Abstract
Congestive heart failure (CHF) is one of the most common diagnoses in the elderly United States Medicare (⩾ age 65) population. This patient population has a particularly high readmission rate, with one estimate of the 6-month readmission rate topping 40%. The rapid rise of mobile health (mHealth) presents a promising new pathway for reducing hospital readmissions of CHF, and, more generally, the management of chronic conditions. Using a randomized research design and a multivariate regression model, we evaluated the effectiveness of a hybrid mHealth model—the integration of remote patient monitoring with an applied health technology and digital disease management platform—on 45-day hospital readmissions for patients diagnosed with CHF. We find a 78% decrease in the likelihood of CHF hospital readmission for patients who were assigned to the digital disease management platform as compared to patients assigned to control.
Keywords
Background
Congestive heart failure is one of the most common diagnoses in the elderly United States Medicare (⩾ age 65) population 1 and the proportion of the population diagnosed with CHF has more than doubled in the last two decades,2–4 due, in part, to increased longevity and subsequent growth in the diagnosis of chronic conditions. 5 Each year over one-half a million patients are diagnosed with CHF and, in total, more than five million Americans are currently living with a CHF diagnosis. 3
This patient population has a particularly high readmission rate, with a 20% 30-day risk-adjusted readmission rate and a nearly 60% (57.2) 1-year risk-adjusted readmission rate (according to a study with 115,000 patients across 416 U.S. hospital sites). 6 Readmissions are a key driver of increased costs 3 and hospital stays are associated with a substantially increased risk of mortality. 7 As such, clinicians and researchers have spent considerable effort devising interventions to reduce the likelihood of readmission for patients with CHF. Traditional interventions consisting of comprehensive education, diet, prescription review, and/or intensive in-person follow-up have been shown to improve life quality as well as reduce the likelihood of any admission, the number of admissions, length of stay, and mortality. 4
The rapid rise of electronic health (eHealth), and in particular the subfield of mobile health (mHealth), presents a promising new pathway for reducing hospital readmissions of CHF, and, more generally, the management of chronic conditions. EHealth, broadly defined, “encompasses the use of information and communication technologies (ICTs) in the support of health and health-related activities,” including electronic medical records, remote/telemonitoring, and mobile health. 8 MHealth can be defined as “the use of mobile computing and communication technologies for health services and information,” including “the use of mobile devices in collecting aggregate and patient level health data providing healthcare information to practitioners, researchers and patients, real-time monitoring of patient’s vital signs and direct provision of care (via mobile telemedicine using smartphones, mobile applications, social media, analytics, and e-Cloud.” 9
A key technological innovation of mHealth, specifically, is the potential to directly and rapidly monitor patients’ vital statistics on an ongoing basis, distinguishing such interventions from more traditional out-of-hospital interventions with slower feedback cycles and, critically, increasing medical professionals’ ability to early identify concerning changes to a patient’s health status and prevent more severe medical record events. Models that integrate remote patient monitoring of patient vitals with applied health technology and digital disease management are a promising new approach for improving care, reducing readmissions, and controlling costs, particularly with respect to the management of chronic conditions. Importantly, the electronic nature of these interventions can help lower the price point for post-discharge monitoring. 10
The quantity of cardiology research examining the impact of eHealth on such outcomes as prevention, early detection, treatment, stabilization, and self-management has blossomed over the last decade. De la Torre Diez et al., 11 through a literature review of over 100 articles in the field, finds a generally positive impact of such “eCardiology” technological uses as remote monitoring and telemedicine on patient outcomes. The adaptation of effective eHealth applications to cardiology are also of enormous interest to and supported by many professional cardiology organizations, with entire professional association meetings having been held on the topic. 8 Yet the pace at which eHealth has been integrated into cardiology remains sluggish. The evidence base has been slow to develop, greater provider and patient feedback is needed to improve ease of use, and further cost-effectiveness studies are required. 8 This study contributes to this growing field by evaluating the effectiveness of one such model by Veta Health, a digital health company that specializes in digital care pathways to improve patient health—for the case of congestive heart failure (CHF).
Materials and methods
Study setting
Patients were recruited for the study from Cardiology and Vascular Associates (CAVA), an urban outpatient cardiology clinic associated with St. Joseph Mercy Hospital Oakland, a private hospital in the Trinity Health System in Michigan. Study participation was voluntary. The hospital’s institutional review board (IRB) granted approval for this study. The clinic attends or consults on approximately 40% of all CHF admissions at the study hospital.
All patients in the study were treated by CAVA physicians and were required to have a primary or secondary diagnosis (Stage C, Class III and IV) of CHF, identified through DRG codes 291, 292, or 293. All patients spoke and understood English. There was no age cutoff for sample eligibility. Patients enrolled in the study interacted with a mobile platform and a Bluetooth wearable device. Patients with a comorbid diagnosis of dementia or cognitive impairment were therefore ineligible for the study. The eligibility window spanned December 2016 to January 2018. Patients were assigned a random anonymous unique patient id. All study analyses were conducted using the deidentified ids. All Veta Health employees underwent HIPAA training and followed data privacy and security standards as stipulated by HIPAA regulation.
Physicians at CAVA and the study hospital partnered with Veta Health (VH), a digital health company that specializes in digital care pathways to improve population health, to provide the front-end platform for patients and providers. The Veta Health platform increases visibility beyond the four walls of the hospital and employs patient self-management strategies.
