Abstract
CuO- and Ag2O-decorated g-C3N4 photocatalysts were prepared by appropriate chemical modification of carbon nitride nanosheets produced from programmed pyrolysis of urea. After comprehensive characterization by powder X-ray diffraction, diffuse reflectance spectroscopy, Fourier transform–infrared spectroscopy, scanning electron microscopy, and Brunauer–Emmett–Teller analysis methods, their photocatalytic performances were examined for the removal of Acid Blue 92 azo dye, as a typical wastewater component from the textile industry. The X-ray diffraction patterns confirmed the presence of CuO and Ag2O nanoparticles on the surface of the sheets. In addition, diffuse reflectance spectra indicated a considerable reduction of the band gap of pure C3N4 by modification. The photoreaction was discussed mechanistically and the best operational parameters were found to achieve the highest efficiency under visible light.
Introduction
Semiconductor photocatalysis proposes feasible strategies for addressing the incoming global energy and environmental crises.1,2 However, most of the widely used photoactive materials have two main restrictions: one is the low solar energy conversion efficiency due to the large band gap, and the other is the high recombination ratio of photo-induced electron–hole pairs. For instance, as the most abundant photoactive material, titanium dioxide works only under ultraviolet (UV) illumination, which accounts for less than 5% of sunlight. As a result, the creation of efficient photocatalysts operating under visible light has been actively pursued as a promising procedure to solve future energy shortages by converting solar energy to electrical and chemical energies. 2 Also, reducing the band gap of the UV-responsive semiconductors by metallic and nonmetallic doping has successfully developed their optical sensitivity to the visible region,3–5 but this route usually reduces the stability of the photocatalysts. Hence, seeking highly active photocatalysts retains much interest worldwide. The use of new semiconductors (e.g. vanadates, orthophosphates, complex metal oxides, nitrides, sulfides, polymeric semiconductors, and oxynitrides) in catalysis has been studied intensively in the search for viable alternatives to TiO2-based photocatalysts;2,6,7 nevertheless, it remains a challenge to design and make use of new photoactive compounds that are stable, abundant, and which act efficiently under low energy irradiation sources. A polymeric semiconductor based on a defective graphitic carbon nitride (g-C3N4), the most stable allotrope of covalent nitride solids at ambient conditions, has been successfully developed as a new metal-free visible light-active photocatalyst for wastewater remediation, owing to its unique electronic band structure.8,9 g-C3N4 has the smallest direct band gap due to sp 2 hybridization of carbon and nitrogen atoms forming the π-conjugated graphitic planes. 10 To progress this sustainable environmentally-friendly compound, many efforts have been devoted to modify its physico-chemical properties and improve the photocatalytic performance such as design and preparation of nano-porous g-C3N4, nonmetal doping, its protonation by strong mineral acids, and constructing suitable heterojunction composites with other semiconductors (ZnO, Ag3PO4, AgBr, TiO2, Bi2WO6, etc.), as well as copolymerization.11–15 Moreover, the use of different precursors that contain C≡N core structures for the synthesis of C3N4-based materials has also been confirmed to be an efficient means of achieving new morphologies and increasing its photocatalytic proficiency.16–18 Introducing and eliminating defect sites has also been widely investigated in semiconductor-based photocatalysis because of the effectiveness of this strategy in broadening the light-responsive range of wide band-gap compounds.19,20 Recently, metallic species based on Ag and Cu have been shown to be promising choices for amplifying the absorption of visible light and suppressing electron–hole recombination on the photocatalyst surface.21–23 Herein, we report two types of heterojunctions comprising visible active semiconductors of CuO-C3N4 and Ag2O-C3N4. CuO and Ag2O nanoparticles were deposited on the surface of C3N4 nanosheets (NS) by a simple coprecipitation method. The photoactivity of the samples was evaluated by remediation of textile wastewaters under visible-light illumination. We also discuss the enhanced photocatalytic performance of the nanostructures based on the sorption capacity of the support and the synergetic effect of the functioned agents.
Experimental
Materials and preparation method
Urea was used to prepare g-C3N4 powder. Cu(NO3)2 and AgNO3 were used to load the Ag2O and CuO nanoparticles on the surface of the supports. Ethanol, tetramethylammonium hydroxide (TMAOH), isopropanol, HNO3, and all the other chemicals were purchased from Merck Co. and utilized without further purification. Acid Blue 92 (AB92) azo dye powder (as a typical textile wastewater component) was provided by Chaprang Co., Tabriz, Iran.
A crucible containing urea, well-covered by Al foil, was transferred to an oven, and the temperature was increased to 500°C at a rate of 10°C min−1. After 180 min, the crucible was cooled to room temperature, and the content was finely crushed, washed repeatedly with 0.1 M HNO3 solution, and kept at 60°C for 24 h. The product (50 mg) was poured into a beaker containing 100 mL of isopropanol, and the vessel was ultrasonicated for 1 hr. The suspension was then centrifuged, and the precipitate was dried at 60°C for 6 h. The product, C3N4 nanosheets was denoted as NS throughout.
AgNO3 solution was slowly added to a solution containing TMAOH and oleic acid, and the mixture was stirred for 1 h. A specific amount of the as-prepared NS powder was poured into the vessel and stirred magnetically for 1 h. The precipitate was filtered off and heated at 60°C for 24 h. The product was denoted as Ag2O-NS. CuO-NS was prepared by the same method, with copper nitrate used as the metallic source.
