Abstract
Diabetic retinopathy is a microvascular complication of diabetes mellitus that is usually asymptomatic in the early stages. Therefore, its timely detection and treatment are essential. First pilot projects exist to establish a smartphone-based and AI-supported screening of DR in primary care. This study explored health professionals’ perceptions of potential barriers and enablers of using a screening such as this in primary care to understand the mechanisms that could influence implementation into routine clinical practice. Semi-structured telephone interviews were conducted and analysed with the help of qualitative analysis of Mayring. The following main influencing factors to implementation have been identified: personal attitude, organisation, time, financial factors, education, support, technical requirement, influence on profession and patient welfare. Most determinants could be relocated in the behaviour change wheel, a validated implementation model. Further research on the patients’ perspective and a ranking of the determinants found is needed.
Keywords
Introduction
Diabetes mellitus (DM), especially type 2, is among the most widespread global chronic health conditions affecting 425 million people worldwide in 2017. Globally, USD 727 billion is spent annually on diabetes-related health care. 1 Diabetic retinopathy (DR) is a microvascular complication of DM which leads to progressive damage of the retina. 2 DR is a leading cause of vision loss in industrialised countries. 3 Many of those cases in which DR causes visual impairment can be prevented by early detection, treatment of risk factors and by photocoagulation. 4 Since DR is usually asymptomatic in the early stages, its timely detection and treatment are essential. In Germany, 1,270,000 people, corresponding to 21.7% of all patients with DM or 1.53% of the whole population, suffer from DR. 5
Regular examinations in the context of e.g. Disease-Management-Programs (DMPs) have been established to standardise the care of patients with common chronic diseases such as DM. Within these programs, regular ophthalmologic check-ups are offered to recognize or avoid complications at an early stage. 6 However, patient compliance with DR screening is not optimal, as shown by attendance rates ranging from 32 to 85%. 7
Considering an ageing population and the increasing significance of chronic diseases such as DM in the years to come, overburdened healthcare systems will increasingly require new concepts for medical care. Against this background, E-health has been presented as a potential helpful addition. E-health is an emerging field in the intersection of medical informatics, public health and business, referring to health services and information delivered or enhanced by the internet and related technologies. 8 For example in the United Kingdom, China, Singapore and India, DR screening projects have been conducted by the help of telemedicine. In these cases, fundus photographs have been taken by non-medical staff (MA) with the help of smartphones which were interpreted by ophthalmologists in an asynchronous manner. 9 With high-resolution cameras in conjunction with certain add-ons, smartphones are capable of taking high quality images of the retina, even in the hands of a non-specialist. 10
Another E-health approach in DR screening has developed in the past years in which retinal photographs are interpreted by Artificial Intelligence (AI). 11 In this scenario, no human specialist would be needed for the analysis and the examination could also take place on a primary care level, making the General Practitioner (GP) responsible for the entire follow-up of patients with DM. Especially in rural areas with a distant ophthalmologist and multimorbid patients, it could improve access to care and enable a regular and timely screening of patients. 12
Deep learning (DL), which constitutes a subtype of AI, is a state-of-the-art machine learning technique that shows a promising performance in image recognition, speech recognition and natural language processing. 13 Meanwhile it has been adopted in various domains such as telecommunications, social media and medicine. Especially for medical imaging analysis, it has achieved good results in several medical specialities such as radiology, 14 dermatology 15 and in particular ophthalmology.16–18 DL takes up a long history of autonomous and assisted analysis of retinal photographs that has existed since the 1990s and before. A metaanalysis from 2020, including 10 studies of DR screening in primary care, attributed a high diagnostic performance to DL-algorithms, with a pooled sensitivity of 87% and pooled specificity of 90% for referable DR. 19 A study from 2019 even revealed that in comparison with human graders, the DL algorithm had a significantly higher sensitivity (97% vs. 74%) in detecting referable DR. Also, the algorithms significantly reduced the false negative rate (by 23%) at the cost of slightly higher false positive rates (2%). 20 Especially in a screening situation, the sensitivity and prevalence are important because it determines the degree of false negative results.
However, such studies have to be evaluated with caution: Most research on AI in patient care concentrates on the technology itself or its diagnostic performance because developers seek to show that the technology is working. 21 Design features that are needed for robust validation of the real-world performance of the AI are missing. Also, most studies that exist on AI-assisted screening methods of DR in a clinical setting are made from retrospective data. Prospective studies that include evaluations of AI models within a clinical environment are beginning to emerge, but are still scarce. Therefore, current research lacks evaluation of true clinical effectiveness because socio-environmental factors that have an impact on the implementation are not sufficiently taken into account.22–24 Another factor is that very few of the studies have been conducted in primary care or in Europe.16,18,25–29 For these reasons, the results cannot be directly transferred to clinical practice.
When it comes to the implementation of a new technology into daily clinical practice, the barriers and facilitators to this implementation are multilayer. So far, an AI-assisted DR screening has not yet found its way into daily practice of GPs in Germany. Thus, little is known about factors that might influence the implementation of such devices in such a setting. Experience during the last few years has shown that a high number of E-health projects had trouble developing beyond a pilot stage without accompanying assistance by implementation science knowledge. 30 Implementation science is a part of health services research that understands and promotes the integration of evidence-based medicine into complex real-world settings. Research on the needs and requirements of future users is the basis for implementation. By identifying potential determinants to implementation, possible barriers e.g. specific reservations of the target group could be reduced.
