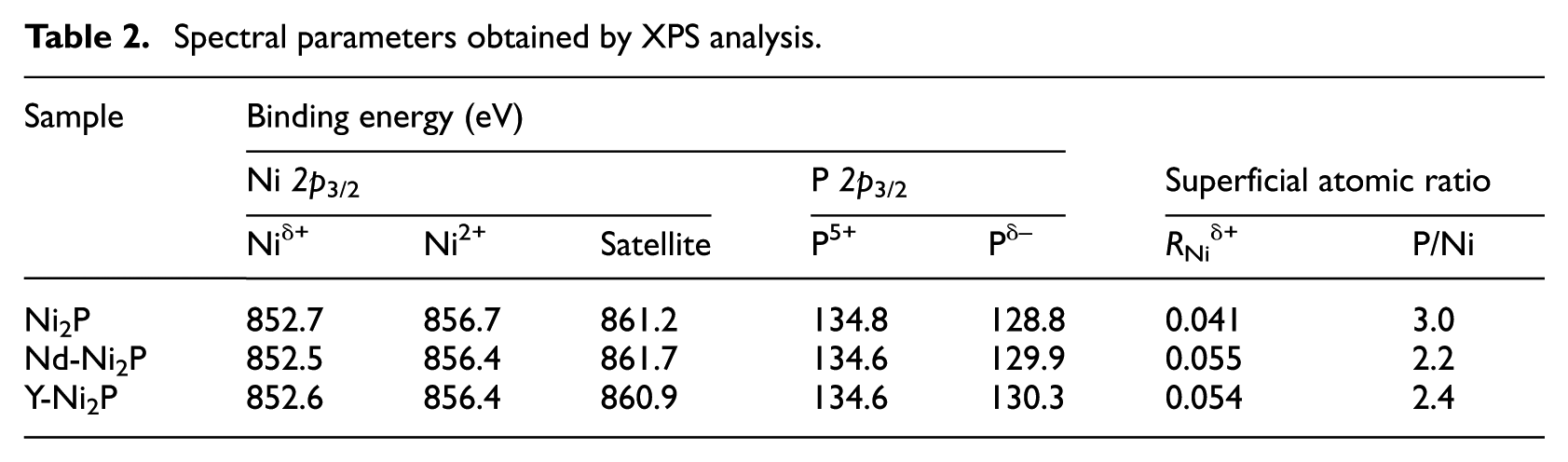
The ratios of the Niδ+ species to total Ni of the Ni2P phase (RNiδ+) are shown in Table 2. The RNiδ+ of bulk Ni2P is only 0.041, whereas those of the Nd-Ni2P and Y-Ni2P catalysts are 0.055 and 0.054, respectively. This result indicates that the addition of a suitable amount of Y or Nd promotes the formation of active Ni2P particles.
XPS data were applied to calculate the surface Ni/P atomic ratios and the results are shown in column 8 in Table 2. All samples showed larger P/Ni values than the initial P/Ni molar ratio (1.0) caused by an enrichment of phosphorus on the surfaces. The surface P/Ni atomic ratios were 2.2 and 2.4 for Nd-Ni2P and Y-Ni2P, respectively, which was smaller than that for bulk Ni2P (3.0). This result suggests that the addition of Y or Nd to bulk Ni2P can suppress the enrichment of P, which would lead to more nickel sites being exposed on the surface. 17
Catalytic results and reaction pathways
Figure 3 depicts the BF conversions over samples as a function of the reaction times. The BF conversion gradually increased and then remained stable with time. The BF conversion over bulk Ni2P was 70.0% after 8 h. Upon adding Nd or Y to the bulk Ni2P, the BF conversion was increased. The BF conversions over Nd-Ni2P and Y-Ni2P were 95.3% and 89.1%, respectively. This was attributed to Nd-Ni2P and Y-Ni2P having smaller sized particles and a better dispersion of the active Ni2P phase, together with the higher surface area and lower enrichment of phosphorus when compared with the bulk Ni2P catalyst.