Photocatalytic tests
The experiments were conducted in a double-layer reactor made of Pyrex which was kept at room temperature. The visible light source was a 125 W OSRAM lamp which was surrounded by a reflective Al jacket to concentrate the beam on the reaction medium. The suspension comprising specific concentrations of the AB92 solution and catalyst powder was stirred magnetically for 15 min and then the lamp was turned on. The total concentration of AB92 (AB92 was provided by Chaprang Co., Tabriz, Iran, as a gift.) at different time intervals was measured using a UV spectrophotometer set at λmax of the azo dye (574 nm).
Results and discussion
Characterization of the samples
Powder X-ray diffraction analysis
The X-ray diffraction (XRD) patterns of the nanostructures are shown in Figure 1. The dominant diffraction peak at 2θ = 27.7°, which can be observed in all the samples, is assigned to the (002) diffraction planes of g-C3N4 and a small diffraction peak at approximately 13° is indexed to the in-plane of tris-triazine units (in-plane of tris-triazine units means in- plane structural packing motif of tristriazine units). 24 As can be seen, the latter peak was surprisingly intensified by decorating the surfaces with CuO nanoparticles. This sharp peak in CuO-NS corresponds to the CuO crystal planes. Moreover, the characteristic peak assigned to the cubic structure of Ag2O in the XRD pattern of the sample appeared around 2θ = 33° (JCPDS file no. 41-1104). From the patterns, the (002) diffraction peak of g-C3N4 is sharper in the NS compared to bulk g-C3N4.
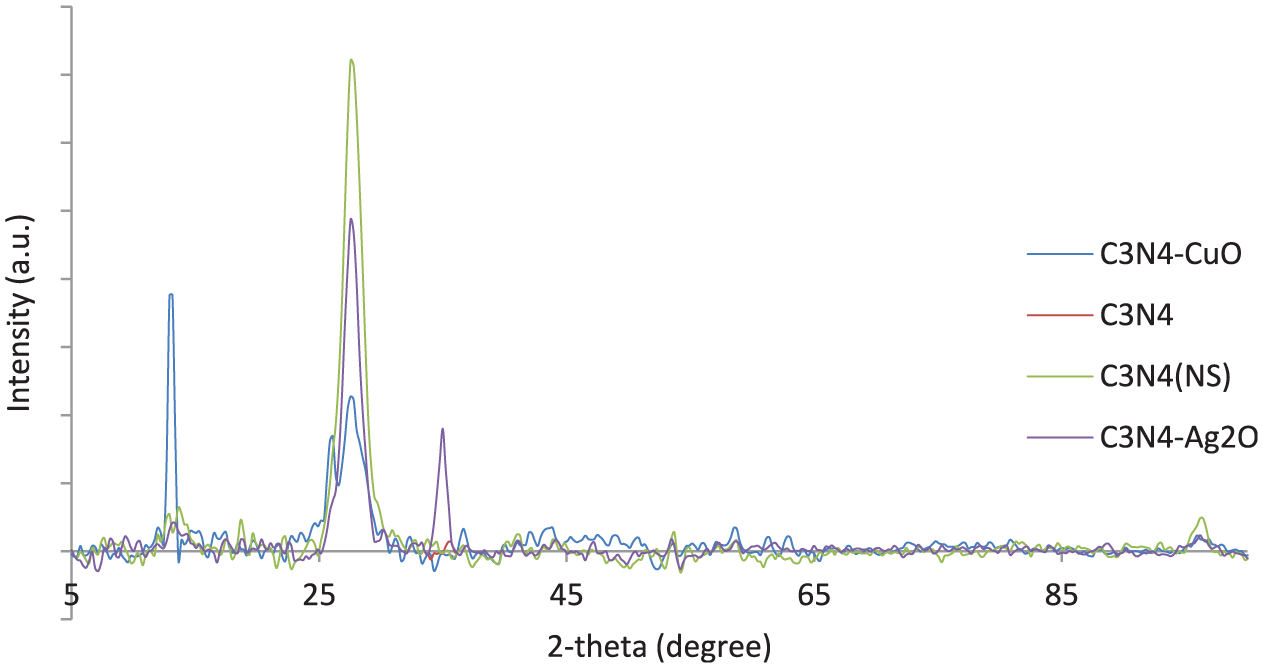
The XRD patterns of the photocatalysts.
