Abstract
Digital medicine programs (DMPs) are emerging technologies that use sensor-enabled medicine to detect when patients have taken their medication and then provide feedback about adherence. We use qualitative methods to understand how patients change their behavioral patterns while participating in a DMP intervention. An influence diagram outlining the factors hypothesized to affect adherence in DMPs constructed from prior scientific research and expert input was created. Subsequently, we conducted semi-structured interviews with 10 patients to see if their experience supported the relationships outlined in the model. We identified three pathways by which DMPs are likely to change behavior around medication adherence: (1) providing patients and providers with accurate, personalized information about adherence; (2) improving patient–provider interactions by structuring them around this information; and (3) facilitating routines and habits for medication use. Chronically ill patients often fail to adhere to drug regimens. Patients in a DMP intervention used the DMP-provided information to better understand drug efficacy and collaborated with their physician to develop adherence strategies. DMPs can promote medication adherence among patients who are willing to use them and may be most effective if physicians are active partners in the DMP.
Introduction
Non-adherence is recognized as a public health problem by the World Health Organization, 1 occurring in up to 50% of patients prescribed medications, even for conditions in which missing a single dose has serious health consequences.2–4 For US patients with chronic conditions that require long-term therapy, such as hypertension, hyperlipidemia, and diabetes, almost one third of patients do not fill their initial prescription (28%, 28%, and 31%, respectively). 5 For those who initiate medication therapy, adherence declines considerably over time.6,7 Among hypertensive patients, as an example, poor adherence is associated with more coronary heart disease, heart failure, and cerebrovascular disease.8–11 Poor adherence also has significant economic costs for healthcare systems, in hospital admissions and readmissions, and for individual patients, due to increases in service utilization.12–14
Medications incorporating ingestible sensors offer a new approach to improving adherence. When patients take these medications, sensors send a signal that is communicated to patients and providers. Preliminary evidence suggests that such digital medicine programs (DMPs) improve adherence,15–18 yet little is known about how DMPs affect adherence behavior, nor about the way they impact patient–provider interactions.
As an example, Proteus Digital Health® is a DMP that includes (1) an ingestible transmitter co-encapsulated with medication, which sends a signal when ingested; (2) an adhesive wearable patch that detects that signal, as well as physiological measures and physical activity; and (3) information provision through a patient app and provider portal, which can be integrated with electronic medical records. The patient-facing app gives patients the ability to access immediate feedback about their medication-taking behavior. Figure 1 illustrates the DMP monitoring and communication process for a hypertension medication. Preliminary results among patients with uncontrolled hypertension and diabetes found that 84% of those who used the DMP met their blood pressure targets after 4 weeks, compared to 33% of usual care patients.
