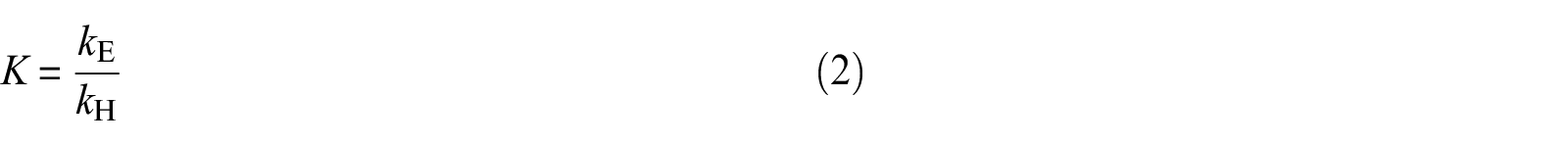Abstract
Equilibrium constants and reaction rate constants for the esterification of secondary alkan-2-ols with acetic acid were measured at 60°C in 1,4-dioxane. Taft coefficients, as single parameter (without inductive effects), and two-parameter correlation (including inductive and steric effects), of the measurements were used for the prediction of esterification rate constants for secondary alkan-2-ols with monocarboxylic acids. For this prediction, previously observed results of linear correlation of rate constants for the esterification of propan-1-ol with monocarboxylic acids measured under identical experimental conditions were applied. Two parameter Taft equations for the correlation of secondary alkan-2-ols and for monocarboxylic acids were combined, resulting in an overall correlation equation usable for the prediction of reaction rates for secondary alkan-2-ols with any monocarboxylic acid. This equation was experimentally verified for the esterification of three randomly chosen alkan-2-ols with three randomly chosen monocarboxylic acids.
Introduction
Esters of secondary alcohols with organic acids, like esters of primary alcohols,1,2 have been applied in industrial production of fragrances and aromas. For example, propan-2-yl acetate is used in the preparation of apple aromas (FEMA 2926), propan-2-yl propionate for plum- or raspberry-type aromas (FEMA 2959), cyclohexyl acetate for raspberry-, banana-, or pineapple-type aromas (FEMA 2349), and cyclohexyl butyrate for pineapple or apple aromas, or jasmine scents (FEMA 2351). Apart from applications in the food industry, esters of secondary alcohols have various usages. The 2-propyl ester of 4-chloro-2-methylphenoxyacetic acid has been utilized as a herbicide. 3 Propan-2-ol and butan-2-ol esters with fatty acids lower the cloud and pour points of biodiesel 4 by up to −17°C in comparison with methyl esters. 5 Esters of propan-2-ol with tetradecanoic (myristic) or 16-methylheptadecanoic (isostearic) acids significantly increase the penetration of various substances through skin. 6 Compounds containing propan-2-yl tetradecanoate are described as carriers of natural antioxidants. 7 Esters of decan-2-ol and tridecan-2-ol are used for the preparation of synthetic oils. 8
This article is an attempt to determine experimentally a correlation equation of Taft-type for one and two parameters (including coefficients for steric and inductive effects) for the esterification of secondary alkan-2-ols with monocarboxylic organic acids. This equation was combined with previous results 1 leading to an equation according to which it is possible to predict the esterification rate constants of secondary alkan-2-ols with any monocarboxylic acid without experimental measurements. This methodology can be applied in ester synthesis and the food and perfume industries for the calculation of reaction time expenses for the production of these materials.
The esterification of secondary alcohols with organic acids is a well-studied reaction. Its reaction rate depends on temperature, concentrations of reactants and products, concentration of catalyst (hydrogen ions), solvent, and the structures of alcohols and acids.1,9,10 The rate of esterification voverall for secondary alcohols (including alkan-2-ols) is described by the following equation
where c is the molar concentration of the corresponding species. Since the equilibrium constant K is defined as the ratio of the forward (esterification) kE and reverse (hydrolysis) kH reaction rate constants
equation (1) may be rewritten as follows
The solution of this differential equation was derived 1 under initial conditions cacid=calcohol=1 mol dm−3 and cester = cwater = 0 mol dm−3 and may be expressed as
where x is the degree of conversion as a function of time t
Experimental
Propan-2-ol, butan-2-ol, pentan-2-ol, hexan-2-ol, 4-methylpentan-2-ol, 1,4-dioxane (Merck, Germany), anhydrous CuSO4, methanol, H2SO4, phenol, acetone, and NaOH (Lachema, Czech Republic) were of AR purity. Ethanol was used for ultraviolet (UV) spectroscopy in all experiments.
First, 1-chloropropan-2-ol was prepared by the rectification of a mixture of 1-chloropropan-2-ol and 2-chloropropan-1-ol (ratio 3:1), which was prepared by the reaction of propene with hypochlorous acid (supplied by CHZ Nováky, Slovakia) using a column with an efficiency of 80 TP at a pressure of 4 kPa. From this precursor (1-chloropropan-2-ol), 1-methoxypropan-2-ol, and 1-phenoxypropan-2-ol were synthesized by condensation with sodium methoxide or sodium phenoxide, respectively, in anhydrous methanol and then purified using the same distillation column. Physical properties related to the synthesized alkan-2-ols are presented in Table 1.