Fourier transform–infrared spectroscopy
The Fourier transform–infrared (FTIR) spectra of the samples are shown in Figure 2. The broadened peak above 3400 cm−1 is due to water molecules adsorbed on the surface. The sharp peak at 808 cm−1 originates from the triazine ring system. 25 The peak at 1636 cm−1 is attributable to C═N stretching vibration modes. Moreover, the other bands appearing at 1240, 1320, and 1403 cm−1 could be related to the aromatic C–N stretching vibrations. 26
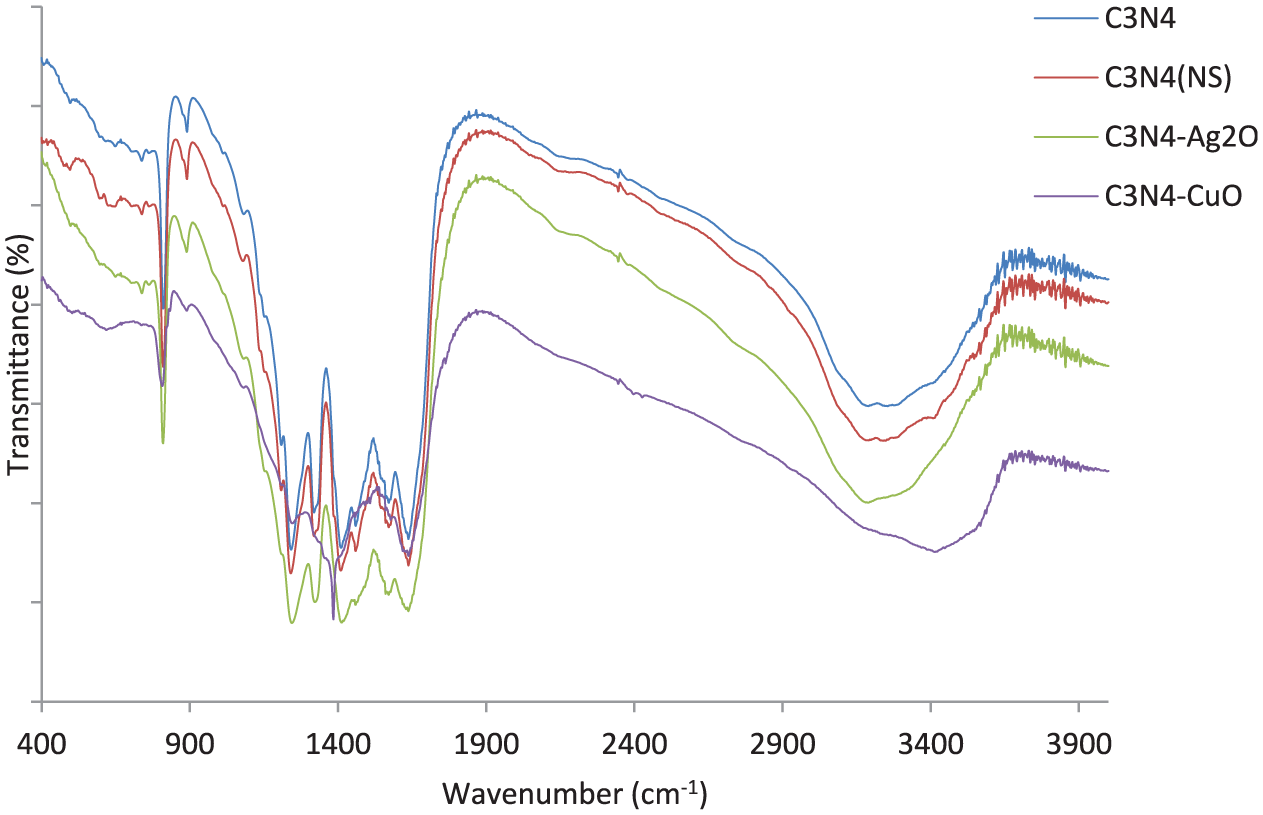
FTIR spectra of the samples.
Scanning electron microscopy micrographs
To characterize the morphology of the nanostructures, scanning electron microscopy (SEM) analysis was performed, and the images are shown in Figure 3. From the results, the micrographs of NS display a lamellar structure with the thicknesses of the sheets below 50 nm (a-1 and a-2). After the introduction of Ag2O or CuO nanoparticles, the presence of new species deposited on the surfaces leading to the formation of heterojunction photocatalysts is observable (b and c). The guest nanoparticles are clear in the SEM images of CuO-NS and Ag2O-NS. Forming this kind of heterojunction can readily increase the half-life of the photogenerated electron–hole pairs by their being transferred between p (CuO or Ag2O)-n type (C3N4) semiconductors which enhances the quantum yield effectively.

SEM images of the prepared photocatalysts: (a-1 and a-2) NS, (b) Ag2O-NS, and (c) CuO-NS.
Diffuse reflectance spectra
To find the effect of the oxide nanoparticles on the band gap of the C3N4 photocatalyst, diffuse reflectance spectroscopy (DRS) analysis was performed, and the results are presented in Figure 4. From the recorded spectra, the absorption step is the same for both bulk C3N4 and NS, but it shifts to higher wavelengths when the support is decorated with CuO or Ag2O nanoparticles. The results convinced us that the CuO junction more effectively influenced the band gap compared with the Ag2O one. According to the DRS results, the energy cut-off for the former case is located at a higher wavelength, which implies its lower band gap.