17
DMP for hypertensive therapy, modified from Noble et al. [2016].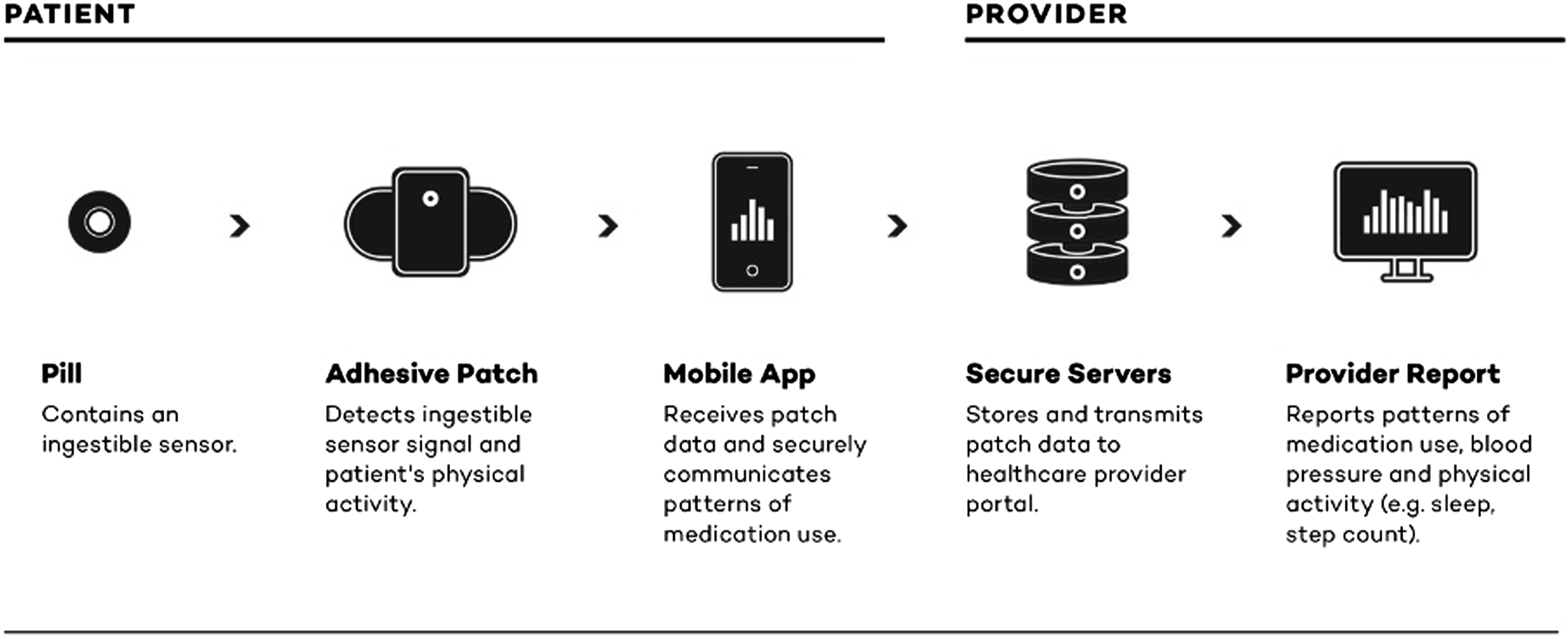
The aim of this work was to develop a conceptual framework based on existing literature, augmented by expert and patient feedback, to understand how DMPs influence adherence. Our initial analysis of how DMP affects patient adherence behavior focuses on a patient’s decision to take the Nth dose of a prescribed medication to treat a chronic disease. The influence diagram that resulted from this analysis was then reviewed and augmented by a set of adherence experts. We then interviewed 10 patients who participated in the Proteus Digital Health® DMP described in Figure 1. All procedures were approved as exempt research by the Western Institutional Review Board.
Methods and materials
Influence model for adherence behavior and DMPs
Drawing on several systematic reviews18–21 and other peer-reviewed studies,22-42 we created an influence diagram of the factors that had empirical or theoretical effects on adherence. We then reviewed the model with a set of experts (researchers and practitioners), including three who worked for Proteus Digital Health® and were familiar with the company’s DMP, three clinical experts working with patients that have used the DMP, and three academic researchers with expertise in medication adherence. These experts were referred by Proteus Digital Health®. Interviews were conducted using video or telephone conferencing, and the audio was recorded for each interview.
Experts were shown the initial influence diagram that included variables affecting each of three adherence pathways: intentional factors, non-intentional factors, and habit formation on behavioral maintenance of adherence. What emerged was a consensus that ingestible sensor-enabled medications are most likely to result in long-term adherence through habit formation.
Research has long focused on patient-related factors that shape both intentional and unintentional non-adherence. More recent work has focused on habit formation, where intentional adherence transforms into automatic behavior. 42 Specific to DMPs, experts who reviewed the model postulated that DMPs allowed providers to address gaps in patient understanding of their condition and treatment. One expert suggested that DMPs enable physicians to review adherence patterns with patients and discuss the effects of taking some doses at the wrong time or skipping them altogether. Another expert mentioned that receiving accurate monitoring information could improve patient understanding of the disease processes. Additionally, using digital medicine with direct patient feedback facilitates personalized care and makes unintentional lapses in adherence visible to providers and patients.