Figure 1 describes the specific care pathway to improve disease management for CHF patients deployed and is the subject of the study’s core research question. Patients were provided with Bluetooth enabled smartphones with the VH application pre-downloaded. Patients were also provided with a Bluetooth enabled blood pressure cuff and Pulse Oximeter to track vital measurements. Patients were trained to use the devices and VH app by Veta Health’s Patient Success Coordinators (PSCs), Veta Health employees (not health professionals). The devices were used to record all of a patient’s vital measures per the study protocol except for weight. Patients were instructed to manually enter weight readings using their own scale into the digital platform. Patients received emails, pop-up notifications, and self-assessment prompts through the VH app on the smartphone for vital measurement, symptom surveys, and to view additional educational content. This allowed for a trending of a patient’s health. Patients also had the option to manually enter other vital statistics information to help them better self-manage their conditions.
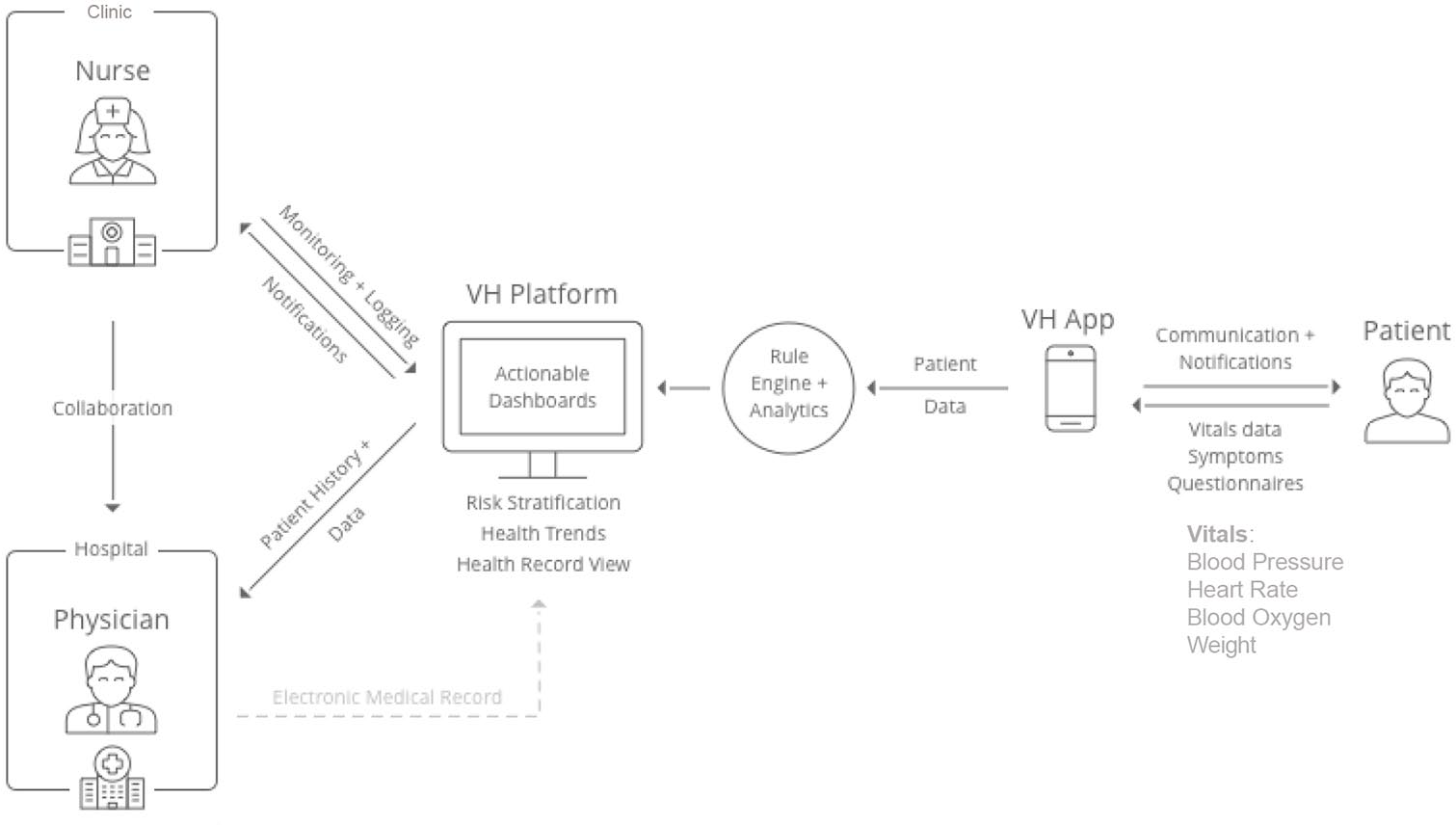
CHF disease management pathway through the Veta Health (VH) platform.
The patient portal (Figure 2) provides on-demand, in-pocket access to a patient’s digital care journey, allowing the patient to understand his treatment plan and how best to optimize his/her health outcomes through active participation and self-monitoring. Automated responses to patient entered data allow the patient to understand how to interpret self-monitoring results and the direct link to the patient’s care coordinator allowed for early intervention in the event of disease exacerbation. Patients could view their data in graphical and tabular form.
