Abstract
Introduction
Developmental disorders in children present a range of challenges, including delays, deviations, and dissociations during development. 1 These conditions, classified under neurodevelopmental disorders in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), encompass intellectual disabilities, communication disorders, autism spectrum disorders, attention-deficit/hyperactivity disorders, specific learning disorders, and motor disorders such as tic and Tourette’s disorders. 2 These disorders often persist throughout the life of approximately 15% of children worldwide, making regular assessment essential. 3 This highlights the critical role of developmental surveillance in treatment approaches, particularly in contrast to other mental health conditions. 4
PHR systems offer significant advancements in managing developmental disorders, allowing for a more personalized and controlled approach to healthcare. These electronic platforms enable individuals and caregivers to store, manage, and control health information securely.5,6 Unlike traditional health records managed by healthcare providers, PHR systems empower patients and caregivers by integrating data from diagnoses, treatments, medication schedules, and developmental milestones.7,8 Given the complex nature of developmental disorders, ongoing diagnostic assessments and interventions across multiple facilities are often needed. PHR systems facilitate this by enabling continuous and periodic management of medical records, thereby supporting detailed tracking of developmental progress and medication adherence. 9 This integration allows timely and appropriate medical and therapeutic interventions.
Despite these advantages, current PHR systems have limitations that can lead caregivers to seek less reliable information sources. They often lack explanations of medical terminology, abbreviations, and symptom interpretations, which can create significant challenges for caregivers.10,11 This gap in professional information delays access to necessary medical data, reducing treatment efficacy. 12 Caregivers may turn to informal communities for interpretations, leading to confusion and inappropriate treatment.
Effective PHR platform development for managing developmental disorders requires overcoming several challenges that can impact PHR functionality and user adoption. One primary concern is the lack of clinical involvement in the design and evaluation phases, which is essential for ensuring that these tools meet medical needs. 13 For example, the ‘Medihand’ project—a PHR application developed for parents of children with developmental disorders—lacked adequate clinical input, resulting in limited effectiveness due to insufficient clinical depth and precision in PHR data tracking. 9
The diagnosis and evaluation of developmental disorders in children involve complex procedures that require expert interpretation by psychiatrists and clinical psychologists.14,15 Over 200 distinct neuropsychological tests are available for assessing children and adolescents, each generating extensive data across numerous subfactors.16,17 The aggregation and representation of these results can vary significantly across institutions due to differences in measurement units and reporting formats. Therefore, clinical expertise and experience are indispensable in the successful development of PHR-based applications.
Moreover, the current approach to providing psychiatric PHR data is outdated and inefficient. In psychiatric settings, medical records for children with developmental disorders are provided primarily in printed formats due to privacy concerns. Digital records pose greater risks of unauthorized access, breaches, and misuse, which can undermine patient trust and discourage full disclosure, potentially affecting the quality of care.18–20 The physical transfer of these printed records between hospitals and other institutions presents practical challenges, especially in managing periodic changes during a patient’s development. If records are lost, the continuity of care relies solely on patient memory, leading to inefficiencies and potentially requiring repeated examinations, resulting in unnecessary costs and wasted resources.
To address these challenges, this study proposes IVORY, a PHR platform specifically designed to enhance the management of developmental disorders. IVORY is intended to bridge the gap between caregivers and healthcare professionals by providing a centralized, digital platform that integrates patient data across multiple institutions, facilitates longitudinal tracking of developmental progress, and supports clinical decision-making. Its primary role in the clinical pathway is to digitize, organize, and interpret medical data for use in consultations and treatment planning, reducing caregiver burden and enhancing clinician efficiency. By incorporating AI technology, the platform offers detailed medical explanations and symptom interpretation, significantly improving comprehension for patients and caregivers, thereby facilitating more informed decision-making. By transitioning from paper-based records to a fully digitalized system, IVORY addresses inefficiencies in traditional PHR systems, improving communication between providers and ensuring care continuity. Additionally, IVORY’s usability was evaluated through targeted testing involving both caregivers and healthcare professionals. These insights were instrumental in refining the platform’s features to ensure clinical relevance and real-world applicability.
