Abstract
A large number of heterogeneous catalysts were tested in the aldol condensation of heptanal with cyclopentanone. There are many possible products from this aldol condensation, among which the most desired is 2-heptylidenecyclopentanone. As heterogeneous catalysts, the following were tested: caesium modified zeolites, functionalized MCM-41, and single (MgO, CaO, ZnO) and double (Mo–Si, Mg–Al, Zn–Al) metal oxides. The activity of modified zeolites and functionalized zeolites in this aldol condensation was low, whereas single and double metal oxides possessed high activity. The highest selectivity to the desired 2-heptylidenecyclopentanone (52%) was obtained using magnesium oxide as catalyst.
Introduction
Aldol condensation of heptanal or pentanal with cyclopentanone giving 2-alkylidenecyclopentan-1-one is a way to produce the precursors of different fragrances (of, for example, lactone type). Several types of catalyst have already been tested in this aldol condensation. Among the homogeneous ones, for example, different amines (proline), sodium hydroxide, titanium tetra-
Experimental
Catalyst preparation
Modification of MCM-41 by silanol modifiers was performed by the post-grafting method
8
using (3-mercaptopropyl)trimethoxysilane (Sigma Aldrich, 95%), (3-aminopropyl)triethoxysilane (Sigma Aldrich, >98%),
Modification of zeolite NaY (Zeolyst) by caesium or alkylammonium ions was performed by ion-exchange (repeated four times) involving 1 M methanolic caesium acetate (Sigma Aldrich, >99%) or 1 M aqueous solution of methylamine or dimethylamine hydrochloride (both Aldrich, >99%). 16 Modification of zeolites by caesium ions was also performed using the impregnation method, again using caesium acetate. 14
Molybdenum oxide-modified silicas were prepared by the impregnation (10 wt%) and sol–gel (20 wt%) methods
17
using tetraethylorthosilicate (Sigma Aldrich, 98%), ammonium heptamolybdate (Chemapol, pour analysi, p.a.), or fumed silica (Sigma Aldrich, p.a. 0.2–0.3 µm). Commercial materials were also used as catalysts: magnesium oxide (Penta), zinc oxide (Fluka), and calcium oxide (Penta). Mixed Mg–Al (Zn–Al) oxides were prepared by a co-precipitation method at a constant pH value (pH = 10,
Aldol condensation
A round-bottomed flask equipped with a condenser was filled with catalyst (20 wt% to mass of heptanal), cyclopentanone and heptanal (2 mL). A mixture of reactants was preheated to the desired temperature and at a specified interval the samples (0.1 mL) were taken. Samples were centrifuged, mixed with ethanol, and analyzed using an Agilent 6890 chromatograph fitted with a nonpolar HP-5 column (30 m length, 0.32 i.d., and 0.25 μm stationary film). Mixed oxides (Mg–Al, Zn–Al, Mo–Si, Wo–Si) were pretreated before reaction by calcination at 450°C (air, 17 h).
Results and discussion
Catalyst preparation
MCM-41 modified by different groups by silanol modifiers have already been characterized in our previous work 8 and mixed oxides in another of our studies. 11 Silicate materials modified by molybdenum have also already been characterized in our earlier work. 16
Modified zeolites
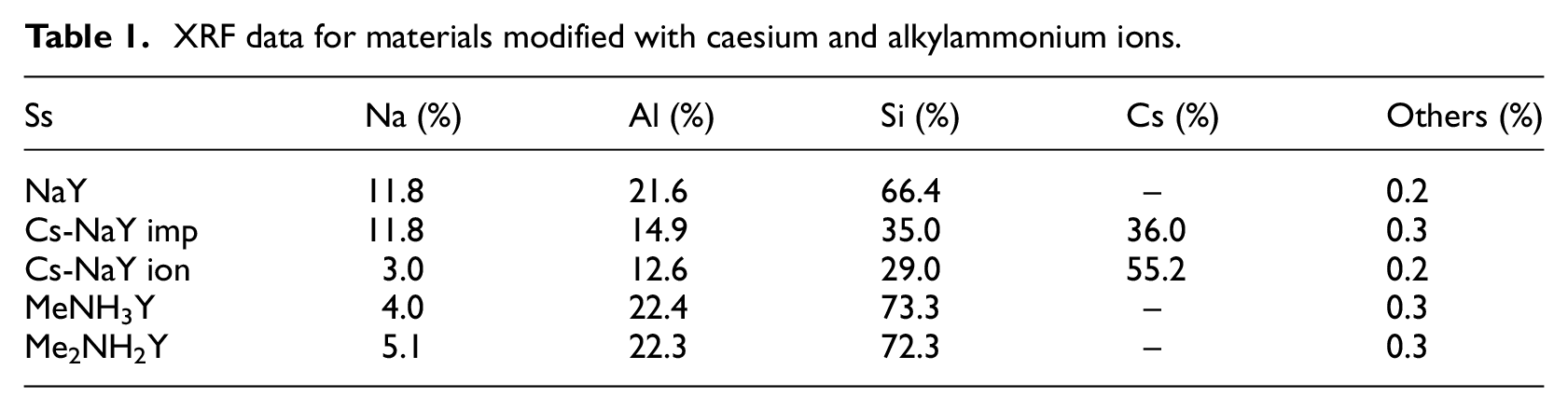
The composition of the catalysts was evaluated by XRF analysis (Table 1). The same amount of sodium before and after impregnation was monitored by XRF analysis in the case of impregnation of catalysts. This indicates that during impregnation, no ion-exchange took place and caesium ions were deposited only on the surface of the catalyst. On the other hand, the sodium content decreased after modification with alkylammonium ions and caesium ions using the ion-exchange technique, which indicates that sodium ions were successfully exchanged with alkylammonium or caesium ions (Table 1).
