Abstract
The electrical and thermal conductivities as well as Lorenz number of polyvinyl alcohol (PVA) composites have been investigated at different concentrations of the copper chloride II (0, 1, 3, 5, 7 and 12 wt-%). The powder of copper chloride II (CuCl2) was casted with the PVA polymer1. The electrical conductivity was measured by using the impedance technique, and it was studied as functions of the CuCl2 concentrations, applied frequency range of 100–7000 kHz, and temperature range of 30–100 °C. The activation energy (
Introduction
Generally, polymers are inactive electrical insulators; the existence of impurities enables them to be electric conductors because of the free movement of the ions of impurities through them. In the recent years, studies of the electrical and thermal properties of polymers have attracted much attention in view of their applications in electronic and thermal devices. 1 However, the electrical conduction in polymers has been studied, in order to understand the nature of the charge transport prevalent in these materials while the thermal properties are aimed at achieving better thermal conductivity and thermal diffusion.
One of the important classes of polymer electrolytes is the polar polymer such as polyethylene oxide (PEO), polypropylene oxide (PPO) and polyvinyl alcohol (PVA). The PVA is a polymer that has been studied intensively due to its several interesting physical properties such as high dielectric strength, good charge storage capacity and damping dependent electrical properties in both forms pure and composite. The PVA has been widely used as a solid polymer electrolyte (SPE) as a stabiliser for producing conducting polymer dispersions. It is observed that doping a polymer with metal salts has significant effect on their structural, optical, thermal, and electrical properties.2,3 These effects on the physical properties of the polymers depend on the chemical nature of the dopant materials and on the way in which they interact with the host polymer. As a result, inorganic additives such as CuCl2 have considerable effect on the structural, electrical, and thermal properties when it is doped with a polymer. 4
The thermal conduction in the solid polymers is produced by phonon, ions transport (as major contributors), electrons and impurities existing in their bulk structure. Measurements of the thermal conductivity of polymeric materials is difficult since its value is relatively small which is in the range of 0.1–0.4 W·m−1·K−1. In addition to, the thermal conductivity is so sensitive to the measuring conditions, physical, and chemical structure of the polymer composite. Thus, the growing needs for materials to be used in thermal applications leads to the design of a new composite material with the appropriate combination of selected polymeric matrices and suitable fillers. 5
In this paper, we used the PVA as a host polymer because the PVA is a semi-crystalline polymer, and it has very important applications due to the role of OH group and hydrogen bonds.
6
The present study could be contributed to understand the effect of different concentrations of copper chloride II (0, 1, 3, 5, 7 and 12 wt-%) on the electrical and thermal properties of the PVA films such as AC conductivity (
Experimental work
Sample preparation
The polyvinyl alcohol composite doped with copper chloride (PVA/CuCl2) films were prepared at room temperature by solution casting method. The PVA composite was dissolved in distilled water and heated gently in water bath to prevent thermal decomposition of polymer. The PVA was stirred by magnetic stirrer for completely dissolved. Whereas the copper chloride with different concentrations (0, 1, 3, 5, 7 and 12 wt-%) were dissolved in distilled water and added to the polymer solution, they were heated until they were completely dissolved. Then the solution poured onto a glass plate and left to dry for 24 h to remove any residual solvent. By using the electronic micrometer, the thickness range of the PVA/CuCl2 films was found to be 60–70 μm.
Impedance measurements
The AC electrical measurements were measured in the frequency range of 100–7000 kHz at different temperatures. The AC measurements were performed using an LF impedance analyser (IA) manufactured by HP model 4192. The IA was a fully automatic, high-performance test instrument designed to measure a wide range of impedance related parameters, such as phase angle, real and imaginary components of the impedance, admittance, conductance and capacitance.