Therefore, the aim of this study was to generate hypotheses for barriers and facilitators of the implementation of smartphone-based and AI-supported devices for the screening of DR within GP practice.
Methods
Study design
We conducted a qualitative study in Schleswig-Holstein, a northern federal state of Germany, using semi-structured interviews to collect data. Through these interviews we explored attitudes to digital technologies and AI as well as barriers and facilitators for the implementation of smartphone-based and AI-supported diagnosis tools for the screening of DR in primary care.
The guided questionnaire was developed by experiences of the researchers (postgraduate trainee, economic engineer and GP), results of a metaanalysis 19 within a project addressing AI and diagnosing DR 31 and brainstorming. It was piloted prior to the interviews with three GPs working at the Institute of Family Medicine in Lübeck. Due to this process, only minor (linguistic) amendments were made.
The final template included the following questions
Warm Up
1. What significance do digital technologies (e.g. smartphone, internet) have for your daily life, in private and during work? 2. For what purposes do you use a smartphone in a professional context? 3. What experiences have you had before with the use of telemedicine for example in the form of video consultations? 4. What experiences have you had before with „artificial intelligence” in a medical context, for example the analysis of an ECG tool?
Prior to the main questions, the following information was given to every participant: “Now to a more specific topic that deals with diagnostic tools to be used in the GP practice. It is about a certain add-on that can be attached to a smartphone with which images of the retina of patients with diabetes can be taken without using mydriatics. These images are then to be evaluated directly by the software using artificial intelligence, so that the general practitioner has a direct answer to the question of whether certain fundus pathologies such as diabetic retinopathy are present, as a kind of screening, in which case some patients could be referred to an ophthalmologist.”
Main Questions
5. What could influence the acceptance of patients of such smartphone-based and artificial intelligence-based screening devices for DR in primary care? 6. In your opinion, what barriers are there to implementing the described devices in the GP’s practice? 7. What facilitators do exist to implement the described devices in the GP’s practice? 8. What influence does the option of AI-supported and smartphone-based screening devices have on the profession of the GP? 9. In your opinion, what should be considered in the technical implementation of such devices? 10. Are there aspects which are important to you that we have not already discussed?
Sampling and recruitment
GPs and medical practice staff (MAs) in Schleswig-Holstein were contacted by letter and email. In the beginning, teaching practices of the University Lbeck were contacted. Furthermore, emails were sent to GPs in Schleswig-Holstein to extend the recruitment to both urban and rural areas. GPs were invited to ask their MAs to participate in the interviews. In a second step, the integrated ophthalmic healthcare corporation eG (Gesellschaft fr integrierte ophthalmologische Versorgung Schleswig-Holstein, GIO) was contacted in order to recruit established ophthalmologists. Furthermore, established ophthalmologists were contacted by letter individually. The addresses of GPs and ophthalmologists were found using the website of the Associations of Statutory Health Insurance Physicians (Kassenärztliche Vereinigung, KV). One ophthalmologist was contacted individually due to personal experience with smartphone-based retinal image creation.
The participants received information regarding the background and the aim of the study. Furthermore, the participants were informed about the handling of the data and their right to withdraw from the study at any moment. Informed consent was received prior to the interviews.
Data collection and handling
The interviews were conducted from September 2020 until February 2021. The participants were interviewed by the first author, a resident in the first year of specialist training, via telephone, due to the COVID-19 pandemic. Interviews were recorded digitally, pseudo-anonymised and transcribed by the interviewer, the second author and auxiliary scientists working in the Institute of Family Medicine in Lbeck. Recruitment and interviews continued until thematic saturation was reached (i.e. no major new themes were emerging).
Data analyses
After transcription, the audio files were checked for accuracy and read repeatedly by the first and second author.
According to Mayring, 32 a structured qualitative content analysis was carried out: Relevant quotes were marked. Themes for analysis were identified through repetition and frequency in the transcripts. In the following step, categories were inductively formed from the quotes. Meaningful segments of the transcripts were units for coding and analysis. These codes were grouped to form overarching themes which were iteratively refined over the course of analysis, forming categories and subcategories. The entire team met virtually several times to review the coding and agree on each category and subcategory. Thereby, further inductive codes emerged from the data.
In a last stage of analysis, the categories were compared to existing implementation models, such as the Behaviour Change Wheel. 33
Reporting
The COREQ (COnsolidated criteria for REporting Qualitative research) guideline was used to guide reporting.
Ethics
A favourable ethical opinion was obtained from the Ethics Committee of the University of Lbeck on 4 June 2020.
Results
Demographic information.

The questions from the interview template were aimed at elaborating on the main aim of this study, working out the potential barriers and facilitators to implementation of AI-assisted screening devices for DR in primary care.
Determinants of the implementation of AI-assisted DR screening tools in primary care.