Another DMP-specific contribution highlighted by experts was the way it could facilitate the development of coherent beliefs about the medication through accurate and personalized feedback. For example, DMPs allow patients with asymptomatic conditions to track the relationship between their adherence and biomarkers, such as blood pressure, a connection that is difficult to make in the absence of an accurate tracking mechanism. Experts also reported that patients may feel a sense of positive reinforcement from this feedback, either from knowing that they are doing the “right” thing, or through simple representations of consistent adherence, such as check-marks or streaks presented in the app. Finally, the accuracy and instantaneous nature of the data can also make patients acutely aware that they are being monitored. Hence, for patients using a DMP, motivation is likely to reflect both the internal drive provided by empowerment and the external drive provided by awareness of being monitored, akin to a Hawthorne effect.43,44 The results of this initial review by experts were incorporated into Figure 2. Adherence influence diagram.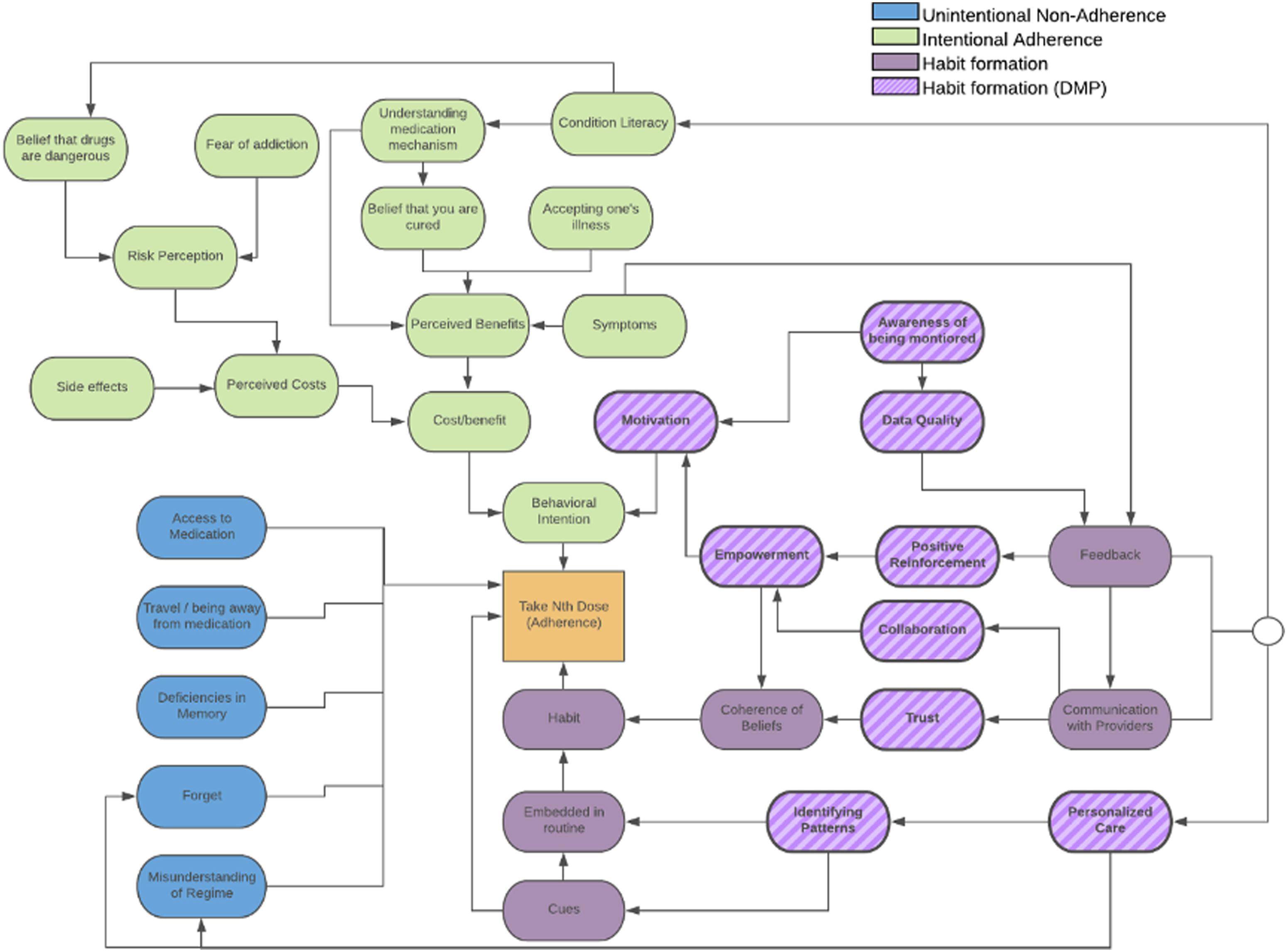
Patient interviews
We conducted semi-structured interviews with patients who opted into participating in a DMP intervention using Proteus Digital Health® (which we refer to as the DMP intervention). These interviews were structured around the influence diagram shown in Figure 2. The interviews were designed to examine whether the potential relationships identified in the influence diagram were supported by the experience of patients using the DMP. The interviews began with open-ended questions, encouraging participants to share whatever was on their minds, then proceeded to ask questions increasingly focused on specific elements in the diagram. Throughout the interviews, participants expressed themselves in their own words, with the task framed as the researcher enlisting their help to improve medical services. The audio of each interview was recorded.
