Abstract
Substitution reactions of square-pyramidal [ZnCl2(terpy)] complex (terpy = 2,2′:6′,2″-terpyridine) with biologically relevant nucleophiles such as imidazole, glutathione, 1,2,4-triazole, and pyrazine were investigated at pH 7.0 as a function of nucleophile concentration. The reactions were followed under pseudo first-order conditions by UV-Vis spectrophotometry. The substitution reactions comprised two steps of consecutive displacement of chlorido ligands. Different reaction pathways for the first reaction step of nucleophilic substitution were defined. The order of reactivity of the investigated nucleophiles for the first reaction was imidazole > glutathione > pyrazine > 1,2,4-triazole, while for the second reaction step it was pyrazine > 1,2,4-triazole > imidazole > glutathione.
Introduction
Design of DNA- and protein-targeting metal-based anticancer agents with potential in vitro toxicity has gained importance in recent years. 1 Some transition metal ions are essential cellular components selected by nature to function in several biochemical processes; they act mainly as Lewis acids and have unique characteristics that include variable coordination modes and reactivity towards biologically relevant nucleophiles, etc. Zinc-based compounds could be promising anticancer agents, especially because zinc is implicated as an important cytotoxic/tumor suppressor agent in several cancers. 2 The mechanism of potential anticancer activity of zinc(II) complexes is assumed to be connected with (1) fast interconversion among its four-, five-, and six-coordinate states and (2) preference of the variable coordination geometries (tetrahedral, five-coordinate, and octahedral) that zinc(II) is able to adopt, towards diverse donor sites of relevant nucleophiles. 3 Knowledge of the mechanism of interaction of zinc(II) ions with bio molecules and other relevant ligands is essential for understanding the cellular biology of delivery complexes to DNA and proteins. The investigation of zinc biology includes at least three types of agent: chelating agents to remove zinc ions or buffer zinc in a certain range of pZn, pharmacological agents to transport through cellular membranes and deliver zinc in a bioavailable form, and as a fluorescent probe to investigate zinc-dependent processes. 4