Physical parameters of synthesized alkan-2-ols.
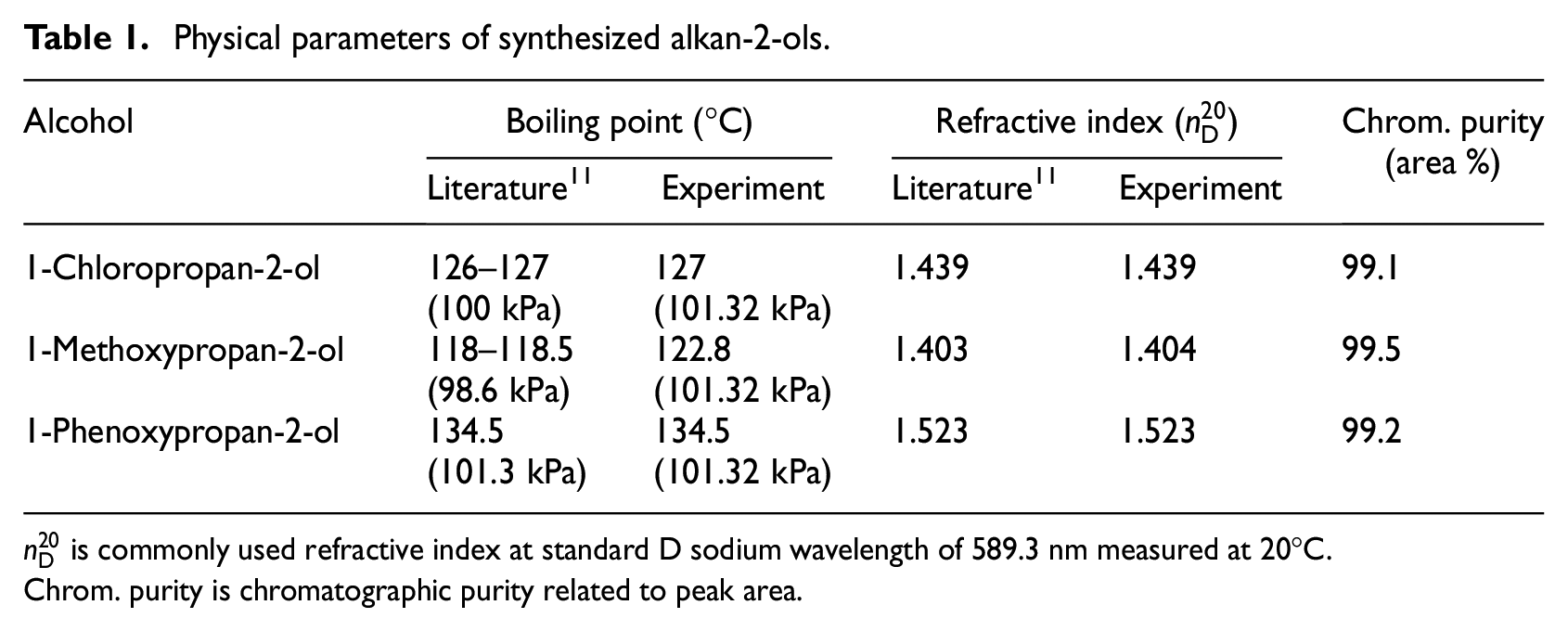
Chrom. purity is chromatographic purity related to peak area.
A static method was used for the determination of the esterification equilibrium constants. Solutions containing equimolar amounts of the relevant alkan-2-ol and acetic acid at a concentration of 1 mol dm−3, 1,4-dioxane (dried using CuSO4), and catalyst (H2SO4, 0.01 mol dm−3) were sealed in glass vials and heated to 90°C for 3 h and then left for 3 months at room temperature. Subsequently, vials were placed into a thermostatically controlled device, keeping the temperature at 60 ± 0.1°C for 7 days. The acetic acid content was determined titrimetrically, and equilibrium constants were calculated according to equation (6).
A discontinuous isothermal reactor, immersed in a bath at 60 ± 0.1°C, equipped with an electromagnetic stirrer, and closed by a cap, was used for the measurements related to esterification reaction kinetics of secondary alkan-2-ols. Samples for analysis, carried out by titration, were taken at selected intervals using a syringe.
Titration was carried out using phenolphthalein indicator in the NaOH titration solution. The purity of synthesized alkan-2-ols was assessed using an Abbe refractometer, a pycno-meter, and capillary gas chromatography.