Participants
All interviewees were recruited from a healthcare practice administering a prescription management program in California. The DMP was made available to eligible patients from this program with one or more chronic diseases. In order to be candidates for the intervention, patients had to (1) be taking one or more prescribed medication for diabetes, hypertension, or high cholesterol; and (2) be identified, by their providers, as having high disease risk, as reflected in elevated A1C (blood test that measures average blood glucose level over the past 3 months), blood pressure, or low-density lipoprotein levels (measure of harmful cholesterol levels). The pool of participants had been using the intervention for at least 1 month and had been identified by providers as having problems with adherence. Researchers received a contact list of 57 patients from the healthcare practice that met the recruitment criteria. Recruitment was completed by calling patients from this pool and asking if they would be interested in participating in the study.
Interviews were conducted in-person or by telephone between April and August 2019. Interviewees were compensated USD 50 for their participation for approximately 1 h of their time. Our sample included eight men and two women. Their ages ranged from 51 to 85 years (mean = 69.7), consistent with the ages of patients most affected by these chronic conditions. About two-thirds (65%) identified themselves as non-Hispanic Caucasian, 30% as Hispanic, and one as African American. Most (70%) indicated an educational level of “Some College” or higher. Participants had been using the intervention between one and 3 months and were planning to continue to use the intervention while completing the study.
Interview procedures
The interviews consisted of open-ended questions, formulated to suggest potentially relevant topics, but not desired answers. They started broadly (e.g., “Can you walk me through the steps of a what a normal day looks like for you?”), then became increasingly focused on adherence (e.g., “Are there any activities that get in the way