Abstract
The main aim of this study is to assess the effectiveness of using a developed asthma mobile application to enhance medication adherence in Jordan. Asthma patients visiting outpatient respiratory clinics and using inhalers were recruited. Patients were assigned into two groups: intervention and control. The intervention group was instructed to download and use the application. Asthma control was assessed using Asthma Control Test (ACT) at baseline and at follow-up of 3 months for both groups. A total of 171 patients (control, n = 83, and intervention, n = 88) participated in the study. After 3 months of usage, patients in the intervention group achieved a significant improvement in ACT score compared to control (p-value <0.05), and reported a significant satisfaction of the application use. Therefore, the asthma mobile application is found as an effective tool to enhance medication adherence in asthma patients.
Introduction
Asthma is a chronic inflammatory disease that affects the airway of the lungs. It is very common and affects people of all ages.1,2 Globally, asthma has affected >339 million individuals and caused around 417,918 deaths per year.3,4 In Jordan, a low-resource country, asthma prevalence is high and has been doubled in the last decade. 5 There is no complete cure for asthma; therefore, the achievable goal of asthma management is to relieve symptoms, decrease the disease progression, and enhance patients’ quality of life. 6 This can be achieved by avoiding triggers such as dust and pollen and using inhaled medication. 6 Inhalers are devices that contain drugs which either relax the airway muscles and/or reduce inflammation in the lungs.2,6 The use of inhaler devices is challenging for most of the patients, in particular, metered-dose inhalers (MDIs) and powder inhalers (DPIs).7,8 However, incorrect inhalation technique and frequent misuse of inhalers would lead to treatment failure and poor asthma control.9,10
Mobile health applications (mHealth apps) are innovative digital interventions that have been developed as an easy and viable way to provide patients and health care providers with a wide range of information to manage various conditions, including chronic diseases.11 –13 Such applications can deliver automated summary reports of patients’ conditions to health care providers and promote adherence to medication regimens.14 –16 A systematic review analysis investigating the impact of mHealth apps on self-management outcomes in asthmatic patients, showed that mHealth apps improved asthma control and lung function and enhanced patients’ quality of life. 17 In addition, the use and effectiveness of mHealth apps in different age groups of asthmatic patients were investigated and showed promising results, such as enhancing medication adherence and self-management.18 –21 These findings highlight the importance of this technology in supporting the existing clinical health care in asthma management and medication adherence.
Studies have shown that asthma control is poor among the Jordanian population,10,22 and this was attributed to multiple factors, including the poor inhalation techniques and the incorrect use of devices.22,23 Interestingly, no previous studies have investigated the use of mHealth technology to promote adherence to the correct use of inhaler devices and asthma self-management among Jordanians. The current study aimed to explore and assess Jordanian asthmatic patients’ knowledge and attitude and design preferences toward using the mHealth app to enhance adherence to the correct use of inhaler devices and asthma self-management. In addition, the study aimed to design an accessible and culturally appropriate mHealth app and to evaluate its usability and satisfaction.
Methods
Study design, sitting, and patients
This prospective interventional study was conducted over a period of 9 months at the outpatient respiratory clinics of the Jordan University Hospital (JUH), Amman, Jordan. The study was carried out according to the Declaration of Helsinki. The study design and protocol were approved by the Institutional Review Board (IRB) at the Deanship of Scientific Research, The University of Jordan, and JUH, reference number (80/2018/1358). Asthmatic patients using inhalers and who visited the respiratory clinics were recruited. Patients were assigned into two groups: intervention and control. Patients who had a smart mobile phone and were willing to participate and download the mHealth app were included in the intervention group. While those who were willing participate but did not have a smart mobile phone or did not wish to download the app were included in the control group. All patients were asked to sign an informed consent form before filling out a paper-based questionnaire and downloading the app on their smartphones. Patients were informed that data and personal information collected as part of the study were going to be treated as confidential. The inclusion criteria were: patients aged at least 18 years, diagnosed with asthma for at least 6 months, have been using controller inhaler devices for a minimum of 1 month before study recruitment. A follow-up after 3 months was considered.
A total of 190 patients were recruited and agreed to participate in the study. Patients were divided into 102 in the intervention group and 88 in the control group. Of the total, 171 patients (88 in the intervention group and 83 in the control group) completed the study. There was 10% drop-out rate (n = 13 were lost from follow-up, n = 3 died, and n = 3 did not use the app after downloading it). A flow chart of patients’ recruitment is shown in Figure 1.