Results and discussion
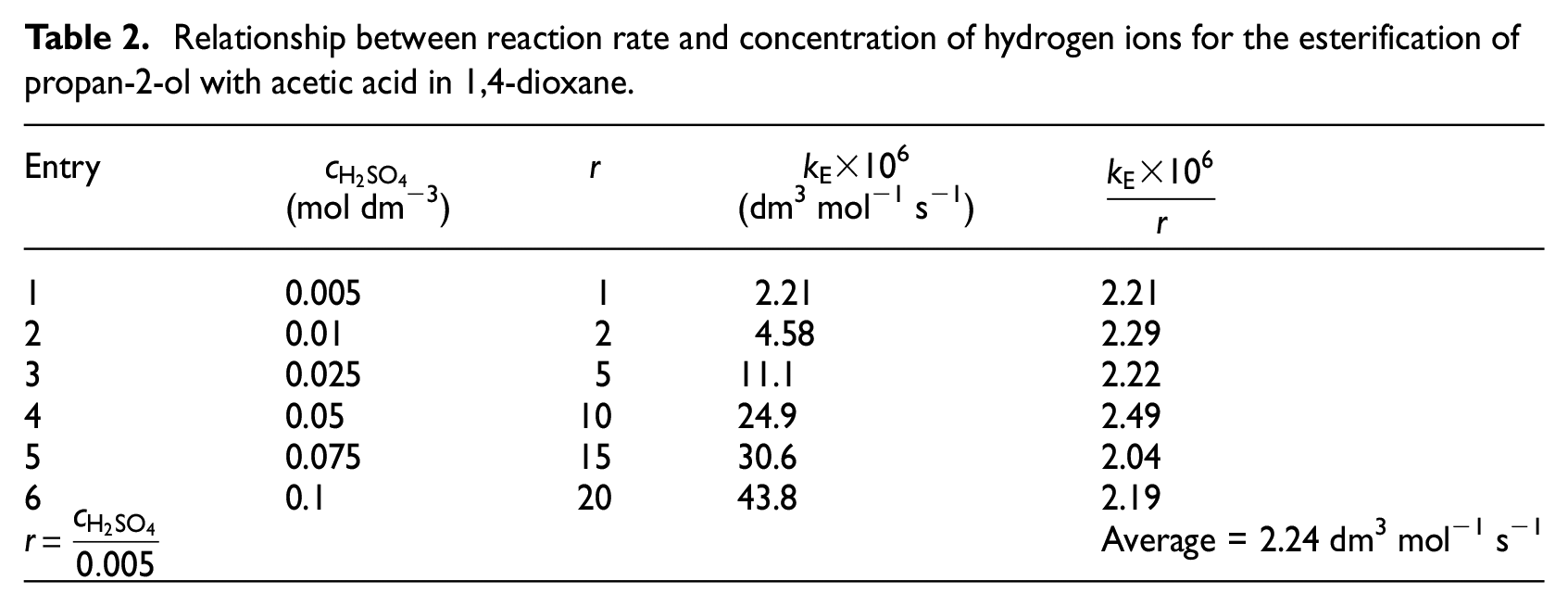
The esterification rate of secondary alcohols is an order of magnitude slower in comparison with primary alcohols; therefore, a larger amount of catalyst has been used for measuring rate constants. The esterification rate constant depends linearly on the hydrogen ion concentration according to equation (5)

where
Relationship between reaction rate and concentration of hydrogen ions for the esterification of propan-2-ol with acetic acid in 1,4-dioxane.
Measured values, x∞, of conversion at equilibrium (degree of conversion x for t = t∞) for the secondary alkan-2-ols are presented in Table 3.
Equilibrium constants and Taft coefficients 12 of secondary alcohols.