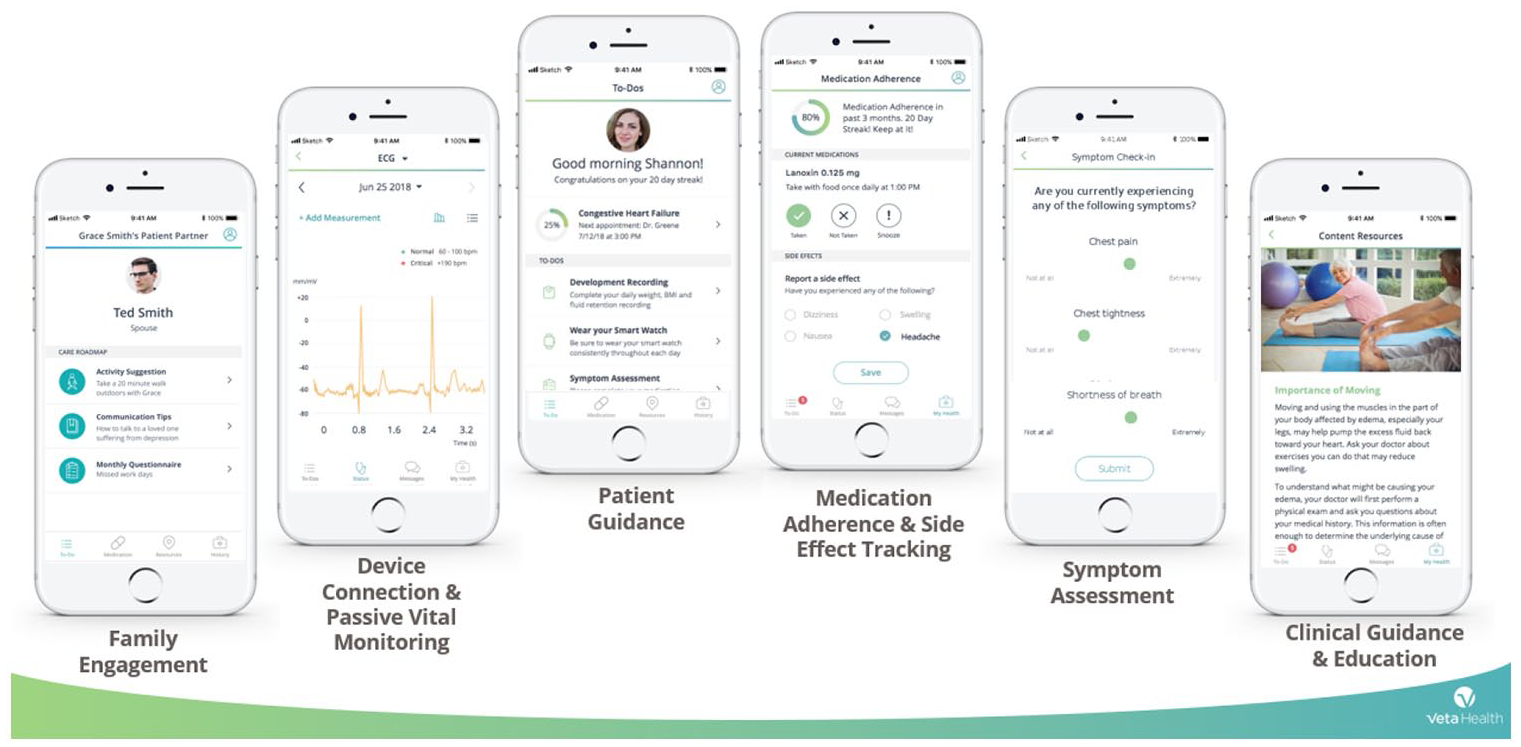
Veta Health patient portal.
Veta Health’s mobile platform tracked real-time information entered and transmitted by the mobile devices throughout the patient journey in order to gather key health indicators at high-risk times for patients. The platform recorded crucial vitals (e.g. systolic and diastolic blood pressure, heart rate, blood-oxygen saturation, weight, and BMI) and condition symptoms through a follow-up assessment, such as shortness of breath, dizziness, and chest pain, which can be predictive of more serious complications, as well as questionnaires. The platform analyzed and translated the clinical information gathered outside of traditional care settings into a digestible, actionable format for clinicians, enabling them to help patients before complications occur (Figure 3(a) and (b)).
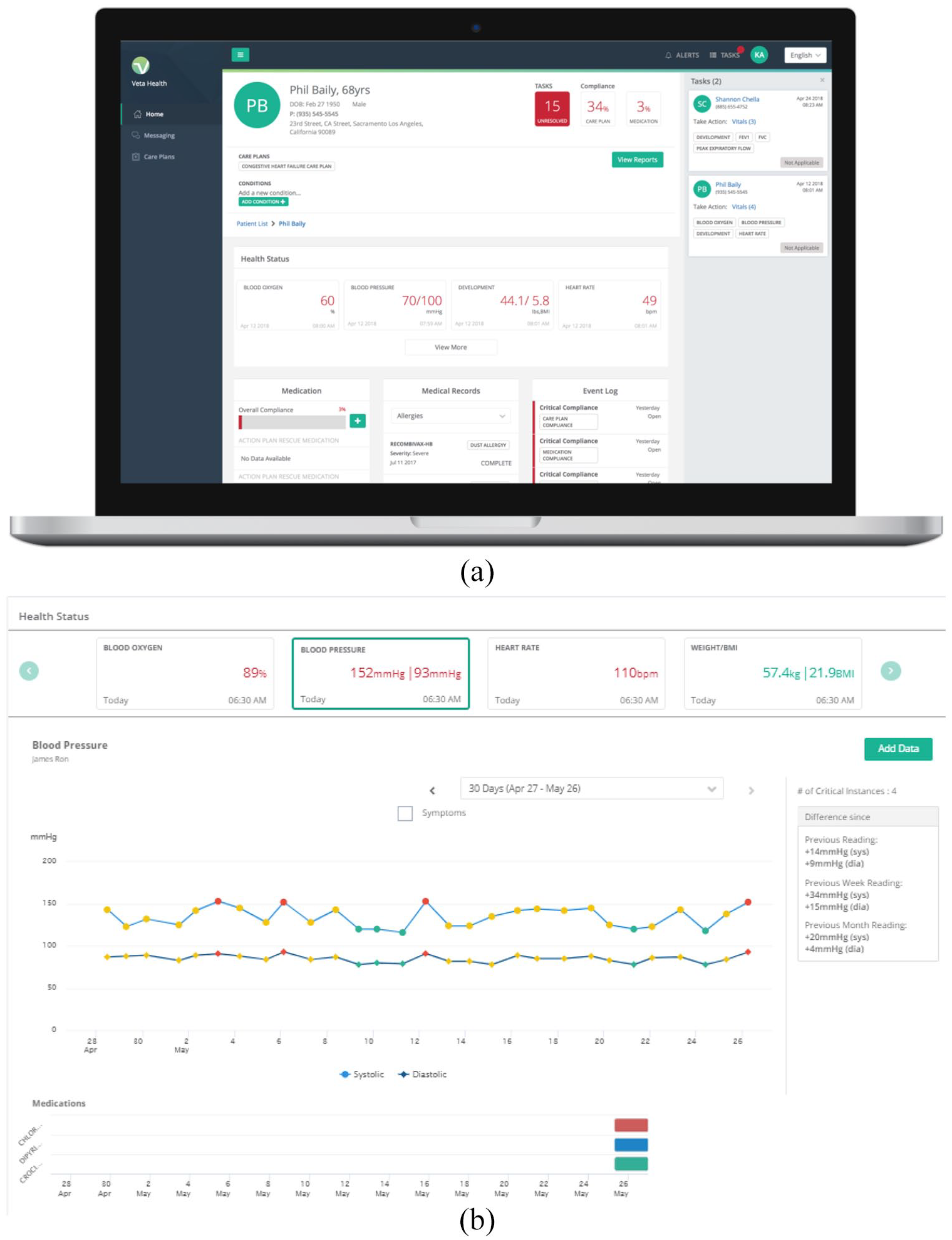
(a, b) Veta Health clinician portal and longitudinal data visualization.
For instance, the platform enabled viewing of longitudinal health trends of patient populations and individual patients, including medical record information, vitals, symptoms, medication regimen, and adherence. Clinicians accessed the platform via a web-based portal and patient health information from the electronic medical record (EMR) was uploaded to the platform and viewed alongside the patient-generated data.
The first line of patients’ care coordination efforts was led by Veta Health’s PSCs. The PSCs received immediate and automated alerts regarding any abnormal health readings and symptoms through the platform. If patient data analyzed in the platform indicated health deterioration, the task list on the right-hand side (Figure 3(a)) auto-populated tasks for the care coordinator to act upon. Upon receiving such notifications the PSCs contacted patients to immediately assess next steps. Based upon a pre-defined set of parameters, they assessed the risk to the patient and whether to elevate the intervention to a member of the physician team in an outpatient setting. If an outpatient visit was recommended, a physician review determined if the patient required a care plan adjustment, outpatient clinic visit, or if the patient required an inpatient admission for further monitoring. Most often, the coordinator was able to communicate with the patient directly to address the potential issue, without escalating care to a physician. The intervention can therefore be thought of as a set of discrete patient assessments of increasing intensity, beginning with utilizing the mHealth technology to automate vital recordings in real-time, complete a symptom assessment upon an abnormal vital reading, and automatically flag abnormal values and trends for immediate review by the Veta Health team. This model of care coordination was designed to effectively identify patients with abnormal health readings in real-time, improve efficiency of diagnosis and care when elevation to the physician team was needed, and reduce the costs associated with preventable inpatient stays. Interventions and outcomes could be tracked alongside to provide insights into treatment success over the long-term. All patient data was hosted on a HIPAA-complaint secure cloud. I