Recently, we have determined the mechanism of interaction between biologically relevant nucleophiles and the [ZnCl2(terpy)] complex in the presence of NaCl.5,6 The excess of chloride did not affect the coordination geometry of the complex. The investigation of the metal–ligand stoichiometry between the [ZnCl2(terpy)] complex and imidazole implied formation of the five-coordinate species [Zn(terpy)(imidazole)2]. 5
The main goal of this study was to determine the complex-formation kinetics between the zinc(II) complex and some biologically relevant nucleophiles in aqueous solution at pH 7.0 in the absence of biologically important anions such as chloride, inorganic phosphate, sulfate, etc. It was envisaged that this study could throw more light on our understanding of the mechanism of interaction of the square-pyramidal complex with N-, O-, or S-donor ligands. Experiments performed under selected conditions enabled us to make comparison with the results obtained previously. The structures of the complex and the selected nucleophiles are shown in Figure 1.
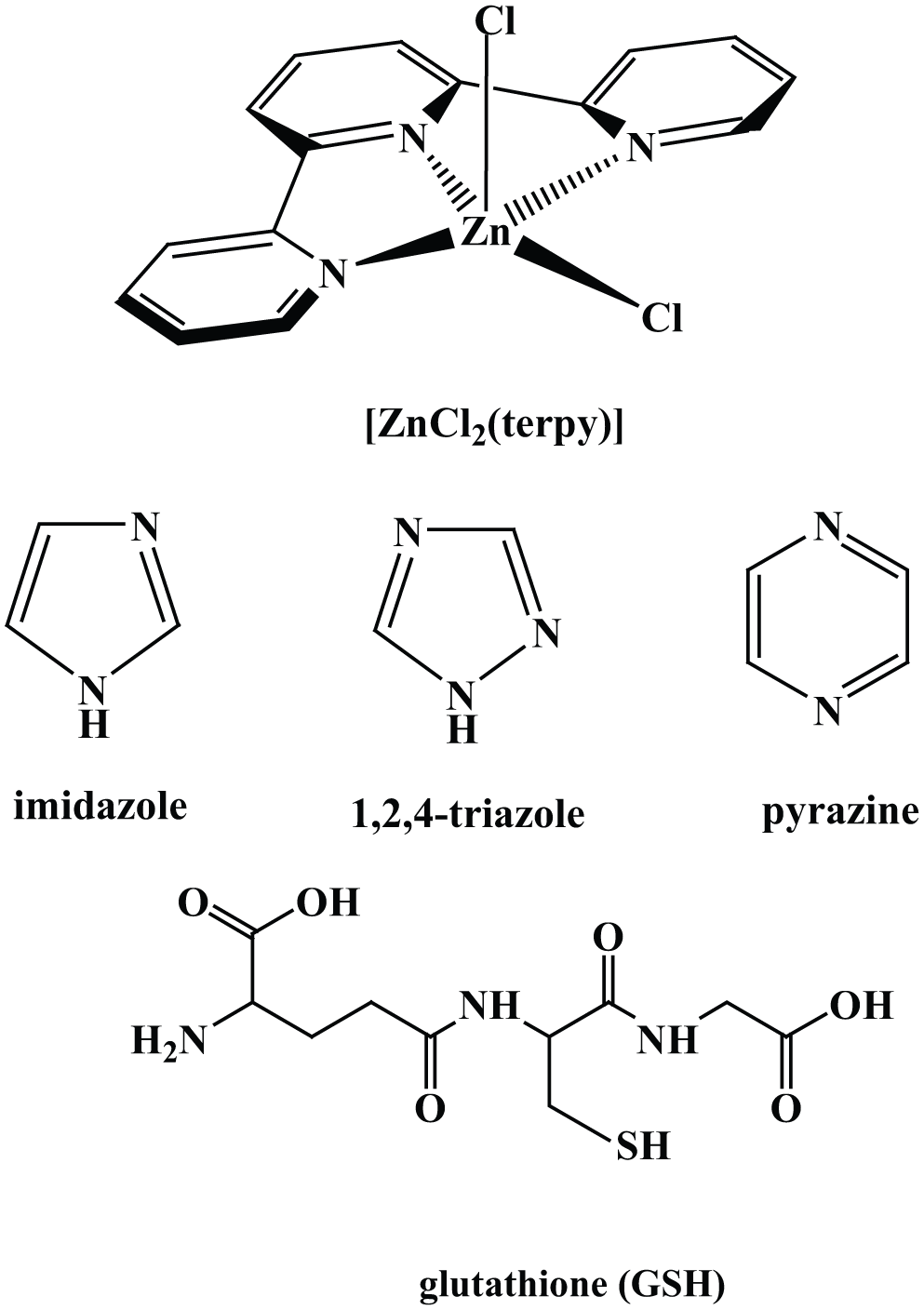
Structures of the investigated complex and nucleophiles along with the adopted abbreviation.
Experimental
Chemicals and instrumentation
The nucleophiles imidazole, glutathione (GSH), 1,2,4-triazole, and pyrazine were obtained from Sigma-Aldrich, Acros Organics, and Fluka. The complex [ZnCl2(terpy)] was synthesized according to the literature method. 7 Chemical analysis was performed on a Carlo Erba Elemental Analyzer 1106: Anal. calcd for C15H11Cl2N3Zn: N, 11.37; C, 48.75; H, 3.00; found: N, 11.31; C, 48.61; H, 3.01%. UV-Vis spectra were recorded on Uvikon XS and Shimadzu UV250 diode-array spectrophotometers in thermostated 1.00 cm quartz Suprasil cells. The temperature was controlled throughout all kinetic experiments to ±0.1°C. The pH measurements were recorded on a Jenway 4330 pH meter with a combined Jenway glass microelectrode that had been calibrated with standard buffer solutions of pH 4.0, 7.0, and 10.0 (Merck). The KCl solution in the reference electrode was replaced with a 3 M NaCl electrolyte to prevent precipitation of KClO4 during use.8,9
Kinetic measurements
Spectral changes resulting from mixing [ZnCl2(terpy)] and nucleophile solutions were recorded over the wavelength range 200–500 nm to establish a suitable wavelength at which kinetic measurements could be performed (see Figure 2 and supplemental material Tables S1–S4). The ligand substitution reactions were studied for the nucleophiles: imidazole, 1,2,4-traiazole, pyrazine, and GSH. All solutions were prepared shortly before use by dissolving the chemicals in highly purified double deionized water.