XRF data for materials modified with caesium and alkylammonium ions.
Aldol condensation
Aldol condensation of heptanal and cyclopentanone can give rise to a large number of possible products. In the reaction scheme in Figure 1, we show the products which were detected and identified within this work. The product of mixed aldol condensation, 2-heptylidenecyclopentanone (
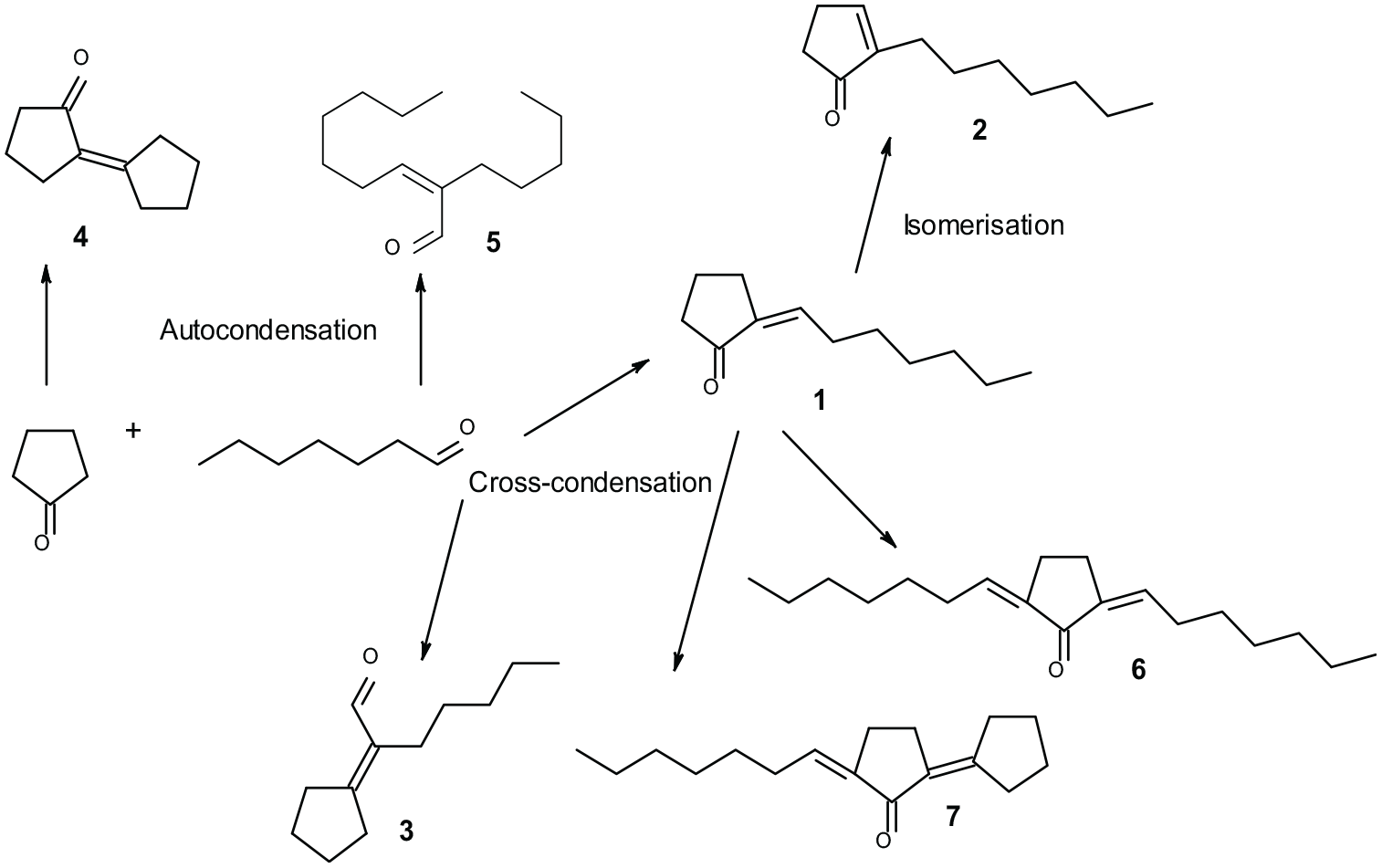
Reaction scheme of aldol condensation of heptanal and cyclopentanone—products identified in the reaction mixture (
Influence of catalyst type
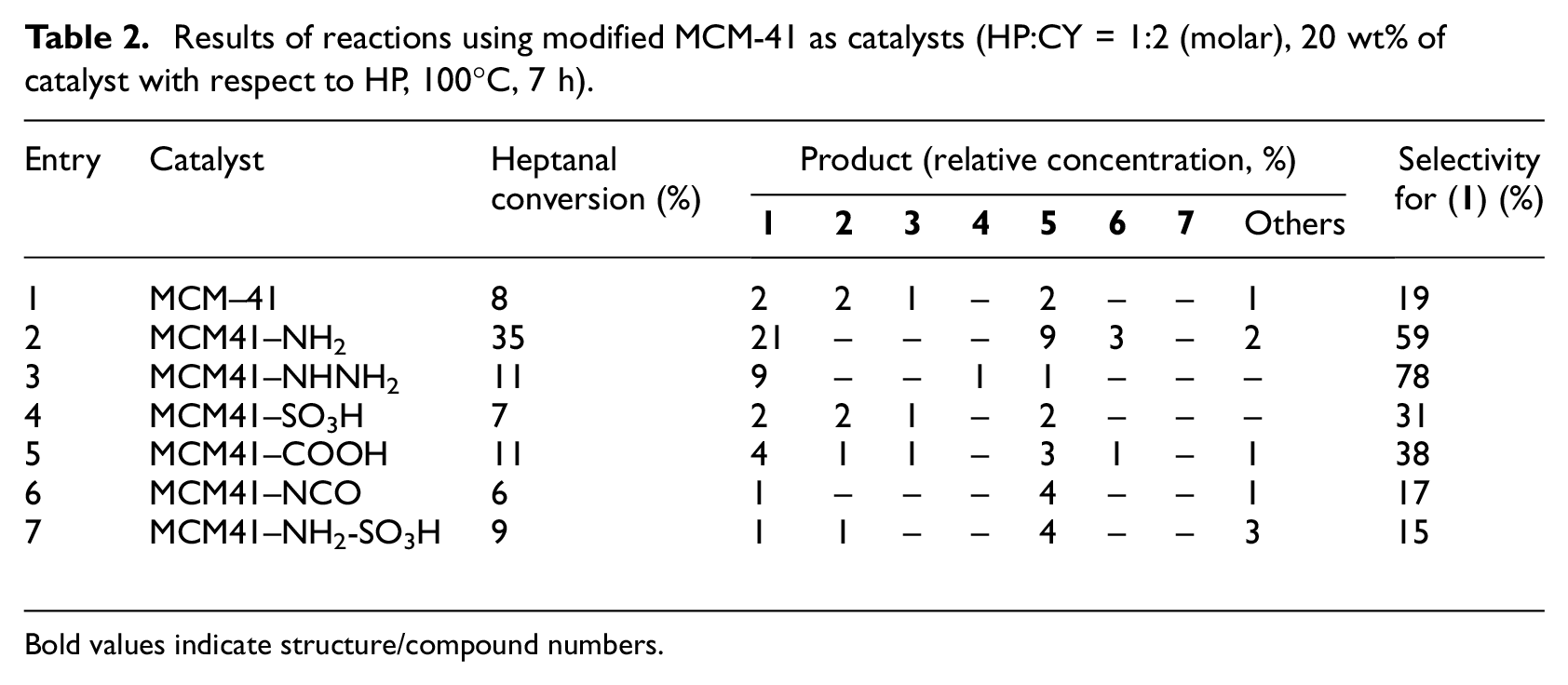
The first catalysts tested in the aldol condensation of heptanal with cyclopentanone were modified silicate materials (Table 2). These materials were chosen because they were already reported to be active in aldol condensation of 4-isopropylbenzaldehyde with propanal or in the reaction between benzaldehyde and acetic anhydride.7,8
Results of reactions using modified MCM-41 as catalysts (HP:CY = 1:2 (molar), 20 wt% of catalyst with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers.