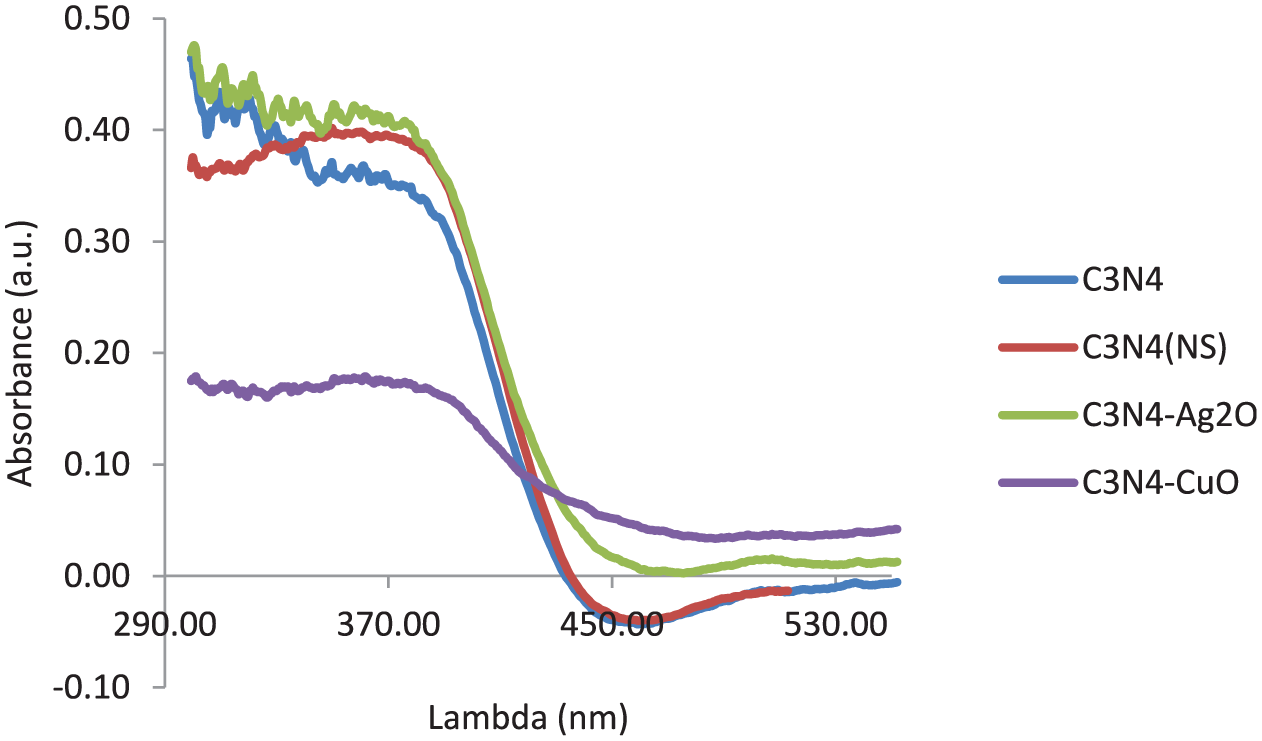
DRS of the samples.
Specific surface area (Brunauer–Emmett–Teller) analysis
As an effective tool for determining the specific surface area and porosity, Brunauer–Emmett–Teller (BET) analysis was conducted. Figure 5 shows a typical hysteresis loop related to the N2 adsorption–desorption curve for NS, which indicates that the support is expected to be porous. Moreover, the results revealed that deposition of the oxide nanoparticles on the surface decreases the specific surface area smoothly and also influences the morphology. The BET surface areas were 37.9, 38.4, and 31.2 m2 g−1 for g-C3N4, NS, and Ag2O-NS, respectively. Furthermore, the total pore volumes were evaluated to be 0.14, 0.16, and 0.17 cm3 g−1 for Ag2O-NS, NS, and g-C3N4, respectively. The pore size distributions of the prepared samples were determined by the Barrett–Joyner–Halenda method which indicated that the maximum distribution for the pores was around 1.6 nm, and there was no significant difference between the results obtained for the products.
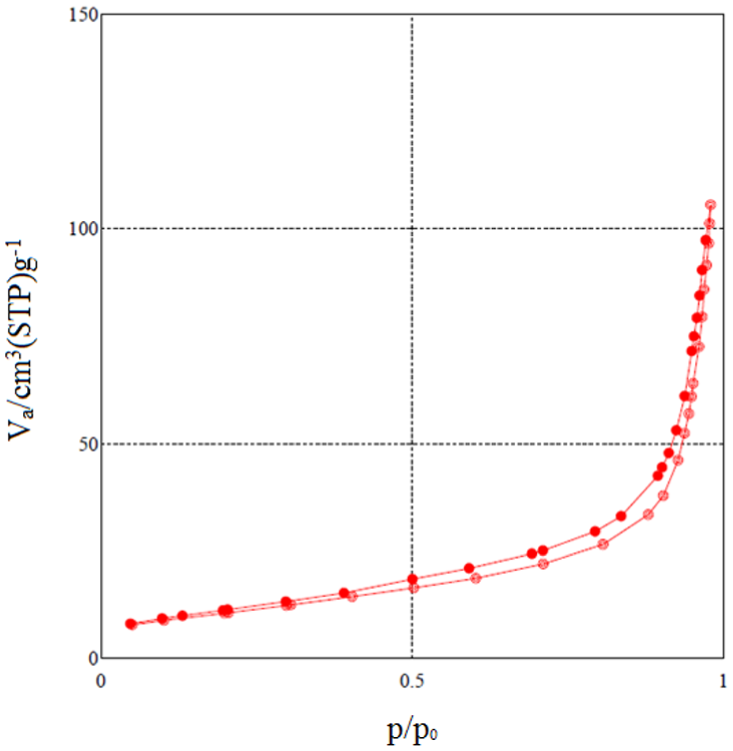
Nitrogen adsorption–desorption isotherm obtained for the pure NS sample.
Photocatalytic remediation of wastewater
The removal of AB92 from wastewater was studied over the prepared nanostructures under different operational conditions, and the results are shown in Figure 6.