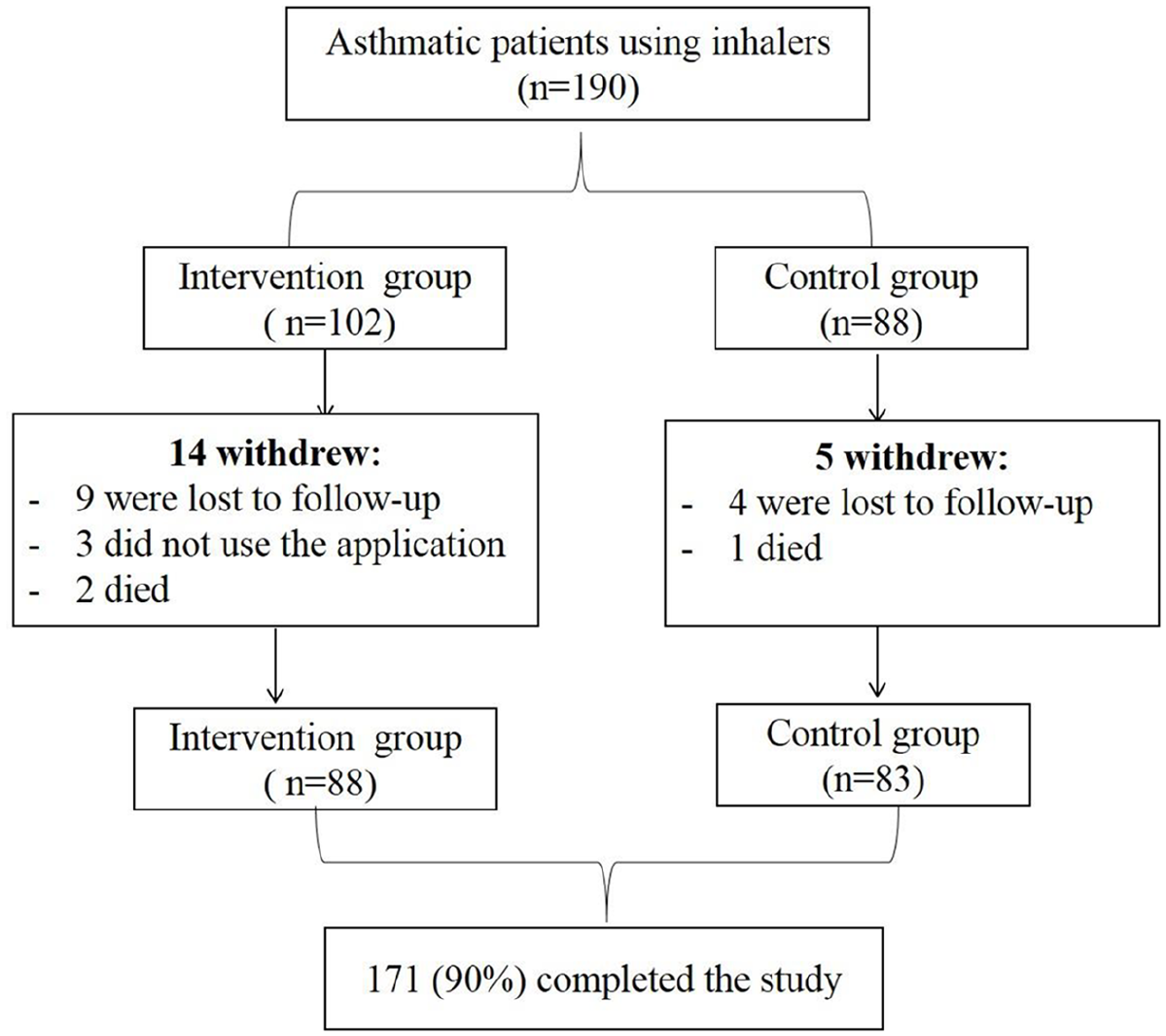
Flow chart of patients’ recruitment. A total of 190 asthmatic patients were approached to participate in the study. About 102 patients in the intervention group and 88 in the control group. At the end of the study, 13 patients were lost from follow up, 3 died, 3 did not use the application, and 171 patients successfully completed the study.
The questionnaires
All patients’ data were collected at baseline using a developed questionnaire consisting of three main domains; the first domain had 12 items regarding patients’ demographics and information about asthma and medication use. The second domain included eight items to assess patients’ knowledge and experience with smartphones and health technology aids. The third domain consisted of six items to explore patients’ attitude toward using mHealth app to enhance medication adherence. Likert scale (strongly agree, agree, neutral, disagree, and strongly disagree) was used to evaluate patients’ attitude toward using the mHealth app. This questionnaire was developed and designed based on an extensive literature review.13,18,20,24 –27
Asthma control for patients from both intervention and control was assessed using Asthma Control Test (ACT). The ACT is a validated patient-based tool that is usually used to evaluate asthma control in clinical practice.22,28,29 It is recommended by the American Thoracic Society/European Respiratory Society Task Force as an assessment tool of asthma control. 30 It is a questionnaire consisting of five questions; four of them are related to symptoms and use of relievers and one question is about patient self-assessed level of control.28,29,31 ACT reflects asthma patients’ status during the last 4 weeks using a total score ranging from 5 to 25, with higher scores indicating a better asthma control.22,29 Asthma patients who had scores between 20 and 25 were classified as well controlled; 16–19 as partially controlled; and 5–15 as poorly controlled. 31 In this study, a validated Arabic version of the ACT test was used. 32 ACT was calculated for all recruited patients at baseline and at follow-up after 3 months.
Patients in the intervention group were asked to download the app and notified to use it via a weekly notification. In addition to ACT scores, patients in the intervention group were asked to fill a questionnaire assessing and scoring their satisfaction with the app (i.e. ease of use, information, usefulness for asthma) at follow-up. This custom questionnaire was modified from previously published mHealth App Usability Questionnaire (MAUQ) and satisfaction questionnaires,19,33,34 which have the reliability and validity required to assess mHealth app usability. In fact, in this study the commonly used questionnaires such as System Usability Scale (SUS) and Post-Study System Usability Questionnaire PSSUQ were not used as these questionnaires were designed to evaluate a software or website, not mHealth app.
The content of all the used questionnaires (at baseline and follow-up) were critically reviewed and validated by distributing initial drafts to several colleagues in the field and then to a sample of 20 patients. The received feedback was considered for reviewing and performing any amendments needed. Full professional demonstration and counseling were provided by the researchers to each patient pertaining to the questionnaire items. A summary of the study design is demonstrated in the flow chart in Figure 2.