of you taking your medication?”), the ingestible sensor-enabled medication (“Can you tell me more about why you decided to take sensor-enabled medications?”), and interactions with their providers (“Is drug adherence a topic you have discussed with your provider?”). If necessary, answers were followed up with prompts for clarity (e.g., “How does that work?” “Can you explain what you mean a little more?”). Figure 3 illustrates how sample questions and follow-up questions map onto the influence diagram. Sample questions and associated links.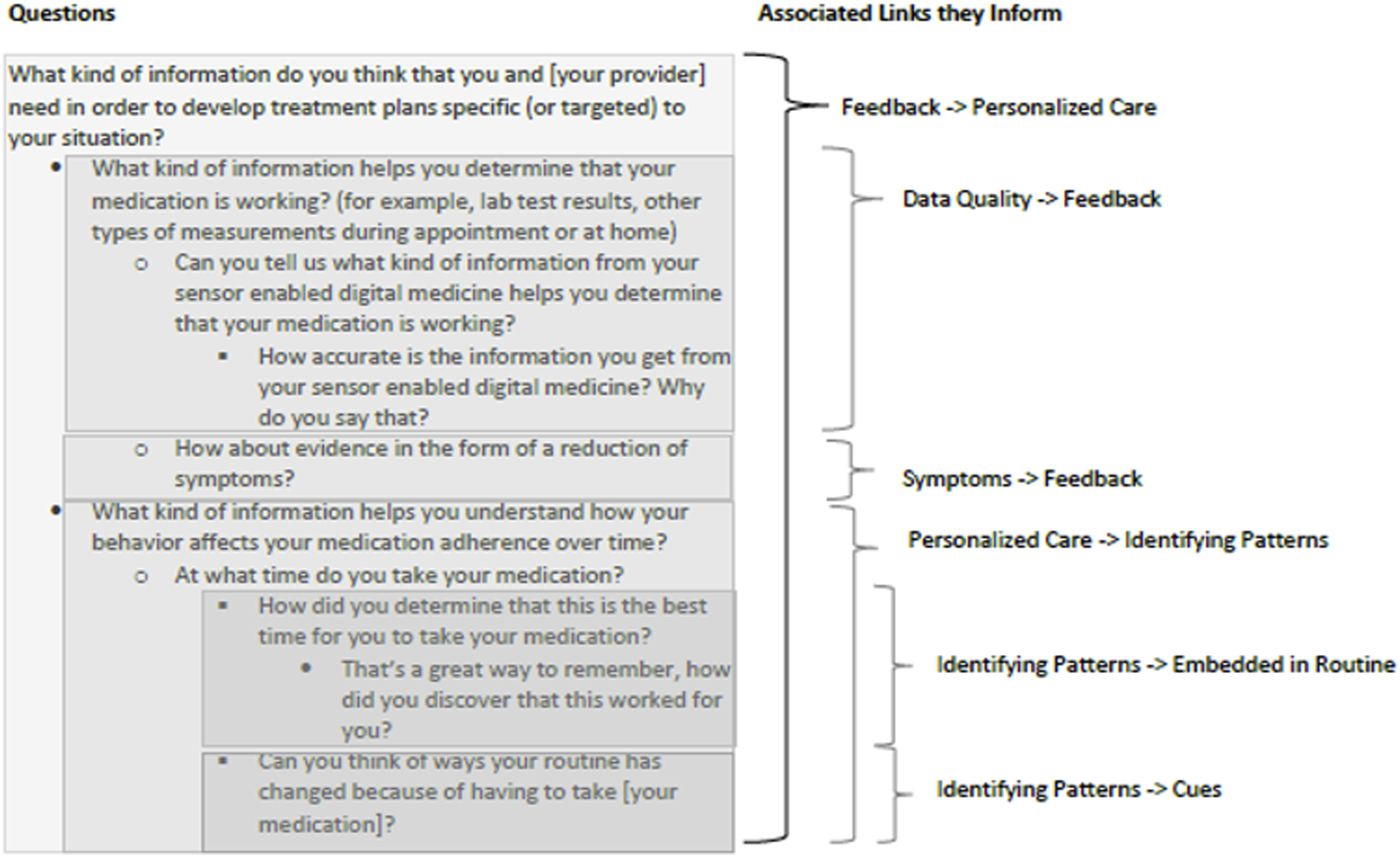
Analysis
The recorded interviews were transcribed verbatim. An initial codebook was developed to map the themes outlined in Figure 2. For the analysis, a behavioral decision research approach was applied to identify any themes or beliefs that diverged from this codebook. This approach uses a systematic method to assess the information needed to make informed decisions and as a result identify how to reduce barriers to desired action. 45 The qualitative coding analysis in this approach is most similar to thematic content analysis. The corresponding author reviewed and read the interview transcripts several times to determine if new themes that emerged after each interview. The final codes were developed after the interviews were completed. The analysis did not show any divergence based on the amount of time the intervention had been used or any other variable, including gender.
Results
Participants described their adherence decisions in terms of the perceived risks, costs, and benefits of non-adherence. For example, they referenced costs such as side effects and possible long-term effects of medication in the body. They also mentioned how they could perceive some benefits when taking medication, such as lowering their A1C or blood pressure. None of the participants in our study reported general cognitive impairments that could explain their occasional lapses in adherence. Hence, a commonly cited element affecting adherence, unintentional non-adherence due to cognitive impairment, was not a factor in this study.