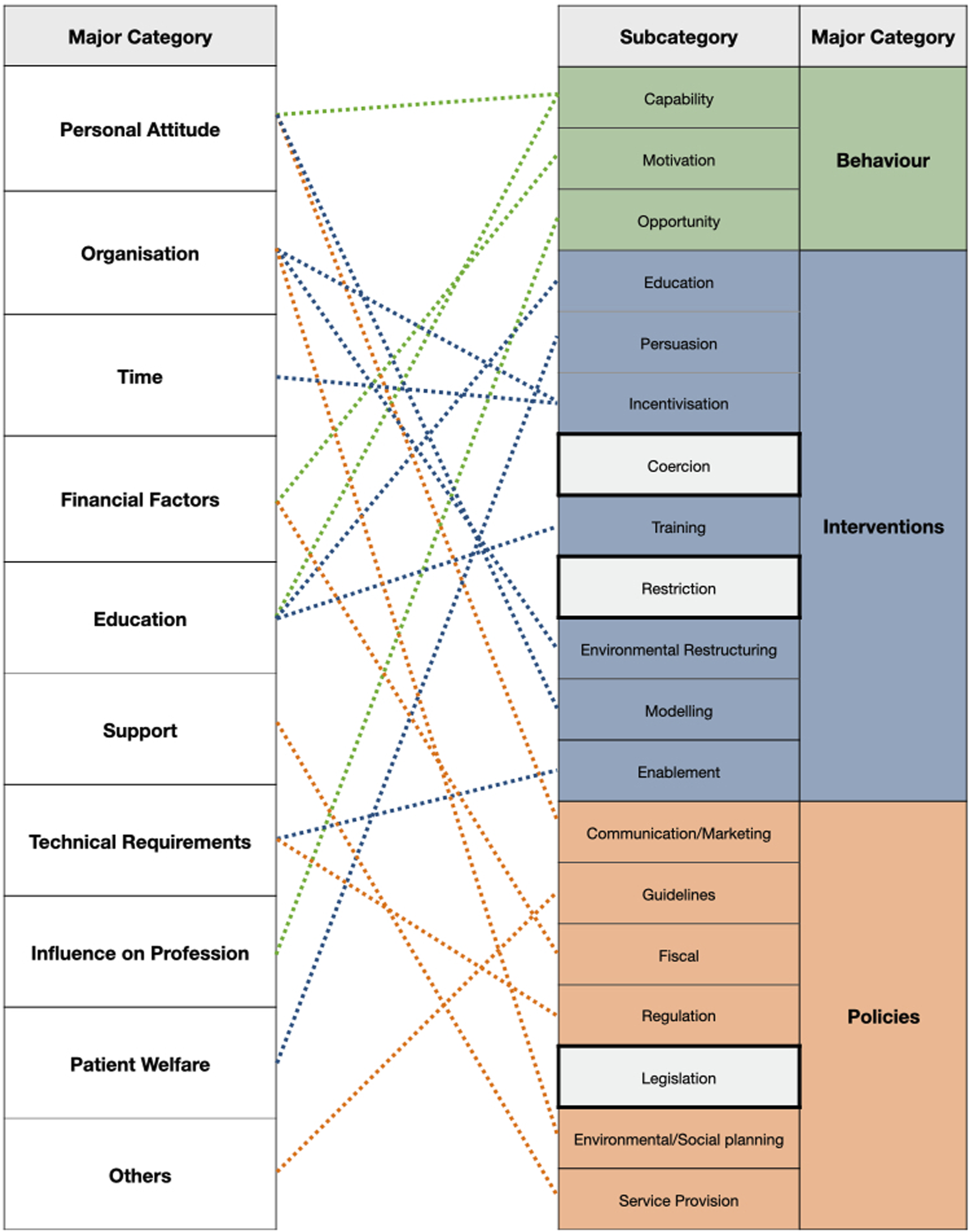
Determinants of the implementation of AI-assisted screening tools in primary care compared to the Behaviour Change Wheel (on the right side).
Personal attitudes
Positive and negative personal attitudes to digital technologies were described as essential influencing factors for implementation. Many participants stated that the internet and smartphones play an important role in their daily lives, both in their private lives and at work.
“A big (…) daily importance, I couldn’t imagine my life without it.” (TN1, GP)
Examples for the various uses of a smartphone in a professional context.
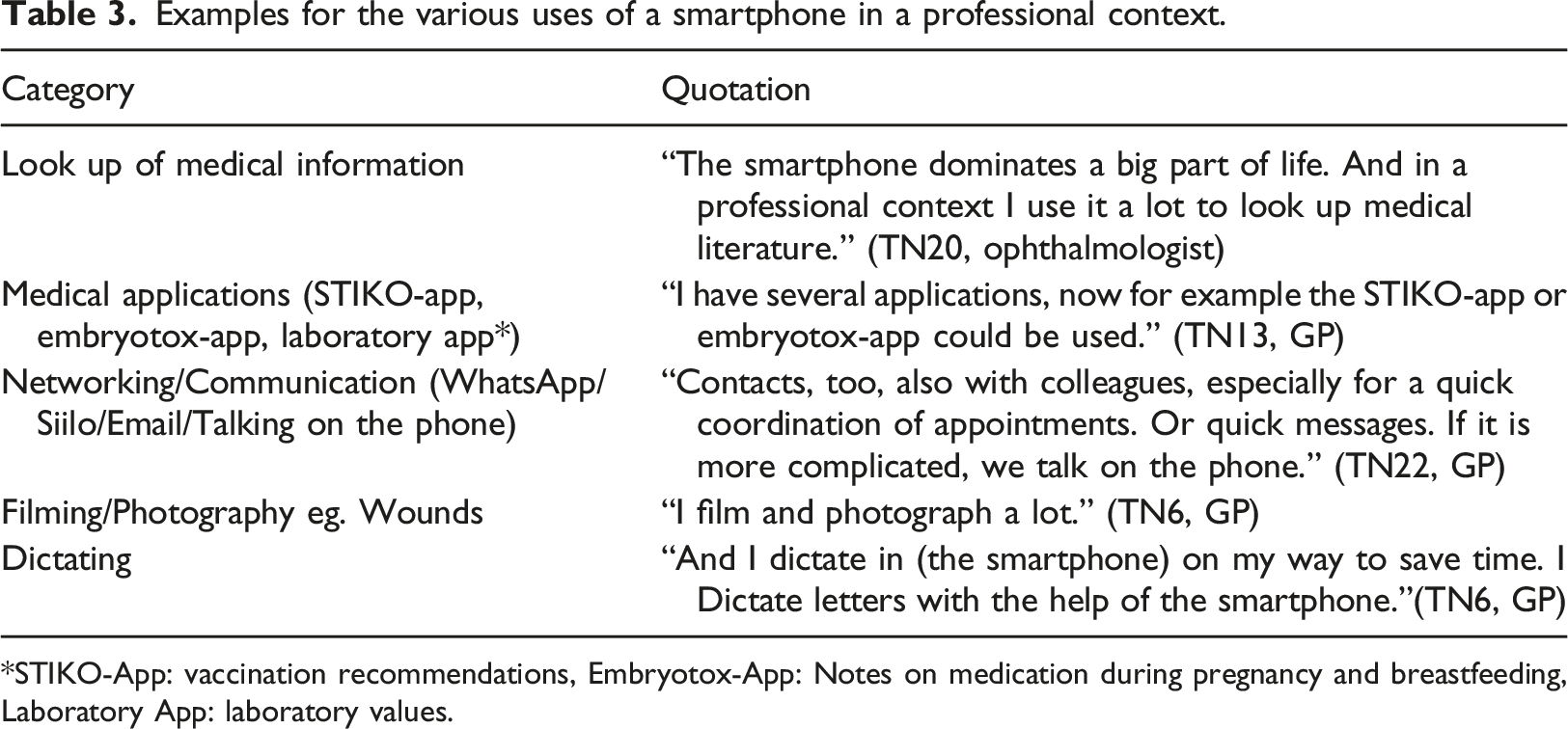
STIKO-App: vaccination recommendations, Embryotox-App: Notes on medication during pregnancy and breastfeeding, Laboratory App: laboratory values.
Furthermore, experience with the implementation of other digital technologies e.g. telemedicine into primary care was mentioned as potentially influencing the attitude to digital technologies. For patients who lived far away from the practice and in the sense of a preliminary stage of conventional medicine, this technology was seen as an enrichment by professionals. Negative experiences with telemedicine were explained with, for example, frequent technical difficulties, a high expenditure of time and little demand from patients for this technology.
The attitude towards AI in medicine was also mentioned as a potential influencing factor. Negative attitudes in this regard were justified for example with a suspicion that there is a lot of commerce behind such AI projects. Scepticism about the quality of the result by an AI was another reason. The fact that one could not fully understand how AI arrived at its result seemed also to play a role. With regard to the automated evaluation of an e.g. which was given during the interview as an example for a form of AI, many participants stated that in their experience the quality of evaluation was often low. At the same time, the question arose as to whether the analysis of an ECG or the retina with the help of algorithms could actually be assigned to the term “artificial intelligence.” “Is an ECG that produces a diagnosis already artificial intelligence? I would doubt that. (…) Intelligence can decide and intelligence also learns. And (…) intelligence also discusses.” (TN18, GP)