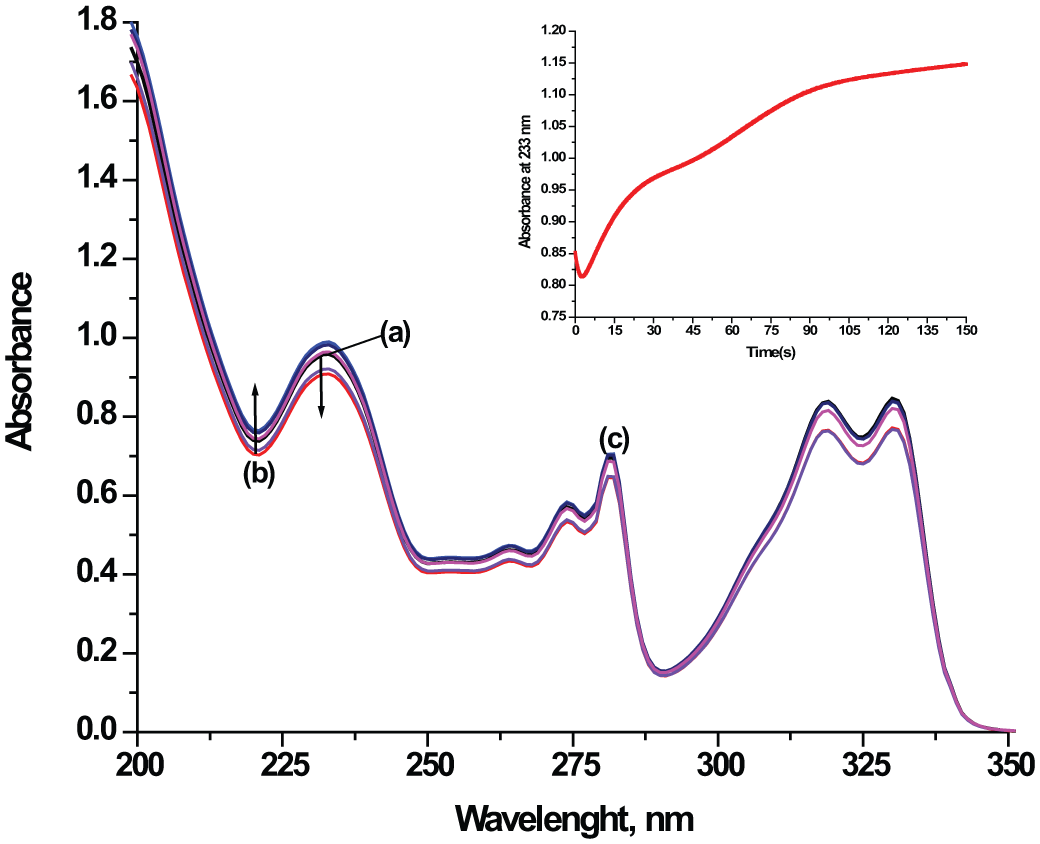
Rapid-scan spectra recorded for the reaction of [ZnCl2(terpy)] complex (0.0001 M) with GSH (0.002 M) at pH 7.0 in aqueous solution at 295 K: (a) spectrum before the reaction; (b) spectrum obtained 3 s after mixing of the reactants and (c) spectrum obtained after 150 s. Inset: time trace obtained for the reaction at 233 nm. A decrease of absorbance and increase after 3 s were observed.
Reactions were initiated by mixing equal volumes of thermostated solutions of the complex and ligand in the UV-Vis spectrophotometric cell and were followed for at least eight half-lives. All kinetic experiments were performed under pseudo first-order conditions with respect to the nucleophile concentration. The initial concentration of the [ZnCl2(terpy)] complex was 0.0001 M. The measured kobs values are summarized in Tables S1–S4 (see supplemental material). All kinetic runs could be fitted to a double-exponential function. The observed pseudo first-order rate constants, kobs, were calculated as the average value from three to four independent kinetic runs. The OriginPro 2016 program was used for the calculations of rate constant values.
Results and discussion
The ligand substitution reactions of the [ZnCl2(terpy)] complex with biologically relevant nucleophiles were monitored in the range of ca. 230–295 nm. An example of the temporal changes in the UV-Vis spectra is shown in Figure 2. In most reactions, an initial decrease of absorbance and then an increase after several seconds was observed.