Study design
To test the efficacy of the digital disease management and front-end integrated health technology platform to reduce CHF patient readmissions, we conducted a randomized control trial of patients recruited from the CAVA clinic with a primary or secondary diagnosis of CHF. Every third patient was assigned to the control group. All other patients were assigned to treatment—receiving 45 days of daily remote monitoring through the integrated health technology and digital disease management platform. II In total, the analytic sample consisted of 47 patients in the treatment (“investigational”) group and 15 in the control group (62 total). III We note that participation in the study was conditional on completing the study on-boarding process within 1 week post-discharge. Three additional patients were unable to complete the on-boarding process within the required timeframe and were therefore excluded from the study. Two other patients volunteered for the study but passed away before the onboarding process could be completed. Of the 47 patients in the treatment group in the analytic sample, eight completed a subset of the 45 days of assessment. We include these individuals in the analysis as they were part of the original research design.
Key outcome of interest and patient characteristics
The key outcome of interest is 45-day hospital (inpatient) readmission (0 no, 1 yes). As a measure of prior medical history, we recorded whether patients had a hospitalization record (CHF and otherwise) one year prior to the study. Patient demographic information is derived from patients’ medical history, including race/ethnicity (0 white, 1 black, 2 other), sex (0 male, 1 female), and age.
Analytic strategy
Our analysis begins with a descriptive examination of patients on demographic information across the treatment and control groups. We examine the overall readmission rate of our analytic sample to historic rates at the study hospital and in the medical literature. We then conduct a multivariate linear probability model to examine the impact of the disease management platform on CHF-related readmissions after accounting for time-invariant patient demographics. As a sensitivity to our findings, we also examine overall readmission rates.
For patient i, Y represents the binary outcome of interest, patient readmission within a 45-day post-discharge time window. Treatment assignment, “Tr,” represents whether the patient was in the control (0) or treatment (1) group. “R” is a categorical variable representing the patient’s race, “F” is a binary indicator of the patient’s sex, and “Age” the patient’s age at discharge.