The questions then focused on the factors that lead to medication adherence, habits, and the role of the DMP in modifying those factors. Here, the interviews showed some deviation from the influence diagram, with differences shown in Figure 4. New concepts and relationships that emerged from the interviews are indicated by wide white stripes and dotted lines. Habit formation and DMP participation.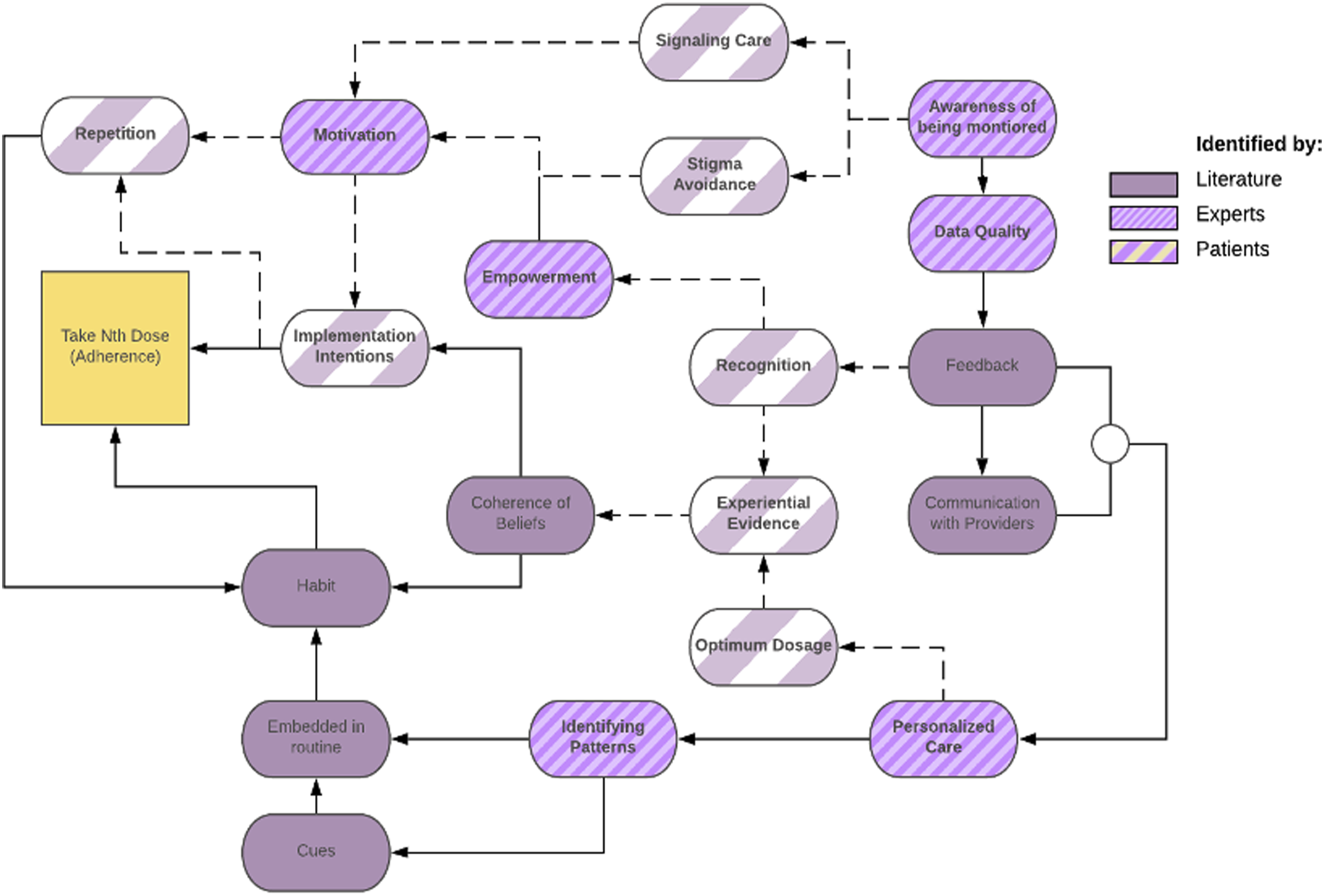
Information availability
The center of Figure 4 shows the connections identified by patients describing how they process feedback received through the DMP. This feedback comes in the form of immediate confirmation that the medication was taken, as well as a tracker that counts the number of consecutive days of successful medication adherence (“streaks”). Additionally, patients are required to input blood pressure and blood sugar measurements, which allows them to correlate their adherence behavior to their physiological outcomes. Patients (6/10) describe that the feedback generates a greater recognition of their behavioral patterns. For example, this user outlines how the intervention affects positive behaviors: “I’d say it strengthens them. Because it’s like when you see for yourself that you’ve taken it and [you] see your results too. And, so I’m able to make the correct decision on my care.”– P013
The interview results suggest that patients accumulate evidence of successful adherence using the DMP, leading to the development of beliefs that the treatment is effective.