7
The LFIA gave the impedance and phase angle of the specimen by varying the applied frequency which was necessary to measure the electrical conductivity (
Thermal conductivity measurements
Measurements of the thermal conductivity (
Results and discussion
The measurements of the AC conductivity of the PVA/CuCl2 films were calculated by using the equation:
8

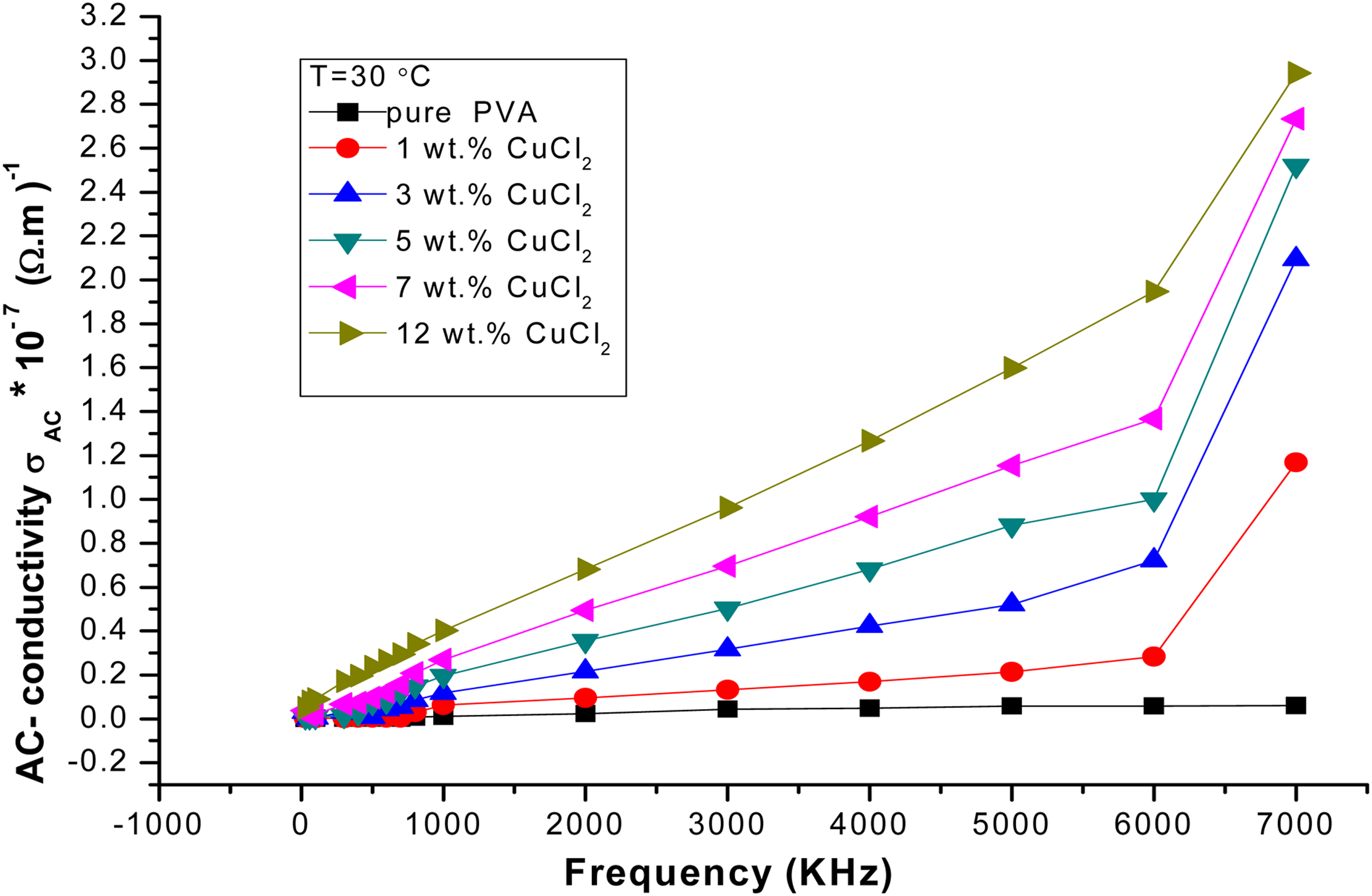
The AC conductivity of PVA/CuCl2 films as a function of frequency.