Except for the material modified with 3-aminopropyl groups, the activity of these materials in aldol condensation of heptanal with cyclopentanone was low. The highest level of heptanal conversion was obtained when using material modified with 3-aminopropyl groups (Table 2, entry 2, 35%). In this case mainly the product of mixed aldol condensation (
The use of materials modified with acid groups, that is, 3-sulfoxypropyl and 3-carboxypropyl (Table 2, entries 4 and 5) resulted in a broader spectrum of reaction products— mixed aldol product (
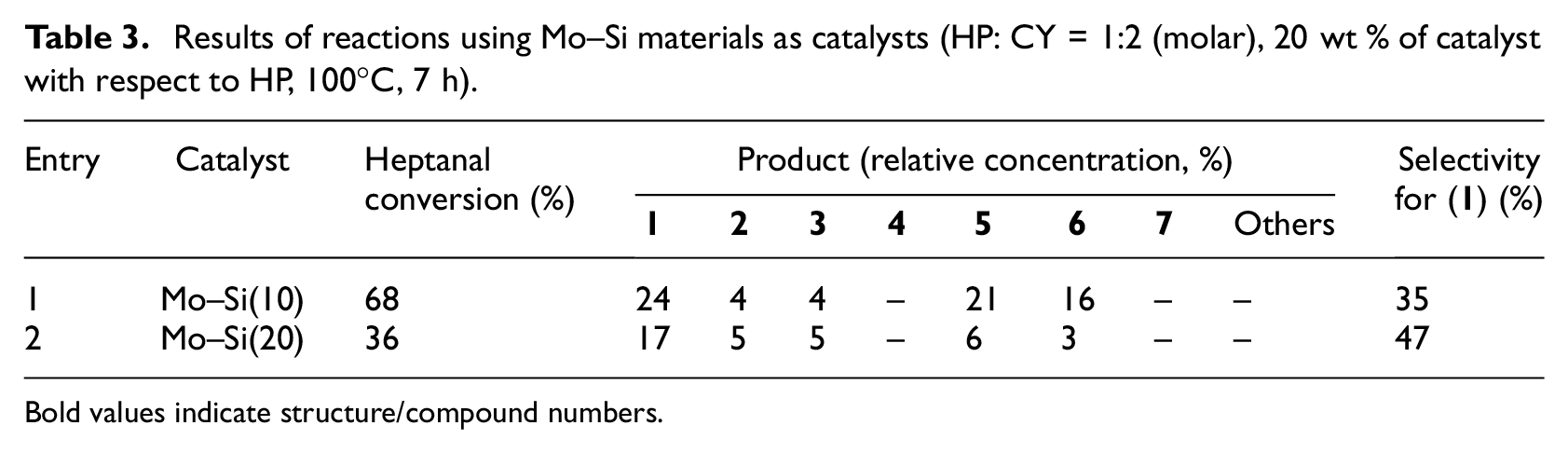
The second group of materials was silicate materials modified with molybdenum(VI) oxide. These materials already showed high activity in the Prins reaction between 3-methylbut-3-en-1-ol and different aldehydes. 16 Materials modified with 10 wt% (impregnated) and 20 wt% of MoO3 (sol–gel synthesized) were chosen to perform aldol condensation of heptanal with cyclopentanone (Table 3).