Solutions were prepared by dissolving known amounts of complex (0.0001 M) in double-deionized water without the presence of excess chloride. Chloride added to the solution to adjust the ionic strength could change the zinc speciation, especially in the absence of other nucleophiles with high affinity for zinc. Our previous mole-ratio research for the [ZnCl2(terpy)] complex in the presence of chlorides showed no additional coordination of chloride.5,6 Ionic strength will not exert an effect on reaction rates, because the charges of the complex and most of the nucleophiles are neutral.
All substitutions proceeded in two consecutive reaction steps, both depending on the nucleophile concentration. All kinetic traces gave an excellent fit to a double-exponential function, typical for a two-step reaction. The pseudo first-order rate constants so-obtained, kobs1 and kobs2, calculated from the kinetic traces, were plotted against the concentration of the entering nucleophiles. A linear dependence on the nucleophile concentration was observed for all reactions studied (see Figures 3 and 4).
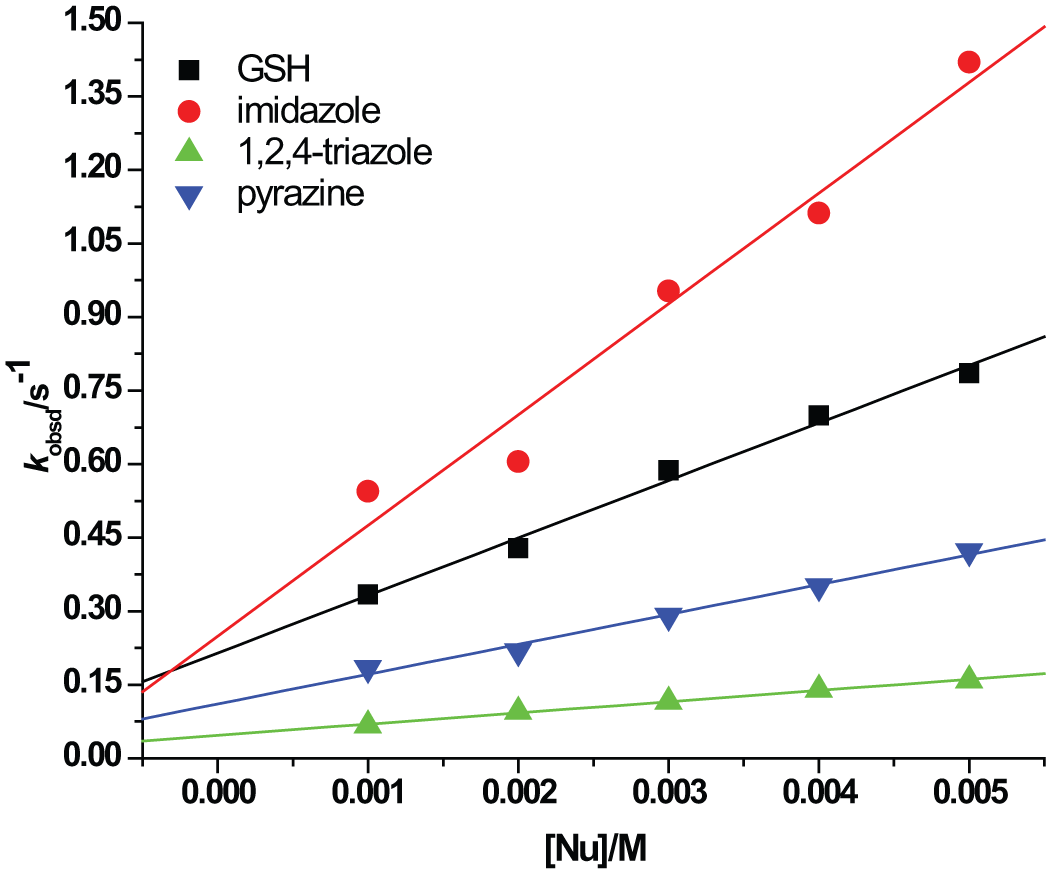
Pseudo first-order rate constants plotted as a function of nucleophile concentration for the first reactions of the [ZnCl2(terpy)] complex with imidazole, GSH, pyrazine and 1,2,4-triazole in aqueous medium at 295 K.