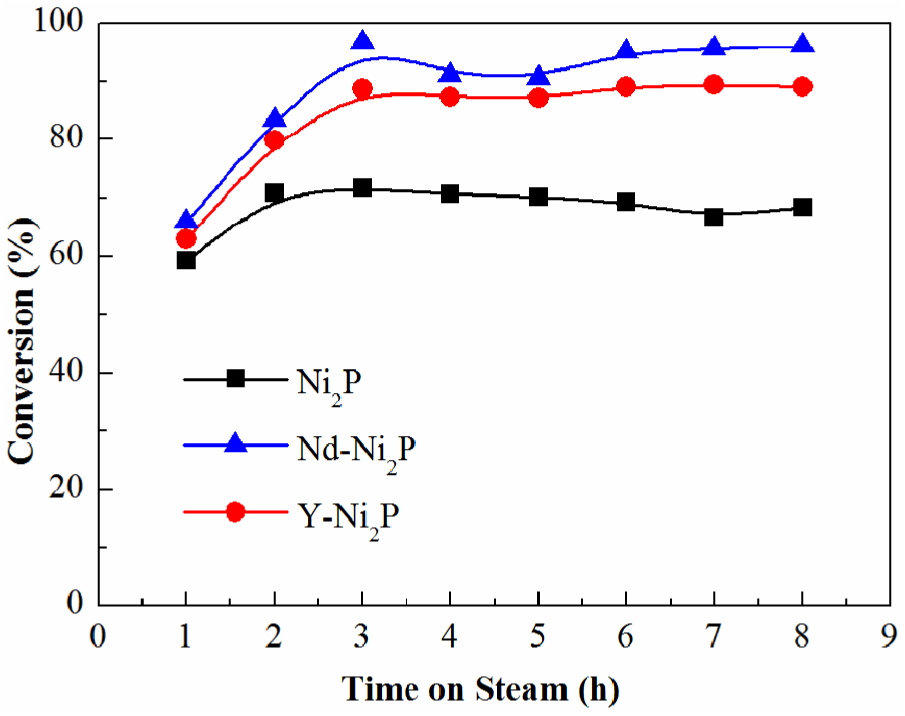
BF HDO conversion over Ni2P, Nd-Ni2P and Y-Ni2P catalysts.
Figure 4 shows the selectivity to reaction products of the prepared catalysts. On the basis of the products and previous work, 17 the reaction route is shown in Scheme 1. The first step involves hydrogenation of the furan ring, which led to the formation of 2,3-dihydrobenzofuran (2,3-DHBF), then 2,3-DHBF is further converted into 2-ethylphenol (2-EtPh) by a C-O bond cleavage of the heterocyclic ring through a hydrogenolysis. Ethylbenzene (EB) is obtained from 2-EtPh by dehydration, then converted into ethylcyclohexane (ECH) by hydrogenation of the benzene ring, followed by demethylation of ECH to give methylcyclohexane (MCH).