Also, the concern was expressed that it could be dangerous to rely too much on AI and that, as a doctor, competencies that actually belong to this profession are at risk. It was stated that the AI cannot take into account the patient holistically like a doctor can. With a strong sense of responsibility on the part of GPs for the well-being of their patients, participating family doctors described an uneasy feeling about advising patients on such a basis. On the other hand, the possibility of making care more cost-efficient was cited as a reason for a positive attitude towards AI. Some participants indicated that AI could be useful, especially in the situation of a screening and therefore a pre-selection of patients. A useful example mentioned from everyday practice was that MAs would get to the doctor more quickly if the automatic interpretation of the ECG device showed an abnormal finding.
“Let’s say we want to do a prioritisation beforehand in emergency rooms, so that a nurse takes this result to sort the patients. That’s how I can imagine it but in the real question of a doctor-patient-relationship, I think the doctor should do the analysis.” (TN18, GP)
Another sub-category is the determinant of openness towards new procedures. Here, the participants addressed the general openness of GPs and practice staff to try out and use new procedures. This also includes a certain curiosity and patience of the individual. “The acceptance of really learning and applying new things again. That could be a barrier.” (TN9, MA)
Organisation
The main category of organisation includes various aspects of further planning for the integration of the new screening examination into everyday medical practice. On the one hand, it is associated with the need to change established practice processes in favour of the new examination. Since it is an additional examination, it must be ensured that the examination runs smoothly so that the practice is not held up for long.
“As with the video consultation, this is also a structural change that you should or must remember at the moment. You definitely have to structure processes.” (TN11, MA)
“It could even be delegated, the doctor wouldn’t have to do it himself, it could also be done by the medical assistant.” (TN23, Ophthalmologist)
Another aspect that affects the organisation of the practice to the outside addresses the creation of established treatment pathways between general practitioners and ophthalmologists. Here, the focus was set on the communication, the agreements and the organisation of the processes between the two practices.