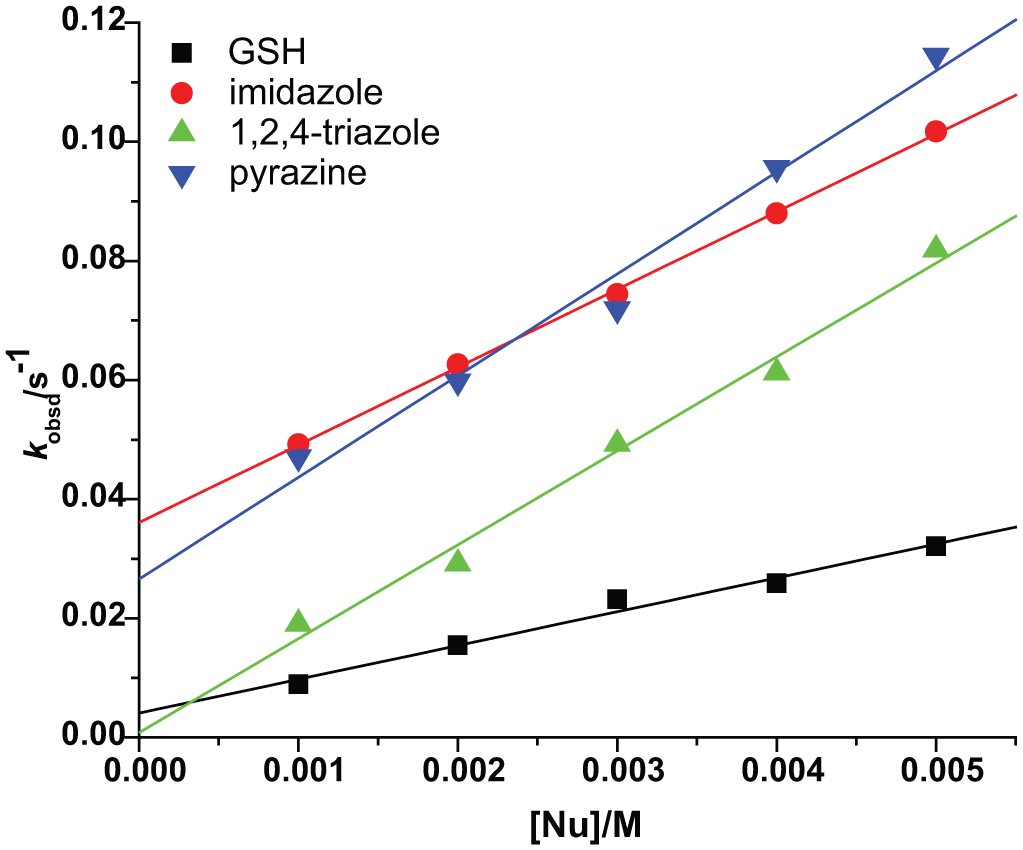
Pseudo first-order rate constants plotted as a function of nucleophile concentration for the second reactions of the [ZnCl2(terpy)] complex with imidazole, GSH, pyrazine and 1,2,4-triazole in aqueous medium at 295 K.
The observed pseudo first-order rate constants, kobs1 and kobs2, depend on the entering nucleophile (Nu) concentration as given in equations (1) and (2)
Linear fits passing through the origin for some reactions in this study indicate that possible parallel reactions are insignificant or absent, that is, k−1 and k−2 are negligible and equations (1) and (2) are simplified to kobs1 = k1[Nu] and kobs2 = k2[Nu]. Thus, in the present systems, direct nucleophilic substitution is the major observed reaction pathway under the selected conditions. The observed intercepts are ascribed to the back reaction with the chloride present in solution (Figures 3 and 4). The derived rate constants are summarized in Table 1.
Second-order rate constants for the first and second substitution reactions between [ZnCl2(terpy)] and imidazole, GSH, pyrazine, and 1,2,4-triazole in aqueous solution.

Analyzing the results, we concluded that the substitution processes happen in two different ways. Hence, when the ratio of kobs1/kobs2 is around 3–6, there are two parallel reaction paths. For reactions of this complex with some incoming nucleophiles, when the ratio is larger, e.g. 10–15 or even 20–40 (see Tables S1–S4 in the supplemental material), the first reaction step was over before the second one starts, which is typical for two consecutive reactions. The rates of the nucleophilic substitution reactions are controlled by the strong π-acceptor ability of the tridentate chelate 2,2′:6′,2′′-terpyridine. The proposed reaction pathways for the first substitution reactions are presented in Scheme 1.