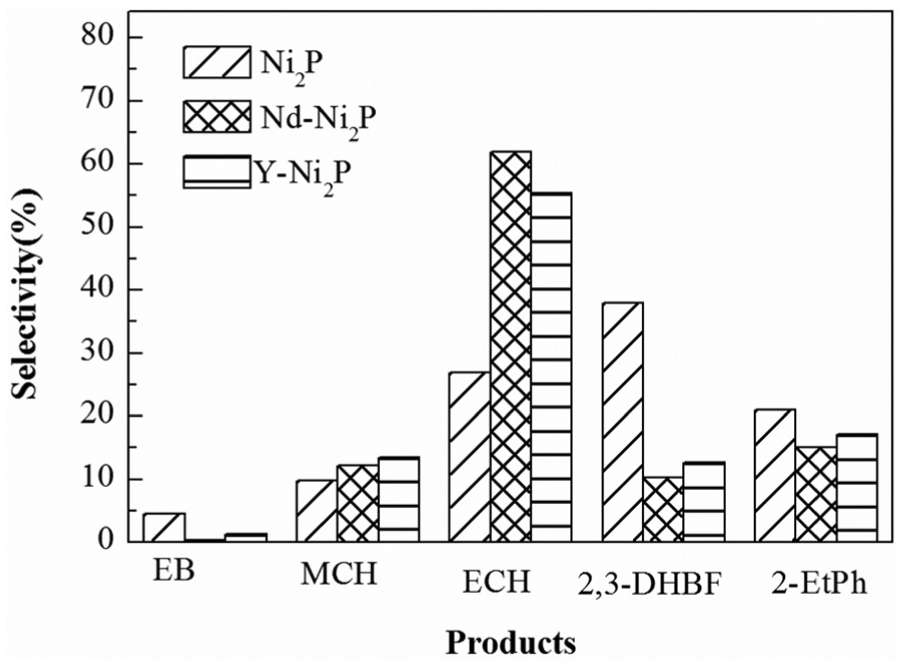
Product selectivities to BF HDO over Ni2P, Y-Ni2P and Nd-Ni2P catalysts.
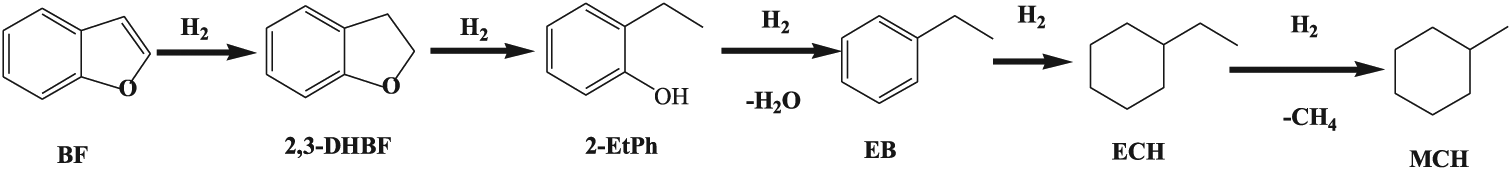
Reaction pathways for BF HDO over Ni2P, Y-Ni2P and Nd-Ni2P catalysts.
For Ni2P catalyst, the order of product selectivity was 2,3-DHBF (37.9%) > ECH (26.9%) > 2-EtPh (20.9%) > MCH (9.7%) > EB (4.6%). The selectivity over Nd-Ni2P decreased in the order ECH (62.0%) > 2-EtPh (15.1%) > MCH (12.3%) > 2,3-DHBF (10.3%) > EB (0.3%). As mentioned above, the conversion of BF over Nd-Ni2P catalyst has increased to 25.3% when compared to that of the Ni2P catalyst, but the selectivity to 2,3-DHBF has decreased to 27.6%. This showed that the addition of Nd could accelerate the rate of C-O bond cleavage of the heterocyclic ring on 2,3-DHBF via hydrogenolysis and lead to the formation of more 2-EtPh. The selectivity to 2-EtPh over Nd-Ni2P has a decrease of 5.8% when compared with that found for Ni2P. This result indicated that the 2-EtPh dehydration reaction was also accelerated by addition of Nd and generated much more EB than did the Ni2P catalyst. However, for Nd-Ni2P, the EB could hardly be detected (0.3%), which showed that the addition of Nd could efficiently accelerate the hydrogenation of the benzene ring on EB to generate ECH. In accordance with the efficient EB hydrogenation, the selectivity to ECH over Nd-Ni2P (62.0%) has an evident increase of 35.1% when compared with that found for Ni2P (26.9%). The selectivity to MCH over Nd-Ni2P (12.3%) had an increase of 2.6% when compared with that found for Ni2P (9.7%). In consideration of the selectivity to ECH over Nd-Ni2P being much higher than that of Ni2P, whether the addition of Nd was beneficial to the ECH demethylation to MCH still needs more research.