Figure 2 shows the electrical conductivity (
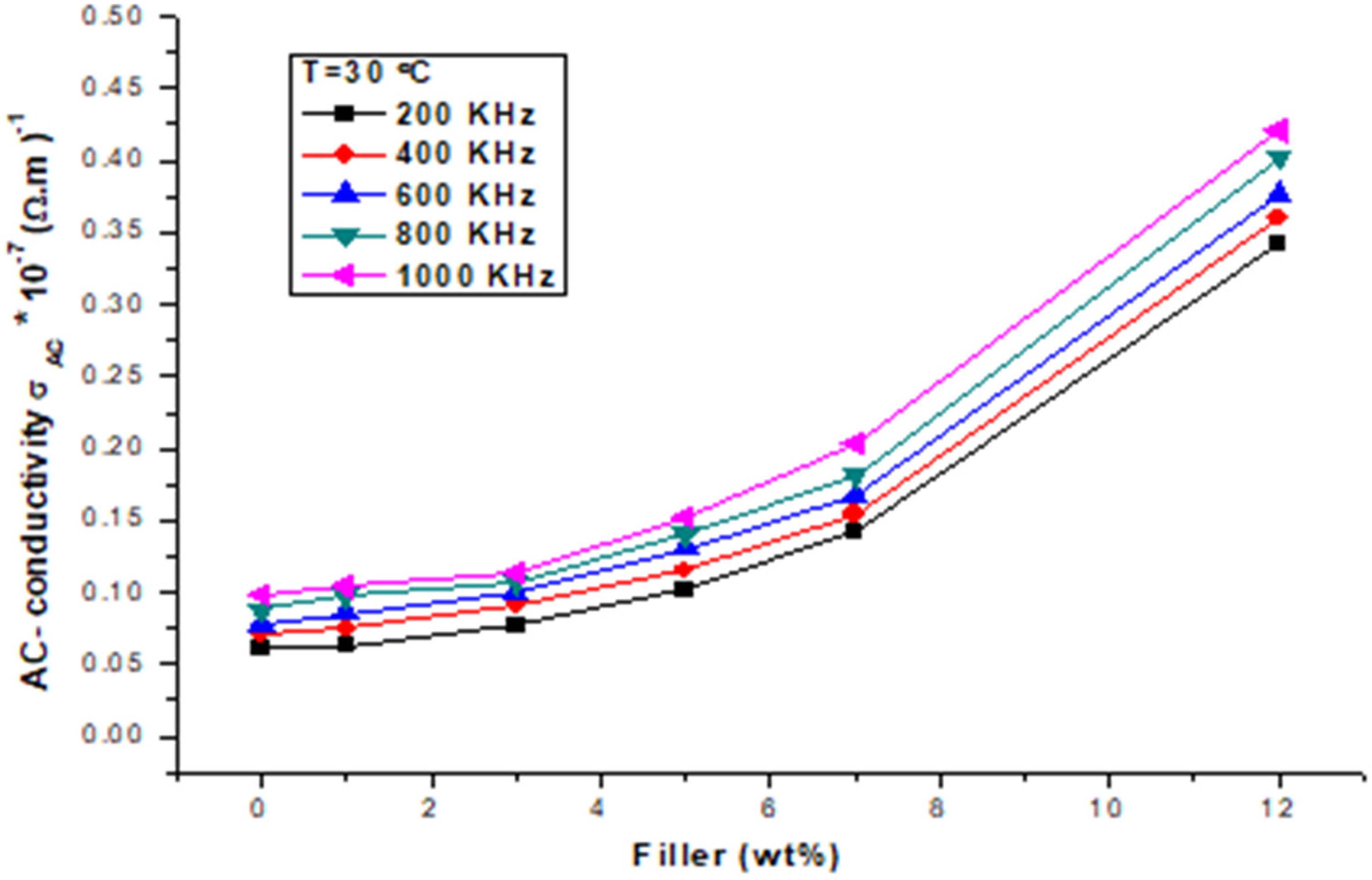
Variation of AC conductivity of the PVA/CuCl2 films with CuCl2 concentrations.