Results of reactions using Mo–Si materials as catalysts (HP: CY = 1:2 (molar), 20 wt % of catalyst with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers.
Materials modified with different amounts of MoO3 were quite active in this aldol condensation. When using material modified with 10 wt% MoO3, similar amounts of mixed aldol product (
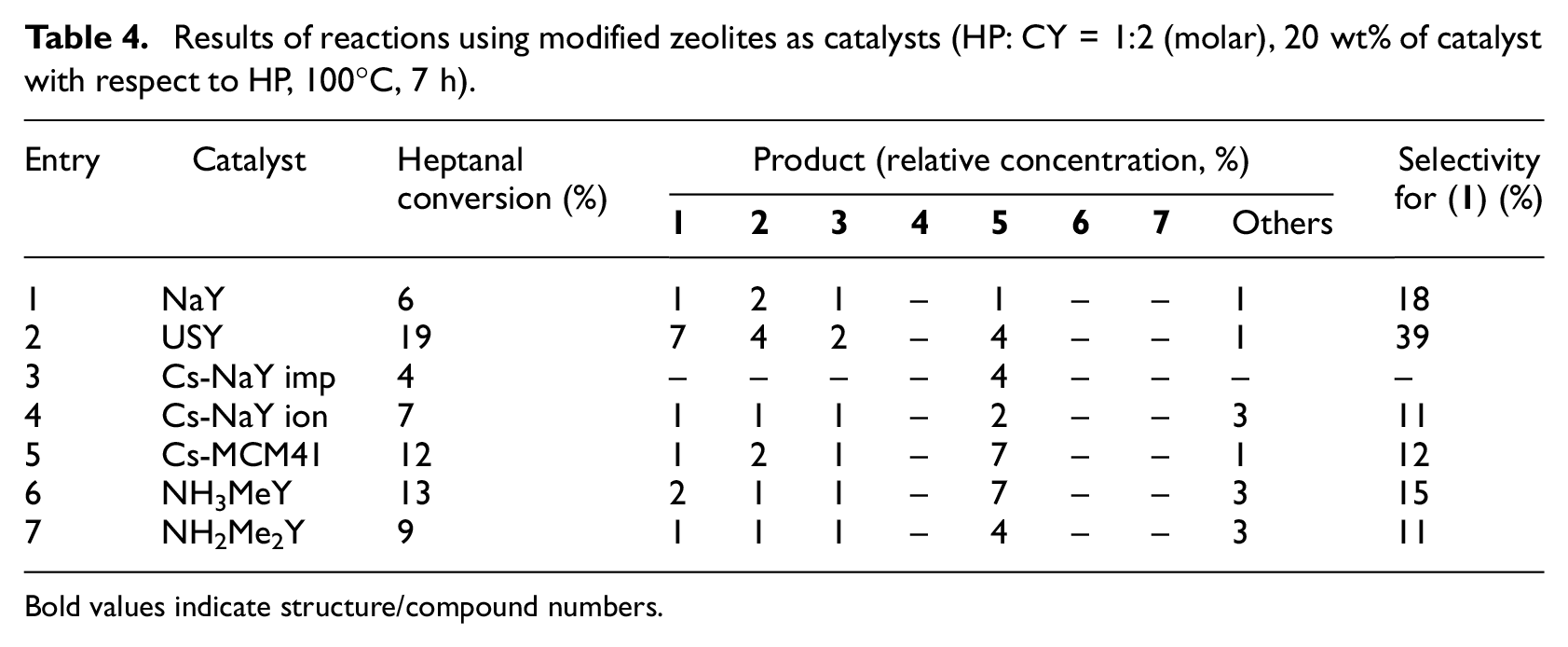
Modified zeolites were chosen as another group of heterogeneous catalysts (Table 4), because they are well known for their catalytic activity in aldol condensations.
Results of reactions using modified zeolites as catalysts (HP: CY = 1:2 (molar), 20 wt% of catalyst with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers.