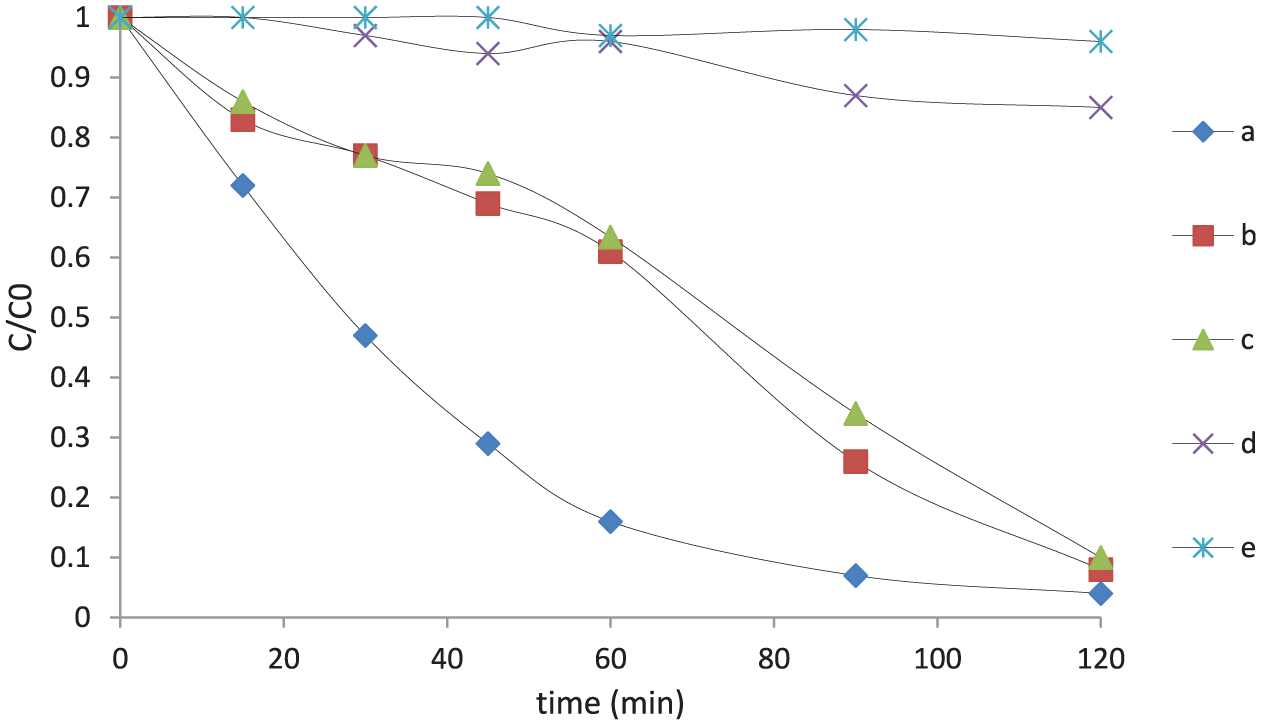
Results for AB92 removal: (a) photodegradation over CuO-NS, (b) photodegradation over Ag2O-NS, (c) photodegradation over NS, (d) photolysis experiment, and (e) dark test over NS.
From the results, the NS powder showed significant photocatalytic activity comparable to Ag2O-NS sample, which implicates the effect of the sonication process in isopropanol during its preparation. This step increases the availability of the active sites responsible for promoting the photocatalysis. The results of the BET analysis confirmed that there is no significant difference between the specific surface areas of these two samples, which convinced us that the higher activity of the NSs cannot be attributed to this parameter. Furthermore, CuO-NS, as the results of DRS analysis implied its lower band gap, was expected to be more active than Ag2O-NS. From Figure 6, the removal efficiency became maximum over the former case in a considerably shorter reaction time compared to the other photocatalysts. Ag2O and CuO are both
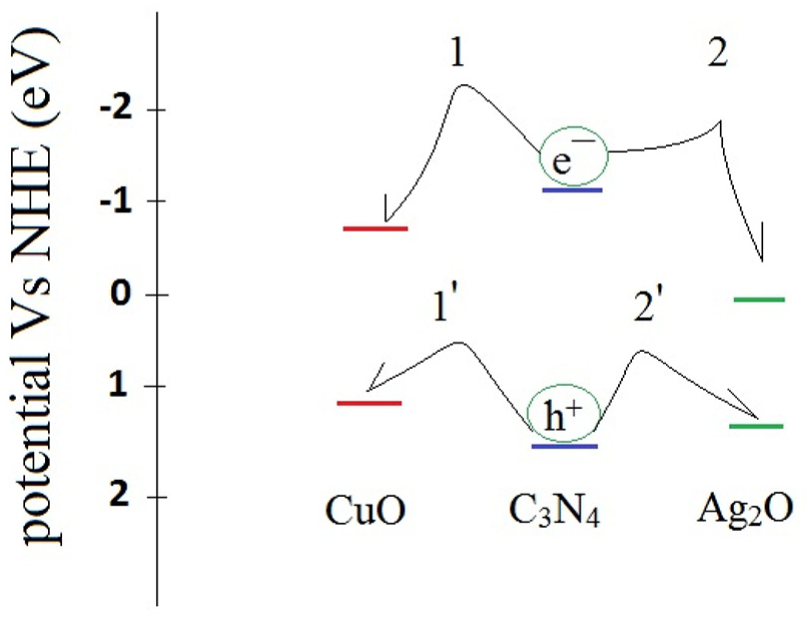
Charge transfer process between the Ag2O, C3N4, and CuO junctions during illumination.