Figure 3 shows the measurements of the AC-conductivity (
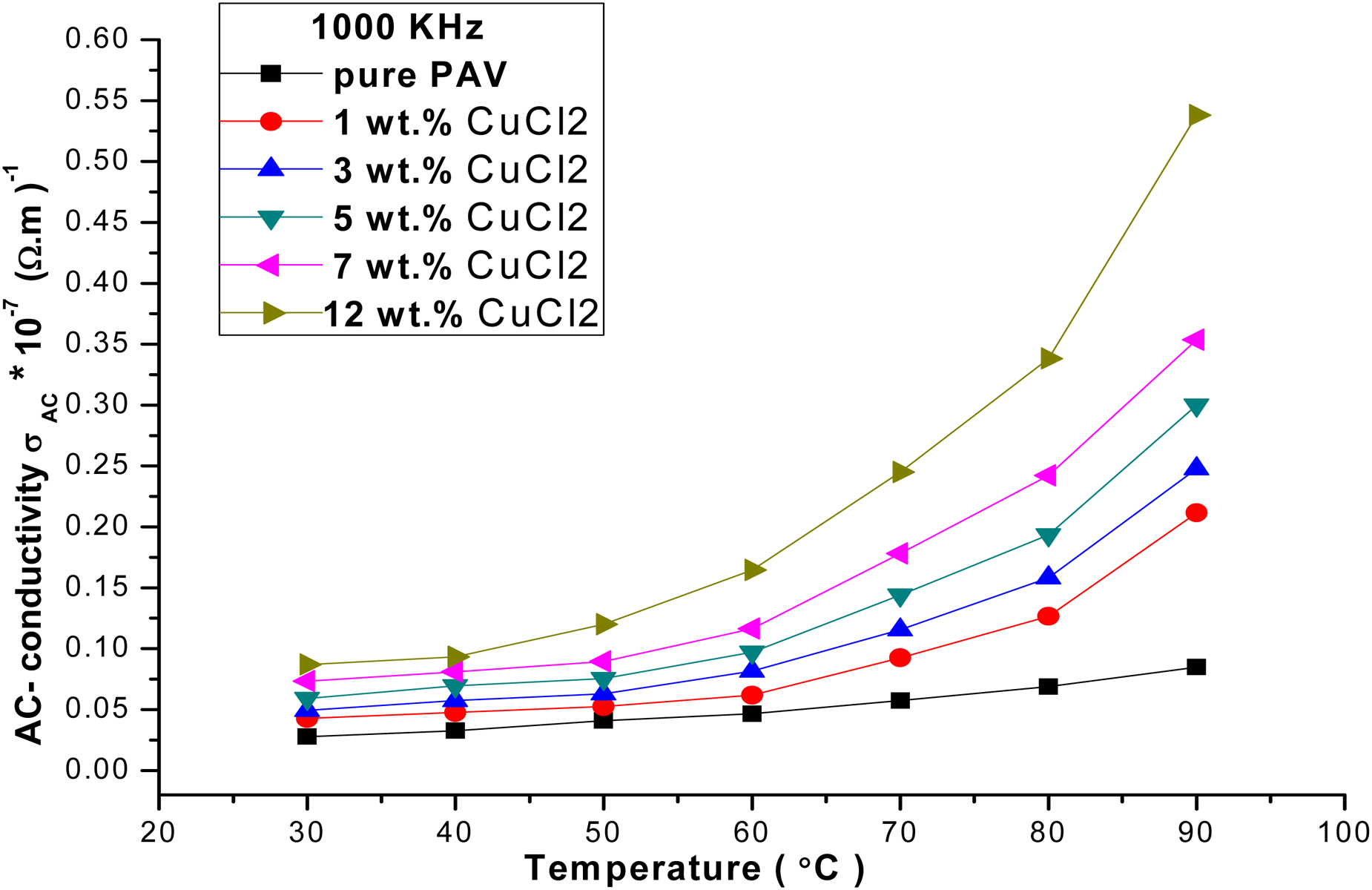
AC-conductivity of the PVA/CuCl2 films as a function of temperature at 1000 kHz.