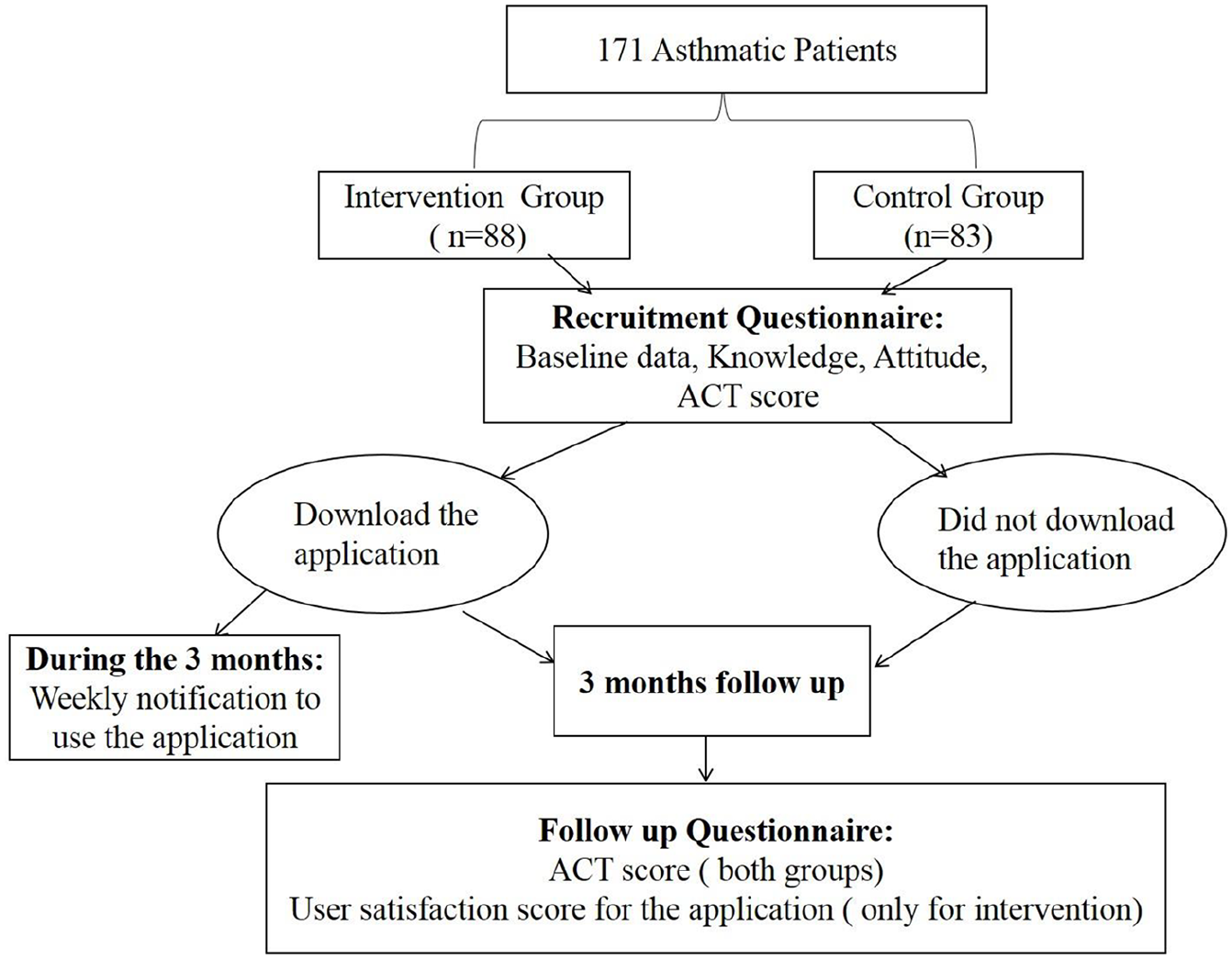
Flow chart of the study design. At recruitment, baseline data was collected from patients in the intervention and control groups. ACT score was calculated for both groups. Intervention group was asked to download the app and was notified weekly to use it. After 3 months, ACT score was re-calculated for both groups. User satisfaction score for the app was assessed in intervention group.
The design mobile health application (asthma mHeath app)
The design science research method 35 was used to design and develop the asthma mHealth app. This method is an iterative process that contains six steps; (1) identifying the problem, (2) defining the solution objectives, (3) designing and developing an artifact: mHealth app, (4) demonstrating the designed artifact, (5) evaluating the artifact’s effectiveness, and (6) communicating the outcomes with patients. A software developer designed a platform smartphone app (asthma mHealth app) that runs freely on Android devices (i.e. Samsung and Huawei). The app was designed and prepared in the Arabic language. It contained several items targeting multiple aspects of the patient’s knowledge about asthma, exacerbating triggers, and adherence to the correct use of different inhaler devices. Home page items were “information about asthma” and “medication.” The information about asthma allowed the user to see multiple pages with simply presented information, including asthma definition, symptoms, triggers, severity, and prevention. At the same time, medication menu provides patients with short demonstration videos about the correct use of different inhaler devices, including MDIs, DPIs, and MDIs with spacer. These videos were recorded by a health care provider in the respiratory clinics, where patients used to receive the instructions about their medications. However, the app does not provide reminders or alerts to patients. An outline of the app screens and examples of app screens are shown in Figure 3 (a and b).
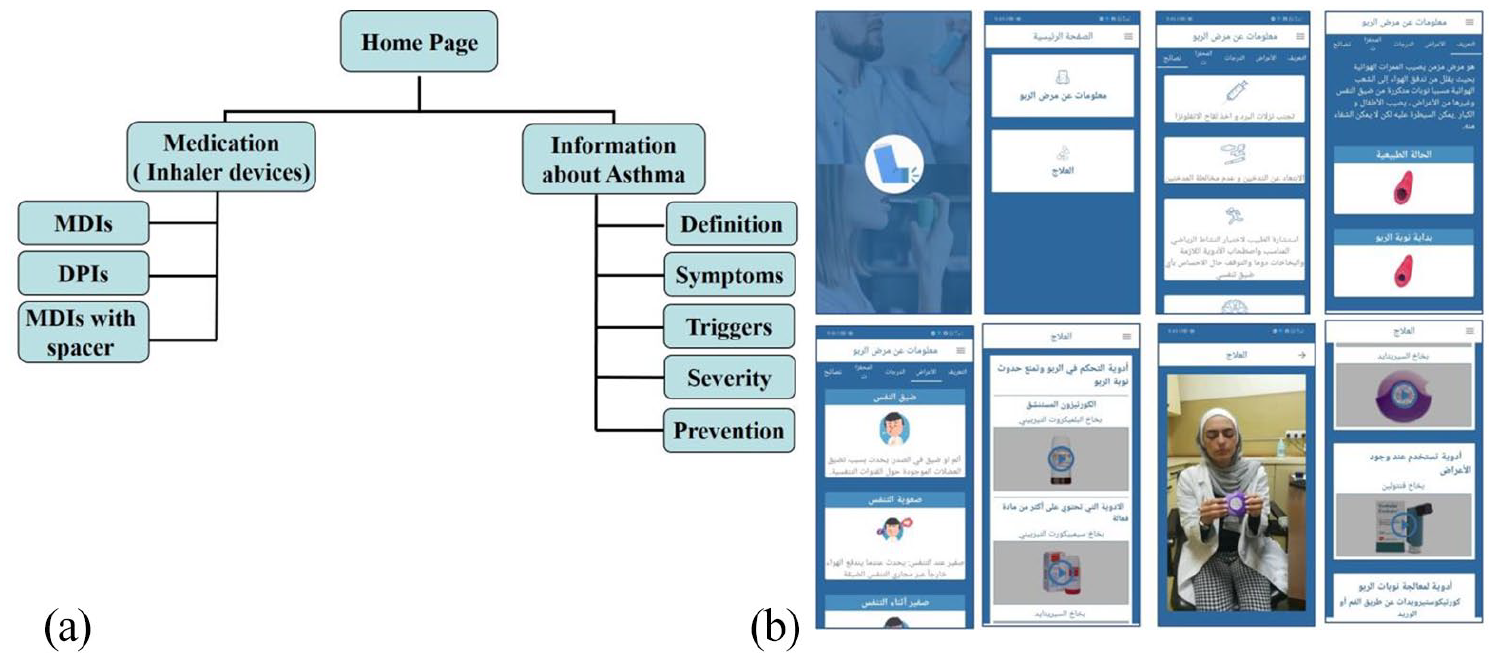
Asthma mHeath app: (a) an outline of app screens and (b) examples of asthma app screens.