46
Additionally, patients (7/10) using the DMP focused on the importance of having a direct way to visualize their adherence behavior and health outcomes for these mostly asymptomatic conditions: “It just made me care more about taking my medicine because I could see the results of what I was doing, where before just somebody told me to take these pills and I’d take them, but I didn’t know why” – P016
Providers receiving accurate adherence information were able to adjust doses more accurately, leading to a more effective treatment, and in turn, more reason for patients to adhere. Similarly, patients were also able to see that medications may not be effective if the dosing is incorrect because the DMP, combined with provider communication, made a direct connection between dosing changes and health outcomes.
Patient–provider interactions
Participants discussed two specific pathways where awareness of being monitored led to a motivation to adhere. The first was stigma avoidance, feeling that they would be in some way negatively judged by their healthcare team for not adhering. In contrast, patients (6/10) also identified that being monitored gave them a signal that their provider cared for them (signaling care) by watching over them through the monitoring system: “Because, sometimes, it was good to know that somebody out there was not with you consistently all the time, but with you, concerned about your health (…) That’s what was really important. It helped me out here.” – P008 “Because they’re seeing everything that I’m entering into the portal and they’re kind of reviewing what I’m doing. If they see something that doesn’t … something might not look good to them that I wouldn’t … I’d think it was okay. It’s the idea of having somebody kind of looking over your shoulder and [that] makes you feel comfortable.” – P016
Routines and repetition
The top left section of Figure 4 shows how patients think about repetition of medication-taking behavior. Participants who believe in medication effectiveness and are motivated to adhere develop implementation intentions to continue their adherence behavior. The DMP then allowed patients (5/10) to come up with concrete ways to put those intentions into action, for example, by embedding their medication into a routine, or creating a plan to take their next dose. As prior peer-reviewed work suggests, the creation of such plans can eventually lead to long-term habit formation even in the absence of DMP, because routine stimulates and reminds the patient to take the medication.
47
“The program, I love the program. It keeps me on track. It’s like, you take your morning meds. Well, if I’m going to take my morning meds, let me have breakfast. I need to take this pill this evening, I need to plan what I’m going to have for dinner” – P004
Patients (7/10) also described how motivation and a plan to execute the behavior encouraged the repetition needed to make adherence a habit: “Yeah. I think [DMP] has caused me to have to take it. In the past, I think there have been times when I might have skipped it before I did [the DMP]. I might have skipped a medication just because it was inconvenient. I don’t think I do that anymore because [the DMP] pretty much forces you to not do that. Chews you out, gives you a good chewing out” – P012
Discussion and conclusion
Discussion
Medication non-adherence is a serious public health problem, and while existing interventions such as patient education, therapy, and support system strategies have had some success in overcoming non-adherence in the short-term, 48 they are resource intensive and often weakened by the limitations of self-reported assessments. Furthermore, very few have shown persistent long-term benefits.49–51
DMPs have the potential to improve both short- and long-term medication adherence by combining sensor-enabled medications with patient- and provider-facing information platforms that enhance both patient–provider interactions and patient ability to manage their own health. These types of interventions improve one-size fits all approaches by offering targeted support to address the specific needs of individual patients. 52 Not only do DMPs communicate individual adherence information that can lead to dosage improvements, but they also allow patients to better take control of their own health, which remains a challenge for other existing interventions. 40
Our results show that DMPs have the potential to influence adherence by providing accurate and personalized information to better monitor and communicate the consequences of non-adherent behavior to patients (rather than providing more generic advice informed by population data). Patients place value on the DMP’s ability to facilitate understanding of the relationship between medication-taking behavior and effectiveness. This highlights how DMPs need to be responsive to the information needs of patients, including ways to make information usable and understandable for different literacy levels. While much focus has been given to data-driven patient–provider communication, these participants show how patients use direct feedback from the DMP to infer the details of their condition, where patients report a large difference between being told to take the medication versus inferring that the medication is effective through experience. 53