Similar results were obtained by addition of Y to Ni2P. The selectivity over Y-Ni2P decreased in the order ECH (56.4%) > 2-EtPh (17.6%) > MCH (12.0%) > 2,3-DHBF (11.7%) > EB (2.4%). However, the HDO performance of Nd-Ni2P was superior to that of Y-Ni2P.
Figure 5 shows the yields of O-free compounds over the catalysts. The yield of O-free compounds over bulk Ni2P catalyst was only 41.2%, while over Nd-Ni2P and Y-Ni2P catalysts they were 74.6% and 67.7%, respectively. These data showed that the benefit of addition of Y was inferior to the addition of Nd as regards the yields of O-free compounds, which corresponded with the characterization of the catalysts.
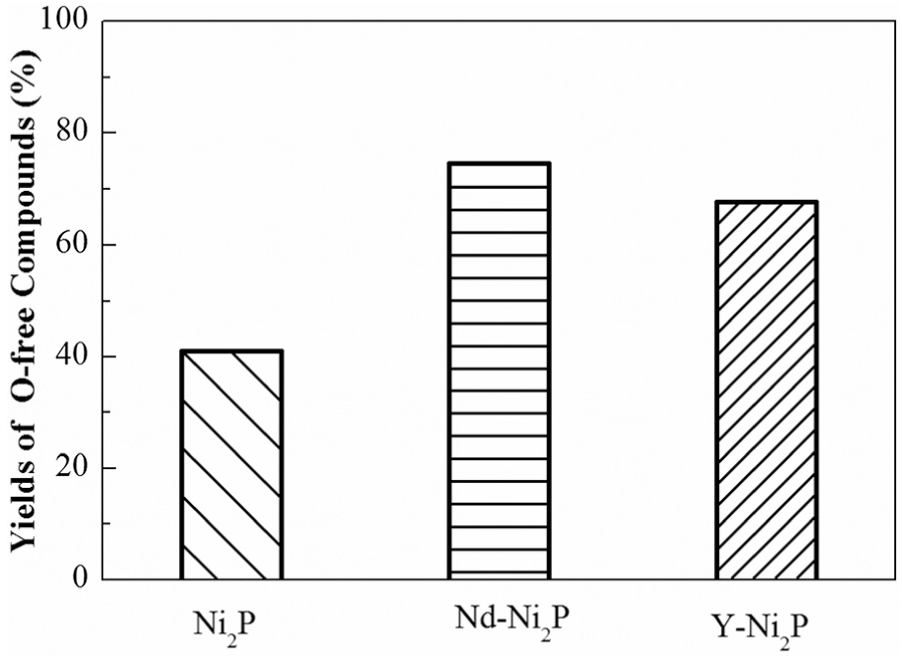
Yields of O-free compounds of BF HDO over Ni2P, Nd-Ni2P and Y-Ni2P catalysts.
Conclusion
In this work, bulk Ni2P, Nd-Ni2P and Y-Ni2P catalysts have been successfully prepared. XRD results indicated that the addition of Nd or Y can restrain the formation of Ni5P4 and thereby facilitate the generation of the Ni2P phase. Nd or Y increased the surface area of catalysts greatly, which was attributed to better dispersion of the Ni2P particles. On the contrary, the enrichment of P on the catalyst surface decreased, leading to the number of exposed nickel atoms being increased and a higher CO uptake. The Nd-Ni2P and Y-Ni2P gave high conversions for BF HDO, and the Nd-Ni2P catalyst reached 95.3%, some 25.3% higher than that of the bulk Ni2P. The yields of O-free compounds over Nd-Ni2P catalyst reached 74.6%, which was an increase of 33.4% when compared with that found for bulk Ni2P and was an increase of 6.9% when compared with that found for Y-Ni2P. The selectivity to the main O-free product MCH over Nd-Ni2P catalyst was 66.0%, indicating that the addition of Nd was highly effective for promoting the BF HDO reaction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support from the National Natural Science Foundation of China (21276048) and Innovative scientific research projects of Innovative Practice Base for Postgraduate Training in Northeast Petroleum University (YJSCX2017-016NEPU).