Statistical analysis
Statistical analysis was carried out using SPSS version 20.0 (SPSS Inc., Chicago, IL). Descriptive statistics were used to describe demographic and general characteristics of participants. Categorical variables were presented as percentages with frequencies, while continuous variables were presented as mean with standard deviation (SD).
An independent sample t-test was used to test for differences in ACT score between control and intervention groups at recruitment and at follow-up. Paired sample t test was used to test differences in ACT score for the intervention group before and after the use of the mobile application. The same comparison was performed for the control group at recruitment and at follow-up evaluation. Pearson chi-square test and Fisher Exact test were used to find associations between categorical variables. All hypothesis testing were two-sided. A p-value of <0.05 was considered significant.
Results
The socio-demographic and clinical characteristics of the study population
The socio-demographic and clinical characteristics of the recruited patients were shown in Table 1. Around 34% of the study population aged between 30 and 49 years (n = 22 control, and n = 36 intervention). In addition, there was a statistical difference in age between the study groups (p-value <0.05). Patients in the intervention group were younger than those in the control group. The percentage of males and females was almost equal between the two groups (males were 30% vs 28.4%, and females were 69.9% vs 71.6% in the control and the intervention groups, respectively). Only 4% (n = 7) of the patients were not educated, and were in the control group. Approximately, 32% (n = 57) of the all patients were either a full-time or part-time workers (n = 22 control, and n = 32 intervention). The majority of the patients have been diagnosed with asthma for >6 months (94.7%, n = 162), and there was no statistically significant difference between intervention and control groups with regards to the time of diagnosis (p-value = 0.801). Even though 78.9% (n = 135) of the patients answered that they take their medications regularly on time and 90.1% (n = 154) of them did not have any technical problem when using inhaler devices, the majority of them had either partially controlled (44.4%, n = 76), or uncontrolled asthma (47.4%, n = 81). Interestingly, only 17% (n = 29) of the patients were smokers and comparably distributed between intervention and control groups (p-value = 0.233). Cigarettes smoking was the most common type of smoking (79.4%, n = 23).
Socio-demographic and clinical characteristics of patients at recruitment.
Asthma Control Test (ACT) scores out of 25; ⩾20, well-controlled asthma; 16–19, moderately controlled asthma; ⩽15, very poorly controlled asthma.
Percentages were calculated out of smokers.
Patients’ knowledge and experience with smart mobile phones and health technology aids
The majority of the patients in this study (96% of the total sample) had a mobile phone, 94.5% of them had a smartphone (Table 2). All patients in the intervention group had smart mobile phones with android platform. In addition, almost >90% of the patients believed that the smart mobile phone is useful in their life (74.2%, n = 115 agree, and 17.4%, n = 27 strongly agree). Furthermore, 95.5% (n = 148) of the patients who had smartphones, usually use their mobiles without any assistant (n = 64 control, and n = 84 intervention). Nevertheless, only 26.5% (n = 41) of the patients who had smart mobile phone, have ever heard about mHealth apps (n = 13 control, and n = 28 intervention). Among these, only 4.5% (n = 4 intervention) had used mHealth apps to manage any chronic conditions before participating in this study. Table 2 presents as assessment of patients’ knowledge and experience with smart mobile phone and health technology aids.
Assessment of patients’ knowledge and experience with smart mobile phones and health technology aids.