We also found the optimum values for the experimental parameters, including catalyst dosage, AB92 concentration, and pH. As shown in Figure 8(a), increasing the NS powder dosage from 5 to 20 mg enhances the photodegradation efficiency considerably while there is no significant gain in changing from 20 to 50 mg.
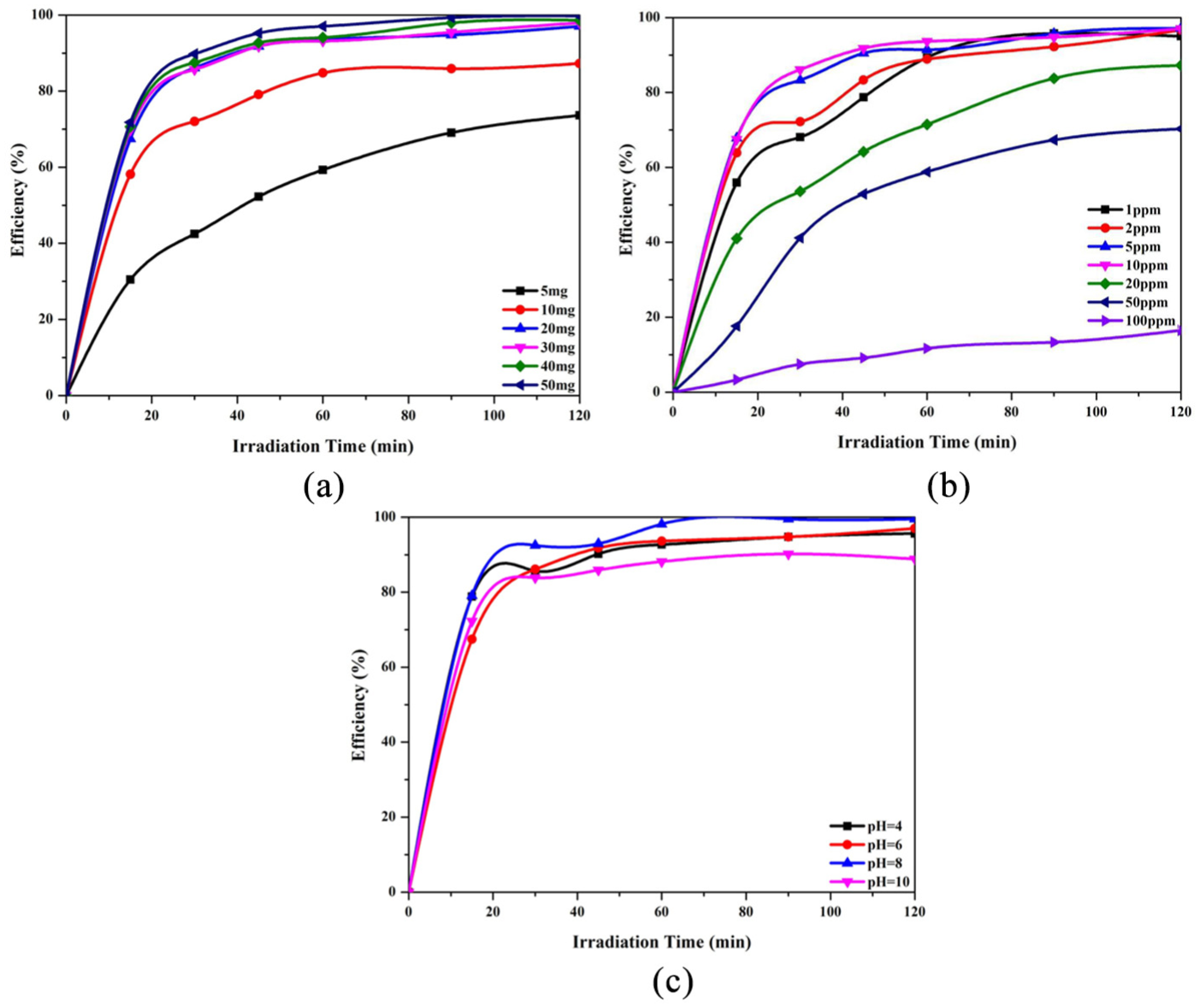
Effect of (a) catalyst dosage, (b) dye concentration, and (c) pH on the photodegradation efficiency of AB92 over NS.
The availability of active sites is strongly affected by increasing the dosage of catalyst particles in the medium in the range of 5–20 mg, as clearly represented in the results obtained. However, we observed that the reaction medium became opaque on adding the catalyst powder in the range of 20–50 mg, thereby reducing the penetration of the light photons to the photocatalyst surface, which controls the photoreaction rate and efficiency. From Figure 8(b), the optimum value for AB92 concentration was evaluated to be 10 ppm. At higher concentrations, the AB92 molecules induce an inner-filter effect. The acidity of the medium was also optimized by following the reaction efficiency at pH values of 4, 6, 8, and 10 (Figure 8(c)). According to the results, the maximum rate and efficiency were achieved at pH 8. Under mild basic conditions, the hydroxyl anions react with photogenerated positive holes and produce OH•, which are the most abundant destroyer radicals in an aquatic environment. Meanwhile, higher concentrations of OH¯, inasmuch as AB92 is a negative azo dye (including sulfonate groups in its chemical structure), interfere with the adsorption of the azo dye molecules on the catalyst surface and reduce their removal efficiency. To reach a clearer understanding of the structural effects, we treated AB92 solely in basic (pH of 8 and 11) and acidic (pH of 2 and 4) solutions. From Figure 9, AB92 showed significant red and blue shifts under basic and acidic conditions, respectively. In addition to having red shift behavior, we observed a new characteristic peak between 350 and 400 nm, which implies the development of a chemical reaction in the basic medium, while under acidic conditions we just had a decrease in the absorption at λmax.