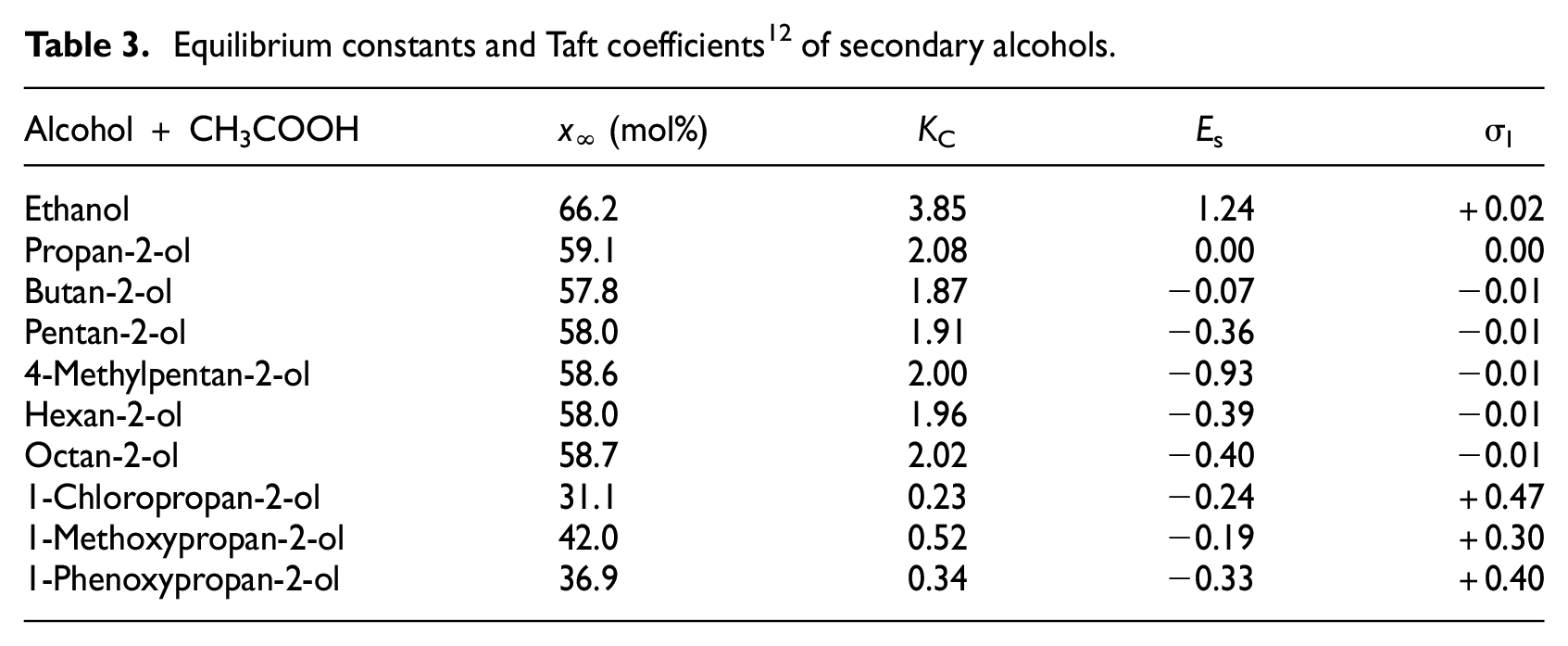
Then, concentration equilibrium constants were calculated according to the following equation

The kinetics of esterification for secondary alkan-2-ols with acetic acid at 60°C were measured as in our previous paper. 1 The equilibrium constants KC (given in Table 3) were used for the calculation of the rate constants kE (refer to equation (4)). The results acquired are shown in Table 4.
Experimental and calculated esterification rate constants for secondary alkan-2-ols with acetic acid at 60°C in 1,4-dioxane.
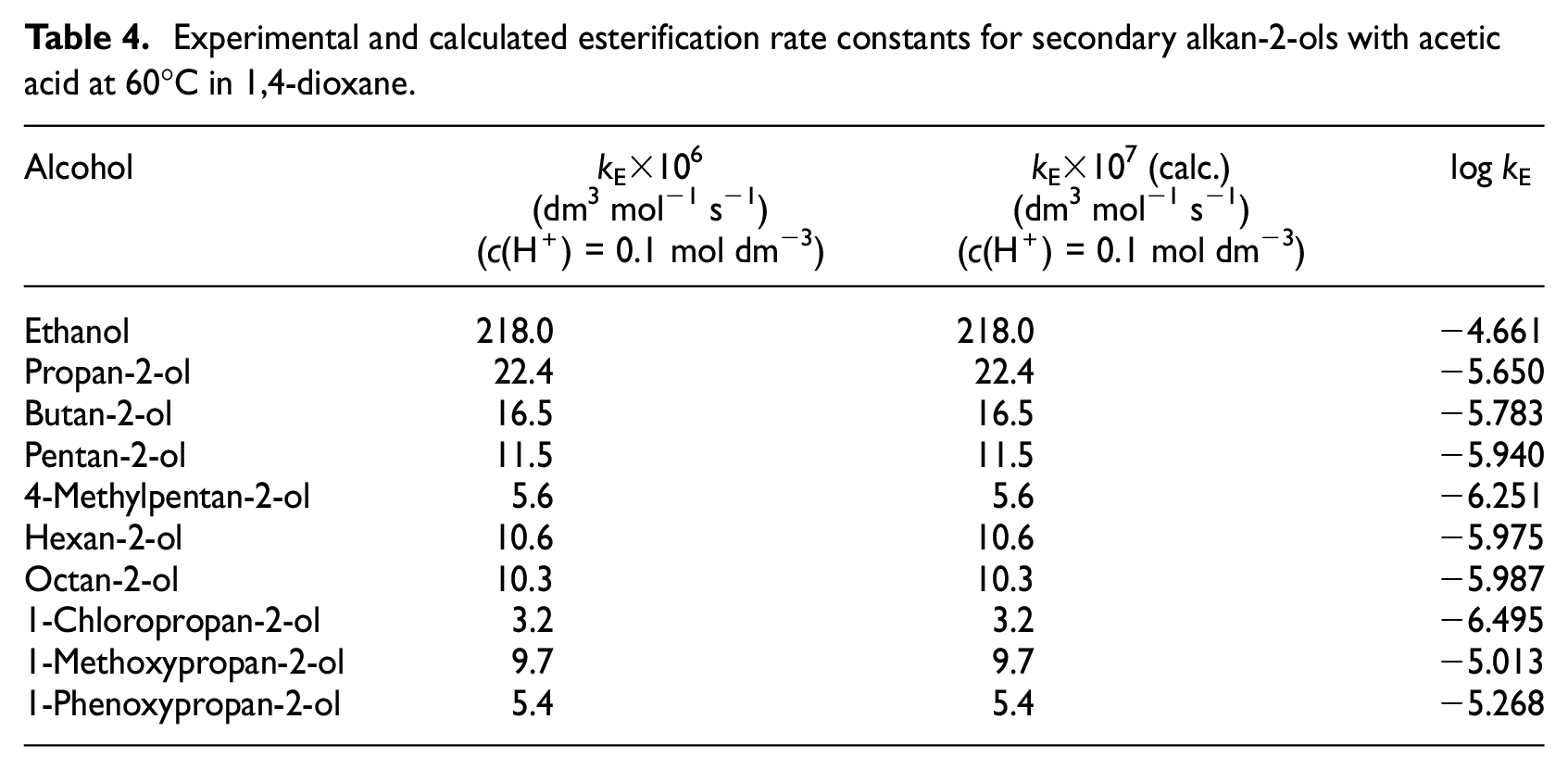