First, the activity of unmodified zeolites NaY and USY was evaluated (Table 4, entries 1 and 2). Activity of these materials was low, comparable with modified MCM-41’s (except MCM-41 modified with 3-aminopropyl groups). Heptanal conversion was very low when using all types of modified zeolites. Modification of material with caesium ions by impregnation caused a change in selectivity—when using this catalyst only heptanal autocondensation product occurred. Modification with caesium ions by impregnation was also performed using MCM-41. In this case, after the modification, a slightly enhanced reaction rate was observed together with a change in selectivity (also heptanal autocondensation preferred, Table 4 entry 5 and Table 2 entry 1). The activity of material modified with methylamino ions was comparable with that of Cs-MCM41 and the activity of material modified with dimethylamino ions with that of Cs-NaY imp. Overall, zeolites as catalysts did not catalyze the formation of disubstituted products (
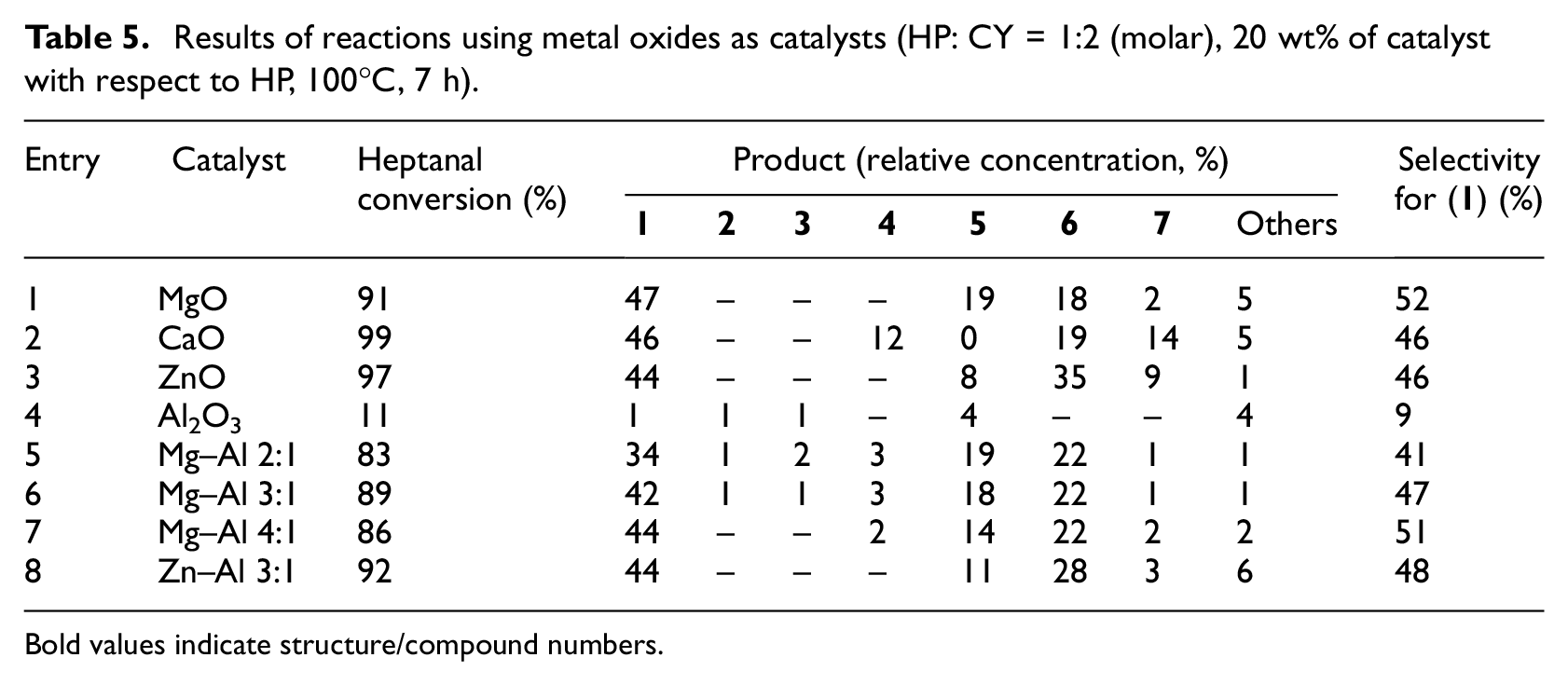
Alkaline earth metal oxides are generally known as heterogeneous basic catalysts, together with mixed Mg–Al oxides.11,12 In this work, we have chosen several metal oxides and mixed metal oxides to catalyze aldol condensation of heptanal with cyclopentanone (Table 5).
Results of reactions using metal oxides as catalysts (HP: CY = 1:2 (molar), 20 wt% of catalyst with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers.
Alkaline earth metal oxides were highly active catalysts (Table 5, entries 1 and 2), together with ZnO; use of all of them resulted in conversions higher than 90% (7 h). In all of the cases, the largest amount of mixed aldol product (
Of all the compared catalysts, the highest conversions were obtained using metal oxides, such as MgO. This was probably caused by the fact that most of the other catalysts used (except Mg–Al mixed oxides) contain predominantly acid active sites rather than predominantly basic active sites. Also, in the case of MgO, the catalytic reaction can be partly heterogeneous and partly homogeneous, because some of the MgO can be dissolved to Mg(OH)2 by water originating in the aldol condensation.