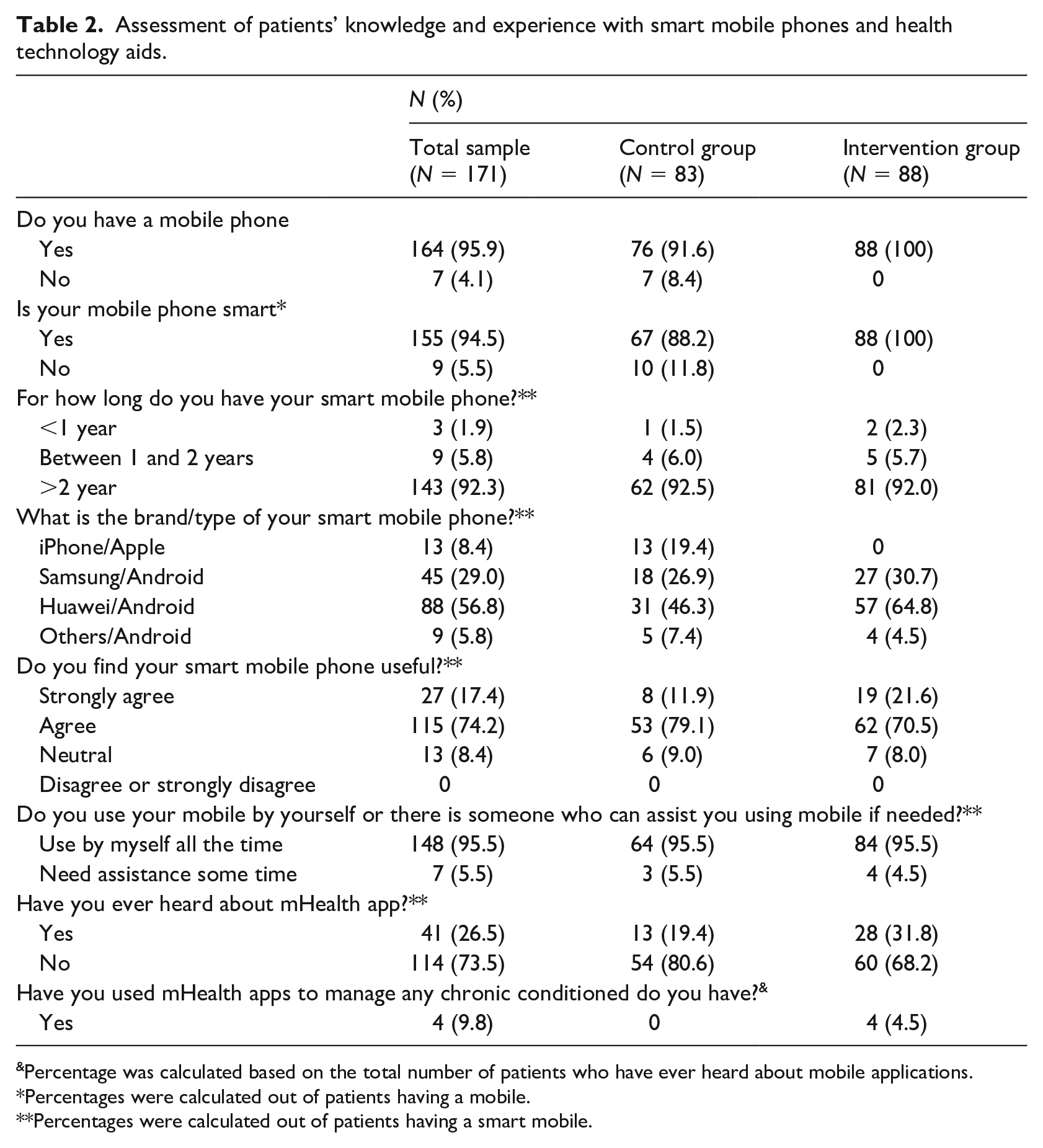
Percentage was calculated based on the total number of patients who have ever heard about mobile applications.
Percentages were calculated out of patients having a mobile.
Percentages were calculated out of patients having a smart mobile.
Patients’ attitude toward using mHealth app to enhance medication adherence
Patients’ attitude toward using the mHealth app to enhance medication adherence was investigated at recruitment, according to the patients’ responses to six items using the Likert scale as demonstrated in Table 3. Before using our designed asthma mobile app, none of the patients believed that the use of the mHealth app would be a useful tool to enhance medication adherence. Similarly, 96.5% of the total sample thought that the use of the mHealth app would not be an effective tool to provide patients with information regarding the correct use of inhaler devices. On the other hand, only one patient from the overall patients indicated that the use of the mHealth app would help to remind patients to take their medications on time. Approximately 96% of the total sample did not agree that the mHealth apps’ use could help patients follow doctor’s instructions regarding treatment plan and medication use. Furthermore, 73.1% of all patients indicated that the use of the mHealth app would not affect the number of times that patients would have to visit the clinic. In addition, there was a statistically significant difference between control and intervention groups’ responses with regards to the effect of the use of the mHealth app on the number of clinic visits, p-value <0.05. Finally, most of the patients at recruitment (97.7%) considered that the mHealth app would provide inaccurate and untrusted health-related information. It is clear from these findings that most of the patients (control and intervention groups) had a negative attitude toward the use of the mHealth app at the recruitment stage and prior to the application use.
Patients’ attitude toward using the mHealth app to enhance medication adherence.
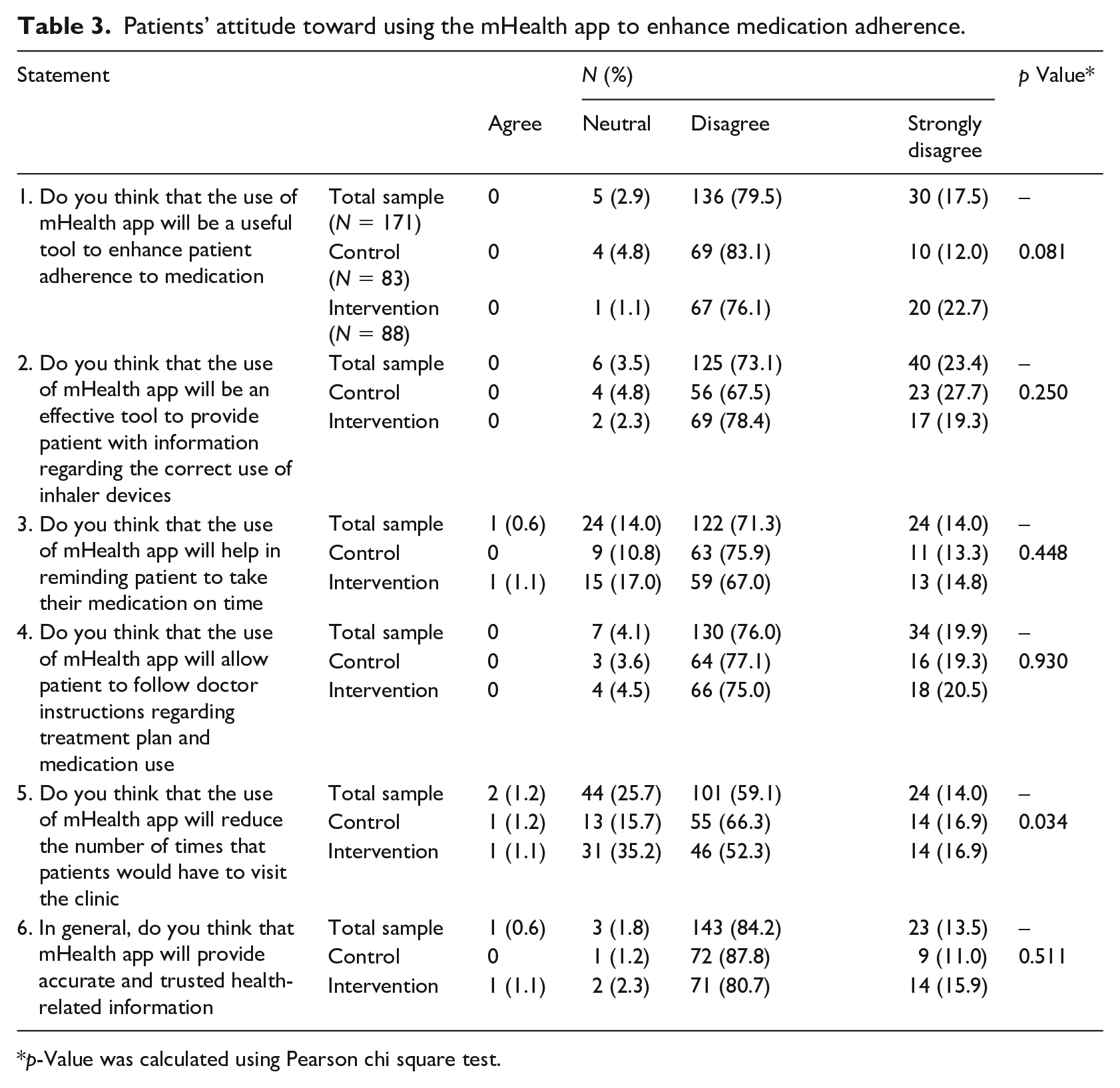
p-Value was calculated using Pearson chi square test.