The esterification rate constants were correlated in two ways. The first correlation was restricted to the secondary alkan-2-ols with nearly zero inductive effects. The esterification rate of these alkan-2-ols is controlled just by the steric effects. The correlation equation with Taft coefficients for the second case was calculated as follows

where kE is the esterification rate constant related to the relevant alkan-2-ols with CH3COOH,
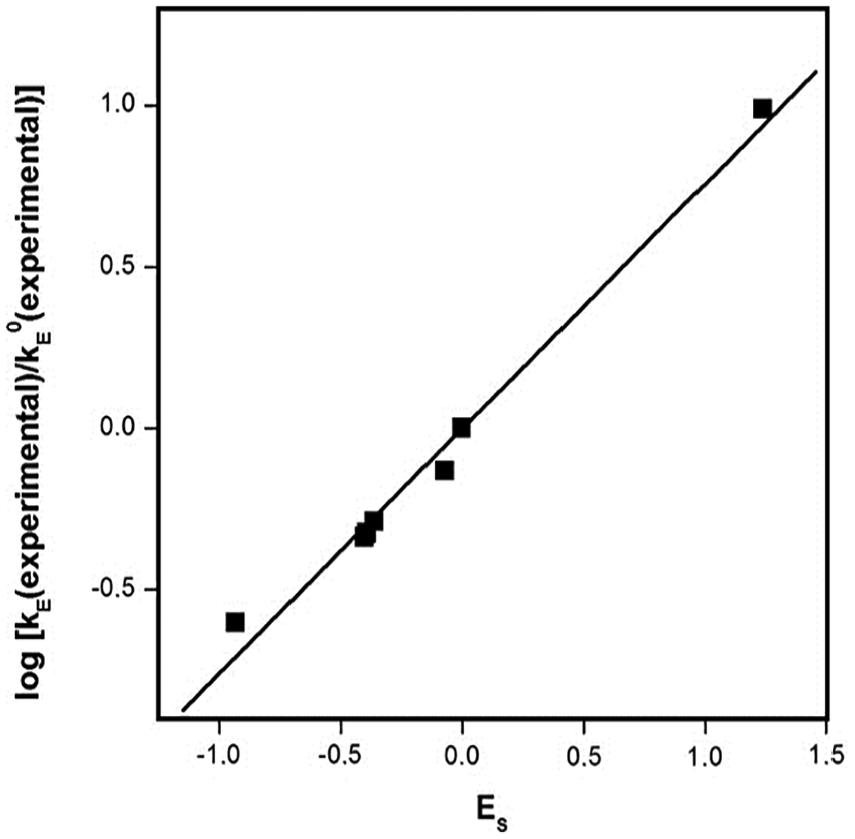
Dependence of ratio log
Relationship between the esterification rate constants of secondary alkan-2-ols and Taft coefficients for the steric effect.