To fully understand the electrical properties of the PVA/CuCl2 films, the activation energy (
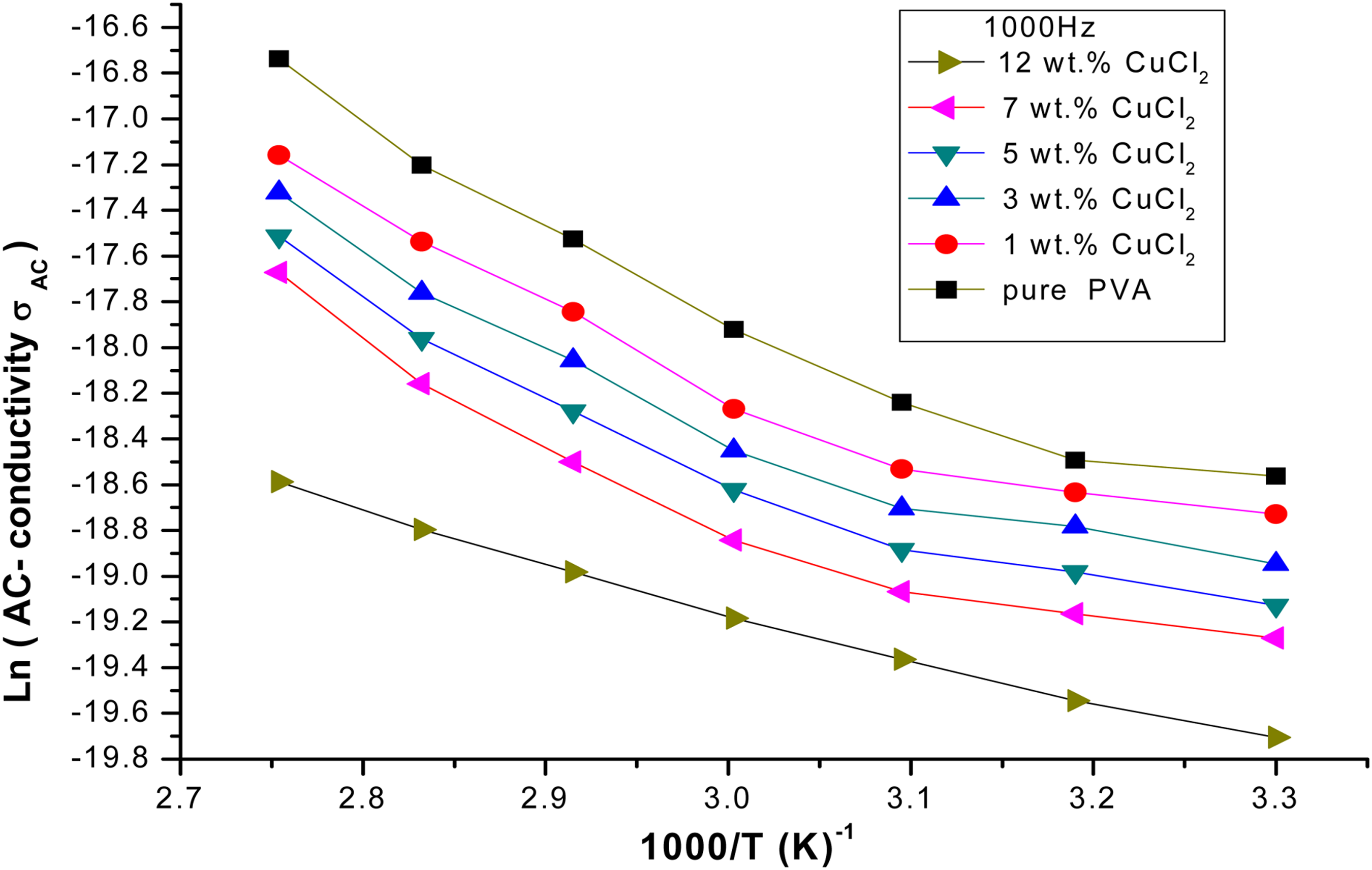
Variation of the Ln (AC-conductivity) of the PVA/CuCl2 films with (1000/
By using equation (2) and the data of Figure 4, the
The activation energy of the PVA/CuCl2 films with the CuCl2 concentrations.