Methods
Development of the application: System implementation and core functions
The research team designed and developed IVORY, a mobile AI system, to enhance PHR management for pediatric patients with developmental disabilities. The platform integrates data from various institutions into a unified format, allowing for trend analysis, longitudinal tracking, and correlation identification to support clinical decision-making and patient engagement. Additionally, IVORY simplifies complex medical information, enabling patients and caregivers to better understand their health status and participate more actively in their care.
System architecture
The platform’s structural diagram is divided into three main stages (as shown in Figure 1): (1) Input stage: Users input demographic data (e.g., age, sex), physical metrics (e.g., height, weight), and developmental history (e.g., prenatal issues) directly into the application. Medical test results, including those from fMRI interpretations, blood/urine tests and various psychological assessments (e.g., intellectual functioning, attention and neurocognitive functioning, and social function), are captured via the camera of the mobile device and digitized through the OCR unit powered by the Image2Text Tool. (2) Storage stage: The OCR-processed data are encrypted and securely stored in a database. The data are then aggregated and systematically categorized via data mining techniques, aligned with specific clinical objectives, and then prepared for AI-based learning in the cloud. (3) Output stage: The processed data are structured into a standardized template, with values converted to percentiles for consistent comparison and easier interpretation. This allows for trend and correlation analysis, enabling users to track developmental progress effectively and predict therapeutic outcomes. Additionally, the platform simplifies medical terminology and uses AI-driven algorithms to analyze and compare similar profiles. Through this process, it provides interpretive insights into analytical results and delivers personalized feedback in a Q&A format, helping to establish potential diagnoses and guide treatment planning. System architecture of IVORY.
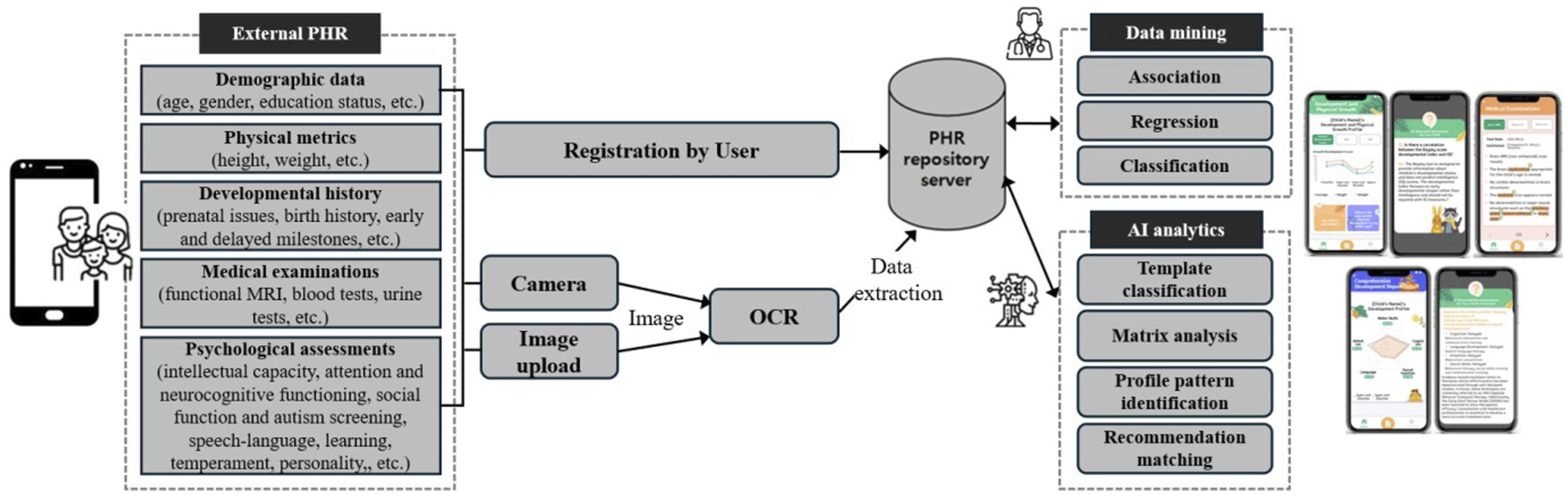
Development environment
The system architecture consists of a user application that interfaces with a server for processing and analyzing images. The server, developed via Python and Flask, operates on an Ubuntu system within the AWS EC2 environment. This infrastructure supports optical character recognition (OCR) requests via the Naver CLOVA OCR API, which outperforms competing services from Google, Microsoft, and AWS in recognizing Korean text. The results are returned in JSON format, which is lightweight and well suited for efficient data exchange and manipulation.