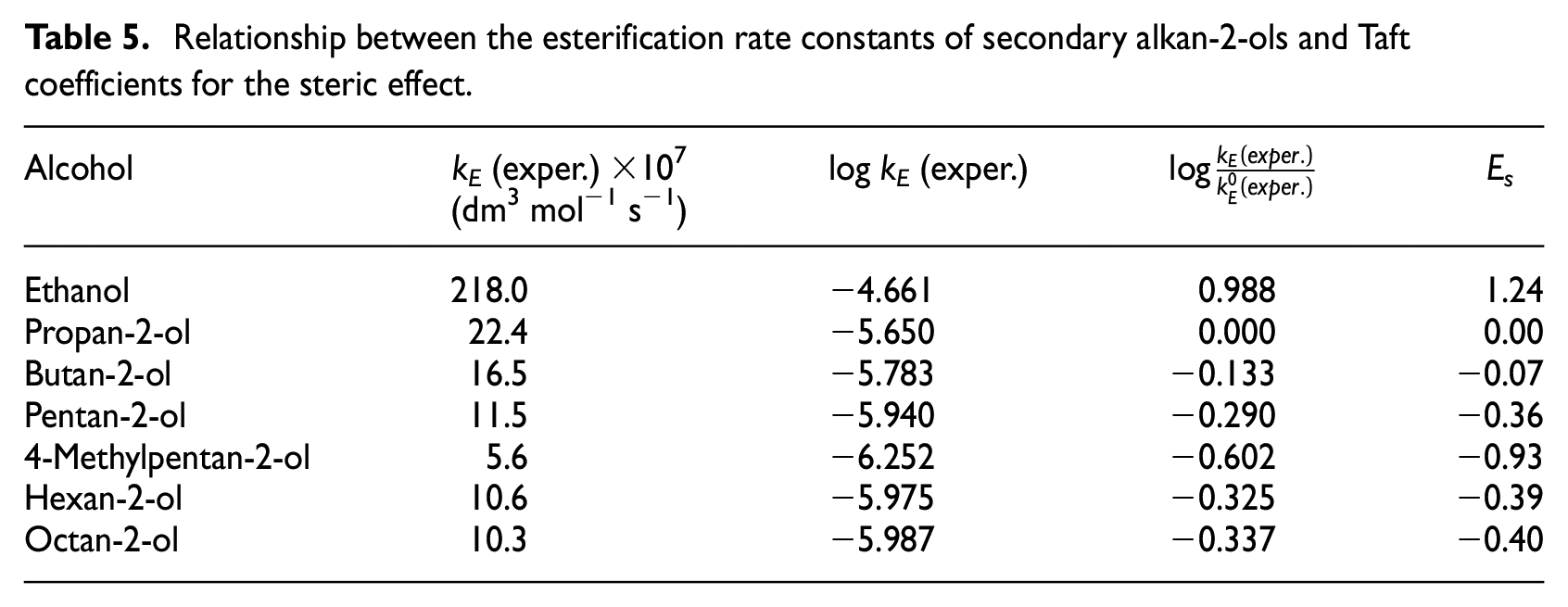
Subsequently, the rate constants of all measured secondary alkan-2-ols were correlated using the following two-parameter Taft equation

where kE,
Comparison of the experimentally acquired and calculated values of the esterification rate constants of secondary alcohols.
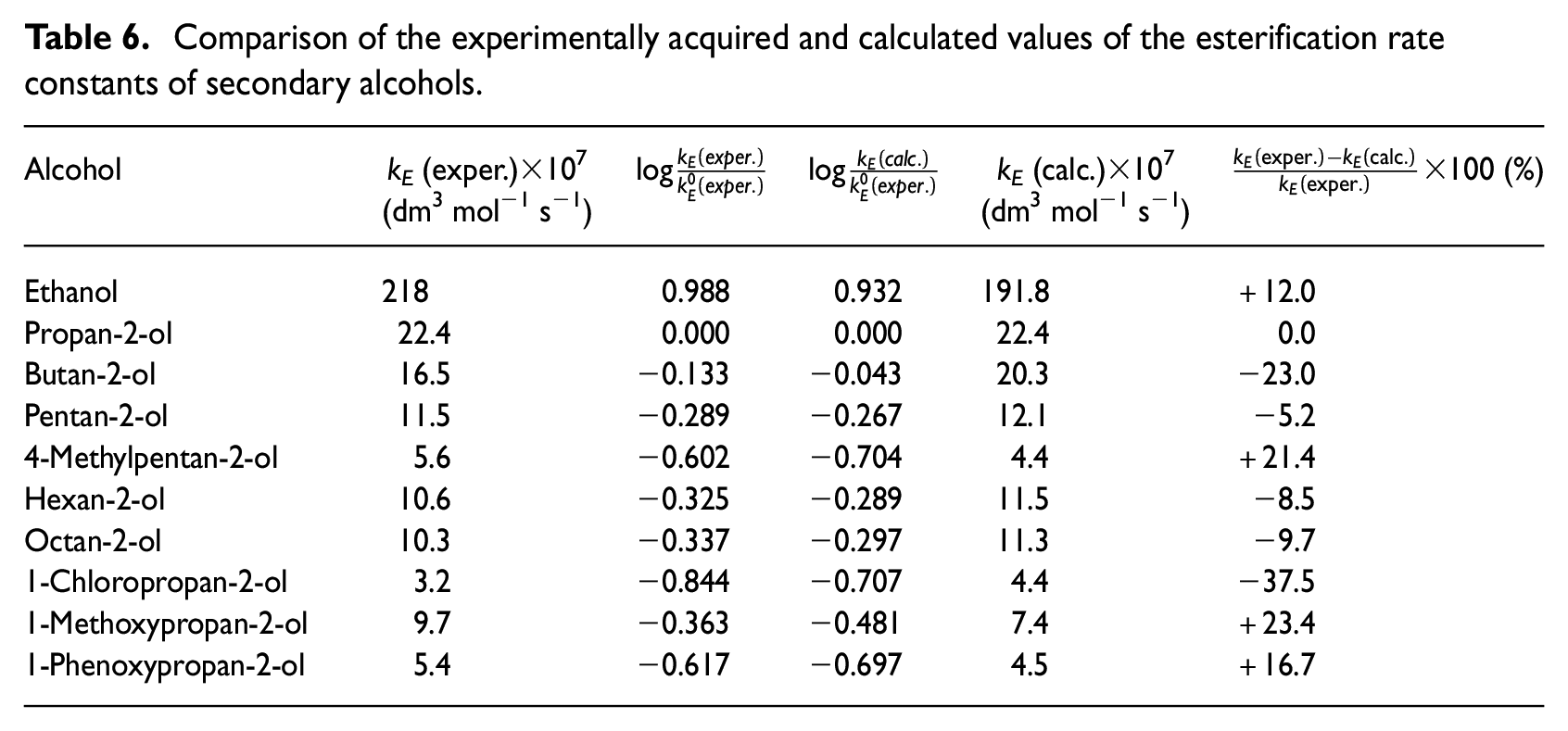
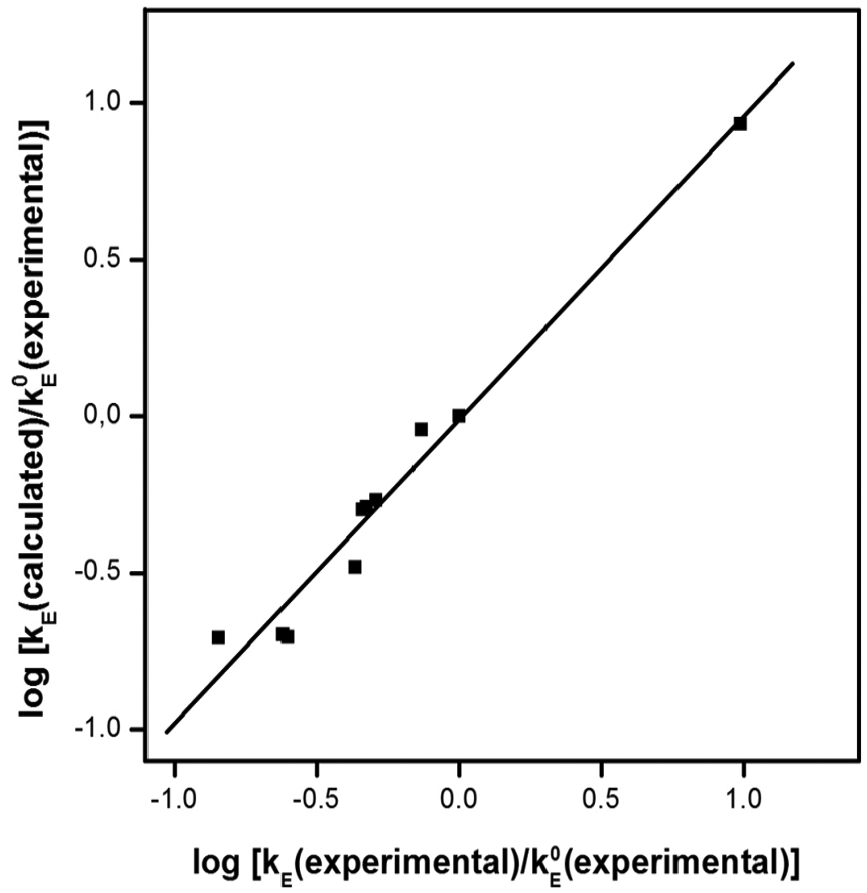
Dependence of
It is interesting to note that the esterification equilibrium constants with acetic acid for the secondary alkan-2-ols lacking inductive effects are practically constant (KC→ 2). This finding confirms the general conclusion9,10 where KC of different acids (KC→ 4) was found in the esterification with propanol. As mentioned above, the esterification rate of the secondary alkan-2-ols (see Table 2) is approximately an order of magnitude slower than the esterification of primary alcohols. 