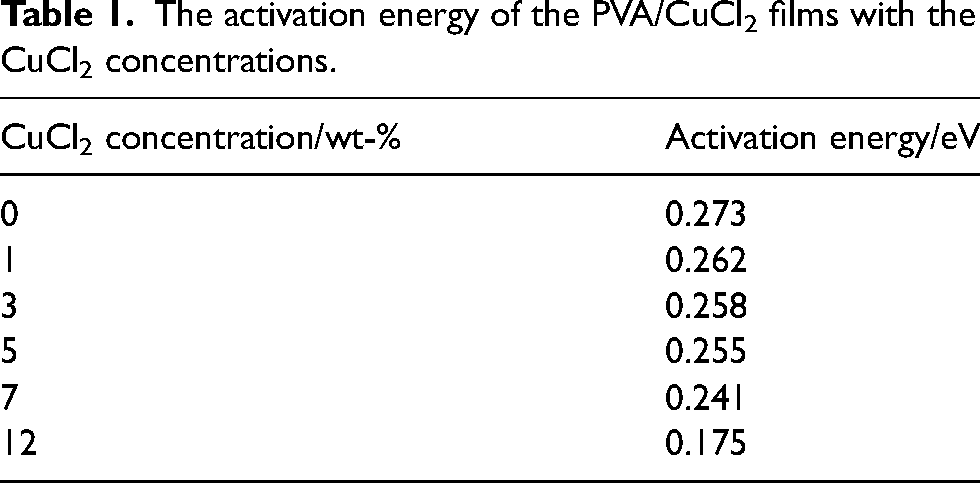
The thermal conductivity (
Figure 5 shows the variation of the thermal conductivity with temperature, the
The variation of the thermal conductivity of the PVA/CuCl2 films with temperature.
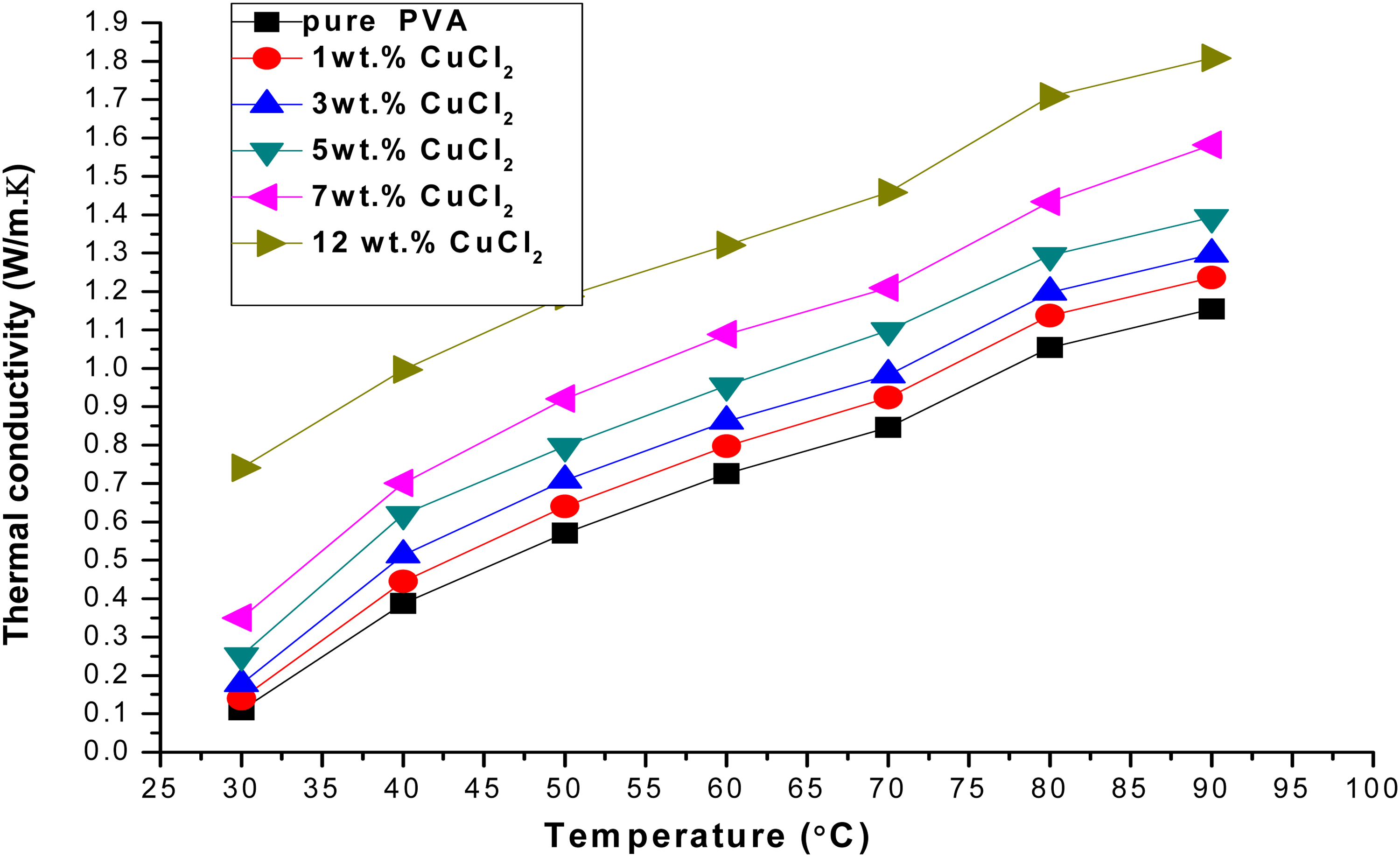
Figure 6 shows the variation of thermal conductivity of the PVA/CuCl2 with CuCl2 concentrations. It can be seen that the thermal conductivity increases with increasing the concentration of the CuCl2 filler content. Thus, the dispersing CuCl2 phase in the PVA matrix enhances the electrolyte thermal conductivity and reduces its thermal resistance, which may be useful to the thermal performance applications. This result may be due to compactness provided by increase in dopants concentration which increase the heat transfer through phonons diffusion. 18
The variation of the thermal conductivity of the PVA/CuCl2 with CuCl2 concentration.
The Lorenz number is a thermoelectrical parameter characterising the effect of temperature on both the thermal and electrical conductivities of a solid material. Knowledge of the Lorenz number is important in the design of the thermoelectric materials and in the experimental determination of the lattice thermal conductivity. The Lorenz number is determined by using the Wiedemann–Franz law:19,20
According to the data shown on the Figures 2 and 6, Table 2 shows a comparison between the electrical and thermal conductivities behaviour of the PVA/CuCl2 films at 30 °C. Both the electrical conductivity and thermal conductivity increase with increasing the dispersed CuCl2 phase in the PVA matrix.
The electrical and thermal conductivities of the PVA/CuCl2 films.