For data visualization, the platform utilizes the MPAndroidChart library, enabling detailed graphical representations of examination trends within the Android application. RxJava is employed to handle asynchronous processing, ensuring that multiple user requests are managed without blocking the user interface, thus maintaining a smooth user experience. Server communication is optimized through the Retrofit library, which is chosen for its efficiency and ease of use, which enhances the application’s responsiveness.
AI-driven processing
OCR and image2text processing
The system employs the pretrained Naver CLOVA OCR algorithm, which is known for its superior accuracy in Korean text recognition. 21 To optimize the OCR performance, templates were developed for 126 unique medical report formats collected from 41 institutions, including 13 university hospitals, 4 general hospitals, 17 local clinics, and additional facilities such as counseling centers. These templates enable the system to identify specific keywords within reports to classify the document type and extract key details. For example, when terms such as “Full-Scale IQ (FSIQ)” are detected, the system classifies the document as an intelligence test report and extracts the relevant key values.
The OCR recognition accuracy was further improved through preprocessing steps, such as unifying expressions and using regular expressions to crosscheck extracted data. A postprocessing stage was implemented to convert extracted data into standardized percentiles for uniform comparisons.
Pattern recognition and rule-based reasoning (RBR)
The system employs an AI-driven profiling algorithm (version 3.7), comprising approximately 2978 structured criteria developed with 19 healthcare professionals, including psychiatrists, clinical psychologists, and speech therapists. These criteria enable pattern detection, clustering, and psychological profiling across key domains such as verbal comprehension, perceptual reasoning, working memory, and processing speed.
The RBR framework was integrated to enhance interpretability, with diagnostic rules structured via IF-THEN logic. For example: • If the FSIQ score is below 70 AND adaptive behavior scores are significantly below average, THEN is classified as an intellectual disability. • If the FSIQ is borderline (70-85) and adaptive behavior scores are within average limits, THEN recommends further developmental assessment.
The RBR framework incorporates a certainty factor (CF) methodology to quantify diagnostic confidence, with CF values aggregated when multiple rules are satisfied. These rules align with expert-reviewed standards and were iteratively tested using diverse datasets for accuracy. The use of CF was chosen because of its intuitive and interpretable approach to handling uncertainty, better alignment with clinical reasoning, and computational efficiency, making it more suitable for clinical decision support in medical applications. 22