“Even if it is already a well-rehearsed team, the general practitioner could always refer to the same ophthalmologist, then there could also simply be collegial communication. (…) if there is a suspicion that something is going on, it should be definitely referred to the ophthalmologist.” (TN23, Ophthalmologist)
Time
The duration of the examination as well as the latency to the test result after the screening were described as relevant factors for implementation. Another advantage of carrying out the screening in primary care would be that GPs would not have to wait for the result or to call the ophthalmologist afterwards if the patients forgot to bring it with them. “It would make it easier because I don’t have to chase after the ophthalmologist’s report myself if the patient has forgotten it.” (TN17, GP)
Financial Factors
A major category that was often mentioned was the financial factors. In particular, in absence of an appropriate remuneration, a successful implementation was considered unlikely by the participants. A participant suspected that GPs might be exploited by such a commercialised project in a way that they work hard and ultimately hardly earn anything themselves.
“Actually we don’t profit from it and the start-up or the large corporation somehow gets 50 euros per photo.” (TN3, GP)
On the other hand, low acquisition costs, low running costs and the simplicity of billing were seen as supportive for implementation.
“In the end one has to say that we can make the best medicine, but (…) with Covid for example, there were seven billing systems for a single examination, with three different cost bearers to whom we had to submit it. This is doomed to fail from the start.” (TN18, GP)
Education
Under this main category, aspects related to education and training have been summarised. Some GPs described an insecurity or lack of knowledge regarding ophthalmological topics.
“I have to honestly say that to myself, I’ve been general practitioner for 40 years, I don’t really trust myself to do that anymore, that’s a [considering noise], a long-ago part of the training.” (TN18, GP)
A MA believed that uncertainties or reluctance to use this technology would regularly resurface among team members. She had experienced this phenomenon already with telemedicine although her practice used a lot of video consultations. Participating ophthalmologists estimated that the handling of the device could be learned within a week in form of a training. Others, however, were more sceptical due to technical challenges and comorbidities.
“One always imagines that it's nice, but (…) that it really works with narrow pupils, for example people often have a slight clouding of the lens, a cataract. The pupils of diabetic patients also typically dilate less well or are often a bit narrower at times and stuff like that. So that the image quality is not quite sufficient.” (TN12, Ophthalmologist)
Support
In this main category, support offers for implementation in general practice were classified. The participants wished for support offers in case of medical or technical problems or questions that arise during implementation.
“First of all, you should have low-threshold support. If it doesn’t work, that you can get help immediately.” (TN22, GP)
Technical requirements
According to the seventh question in the interview template, this main category dealt with the technical challenges in the development of the diagnostic tool. Numerous inductively generated subcategories or determinants could be assigned to this deductive main category. A practical and user-friendly screening device and software was primarily requested by numerous participants.
“(…)if you had such an error message popping up all the time somehow every third time or so, then at some point you wouldn’t want to anymore.” (TN8, GP)
As part of this aspect, a certain robustness, handiness, and disinfection ability was mentioned. Also, the patient data input into the diagnostic software should be of short duration. Ideally, the software should directly fetch the patient data from the practice information system. Likewise, the installation of the software as well as the integration of the software into the current practice information system was demanded to be simple. In this way, the fundus images or preliminary diagnoses could be easily called up in the digital patient file after the examination. The precondition for this would be that the interfaces work well.
“And I also see from other experience that there is often an interface problem. That means that the data that is obtained via my smartphone analysis cannot be entered into my system, that I can also access it well.” (TN4, GP)
Many demanded compatibility with different smartphone systems, e.g. Android, IOS. The desire for a cloud-based solution and the possibility to transmit data directly to the ophthalmologist was also mentioned. In addition, the AI should be able to automatically analyse the image quality. From the point of view of participating ophthalmologists, several technical challenges to acquire high-quality retinal images would have to be faced: The fact that the image is generated without the aid of a mydriatic makes it more difficult to capture a sufficiently large section of the retina. In the case of a patient clientele suffering from DM, frequent comorbidities such as cataract were also assessed to further complicate image creation.
“At an older age, patients often have comorbidities, such as a cataract, so that such simple solutions with a cell phone camera are probably a bit more difficult.” (TN23, ophthalmologist)
Furthermore, it was rated as an enabling factor for the implementation if the diagnostic tool would recognize more than one disease.
Influences on the profession
According to the sixth key question, this main category deals with the influences on the profession of a GP. Various determinants could be subordinated to this deductive main category, which in turn could be divided into positive and negative influences.
A positive influence and thus a supporting factor for the implementation could be an extension of local care and expertise and the projection of a modern image through the usage of new technologies. Some participants believed that the implementation of AI screening could mean an image boost for a practice.
“The more technical and the more colourful blinkers and beeps and flicker, the more patients think that’s cool. And in this respect, such a technically popped-up procedure at the family doctor could rather contribute to an image boost.” (TN6, GP)
Another mentioned factor was that it might add to the GP’s role of a mediator. Even though he may not be the one who conducts the examination, nor the one who evaluates it, he remains the one who discusses the result and its individual significance with the patient and is available for questions of understanding.
On the other hand, potential tensions between GPs and established ophthalmologists were deemed possible by both sides due to potentially lower referral rates to the ophthalmologist if DR screening was implemented into primary care.