Results
Descriptive results
As described in Table 1, the analytic sample consisted of 29 females and 33 males. The average patient was 68.6 years old. The study included 32 whites, 18 blacks, and 12 patients who declined to specify or were of another race/ethnicity. There were no statistically significant differences by gender, race, or mean age between patients in the treatment and control groups. For instance, the average age in the control group was 70 and the average in the treatment group 68.1 (p < 0.57). 70% of the sample was above the age-65 cutoff for Medicare eligibility (the age in which individuals typically become eligible for U.S. government Social Security Administration sponsored health insurance). There was no significant difference in having a CHF readmission for patients in the treatment and control groups 1 year prior to the study (t(60) = 1.25, p < 0.216). The two groups did vary on their overall admissions 1 year prior, but patients in the treatment group were actually the group with the higher rate of hospitalization (100% vs 80%, t(60) = −3.37, p < 0.001)), which suggests that treatment patients may have been, if anything, in slightly worse average health than patients in control, prior to the study.
Patient demographic characteristics a .
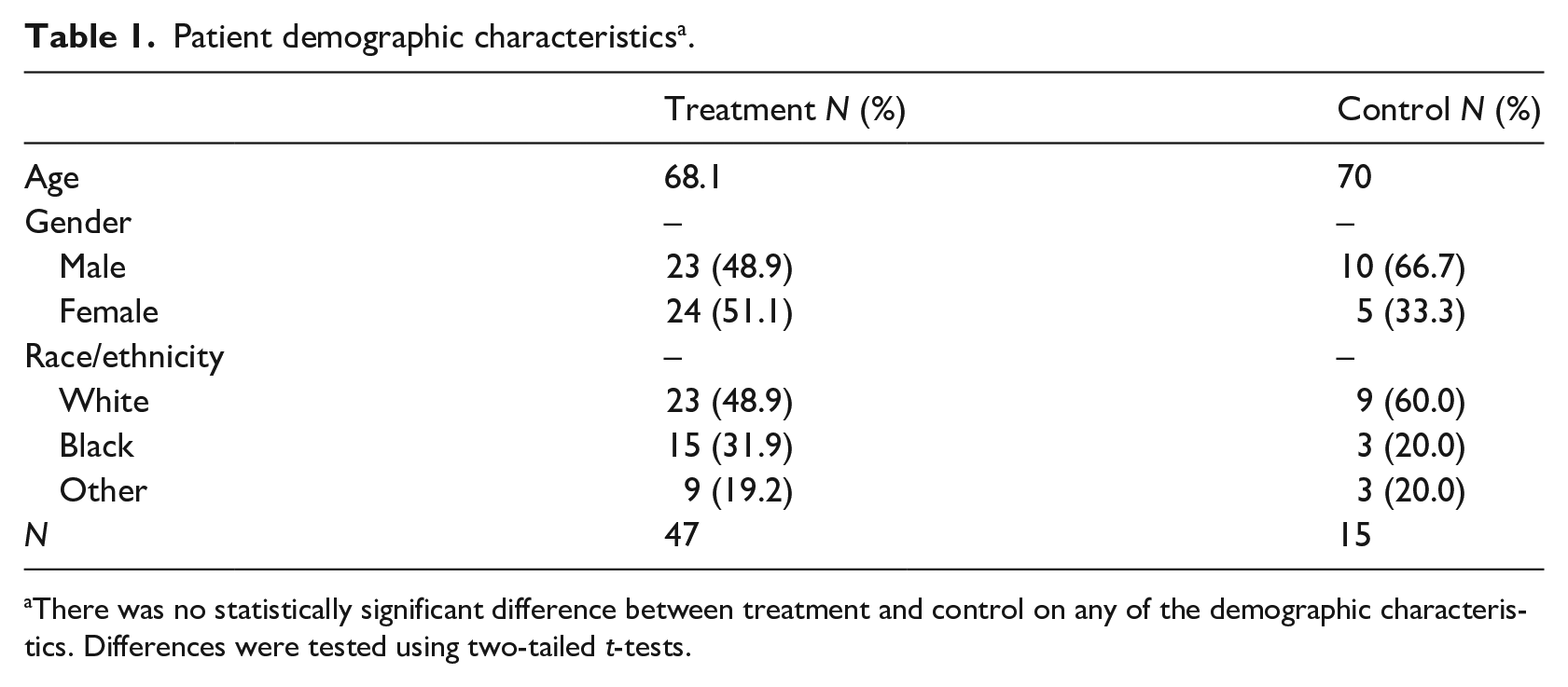
There was no statistically significant difference between treatment and control on any of the demographic characteristics. Differences were tested using two-tailed t-tests.
From 2015 to 2017, the percentage of all Medicare patients readmitted to St. Joseph Mercy Hospital Oakland for CHF (DRG 291–293) within the year averaged just under 25%. 12 The average CHF readmission rate in this study, specifically, was 14.5% (see Table 2—four treatment and five control, i.e. 9 of 62 overall). During the 45-day study window 17 patients in the study sample were readmitted for any reason (eight treatment, nine control). About 8.5% of the treatment group was readmitted for CHF and 17% readmitted for any reason. One-third of the control group was readmitted for CHF and 60% for any reason (see Table 2).
Descriptive patient readmissions within study window.