Influence of solvent, reactant molar ratio and temperature
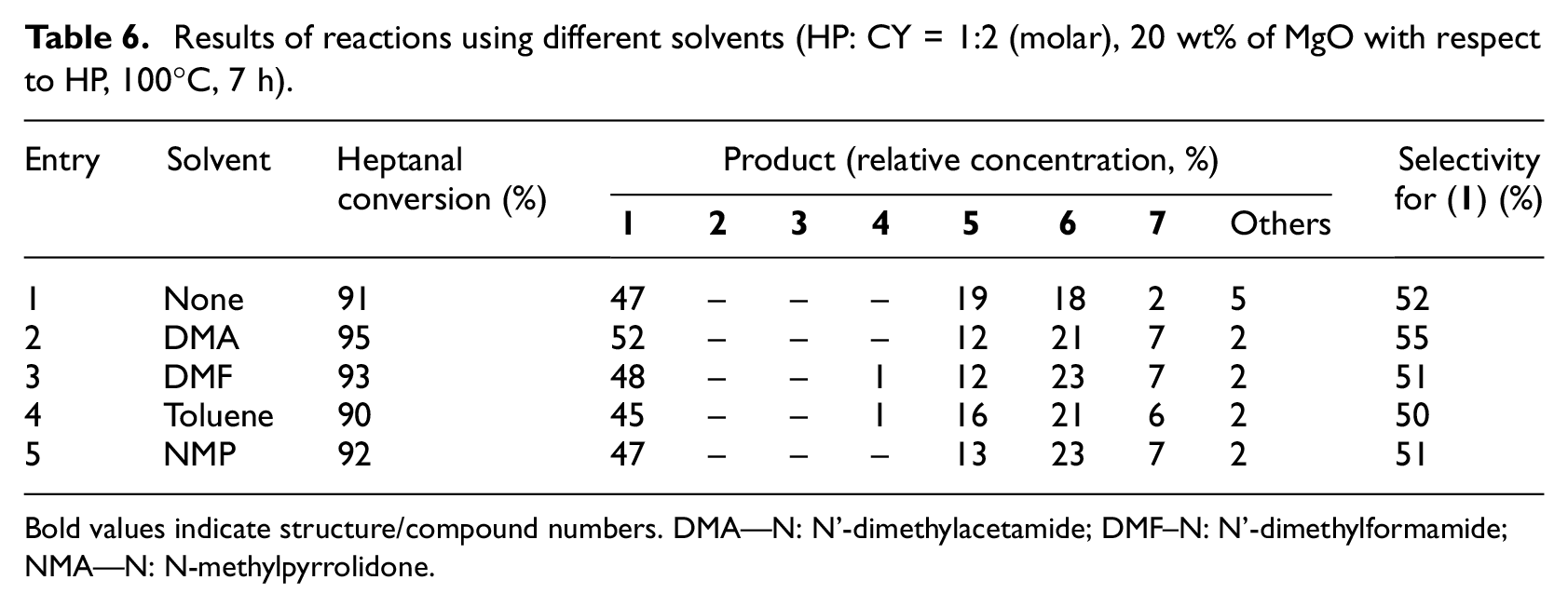
Influence of solvent on reaction course was also evaluated (Table 6). For this measurement the catalyst MgO was chosen, because it possesses one of the highest activities in this aldol condensation. The reaction rate was not significantly influenced by using different solvents. When comparing the reaction result in solvent-free or solvent-based aldol condensation, it was found that the use of solvents decreased the formation of the heptanal autocondensation product, but on the other hand it increased the formation of disubstituted products (
Results of reactions using different solvents (HP: CY = 1:2 (molar), 20 wt% of MgO with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers. DMA—N: N’-dimethylacetamide; DMF–N: N’-dimethylformamide; NMA—N: N-methylpyrrolidone.
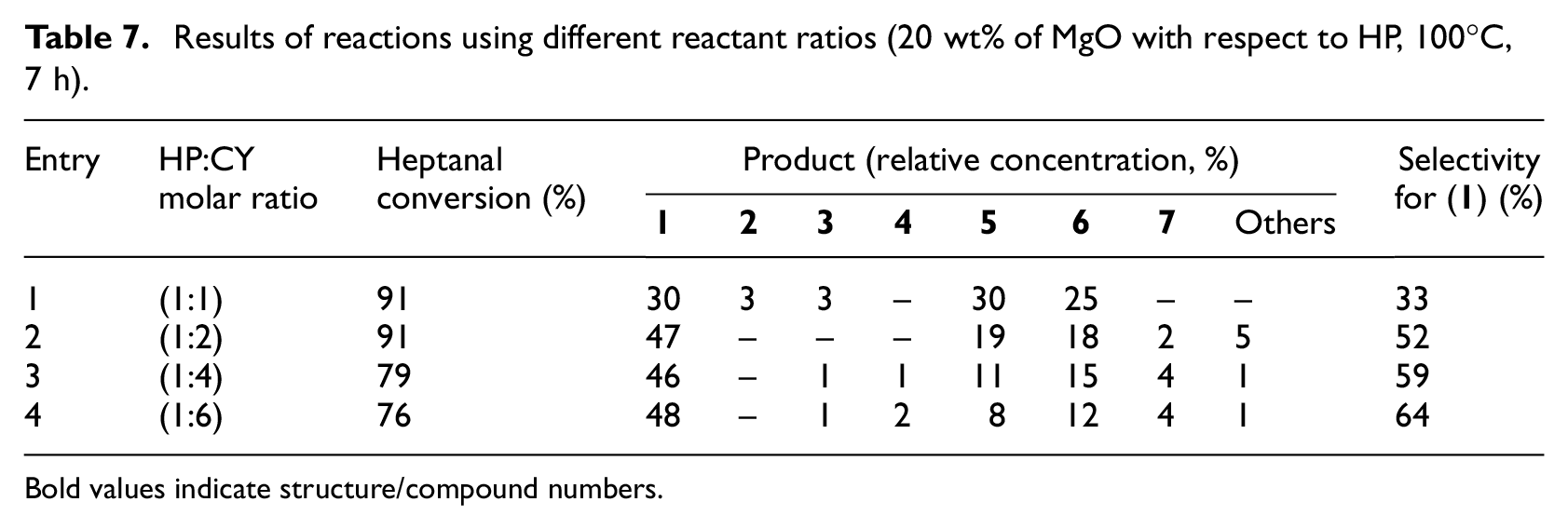
The influence of reactant molar ratio on the reaction course was monitored in aldol condensation catalyzed by MgO (Table 7). Decrease in the reaction rate was observed with excess of cyclopentanone. This decrease was probably caused by the dilution of the reaction mixture. Use of equimolar amounts of heptanal and cyclopentanone resulted in almost the same amounts of mixed aldol product (
Results of reactions using different reactant ratios (20 wt% of MgO with respect to HP, 100°C, 7 h).