“What is of course also viewed with a bit of scepticism among the ophthalmologists is when, as a result, far fewer people are referred to the ophthalmologists and the general practitioners think that they can do everything themselves.” (TN23, ophthalmologist)
Some stated that the collaboration between ophthalmologists and GPs was already not ideal from the beginning. Reasons were a long latency until GPs receive the fundus examination results, a lack of exchange between the specialities and the use of many specialist terms by ophthalmologists which are incomprehensible to GPs. Others, on the other hand, regarded this situation as an opportunity for closer and better cooperation between specialists and GPs. Another concern was that GPs might feel incapacitated by the use of AI for diagnostics.
“The question is what does it do with the profession? Do you feel restricted (…) in your professional practice or patronized by artificial intelligence(…)in the diagnosis?” (TN1, GP)
Some believed that it would have little impact.
Patient welfare
Under this main category were brought together determinants affecting the patient’s well-being. These can be divided into positive and negative consequences:
A strengthening of the primary doctor’s filter function and an associated closer relationship between patient and GP was anticipated and assessed as a positive influence. The chance of a more targeted referral to a specialist was also mentioned.
“I think that would be rather positive, because the general practitioners (…) can perhaps also collect preliminary findings in a more targeted manner and also refer them in a more targeted manner.” (TN8, GP)
This could reduce the pressure on ophthalmologists and provide them more time for more complex cases. Depending on the degree of morbidity and mobility of the patients as well as the distance to the ophthalmologist practice, an inclusion of DR screening in regular visits with the GP could save a lot of effort for patients and their caring relatives.
“The effort is often associated with a lot of time for the patients (…) it usually takes a long time, then with the wide drop, then they are not allowed to drive themselves, then they have to organize someone.” (TN17, GP)
Therefore, more patients could potentially be reached overall compared with the current screening system at the ophthalmologist. This way, a regular screening of DR could be ensured and patient care in the long run could be improved. Another factor that was mentioned by some GPs is that a DR screening in primary care could prevent patients from being charged additional costs by offering a list of individual health services (IGeL-Leistungen) that are not necessary from an evidence-based medicine point of view.
“Every time we send a diabetic patient there, we make the ophthalmologists’ cash register ring by individual health services he additionally sells, and we don’t want that anymore. (…) for me it is part of patient protection.” (TN6, GP)
On the other hand, a concern raised was that pathologies that would have been detected by the ophthalmologist could be missed by the device.
“It would have to be ensured that the patient’s care is not worsened to the point that an ophthalmologist visit which also reveals other pathologies is cancelled altogether.” (TN23,AA)
Other
Three individual inductive determinants that could not be assigned to the other main categories were summarized under this main category. Many participants called for studies that demonstrate high validity of the screening device.
“If that really is a valid instrument, I think it would be adopted.” (TN9, MA)
At the same time, scepticism was expressed as to whether an AI screening with the family doctor could replace the appointment with a specialist in the first place. Furthermore, the question of how to deal with legal liability issues was raised. In the event of possible harm to patients, the concern was also mentioned that patients could subsequently sue and hold the GP responsible.
“That (…) the communication also has a certain legal basis. (…) questions will arise. The patients say: Yes, I was with you and had it done and now I suddenly have the eye disease. So what do we do now? Who did what wrong?” (TN24, ophthalmologist)
Some participants mentioned data security concerns related to smartphones.
Ideas for implementation strategies
Some participants made concrete suggestions for an implementation into primary care: With regard to the organisation of the screening, it was suggested that a planned alternating system could be introduced, with regular, annual check-ups at the family doctor and fixed, scheduled multi-annual check-ups at the ophthalmologist, regardless of inconspicuous findings by the AI.
“You could also say that we take the AI after a year and after 2 years in the general practice with a report to the ophthalmologist, we go back to the ophthalmologist’s practice. And only then after 3 years does he go back to the ophthalmologist.” (TN24, ophthalmologist)
The demand arose that in the planning of such a screening, everyone affected should be involved from the outset. This should also include: The KV, health insurance companies, the state, the university, etc. Another idea when planning the implementation was that such a diagnostic tool should be made available mainly for practices specialising in diabetes, preferably in rural areas, for reasons of cost effectiveness since it would not be worthwhile if the number of patients with diabetes is small.
“I would do a cut (…) for the first introduction. These devices are made available to all general practitioner practices and practices specialising in diabetology that want to take part and treat at least 50 diabetes DMP patients per quarter. And then I would probably bring in a country doctor aspect for the time being for cost reasons.” (TN3, GP)
The idea came up that it would be financially attractive for a GP’s practice if such a screening device could be borrowed, for example from the KV.
“Well, I would also rather present it as a rental device that is provided by the KV or something like that. That as a practice you are not responsible for the costs of these things.” (TN3, GP)
Alternatively, it was proposed that financial incentives are provided by the KV to increase the motivation for GPs. The offer of further training on the topic of DR and fundus images to expand the background knowledge of GPs might help overcome the insecurity GPs perceived when being confronted with ophthalmological topics. Accordingly, training in the handling of the device could also lead to greater routine and reduce the feeling of insecurity when using it. The desire for a specific IT contact person as well as a contact person with ophthalmological expertise became loud.
“That when you do something like that, you have a specific contact person who is easy to reach.” (TN8, GP)
Additionally, it was rated as helpful that a person should accompany the first few days of practical operation of the diagnostic tool in the practice.
Discussion
This study provides insight into perceptions of different healthcare professionals about the implementation of AI-assisted screening of DR in primary care. We have identified the following main influencing factors to implementation: personal attitude, organisation, time, financial factors, education, support, technical requirements, influence on profession and patient welfare.