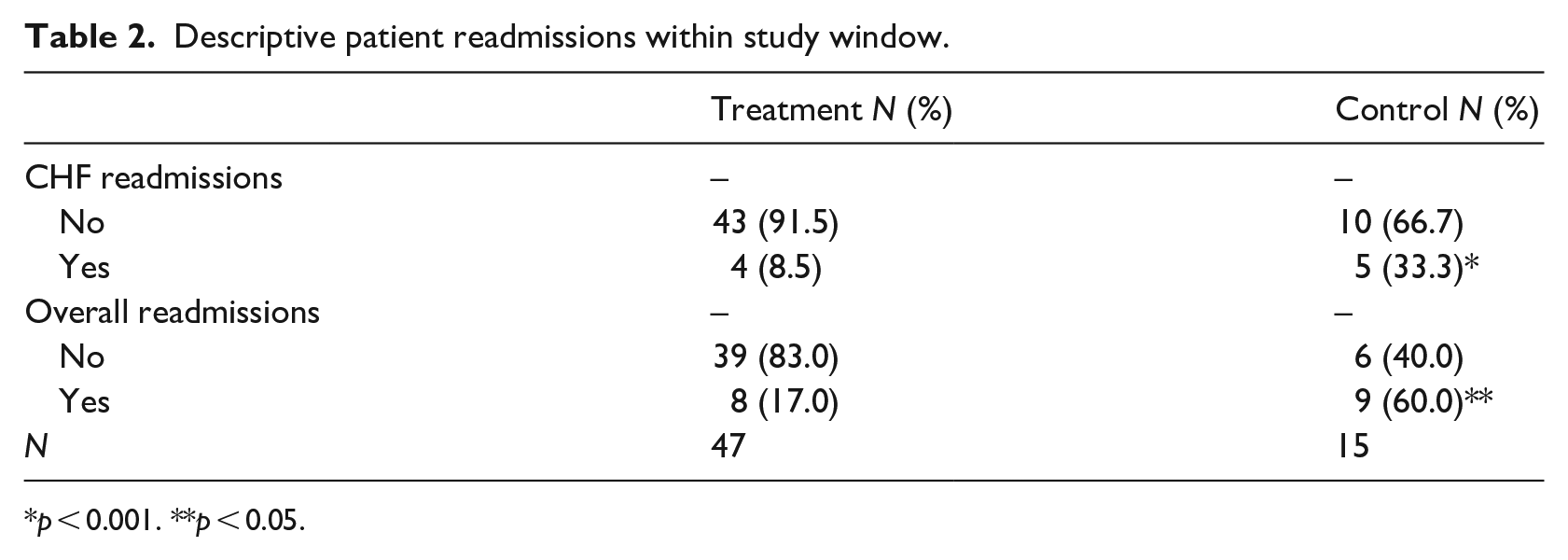
p < 0.001. **p < 0.05.
Figure 4 presents Kaplan-Meier CHF readmission curves across the 45-day study period for patients in the treatment and control groups. The chance of being readmitted was substantially higher for patients in the control group, with over a 25% chance of CHF readmission within the first 10 days. Bivariate t-tests by treatment status indicate that this difference is statistically significant, both for CHF (t(60) = 2.45, p < 0.017)) and overall readmission (t(60) = 3.51, p < 0.001). Appendix Figure A1 presents the results for overall readmissions.
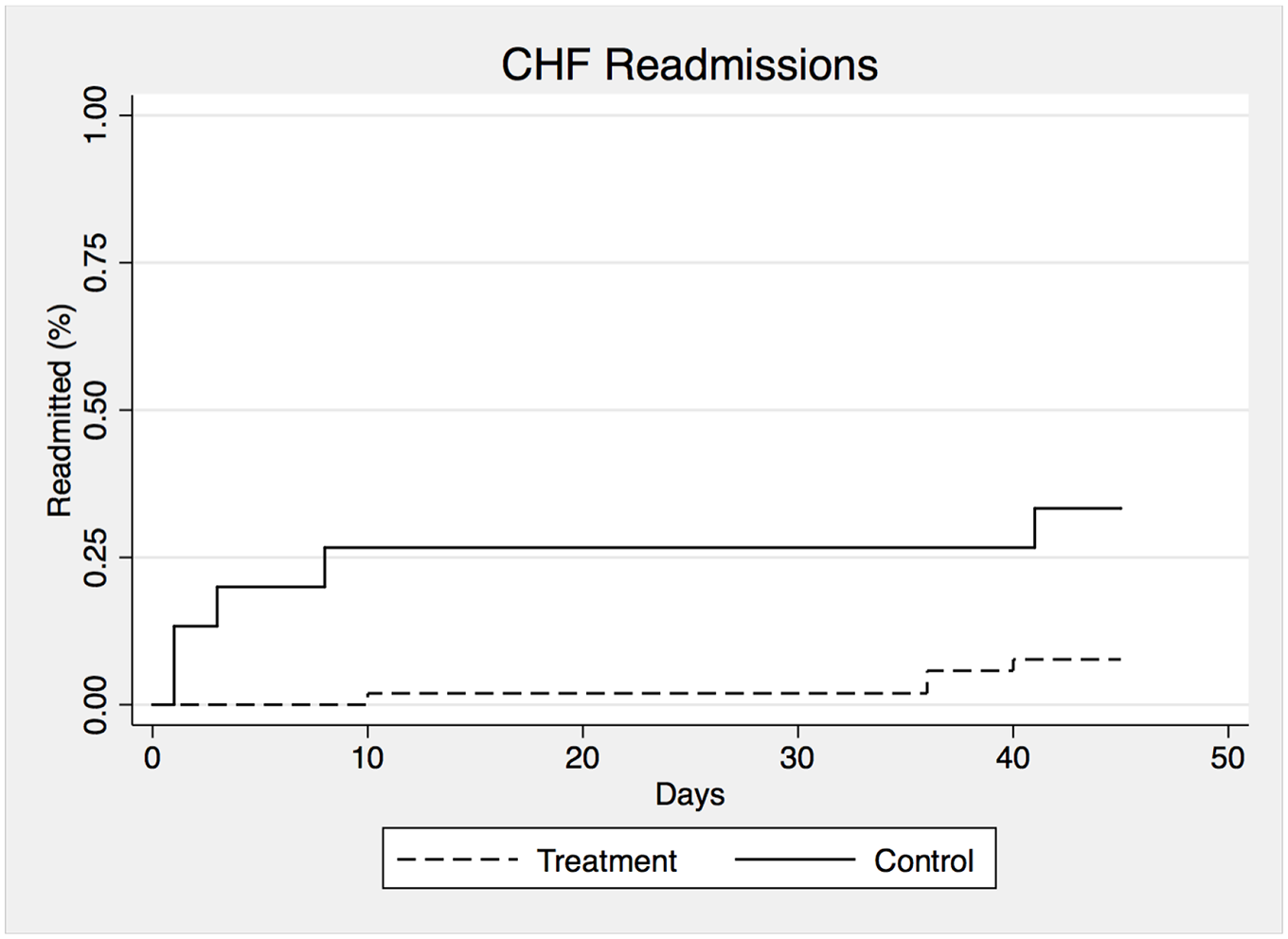
Kaplan-Meier 45-day congestive heart failure readmissions.