Bold values indicate structure/compound numbers.
The influence of temperature on the reaction course was monitored using MgO as a catalyst (Table 8). Increase in the reaction rate with increasing reaction temperature was observed as predicted. At 60°C reaction almost did not take place (heptanal conversion 6%, 7 h). At 80°C, heptanal conversion of 51% was reached after 7 h—selectivity for mixed aldol product (
Results of reactions using different reactant temperatures (HP: CY = 1:2 (molar), 20 wt% of MgO with respect to HP, 7 h; HP-heptanal, CY-cyclopentanone).
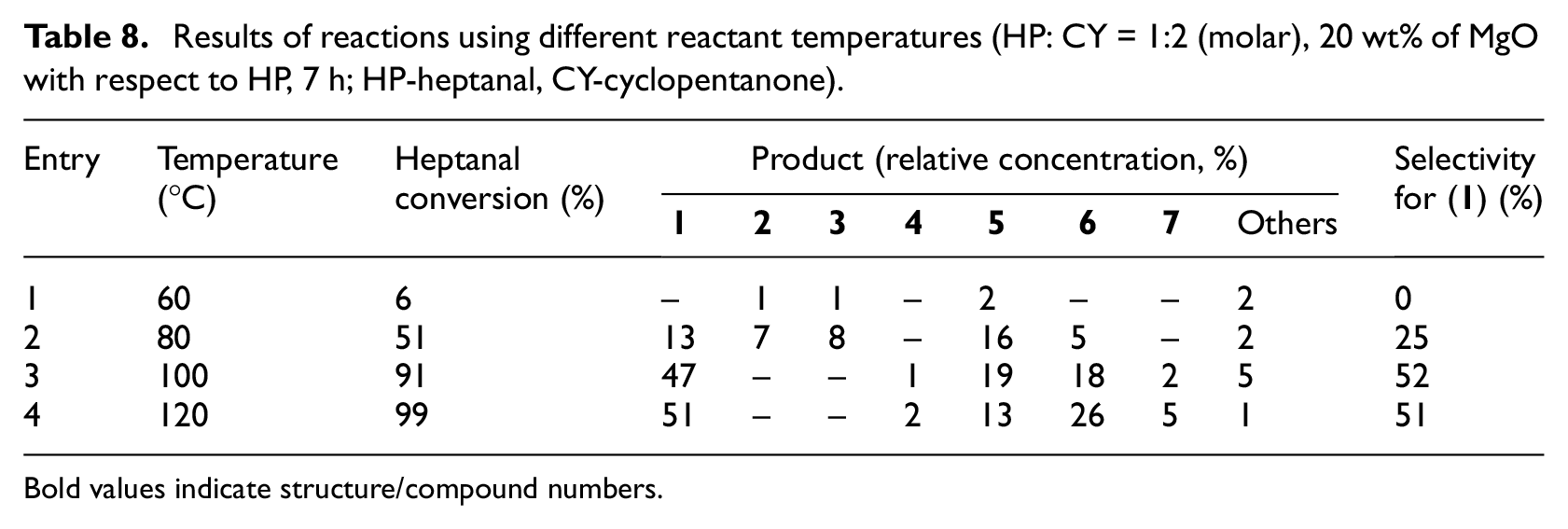
Bold values indicate structure/compound numbers.