1 This may be caused by higher steric shielding of the secondary alcohol by two alkyl groups. Hence, a 10 times higher catalyst concentration (c(H+) = 0.05 mol dm−3) was used for the measurement of the esterification rate constants to effect the esterification at an appropriate rate. Subsequently, the acquired esterification rate constants were divided by five because equation (3) is valid within the range of catalyst concentrations used as experimentally demonstrated (see Table 2), so that the correlation equation used previously for organic acids 1 is applicable

where kE is the esterification rate constant for the organic acid with propan-1-ol;
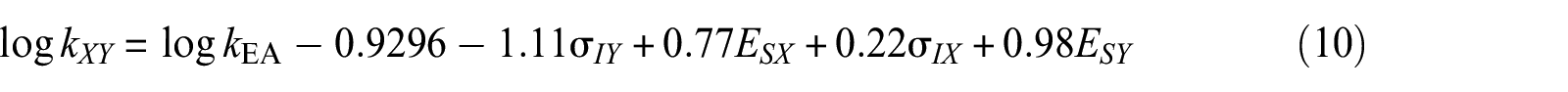
where kXY is the esterification rate constant of any alkan-2-ol in the absence of mesomeric (resonance) effects with any organic acid also without mesomeric (resonance) effects; kEA is the esterification rate constant of ethanol with acetic acid; σIX and ESX are, respectively, the inductive and steric Taft coefficients of secondary alkan-2-ols related to propan-2-ol; and σ IY and ESY are, respectively, the inductive and steric Taft coefficients of organic acids related to acetic acid.