Empirical results
We next turn to a multivariate regression model to examine differences in readmissions after accounting for patient characteristics. Results for CHF readmissions and overall readmissions are displayed in Table 3. As described above, the analytic sample (n = 62) includes all patients eligible for the study that completed the onboarding process within a week after discharge and did not pass away prior to completing the onboarding process. After accounting for gender, race, and age, there is a statistically significant reduction in the likelihood of being readmitted for CHF as well as for overall readmissions for patients in the treatment group. The first column in Table 3 displays the results for CHF readmissions. These patients had a 26 percentage point reduction in the likelihood of being readmitted (p < 0.05). The second column is for overall readmissions. The results are stronger for overall readmissions, with a 45 percentage point decrease in the readmission rate for patients that engaged with the platform. In terms of percentages, patients in the treatment group had a 78% reduction in the likelihood of being readmitted for CHF and a similar 75% reduction in the likelihood of any readmission. IV
Linear probability model predicting hospital readmissions. a
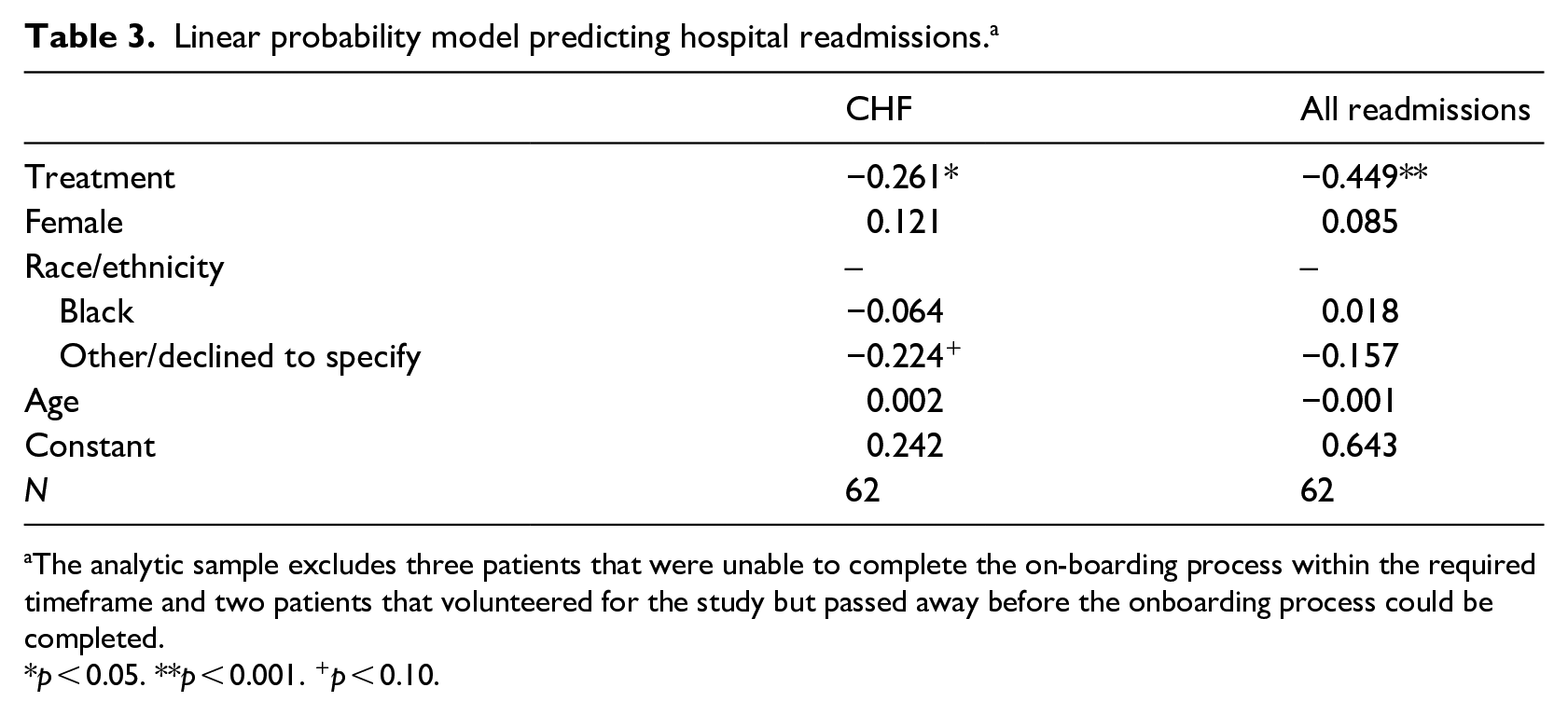
The analytic sample excludes three patients that were unable to complete the on-boarding process within the required timeframe and two patients that volunteered for the study but passed away before the onboarding process could be completed.
p < 0.05. **p < 0.001. +p < 0.10.