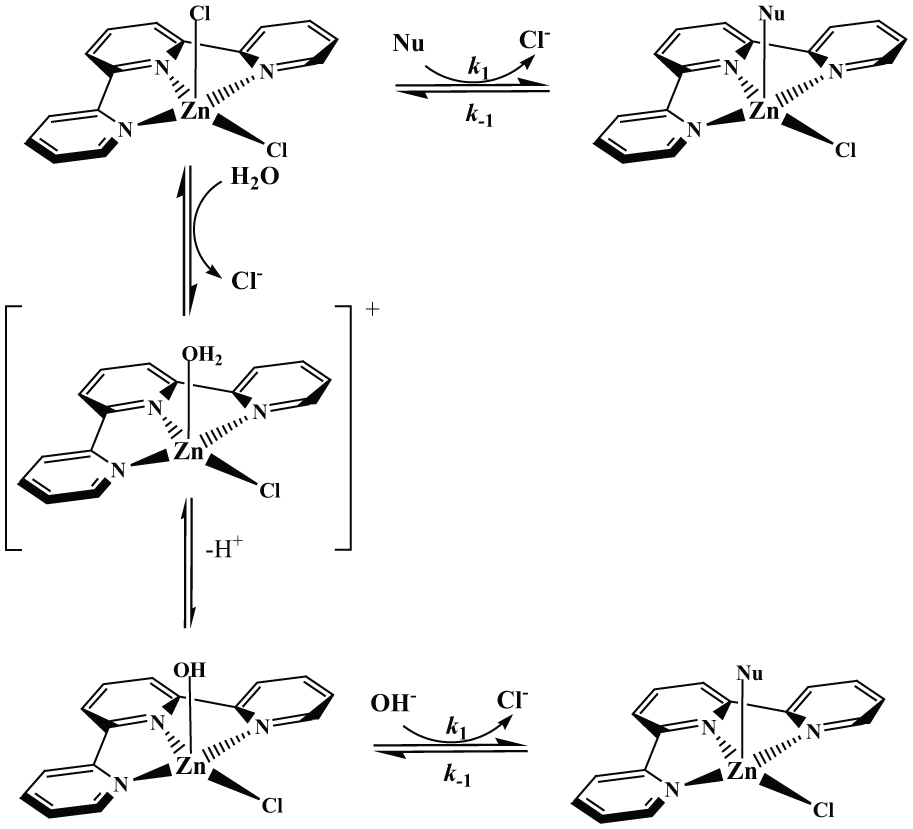
Proposed pathways for the first reactions between [ZnCl2(terpy)] complex and biologically relevant nucleophiles in aqueous solution.
Besides direct nucleophile attack, we propose that hydrolysis occurred in parallel followed by deprotonation of a coordinated water molecule. In the solution, we have two different species which are equally reactive for the substitution. The presence of various species is indicated by the initial decrease and then increase in absorbance in the rapid-scan spectra as is shown in Figure 2. We assume that the substitution of [ZnCl(OH)(terpy)] is the reason why we obtained lower second-order rate constants, k1, for 1,2,4-triazole and pyrazine (see Table 1). The coordinated hydroxide is a slightly poorer nucleophile than the free OH− ion, but it is better than water. It is known that the pKa of coordinated water in zinc complexes is controlled by the coordination number and by the total charge of the complex. Thus, it decreases with decreasing coordination number and with increasing positive charge, because a zinc ion, bearing in effect a more positive charge, will have greater attraction for the oxygen lone pair, thus lowering the pKa. 1 Charged ligands affect water pKa values more than do the number of ligands.
The second reaction step could be interpreted as substitution of the last chlorido ligand. The proposed reaction pathway for the second substitution reactions is given in Scheme 2. The values of second-order rate constants, k2 (Table 1), demonstrate that the second substitution step is slowed almost 20 times after substitution of the first chloride in the Zn(II) complex by imidazole and GSH.