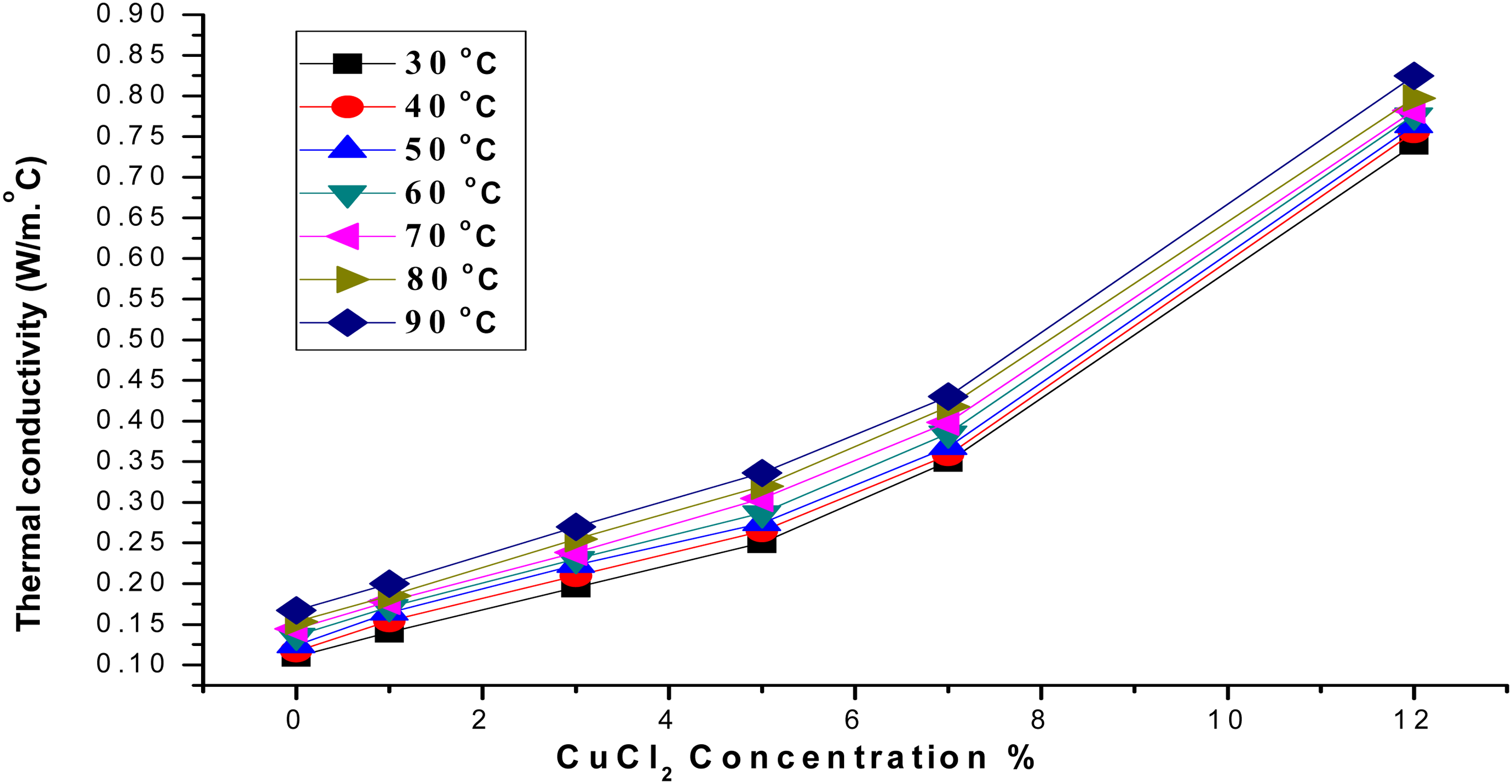
Figure 7 shows the variation of Lorenz number of the PVA/CuCl2 films with CuCl2 concentration at 303 K. It is clearly observed that the Lorenz number increases with increasing CuCl2 concentrations. This can be attributed to the difference in the rate of increasing of the
Variation of Lorenz number of the PVA/CuCl2 films with CuCl2 concentration at 303 K.
Conclusions
The electrical, thermal conductivities, activation energy and Lorenz number of composite films based on copper chloride(II) in PVA matrix have been investigated as functions of the filler concentrations, applied frequency and temperature. The AC conductivity increased with frequency, CuCl2 concentration and temperature. Whereas the thermal conductivity increased only with both temperature and CuCl2 concentration in the studied films. The values of the activation energy (
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