For the experimental verification of equation (10), the formation of esters from some relevant alcohols and acids was used by measuring the esterification rate constants relating to the formation of pentan-2-yl butyrate, 1-chloro-2-bromopropylacetate, and 1-methoxypropan-2-yl propionate. Measured rate constants relating to the formation of these esters are given in Table 7.
Comparison of measured and calculated rate coefficients related to some esters using equation (10).
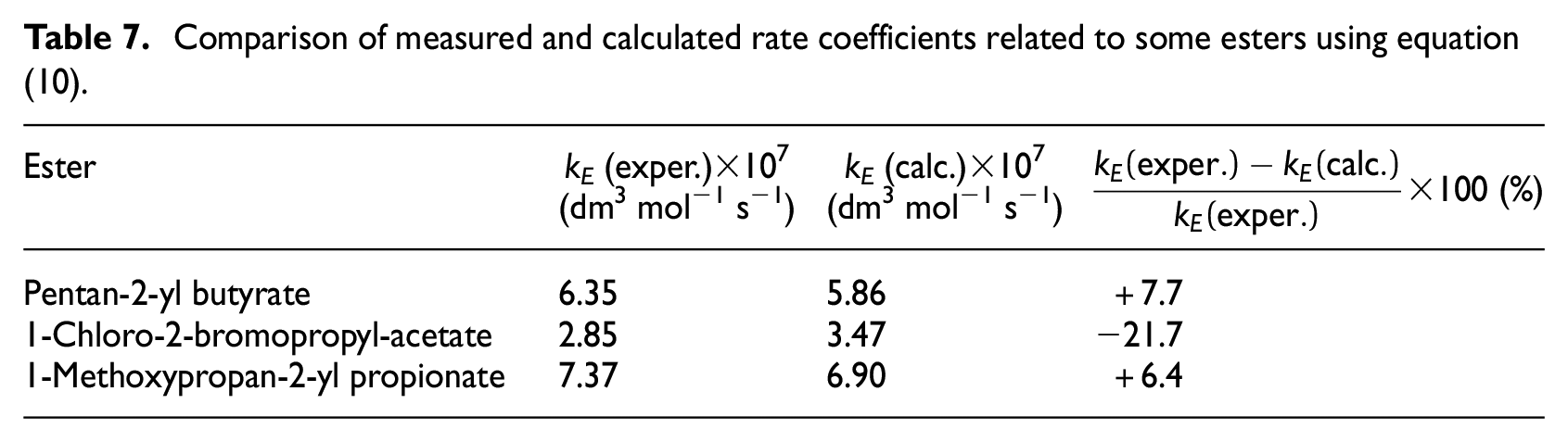
As can be seen from this table, the measured rate constants are in reasonably good agreement with the values calculated according to equation (10).
Prediction of the esterification rate constants for the secondary alkan-2-ols according to equation (10) is justified up to a point as can be seen from these results. However, the accuracy is lower than with the alkan-1-ols, 1 which is presumably due to the limited number of measured alkan-2-ols.
The methodology could probably be applied also for the secondary alkan-3-ols (one of the constant alkyls would be ethyl). However, this test comes up against the lack of suitable model compounds.
Conclusion
Here, we developed a simple and rather reliable tool for the prediction of reaction rates for the esterification of secondary alkan-2-ols. Equilibrium and rate constants for the esterification of chosen, easily accessible alkan-2-ols with acetic acid were measured in 1,4-dioxane as a suitable solvent. The above-mentioned procedure is based on fitting a one parameter Taft equation containing only a Taft steric coefficient and a two-parameter Taft equation containing Taft coefficients for inductive and steric effects. This equation was combined with the two-parameter Taft equation from the study of Vojtko and Tomčík 1 under the same experimental conditions. The results of this combination are a two-parameter equation according to which it is possible to estimate esterification rate constants for any secondary alkan-2-ols with any monocarboxylic acid without experimental measurements under compatible experimental conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the KEGA Agency (project no. 006KU-4/2017) and the GAPF Agency (project no. 1/3/2017).