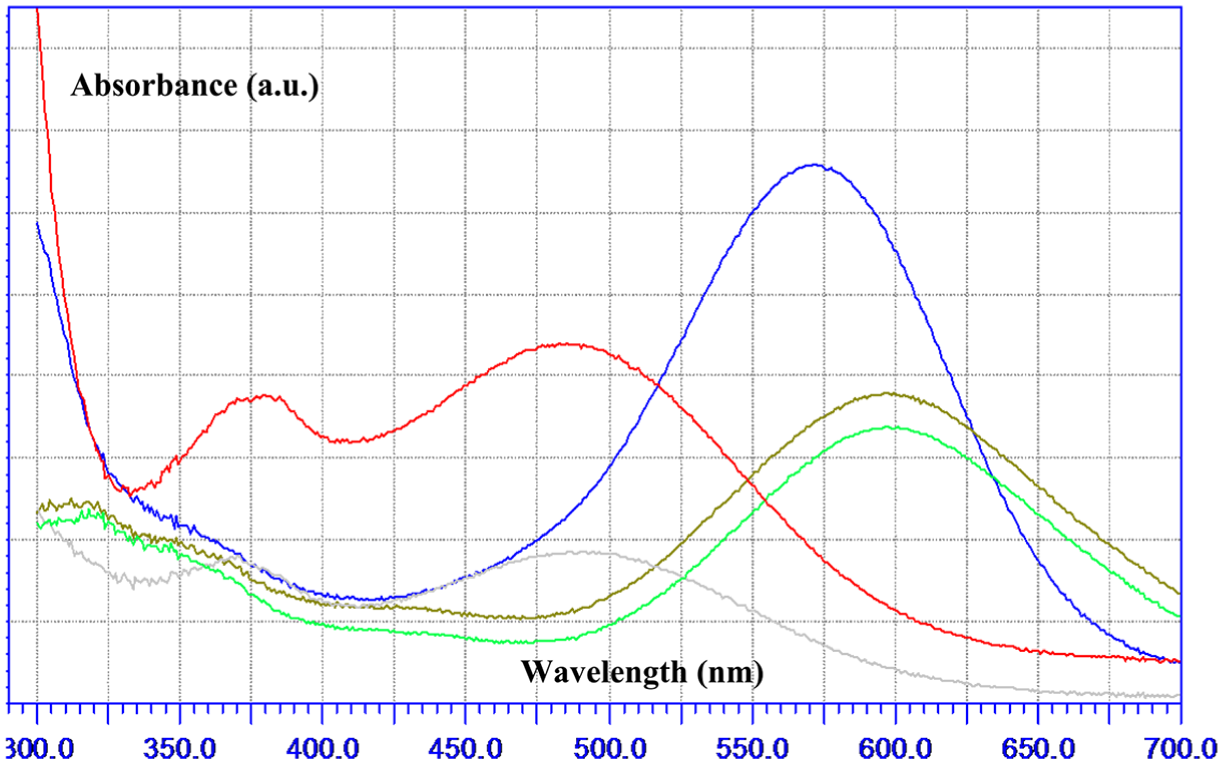
The UV spectra of AB92 solutions at different acidities of the medium (blue: neutral, red: pH = 11, gray: pH = 2, green: pH = 4, light gray: pH = 8).
The most likely event in an acidic medium seems to be protonating the sulfonate groups, which is without effect on the azo chromophore. On the other hand, a high concentration of OH¯ could deprotonate two distinct positions of the structure: OH in the naphthalene ring and NH of the aniline group; the former case is preferred due to the higher electronegativity of oxygen. Thus, O¯ can attach to the N═N group and produce a new species, as schematically represented in Figure 10.
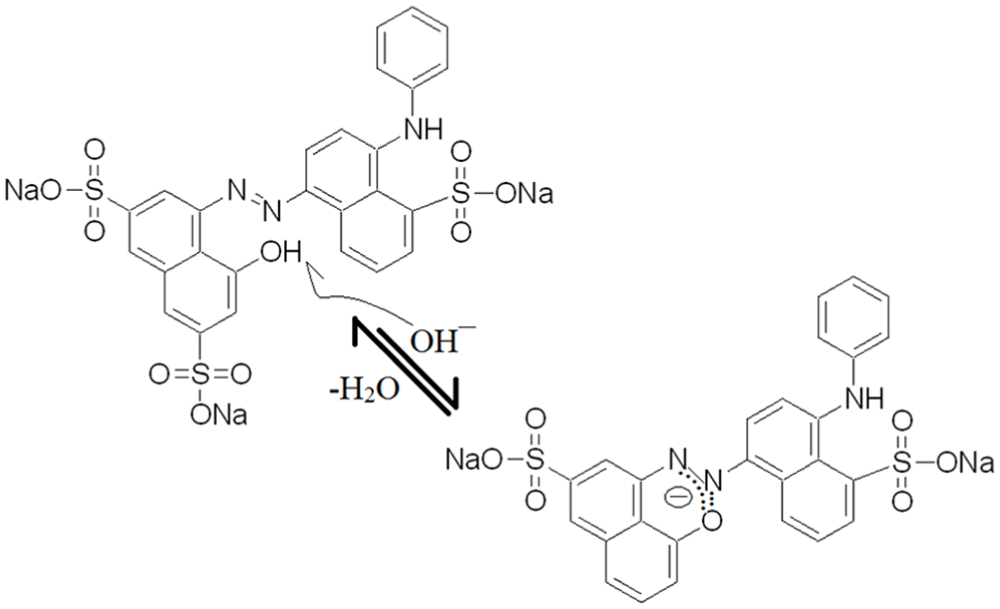
Effect of strong basic conditions on the structure of AB92.