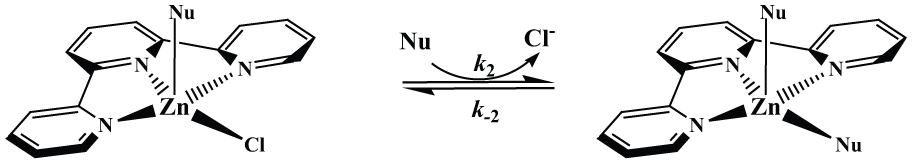
Proposed pathways for the second reactions between [ZnCl2(terpy)] complex and biologically relevant nucleophiles in aqueous solution.
Nucleophiles imidazole and triazoles are well known as inhibitors, which bind zinc enzymes with similar affinities over a large range of pH. Both the imidazolate anion and the neutral imidazole can bind to the hydrated forms of the enzyme with essentially the same affinity, and the reaction of imidazole with the Zn–OH species cannot be distinguished thermodynamically from the reaction of imidazolate with the hydrated forms. The concentration of the zinc hydroxo complex increased above pH 7.5 and became the predominant species at pH 8. 1
If we compare the results obtained in this investigation with our previous results (Table 2), we can conclude that the presence of chloride has a great influence on the reaction rates. The presence of chloride suppresses the hydrolysis of the [ZnCl2(terpy)] complex. Hence, in the presence of chloride, the substitution reactions are faster, which is associated with the absence of different reactive species and parallel reactions that involve them.
A different reaction mechanism of ligand substitution reactions with GSH has been obtained in the presence of 10 mM chloride in 0.005 M phosphate buffer at pH 7.38 (Table 2). The first step is a very fast pre-equilibrium formation of an intermediate (pseudo octahedral complex), followed by rearrangement to the final complex; whereas one chloride is substituted by GSH, the second substitution step was independent of glutathione concentration which indicates that a chelation process takes place. 6 Glutathione participates in various complexation processes with zinc as is well-known, because of its many donor atoms.4,10 In this research, both reaction steps depend on glutathione concentrations and pre-equilibrium formation has not been observed. The coordination of glutathione in the square-pyramidal [ZnCl2(terpy)] complex occurs via O-carboxylate.6,11 The observed second substitution step was dependent on glutathione concentration which indicated that a chelation process did not occur but, due to steric hindrance of the first coordinated glutathione, the second reaction step was slowed down (Table 2).
Based on the results obtained, we consider that the slower substitution reactions are the consequence of unsuppressed parallel hydrolysis. For pyrazine and 1,2,4-triazole, we propose that substitution via the [ZnCl(OH)(terpy)] complex has taken place (Scheme 1). The square-pyramidal structure of Zn2+ in biological systems prefers O-carboxylate, carbonyl, and N-imidazole donor bioligands. 11 With variable coordination geometries (tetrahedral, five-coordinate, and octahedral) that zinc(II) is able to adopt, its balance in donor site preference (−N, −O) may account for the order of reactivity of biologically relevant nucleophiles in the absence of excess of chloride. 12 Steric hindrance could also be a reason for the diverse reactivity of nucleophiles for the second reaction steps. 13
Conclusions
The ligand substitution reactions of the square-pyramidal [ZnCl2(terpy)] complex at pH 7.0, in the absence of excess of chloride, comprised two steps of consecutive displacement of chlorido ligands. In this research, both reaction steps depend on the concentration of entering ligands. The order of reactivity of the investigated nucleophiles for the first reaction is imidazole > glutathione > pyrazine > 1,2,4-triazole, while for the second reaction step it is pyrazine > 1,2,4-triazole > imidazole > glutathione. In the solution, due to hydrolysis, two different complex species are present that are equally important for the substitution in the first reaction step. Chelate formation and pre-equilibrium have not been observed for the substitution process between the [ZnCl2(terpy)] complex and glutathione in the absence of excess of chloride.
Supplemental Material
Supplementary_Material – Supplemental material for Study on the reactions between dichlorido[2,2′:6′,2″-terpyridine]zinc(II) and biologically relevant nucleophiles in aqueous solution
Supplemental material, Supplementary_Material for Study on the reactions between dichlorido [2,2′:6′,2″-terpyridine]zinc(II) and biologically relevant nucleophiles in aqueous solution by Enisa Selimović and Tanja Soldatović in Progress in Reaction Kinetics and Mechanism
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge financial support from the State University of Novi Pazar, Novi Pazar, Republic Serbia and T. Soldatović also gratefully acknowledges financial support from Ministry of Education, Science and Technological Development, Republic of Serbia (Project No. 172011).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