Workflow and implementation
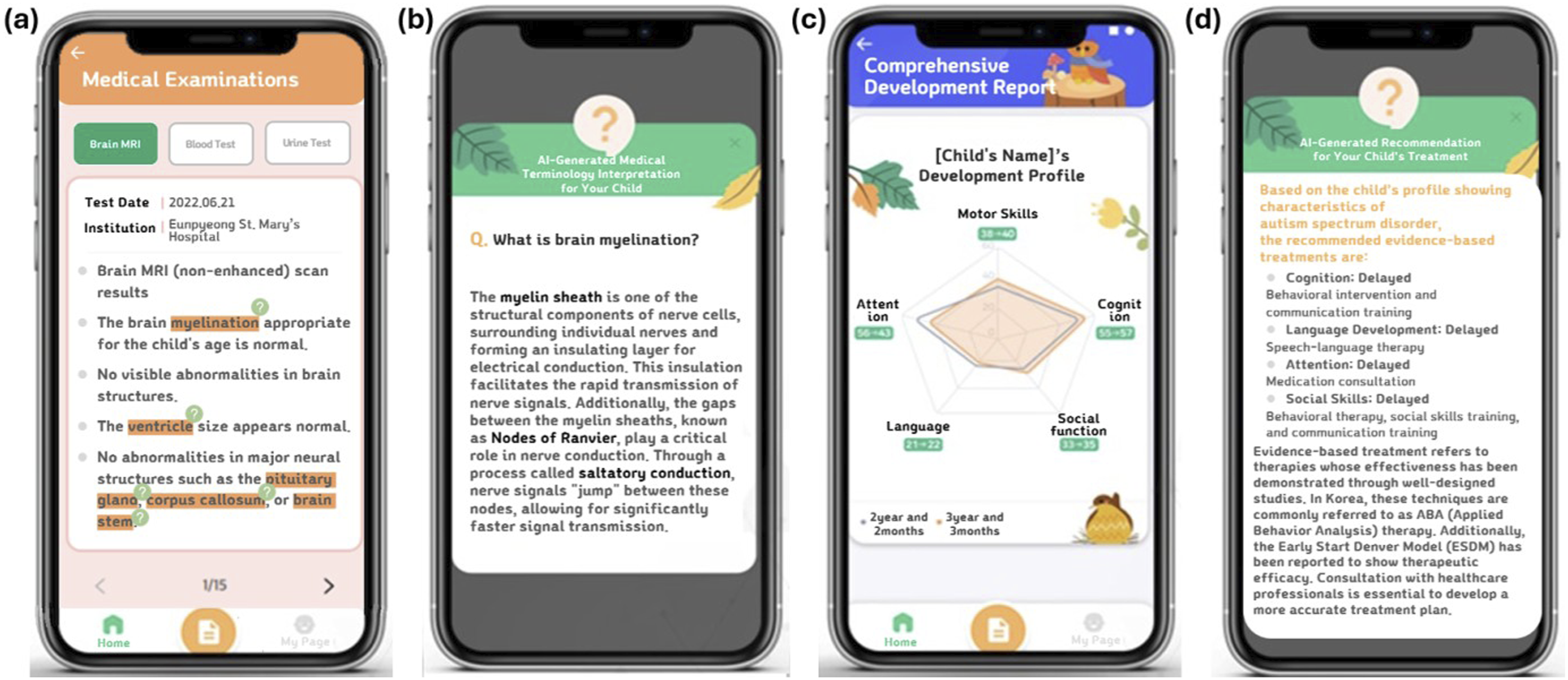
The IVORY mobile application was developed via Android Studio, which leverages the Android architecture component library to increase system reliability and maintainability. The development process involved an 8-month planning phase, followed by 2 months of UI/UX design and 3 months of application development. This was succeeded by a 2-week demonstration and debugging phase, culminating in a 3-month usability validation period. The overall development process was conducted in close collaboration with healthcare professionals, including psychiatrists and clinical psychologists, to ensure its practical utility and seamless integration into clinical workflows. Furthermore, the data analysis workflow underwent iterative refinements on the basis of expert feedback to enhance clinical relevance and accuracy. The specific workflow of the application is as follows. Patients first capture various medical test results via a smartphone camera, and the extracted data are processed via OCR technology (as described in Figure 2(a)–(d)). The processed data are then categorized and stored via data mining techniques (as shown in Figure 2(e)), after which AI algorithms analyze the information and present it in a standardized format. This structured approach enables the provision of personalized recommendations and clinical guidance (as described in Figure 3), supplemented by evidence-based treatment modules for significant clinical findings (as shown in Figure 4). IVORY application workflow for data input. (a) The initial screen is displayed when users access the application, allowing them to input basic information or start using the application directly. (b) The home screen provides access to assessment categories, data input, and my page. (c) Users can upload medical records via camera, album, or direct input. Uploaded data are automatically categorized or manually assigned when directly input. (d) Users select and save specific test results for digitization via OCR technology. (e) OCR recognizes and extracts the required data, classifies templates, and stores the content in the category via data mining functionality. Visualization in a standardized format with personalized information. The OCR-processed data are analyzed via AI algorithms and presented in a standardized format, providing personalized advice and tailored recommendations for each child’s profile. Tailored analysis and recommendations using AI-driven algorithms. (a) Brain MRI results with AI-suggested medical terms for better understanding. (b) AI-provided medical terminology interpretation for user clarity. (c) Development profile integrating heterogeneous data. (d) AI-based recommendations for therapy and consultation.