The effect of designed asthma mobile app use on asthma control
The ACT score was calculated for each patient at recruitment and after a follow-up of 3 months, as an objective measure for asthma control. As shown in Table 4, during recruitment (baseline), there was no statistically significant difference in ACT scores between intervention and control groups (19.4 ± 4.4 versus 18.3 ± 4.6, respectively, p-value = 0.134). For 3 months, the intervention group used the designed asthma mobile app as an educational tool on how to use their inhaler devices and as an additional source of information about asthma. On the contrary, the control group did not use the app and only received traditional patient education from the health care providers. Interestingly, the intervention group had significantly higher ACT scores than control at follow-up (21.1 ± 3.7 versus 19.7 ± 4.0, respectively, p-value = 0.016) (Table 4), indicating that their asthma symptoms were enhanced after using the app and following the instructions on how to use the inhaler devices properly. In addition, the ACT scores of both control and intervention groups were increased (enhanced asthma symptoms) significantly at follow up, p-value <0.001 (Table 4). However, the extent of the increase was higher among the intervention group, from 19.4 ± 4.4 to 21.1 ± 3.7 (Table 4). Differences in ACT among control and intervention groups before and after app use were 1.4 versus 1.7, respectively (Table 4).
Comparison of ACT score (out of 25) between control and intervention groups.
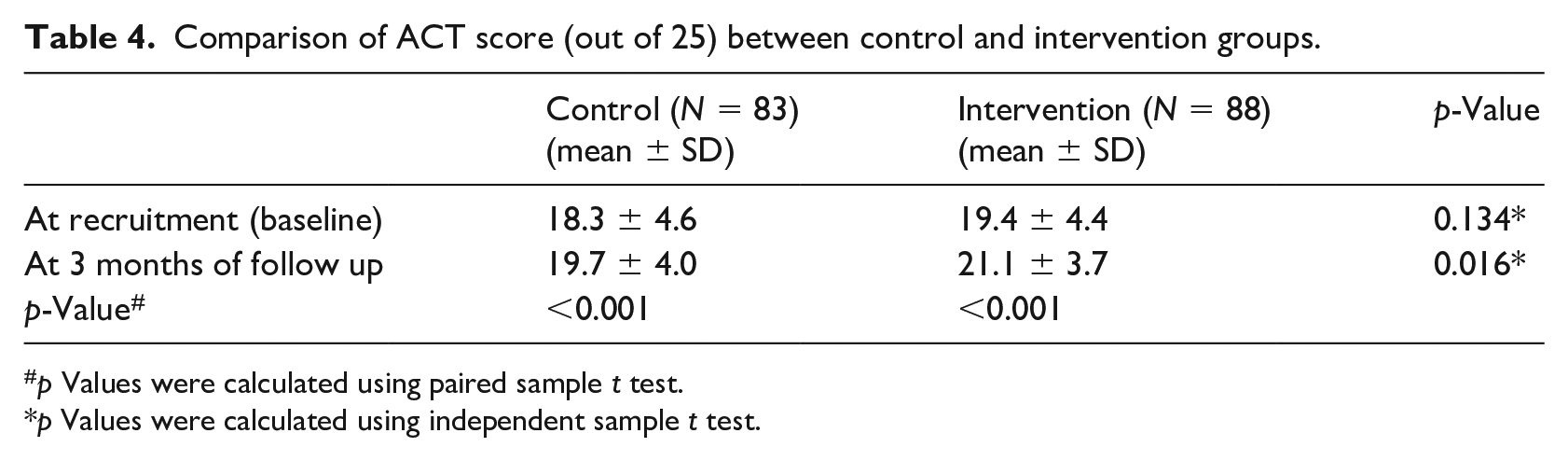
p Values were calculated using paired sample t test.
p Values were calculated using independent sample t test.