When comparing the established barriers and enablers of our study to an established implementation model, the behaviour change wheel (See Figure 1), it can be stated that except for the categories restriction, coercion and legislation, all categories could be relocated in the model. 33
As for the missing categories, it is possible that they play a minor role in the implementation of this specific technology in the context of current healthcare policy. Another reason why coercion was not named could be an increased internal resistance of physicians when they feel too pressured from the outside to change something. This phenomenon has been witnessed during the implementation of Electronic Health Records (EHR) in the USA. It was observed that physicians’ willingness to adopt such systems decreased while the government’s efforts to promote the use of EHR technology increased. Some physicians perceived the implementation as a possible threat to their professional autonomy. Also, they were concerned that “policymakers, insurers and administrators would use EHRs as a proxy mechanism to influence, restrict, or dictate how medicine is practised.” 34 Furthermore, changes are more likely to succeed “when health care professionals have the opportunity to influence the change, feel prepared for the change and recognize the value of the change, including perceiving the benefit of the change for patients.” 35
Even though this screening is a specific example of a new technology that might be implemented into practice, it raises general discussions about the benefit and danger of digital technologies in medicine. Attitude and experience with digital technologies seems to play a vital role in the course of implementation. 36 In former research, medical professionals feared that technology might interfere with their ability to make independent diagnoses and their relationships with patients, whereas other medical staff welcomed it because it provided them with more opportunities to interact with patients and their carers. 37
In particular the use of AI for analysis in medicine remains a controversial issue, in our study as well as in literature. 38 In a Korean study, it was acknowledged that AI would not be able to assist in unexpected situations owing to inadequate information. Additionally, less than half of the participants believed that AI was diagnostically superior to human doctors. 39 A reason which was suggested by participants of our study for a negative attitude to AI was that the less the user comprehends the process by which a machine arrives at a certain result, the stronger the feeling of insecurity towards the diagnosis becomes. This is also called the “black box phenomenon”. 40 Doctors, therefore, could experience difficulty in delegating expertise and responsibility for their patients’ welfare to a machine. Another factor which might add to the potential scepticism of GPs is that AI can only detect what it has been trained to do and lacks the ability to consider the patient holistically.
Furthermore, it was questioned in the interviews whether the screening in this study can actually be assigned to the term “AI.” Other forms of automatic analysis exist in health care, such as blood analysing devices, that doctors are confronted with on a daily basis. Despite the fact that the analysis is undertaken by a machine and the results have an influence on the decision of the treatment for each individual patient, blood test results are randomly challenged. To find an answer to the question where AI begins and where it ends, an exact definition of ‘AI’ is required. However, even though it is a term which is broadly used, it becomes apparent that no exact definition exists. 41 In some cases, AI is interpreted as ‘anything associated with informatics’, whereas in others a precondition is that it is a self-learning system that is always in development and that is capable of making considerations like real intelligence is. This phenomenon is already known from other terms such as “big data”. 42 It therefore remains difficult to find a clear answer to this issue.
Organisational factors are found in various studies as being an important determinant in the course of implementation. Some studies reported that healthcare workers may be resistant to accept a new advanced technology as it may change work processes and routines, in fear of a potentially heavier workload. 43 In other studies, time as a determinant to implementation was categorized under organisational factors. The reason we created a separate category was because it was often separately highlighted by our participants.
Furthermore, a lot of evidence can be found which proposes that in order to incentivize healthcare workers to use AI, current payment systems should be reviewed. Adequate funding can overall encourage healthcare organisations as the introduction of AI requires significant financial expenditures and changes to operational processes.44,45 These results are in line with our study.
One of the most frequently reported factors influencing the implementation of AI tools is the education as well as the competencies of healthcare workers as end-users. 44 According to the interviewed health care professionals, most individuals are already familiar with the handling of a smartphone, both in a private and professional context. This can be confirmed by other studies46,47 and could be an explanation for why many participants did not perceive the training of the handling with the smartphone to create retinal images as a significant barrier.