Participants suggested multiple ways that patient–provider relationships are affected by the use of the DMP. Patients discussed the perceived investment of the provider in their well-being as a motivation to adhere, which was signaled by recommendations and dose adjustments based on DMP-provided feedback. Prior research has found that without consistent provider feedback, patients perceived that self-monitoring was an undue burden.54–56
DMPs were seen by participants as playing a large role in habit formation. Using the data-driven feedback, patients stated that the DMP allowed them to make plans that facilitate repetition of medication-taking behavior. Existing research has found that the use of plans in the form of “if-then” statements, such as “If I have finished breakfast, then I will take my medication,” is effective in circumstances where a patient’s ability to translate intentions into action is impaired (such as during travel or when under cognitive load).57–59 Without requiring prompting, patients recognized the need to create if-then plans in order to comply with the requirements of the intervention. This shows both intrinsic and extrinsic motivation to continue this repetition consistently, reinforcing the creation of habits over time. 48 Repeating a behavior in a stable context is known as a key to developing habits, both good and bad.47,60 However, an important unknown is how much monitored repetition time is needed for a patient to develop stable habits when using a DMP, and how that stability relates to some of the adherence mechanisms discussed in this work.
Limitations
Due to the labor-intensive nature of qualitative research using interviews, we are limited by our sample size to estimate the prevalence of these beliefs in a broader population. While previous studies have found that 15 is a reliable number of interviews for thematic analysis, for more homogeneous populations samples as small as six could offer a reliable thematic saturation.61–63 In this study, we found a remarkable level of data saturations as we reached our sample of 10 participants with consistent patterns starting to emerge in the midst of the interview process. Additionally, this was a sample recruited from a specific clinic with older patients with three specific chronic conditions. Older patients can be difficult to recruit due to additional constraints to participate in research such as lack of mobility or understanding of research purposes. 64 We did not have the opportunity to discuss this with younger patients that may be more familiar with digital interventions and health tracking devices. While the sample frame was slightly skewed toward males (57%), our sample is strongly skewed toward male participants (8/10), which may be the result of using phone calls as our recruitment method, with men being more likely to answer unsolicited phone calls. 65 Additionally, by focusing both our expert and patient interviews on those with familiarity with one specific DMP, we cannot speculate on the receptiveness of naive patients or providers to DMP interventions. All experts were identified and recruited by Proteus Digital Health®. At the time of the interviews, two clinical experts were working on collaborative implementations with the company, the remaining clinical expert and the three academic researchers had also collaborated with Proteus Digital Health® in the past.
Conclusion
The results of the study show at least three mechanisms by which DMPs could potentially support changes in medication-taking behavior. These mechanisms are as follows: (1) providing accurate and personalized information, (2) improving the quality of patient–provider interactions, and (3) creating and repeating patient routines around medication use. Reports from patients affirmed results found in the research literature and suggested by experts, although there are some notable deviations. Specifically, patients were focused on how the DMP gave them information to form their own experiential evidence, how the intervention and provider involvement signaled care for their well-being, and how being watched over motivated them to repeat the behavior.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Anabel Castillo is a full-time employee of Naima Health LLC, a recipient of funding from Proteus Digital Health. Alexander L. Davis and Tamar Krishnamurti are co-founders of Naima Health LLC. Baruch Fischhoff is a faculty member at Carnegie Mellon University. He received no payment or research support from this project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this data collection was provided by Proteus Digital Health.® The funding agreement ensured the authors' independence in designing the study, interpreting the data, writing, and publishing the report. The sponsors provided a list of patients for recruitment from a clinical trial of the product studied here which they selected. However, recruitment from this pool of patients was conducted by the researchers.