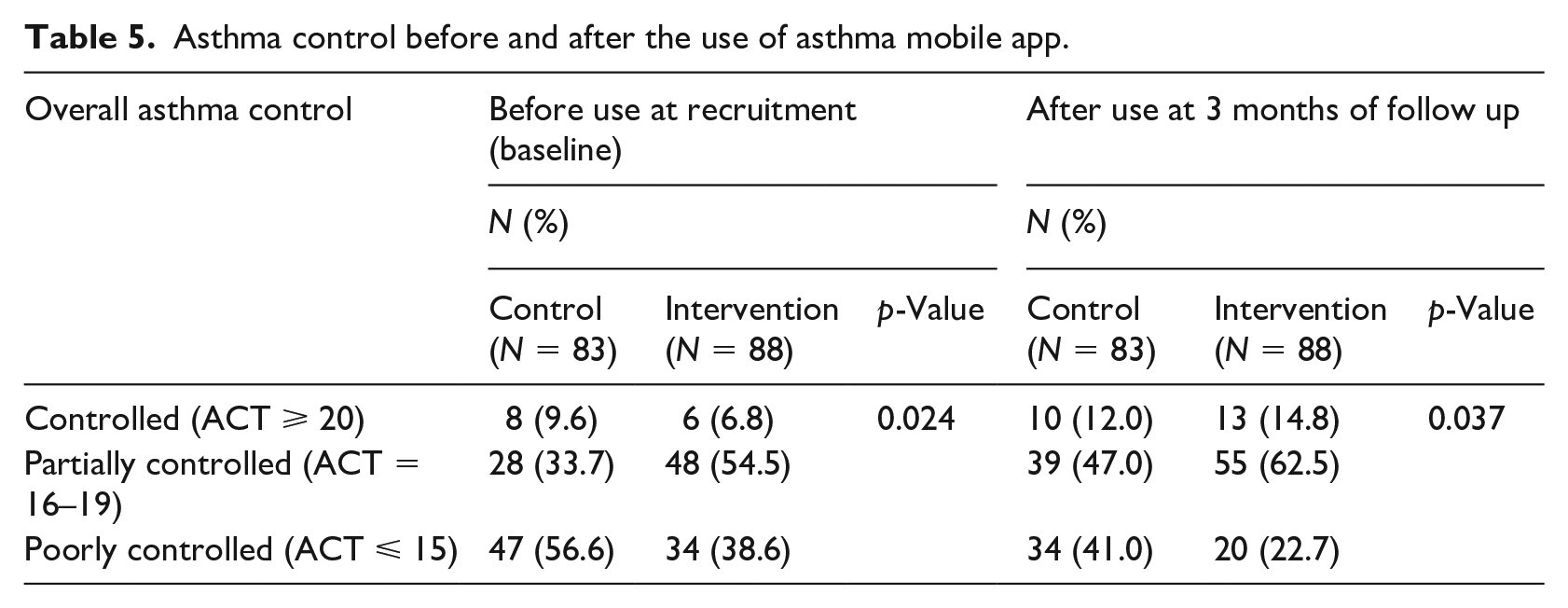
Furthermore, the asthma control of the study population was investigated as numbers and percentages of patients in each category according to the patients’ ACT scores (Table 5). Consistent with data in Table 4, asthma symptoms were improved among study groups at follow up. The number of patients with either controlled or partially controlled asthma had increased among both control and intervention groups at follow-up. However, the percentage of increase in controlled asthma category among intervention group was significantly higher than control (difference in increase was; 8% versus 2.4%, respectively, Table 5).
Asthma control before and after the use of asthma mobile app.
Users’ satisfaction with asthma mobile app
The satisfaction with the designed asthma mobile app use was investigated among patients who used it (intervention group, n = 88). As shown in Table 6, results revealed mostly high satisfaction with app’s ease of use, content, and usefulness for asthma. Even though 8% (n = 8) of the users indicated that using the app requires time, the majority of the users indicated that they were satisfied with the app and they would like to use it again.
Users’ satisfaction for asthma application (N = 88).
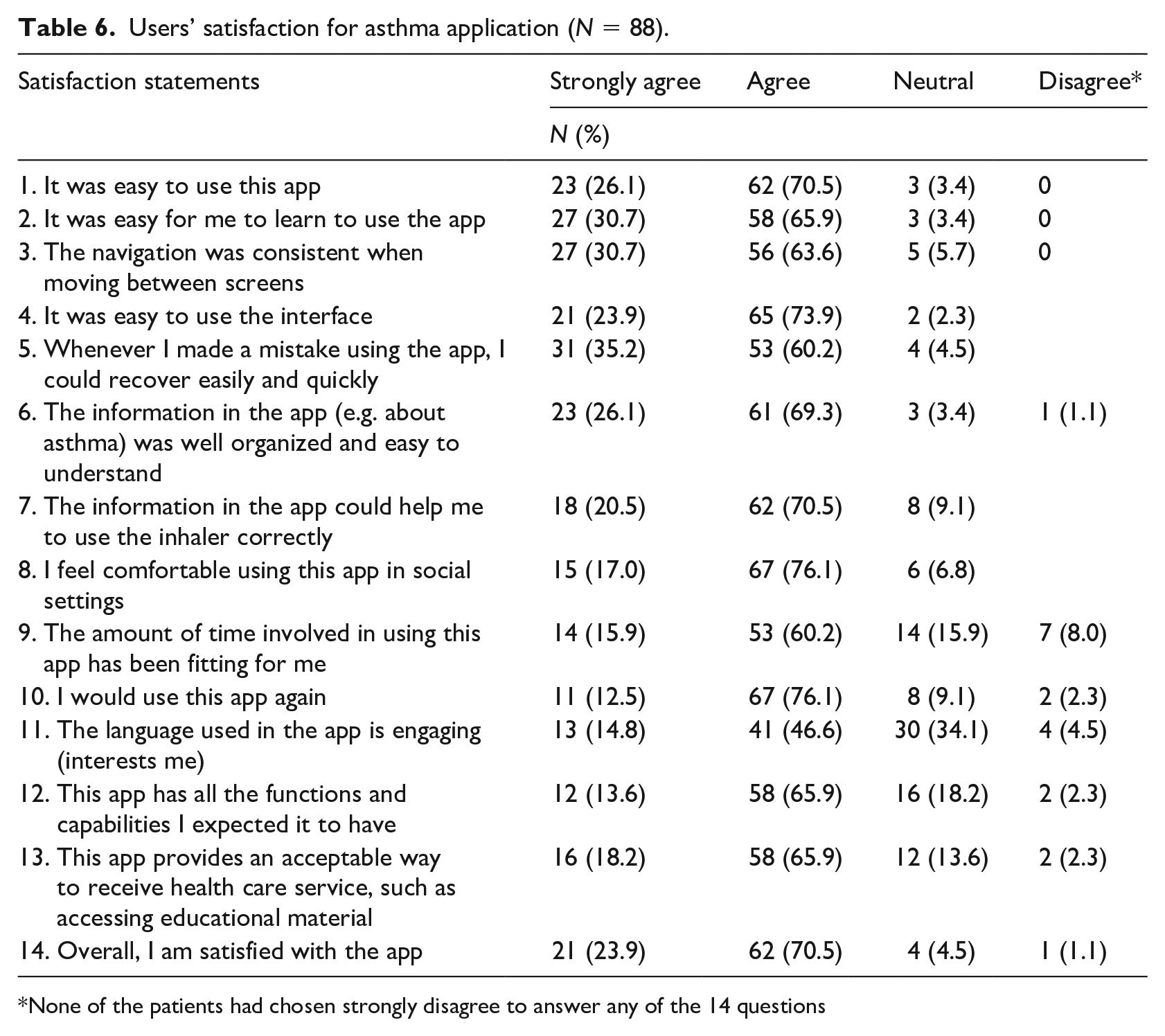
None of the patients had chosen strongly disagree to answer any of the 14 questions
Discussion
This prospective interventional study presents and evaluates a new platform in the management of asthma. It introduces the use of mHealth technology to enhance asthma management and medication adherence. An important aspect of medication adherence in asthma is the correct use of inhaler devices, as poor technique might directly affect asthma control. To the best of our knowledge, this study is the first to investigate the use and effectiveness of a designed mHealth app that delivers evidence-based informed asthma content and demonstrates the correct use of different inhaler devices through videos recorded by a health care provider.