Many studies also confirm that technical factors, especially problems of interface between new and existing IT, seem to be a common source of complications during implementation of a new technology.45,48
An additional reason to add to barriers in intersectoral corporation has been given by study participants. The circumstance that GPs feel that ophthalmologists speak a different language can also be confirmed in literature: A study with GPs found that more than half of the acronyms in common usage by ophthalmologists were either unknown or misunderstood by the GPs. 49
A clear legal framework and certain political decisions, particularly in relation to data protection and liability issues, are essential factors for successful AI adoption.50,51
Even though we found that validity plays a role during implementation, a study in primary care discovered that instead of relying on explicit evidence from research directly, physicians were influenced more by their own and their colleagues' experience, interactions with opinion leaders, patients, and pharmaceutical representatives, and other sources of mostly tacit knowledge. 52
According to the results from our study and what we can learn from research on the topic so far, the following strategies to overcome some of the identified barriers seem to be essential before or during implementation:
To meet concerns about the validity of an analysis by AI instead of an eye specialist, several strategies could be undertaken. One idea is an alternation of a DR screening in primary care with a screening at the ophthalmologist. Thereby, patients would attend a screening with the ophthalmologist e.g. every three to 5 years and in the meantime, the screening could take place at the GPs practice. Here, the risk of overdiagnosis has to be taken into account. The fear of a false diagnosis by AI might be attempted to be reduced by shorter intervals of screening in the beginning of implementation. This, however, might lead to more false-positive results which need to be re-examined by the ophthalmologist and create an unnecessary feeling of insecurity for patients due to a false-positive pre-diagnosis. 19
Another strategy could be to emphasize the point that the use of AI is a question of indication. In the case of a screening situation, AI might constitute a comfortable, cost- and time-efficient solution for a pre-selection of patients. 53 The final diagnosis, however, is to be determined by a doctor. Moreover, the GP remains irreplaceable in the role of a mediator who can explain the result of the AI to the patient. This might also reduce the feeling of incapacitation of GP due to transfer of competencies to AI. During implementation it is important to consider further potential positive and negative consequences to the profession as well as patient welfare which matters to physicians and future users of the new technology. Former research showed that healthcare professionals were more resistant to using AI if they did not see the benefit for the patient outcome. 54
The concept of “Explainable AI'' which refers to modelling and reasoning tools to explain why and how a result was achieved could be a means in the development of AI to improve the feeling of trust of GPs to the prediagnosis of the AI. 55 Nonetheless, a recent study cautions against having explainability be a requirement for clinically deployed models. The study provides an overview of current explainability techniques and explains several examples how they can cause problems in the process of clinical decision making instead of inducing trust, providing transparency and reducing bias as originally hoped. Alternatively, a rigorous internal and external validation of AI models is advocated to engage trust with the health-care workforce. 56
At the same time, it appears to be essential to include all participating parties in the process of implementation from the beginning. 57 To prevent tensions between ophthalmologists and GPs, it is essential to enhance communication between both parties and to share information. It should be communicated that a DR screening in primary care should not be a replacement but an addition to the existing system. Moreover, its potential should be emphasized: this screening method has the capacity to reach patients that would otherwise not have attended a screening, improve screening intervals, prevent unnecessary additional visits to the doctor for patients and to provide useful referrals of patients to the ophthalmologist by a preselection of patients in primary care. Therefore, a consensus between primary care physicians and ophthalmologists is the basis to gradually change the existing system together and to develop new adapted established pathways.
To relieve concerns due to financial factors, it is important to offer appropriate financial incentives to GPs. In order to prevent frustration due to technical problems during the use of the device, developers should collaborate with future users prior and during implementation.
In order to meet the described feeling of insecurity that some GPs feel when being encountered with medical expertise they do not need in their daily routines, it is necessary to provide training in the handling of the device and information on the disease of DR and the possible accompanying changes of the ocular fundus. On top of increasing skills in the handling of the device, training together as a team can deepen trust in each other and consequently lead to improved teamwork.44,58 Another strategie could be to provide support during implementation, in case of technical or ophthalmological questions and problems with the integration in practice procedures.
Considering high financial expenditures and a necessary routine in the handling of the device which can solely be established with frequent use, it seems logical to implement the screening device with priority in practices with many patients with DM.
Some limitations exist to this study. First, the fact that two thirds of the interviewed people had experience with telemedicine shows a certain selection bias of the interviewed participants. Thus, there might be an overrepresentation of people who have a positive attitude towards the topic. Prior to the interviews, an attempt to minimize this bias was made by emphasising that participants with positive or negative attitudes to the topic are equally welcome to participate.
Second, most participants did not have experience with AI in their daily life. Even though some general information on the research question was given prior to the interview, the term “AI” was intentionally not further defined in the interviews to minimize any bias from our side. Therefore, the participants’ idea of what is meant by that, might have varied. Furthermore, the described screening examination does not yet exist in Germany so the interviewed individuals were forced to use their imagination to picture the process and the factors which might be of importance. This element of required imagination could have led to misunderstandings.
The results of a qualitative study on expectations and experiences of eHealth in primary care indicates that there do exist big differences between patient and GP viewpoints on various issues concerning future eHealth services. 43 In this study, only the view of professionals was addressed. To complete the iterative implementation process, the patient view is essential to include. However, the willingness of professionals to implement an AI screening is the condition to make patients experience such a screening in the first place.
Whilst this study does exhibit limitations, several strengths exist. To our knowledge, this study is the first to explore the barriers and enablers of AI-supported screening of DR in primary care. It also takes into consideration multiple perspectives.
Conclusion
In summary, an AI-assisted screening of DR in primary care is a new idea in the healthcare domain that has not yet found its way into practice in Germany.
Potential influencing factors to implementation include the personal attitude to digital technologies and AI, the organisation of practice processes, established treatment pathways between GPs and ophthalmologists, time factors, financial factors, the education, a provision of support in the event of problems or questions, technical requirements of such a screening device, the influence on the profession and patient welfare. Several ideas of implementation strategies can be derived from these determinants.
Further studies on the implementation of AI-supported screening devices for DR in primary care are needed. In the sense of a holistic approach, it is essential to include different perspectives in the development of e-Health services. Especially a study that explores the patients’ perspective is needed because they might have opinions that are not known by healthcare practitioners. Furthermore, a quantitative study could help to establish a further prioritisation of the various determinants. This might improve understanding about how to proceed in the daily practice of e-health solutions in medicine.
Footnotes
Acknowledgements
The authors would like to thank all healthcare professionals who participated in this study.
Author contributions
The first author conducted the interviews and wrote the initial version of the manuscript. The first and second author performed the data analysis, supervized by the third author. All authors contributed substantially to the manuscript and approved the final version for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: his research was funded by the Joachim-Herz-Foundation within the PASBADIA-Project (grant no. 120002).