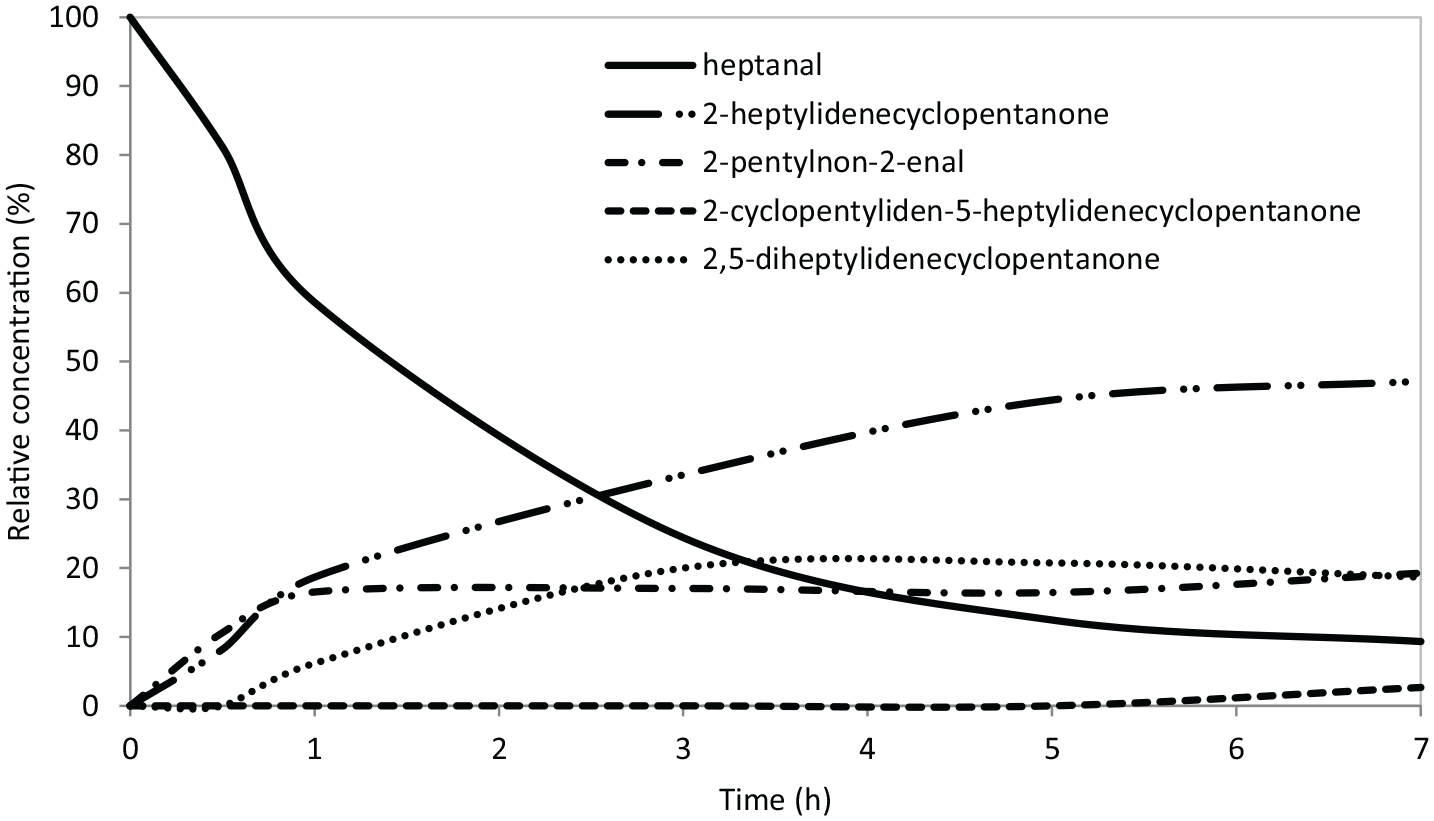
Reaction course (HP:CY = 1:2 (molar), 20 wt% of MgO with respect to HP, 100°C, 7 h; HP - heptanal, CY - cyclopentanone).
Conclusion
Several types of MCM-41 modified with different silanol modifiers and caesium and alkylammonium NaY-modified zeolites were prepared and characterized. The activity of these materials in aldol condensation of cyclopentanone with heptanal was compared with other heterogeneous catalysts—mixed Mg–Al, Zn–Al, and Mo–Si oxides and alkaline earth metal oxides. Modified MCM-41 showed low activity, which ranged from 6 to 11% heptanal conversion (7 h), with the exception of MCM-41 modified with 3-aminopropyl groups (35% heptanal conversion, 7 h). The reaction catalyzed by functionalized MCM-41’s gave mostly heptanal autocondensation product together with 2-heptylidenecyclopentanone. Modified zeolites also possessed low activity comparable to amino functionalized MCM-41 (heptanal conversions 6–13%, 7 h) and gave mostly heptanal autocondensation product. Activity of Mo–Si (20) mixed oxide was significantly higher (68% heptanal conversion, 7 h) and the main products were heptanal autocondensation product, 2-heptylidenecyclopentanone, and 2,5-diheptylidenecyclopentanone. Even higher heptanal conversions were obtained using Mg–Al and Zn–Al mixed oxides (83–92%, 7 h) giving the same products as when using Mo–Si oxides. Mg, Ca, and Zn metal oxides were very active in this aldol condensation (heptanal conversion >90%, 7 h) with the exception of Al2O3, which possessed only low activity (11% heptanal conversion, 7 h). The use of magnesium oxide and zinc oxide resulted in a large amount of 2,5-diheptylidenecyclopentanone and the use of calcium oxide in the formation of cyclopentanone autocondensation product and 2-cyclopentyliden-5-heptylidenecyclopentanone. Overall, the highest yields of the desired 2-heptylidenecyclopentanone were obtained when using magnesium oxide and zinc oxide and mixed Mg–Al and Zn–Al oxides, and these catalysts could be successfully used as efficient catalysts in 2-heptylidenecyclopentanone synthesis.
Footnotes
Acknowledgements
The authors gratefully thank Prof F. Kovanda from the University of Chemistry and Technology, Prague, for helping us to prepare samples of mixed Mg-Al and Zn-Al oxides.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the specific university research (MSMT No 20-SVV/2017).