Prior to implementing the mHealth app, patients’ knowledge and experience with smart mobile phones and health technology aids were assessed. Results indicated that the majority of the study population had a smart mobile phone available for at least 2 years. In addition, <90% of them used their smart mobile phones without any assistant. Clinically, most patients had either partially controlled asthma or poorly controlled asthma. These findings were in accordance with several previous studies that have shown poor asthma control among the Jordanian population,10,22 and the incorrect use of different inhaler devices was one of the associated factors.22,23
Only 26.5% of the patients had heard about mHealth apps before conducting this study. Therefore, implementing the mHealth app to enhance medication adherence and asthma self-management is necessary and would be a new platform for managing asthma among the Jordanian population. Initially, the patients (n = 171) were assigned into intervention and control groups. Elderly, not educated patients, and those who have mobile platforms other than Android, were included in the control group and received the traditional patient education from the health care providers as they could not use the designed asthma app. As a result, patients in the intervention group were younger, more educated, and employed. More than half of the intervention group had partially controlled asthma. However, at baseline, there was no statistically significant difference in ACT scores between control and intervention groups (p-value = 0.134), indicating that both groups had a similar level of asthma control at the beginning of the study.
Indeed, all the patients (control and intervention groups) had a negative attitude toward using mHealth apps at the recruitment stage and before the use of the designed asthma mobile app. This was expected as only 4.5% of them were familiar with mHealth apps. Additionally, the idea of using the mHealth app for asthma management has not been previously investigated among the Jordanian population. Most previous studies focused on pharmacists’ role in enhancing patients’ use of inhaler devices.22,23 Moreover, studies evaluated the attitude of the general population on mHealth apps’ use indicated that older individuals had a negative attitude and were less receptive to using these apps. 24 Consistently, in our study, <45% of the patients aged 50 years and above; they were to have concerns about using a new non-traditional way to deliver medical advice.
Interestingly, at the follow-up of 3 use of the designed asthma mobile app asthma mobile app, a significant change in users’ attitude was noticed, particularly in the aspect of users’ satisfaction. The majority of the users reported high satisfaction with the ease of use, content, and usefulness of the app. Moreover, most of the users showed an interest in using the app and reported their satisfaction with it. This satisfaction with our designed asthma mobile app was translated clinically as a significant improvement in the ACT score among the intervention group.
The use of the designed asthma mobile app enhanced patients’ adherence to the proper use of inhaler devices and thus improved their symptoms and overall asthma control. Although there was an improvement in symptoms in the control group who did not use the app, this was due to the traditional patient education provided by the physician and the clinical pharmacist to all of the study groups. The results showed that patients who had used the designed asthma mobile app for 3 months had better asthma control as the number of patients with poor asthma control decreased. This is in line with previous studies that have shown that adherence and correct medication use were related to better asthma control.36,37
Strength and limitation
A strength of our study was the relatively high number of patients with a low drop-out rate. The low drop out in this study might be attributed to the socio-demographic and behavioral characteristics of the participants. First, most of the patients who used the app were young, and this group of population has been shown to have less drop-out rate. 38 Second, using a mHealth app to support asthma patients to adhere to their medication was new in Jordan as no previous studies have investigated this before. Therefore, patients lack such a needed functionality, and thus they enthusiastically adopt it. Third, asthma is a chronic disease, so patients used to visit the clinic regularly where they were notified to use the app by the physician, which might potentially maintain patients’ interests to use the app. Moreover, the designed asthma mobile app was aligned with patients’ requirements, as it presents different demonstration videos in the Arabic language on how to correctly use several types of inhaler devices and preventers. In addition to its ease of use, the app could be used by the patient in the mode of no internet availability. On the other hand, the app lacks interactive approaches between patients and healthcare providers, including asthma action plans and monitoring of asthma control with feedback. A modification on the used app utilizing interactive approaches between patients and health care providers is under investigation by our research group and would be focusing on the engagement of pharmacists in supporting asthma patients, especially those who were non-adherent to their medications. Among the limitations of this study was a response bias. At the recruitment, motivated, young patients and those having an android platform were probably more interested in participating in the intervention group and more willing to use the app. Therefore, age was not matched between intervention and control groups, which is a limitation in this study. In addition, notifying patients in the intervention group to use the app could be a limitation in this study. However, these patients were supposed to get the instructions on how to use the inhalers by using the app, not by the traditional way by health care providers.
Conclusion
In this study, a smart mobile phone mHealth app for asthma patients was developed. This app incorporated several features that have significantly improved patients’ adherence to medication use and overall asthma control. Therefore, healthcare providers may consider the implementation of mHealth approaches in the treatment of patients with asthma and enhancing medication adherence, especially in the current corona virus disease (COVID-19) pandemic that has affected the proper interaction between patient and healthcare providers.
Footnotes
Acknowledgements
We would like to thank Sujood Nawash, Marwa abdeen, and Batool Alkhawldeh for their help in data collection.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research at the University of Jordan.