Discussion
In this research, we utilized a randomized control trial design to evaluate the effectiveness of Veta Health’s digital disease management platform on 45-day hospital readmissions for patients diagnosed with CHF. Our analyses reveal that patients who engaged with the platform had a 26 percentage point reduction in the likelihood of being readmitted for CHF as compared to patients in the control group. They were also 45 percentage points less likely to be readmitted overall. Both outcomes translate to an approximate 75% reduction in readmissions.
We calculate that the benefits of employing the digital disease management greatly exceed the costs. One estimate of the national average cost of CHF readmissions is approximately $13,000 (Qasim, 2012), while another places the estimate closer to $15,700.13,14 The realized cost-savings per prevented readmission could be substantial.
We acknowledge several limitations with the study. Although the reduction in CHF (and overall readmission) for patients with CHF is quite large, the study was conducted for a relatively small sample, and with different size treatment and control groups. Because results based on small samples have less precision, our findings and cost savings analysis should be interpreted with this in mind. Although there were no statistically significant differences between the treatment and control groups on demographic characteristics at baseline and there was no difference in the likelihood of CHF admission 1 year prior to the study, the treatment group was more likely to have had a hospital admission within 1 year prior to the study overall, which could suggest slightly worse average health at the onset of the study. In addition, as a hybrid mHealth model, we are unable to decompose the findings into the intervention’s various component parts—remote patient monitoring, applied health technology and a digital disease management platform, and patient success coordinators.
The research took place at a hospital that serves a highly educated population and is therefore not generalizable to other settings, including urban areas with large low-income populations. The study results are not generalizable to CHF patients who have a comorbidity of dementia or cognitive impairment. Patients were only followed for 45 days, though CHF is a chronic condition. Future research in this area is needed to explore the application of such technology over a longer timeframe, to other chronic conditions, and to unpack the mechanisms driving the reduction in readmissions.
Conclusion
This research builds upon earlier work in the area and adds a unique contribution to the growing mHealth, as well as the broader eHealth fields. We evaluated the effectiveness of a hybrid mHealth model—the integration of remote patient monitoring with an applied health technology and digital disease management platform. We utilize a randomized control trial research design to examine the impact of the technology and platform on 45-day hospital readmission for patients diagnosed with CHF. We find a 78% (26 percentage point) decrease in the likelihood of hospital readmissions for patients who were randomly assigned to the digital disease management platform as compared to patients assigned to control. The results hold after accounting for patient demographic characteristics in a multivariate linear probability model. Our findings are robust to alternative model specifications and not sensitive to changes to the inclusion/exclusion restrictions to our analytic sample.
While traditional outpatient interventions have failed to bend the cost curve of chronic disease management, mHealth presents a promising alternative model. Such technology offers healthcare providers the ability to track real-time vital statistics and health information of patients outside the hospital, which can immediately alert providers to any abnormal vital readings, expedite the care delivery process, and more accurately allocate patients to the appropriate intensity of care. As Saner 8 describes, an underdeveloped evidence-base is a key barrier to the adaptation of mHealth technology in cardiovascular disease management. Our findings help fill this gap by suggesting that the use of a digital disease management platform and remote patient monitoring could substantially reduce patients’ hospital readmissions in the case of CHF, and therefore decrease the cost of cardiovascular disease management. In particular, care coordinators, such as Veta Health’s Patient Success Coordinators, can play an important role in the platform implementation in instances where the partner institution does not have care management teams in place to monitor the platform. The integration of mHealth technology to the management of CHF and other chronic conditions may increasingly become a key component of care delivery and cost containment as the proportion of elderly Americans diagnosed with chronic conditions continues to increase.
Footnotes
Appendix
Acknowledgements
The authors would like to thank Dr. Augustine George for his assistance with this work.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Veta Health provided free access to the Veta Health mobile platform and Patient Success Coordinators to all patients enrolled in the study at the CAVA clinic, a cardiology clinic associated with St. Joseph Mercy Hospital Oakland. With IRB approval, Veta Health planned and implemented the research trial. Veta Health was responsible for gathering all data through the health platform and worked with medical professionals at St. Joseph Mercy Hospital Oakland to gather and verify patient medical history, demographics, and readmissions information. The authors received no remuneration (financial or otherwise) for the study. Veta Health hired an independent academic research consultant at the University of Pennsylvania to analyze the data. The consultant was compensated by Veta Health for their time. The independent academic consultant had complete independence and autonomy for data analysis decisions and reporting of the findings. Veta Health verifies this independence, had full access to all of the data in the study, and takes complete responsibility for the integrity of the data and accuracy of the data analysis. Both Veta Health and the academic research consultant contributed to the interpretation of the data, writing of the research report, and decision to submit the manuscript for publication. One of the authors of this study, is a co-founder of Veta Health.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical standards
This research was approved by St. Joseph Mercy Hospital Oakland’s institutional review board (IRB), a copy of which was provided to the Editor of this journal.