Performance improvement and validation
To verify the functionality and usability of the IVORY application, healthcare professionals, including pediatric psychiatrists, clinical psychologists, researchers, and technical experts, conducted a predeployment verification. A total of 126 medical report forms were collected and digitized via a mobile terminal, with the OCR used to recognize predefined information. The OCR recognition success rate was 81%, whereas the remaining 19% failure rate was due to issues such as slanted characters, small or blurred text, and data in graphic form without numerical entries. These limitations reflect inherent OCR challenges, such as image resolution and font size. 23 The OCR text recognition accuracy was 97%, with an average 3% error rate caused by overlapping text. To address these issues, manual input and a flagging system were introduced, alongside ongoing improvements to the OCR algorithm. The flagging system identifies problematic data, prompting reanalysis or manual input to ensure critical information accuracy. A quality control process assesses poor-quality data, categorizing flagged inputs as recoverable or nonrecoverable. Low-resolution images were flagged for manual review, and graphical reports lacking numerical data were excluded. Clinicians provided supplemental information to ensure the accuracy of clinical decision-making. A human-AI interaction protocol was established to balance automated processing with expert oversight. All errors and failure rates were logged, guiding continuous OCR and AI improvements. To further increase the system’s accuracy, robust data quality assessments were conducted, ensuring that low-resolution or incomplete data were excluded. Standardized report templates ensured that the extracted data were aligned with clinical workflows. Iterative workflow refinements based on feedback improved clinical relevance. The RBR framework underwent rigorous testing to align with clinical standards and provide reliable diagnostic support. Various IF-THEN rules in the RBR framework were reviewed and validated via datasets from 126 digitized report types. These tests confirmed the RBR framework’s high accuracy in identifying key patterns.
On the basis of OCR-recognized data, the accuracy of RBR rule application was 100%. Issues from incomplete or ambiguous input were mitigated via CF score calculations, which assign confidence scores to partially matched rules. The flagging system was extended to the RBR framework for manual review and adjustment of cases where the CF score fell below a predefined threshold. The dual verification process of OCR and RBR ensures high reliability in both text recognition and rule-based decision-making, effectively supporting clinical workflows, even in extreme cases.
Usability study of the application
Participants and sampling
General characteristics of participants.

A separate usability evaluation was conducted with healthcare professionals, including psychiatrists and clinical psychologists. For this group, usability studies typically require smaller sample sizes for exploratory purposes. On the basis of prior research, 12 participants aligning with recommendations that 10–20 participants can effectively identify usability issues. 26 These professionals provided critical insights into AI-driven clinical relevance, interface clarity, navigation ease, and workflow integration, offering constructive feedback for further refinement.
Eligible input data included fMRI interpretations, blood and urine test results, and psychological assessments. Images were required to meet quality standards of a resolution of ≥150 dpi as recommended by CLOVA OCR to ensure optimal processing by the OCR algorithm. Data with illegible font sizes or missing numerical entries were excluded.