This kind of interaction can weaken the strength of the azo bond and makes it much more vulnerable to attack. Accordingly, the efficiency of photocatalytic removal reached its highest point at pH 8. 29 Finally, the degradation of a real wastewater provided by Chaprang Co. was studied over the best photocatalyst and under optimized conditions. This sample contained different organic waste compounds from textile activities rather than AB92 and could be an appropriate choice for evaluating the promising photocatalytic capacity of the prepared nanostructures. The results for this experiment are shown in Figure 11, which indicates an effective decrease at all wavelengths after 2 h of irradiation time.
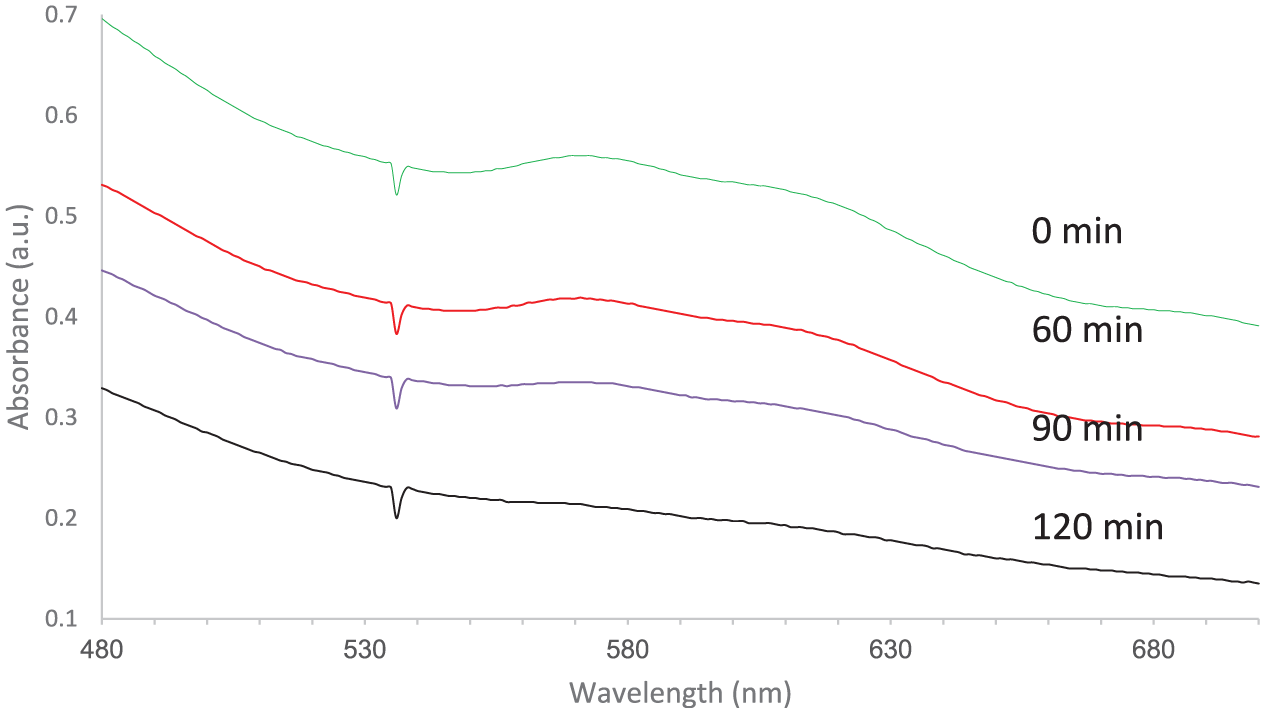
Photocatalytic treatment of the real waste sample under the optimized operational conditions.
Conclusion
CuO-C3N4 and Ag2O-C3N4 nanoframeworks were prepared and thoroughly characterized by different analysis methods including XRD, BET, FTIR, DRS, and SEM. The products showed extraordinary photocatalytic performance for the removal of AB92 solution under visible light. The hysteresis loop of the N2 adsorption–desorption isotherm indicated that the NS sample is rather porous. CuO proved a better option than Ag2O to reduce the band gap of g-C3N4 and improve its photocatalytic properties. 20 mg, 8, and 10 ppm were determined as the optimum values for the catalyst dosage, pH, and AB92 concentration, respectively. We found that AB92 behaves differently on changing pH, and this might influence the photocatalytic routes for its degradation. Its solution showed remarkable red and blue shifts under basic and acidic conditions, respectively. We utilized this observation to propose a suitable mechanism for achieving the highest efficiency at pH 8. Finally, the ability of the products to degrade a real waste water sample was investigated under the optimized conditions, and the results indicated that the absorption at all wavelengths decreased considerably after 2 h of treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