Procedure
The participants installed the IVORY application on their personal mobile phones and uploaded scanned or captured images of their medical records through the application. These images were processed on a cloud-based repository server to generate analyses, enabling both onsite clinicians and remote researchers to access and utilize the data in real time. The researcher team tailored templates for a new report format to enhance the performance of the CLOVA OCR algorithm in classifying templates, extracting key values, and generating standardized report formats. The data were preprocessed to improve compatibility with the AI system, ensuring consistency in the analysis. Low-quality or illegible data were flagged for manual review. Recoverable data were corrected, whereas nonrecoverable data were excluded to maintain reliability. The flagged data were reviewed and managed by healthcare professionals, including psychiatrists and clinical psychologists. The AI system provided outputs in a standardized report format, which included key values such as percentile-based analyses, developmental trend visualizations, and symptom interpretations. The participants reviewed the recognition of medical records and evaluated the usability of various system functions. The IVORY team also assessed the performance of the CLOVA OCR algorithm, measuring the average success rate of OCR recognition, including the successful extraction of key values per template and the average character error rate among recognized key values. The AI outputs supported clinical decision-making by identifying trends, analyzing developmental data, and providing actionable recommendations. To ensure smooth operation and user familiarity, training sessions were conducted for caregivers and healthcare professionals prior to usability testing. These sessions covered data input procedures, system navigation, and troubleshooting. During the trial, ongoing technical support, including onsite assistance, was provided to address any challenges encountered by the participants.
Measures
The participants used the application and completed a structured survey assessing usability. The survey was based on a modified version of the health information website evaluation model by Woo and Cho (2006). 27 The scale itself is structured to capture a range of user experiences across five key evaluation criteria: content composition (5 items), clarity of information and information protection (3 items), content management (2 items), information source (2 items), and overall satisfaction (1 item). The scale consists of 13 items, each rated on a 5-point Likert scale ranging from 0 (Strongly Disagree) to 4 (Strongly Agree), ensuring a clear and intuitive rating system for participants. Scores were calculated for each domain, and domain-level results were aggregated to assess overall usability. In addition to the structured survey, free-text responses were included to gather qualitative feedback, allowing participants to elaborate on specific strengths or challenges they encountered while using the application.
Statistical analysis
The data were analyzed via descriptive statistics to compute means and standard deviations for the usability metrics. All the statistical analyses were conducted via IBM SPSS Statistics 25.0 (IBM Corp, Armonk, NY).
Results
Evaluation outcomes
User satisfaction results for caregivers.

User satisfaction results for healthcare professionals.

Applications and comparisons
Applications with an average satisfaction rating of 3 or above are generally regarded as providing high user satisfaction. IVORY not only met but also exceeded this benchmark across all evaluated subscales, indicating its strong usability and effectiveness. Although direct comparisons with other applications are limited owing to the absence of similar benchmarks, IVORY enhancements are anticipated to substantially improve the management of chronic symptoms and behavioral issues. Future evaluations should include head-to-head comparisons with similar platforms to establish standardized benchmarks for usability and performance.
Discussion
This study proposed IVORY, a PHR-based mobile AI platform, which revealed high user satisfaction, with average scores of 3.52 for caregivers and 3.50 for healthcare professionals. These results suggest that IVORY effectively integrates essential features for managing and monitoring medical data, demonstrating its ability to handle chronic symptoms and behavioral issues efficiently. The platform’s data integration facilitates both longitudinal and cross-sectional analyses, enhancing diagnostic accuracy and supporting personalized treatment plans. AI-generated outputs, such as growth analyses, symptom trend visualization, and customized treatment recommendations, were found to enhance clinicians’ decision-making and caregivers’ understanding. IVORY’s holistic approach allows for the evaluation of symptoms before and after specific interventions. For example, clinicians using IVORY identified correlations in longitudinal data, enabling earlier interventions and treatment adjustments. The effective use of IVORY in clinical settings can significantly improve decision-making for both patients and clinicians while also fostering more informed and proactive caregiver involvement through AI-driven, customized insights.
For caregivers, IVORY streamlines access to integrated data from multiple healthcare institutions, enhancing the management of complex medical information. Providing AI-driven, customized insights helps improve treatment outcomes, particularly for caregivers managing developmental disabilities. Prior research has indicated that access to reliable health information enhances therapeutic discovery, reduces caregiver stress, and supports more informed healthcare decisions.28,29 IVORY’s role in securely aggregating and managing PHR data could significantly advance medical AI technologies, enhancing data-driven decision-making and optimizing healthcare solutions.30,31
For clinicians, IVORY’s AI-driven insights facilitate the early identification of developmental risk, enabling more timely interventions. Given the complexity of developmental disorders, a comprehensive analysis of patient data is essential for improving diagnostic accuracy and optimizing treatment strategies.32,33 By supporting early intervention, IVORY helps prevent symptom progression and mitigates the risk of secondary behavioral challenges. 34 Furthermore, a significant portion of IVORY’s input and output data consists of standardized medical and psychological assessment data that are widely used internationally, ensuring broad applicability. The platform’s use of standardized neuropsychological and behavioral assessments provides norm-referenced scoring, allowing for consistent interpretation across diverse healthcare settings. Since these standardized measures maintain their validity across translations, IVORY’s framework can be seamlessly adapted for international use with minimal modifications, expanding its clinical applicability.
Despite the study’s positive results, there are limitations. While this study highlights IVORY’s high satisfaction scores, the evaluation focused primarily on satisfaction as a usability metric. According to Frøkjær et al. (2000), usability encompasses effectiveness, efficiency, and satisfaction, which should be independently assessed. 35 However, this study did not evaluate effectiveness (e.g., system accuracy) or efficiency (e.g., time/effort required) separately. Future research should address these aspects for a more comprehensive usability evaluation. Additionally, the cross-sectional design may not fully capture long-term shifts in user attitudes toward PHRs. While platform usage can be tracked, it does not directly reflect evolving perceptions. Longitudinal studies are needed to assess these changes as adoption increases. Despite advancements in OCR, challenges such as low resolution and small text persist. To mitigate this, a multistage quality control system integrating automated error detection and clinician oversight was implemented to prevent critical errors. While manual data entry options exist and algorithms continue to improve, ongoing refinements in OCR and data processing are essential for optimizing usability. AI algorithms assist in interpreting developmental disorder assessments but are intended to complement, not replace, clinical expertise. RBR relies on predefined rules, limiting adaptability to nonstandard data or unexpected cases. Although this study demonstrated high accuracy, rule-based approaches may struggle with atypical patterns, potentially increasing the risk of misclassification. Therefore, further research is needed to validate their clinical reliability and enhance their practical applicability. Future studies should focus on improving the precision of these tools and ensuring their adaptability across diverse clinical environments by enhancing scalability. To achieve this, continued collaboration among institutions and researchers is essential. Discussions are currently underway to facilitate access to anonymized data and algorithm updates through the research consortium. If successful, this initiative will not only enable additional validation efforts but also foster cross-institutional collaboration to further refine and advance the algorithm.
Conclusions
IVORY represents a groundbreaking approach to managing pediatric developmental disorders, supporting comprehensive clinical decision-making and enhancing caregiver engagement. As the first platform to integrate AI-driven recognition technology for consolidating PHR data in pediatric developmental disorder management, IVORY offers an innovative solution for standardizing and analyzing heterogeneous medical data across institutions. This capability enables seamless longitudinal tracking, AI-powered diagnostic support, and personalized treatment recommendations, distinguishing IVORY from existing systems. Rather than replacing medical professionals, IVORY enhances clinical workflows by providing AI-driven insights that facilitate more efficient and informed decision-making. Further research is needed to validate its clinical effectiveness and expand its applicability across diverse healthcare environments.
Footnotes
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Technology Innovation Program (grant number PC20ONDI0074; an AI-driven Global PHR Pediatric Developmental Disability Management/Treatment Platform) funded by the Ministry of Trade, Industry & Energy, Republic of Korea. Additionally, this research received support from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number RS-2023-00266131).
Ethical statement
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated or analyzed during the current study are available from the corresponding author upon reasonable request.
