Abstract
Worldwide, Chronic Kidney Disease (CKD), directly or indirectly, causes more than 2.4 million deaths annually with symptoms generally presenting late in the disease course. Clinical guidelines support the early identification and treatment of CKD to delay progression and improve clinical outcomes. This paper reports the protocol for the codesign, implementation and evaluation of a technological platform called Future Health Today (FHT), a software program that aims to optimise early detection and management of CKD in general practice. FHT aims to optimise clinical decision making and reduce practice variation by translating evidence into practice in real time and as a part of quality improvement activities. This protocol describes the co-design and plans for implementation and evaluation of FHT in two general practices invited to test the prototype over 12 months. Service design thinking has informed the design phase and mixed methods will evaluate outcomes following implementation of FHT. Through systematic application of co-design with service users, clinicians and digital technologists, FHT attempts to avoid the pitfalls of past studies that have failed to accommodate the complex requirements and dynamics that can arise between researchers and service users and improve chronic disease management through use of health information technology.
Keywords
Background
Chronic kidney disease (CKD) directly or indirectly causes more than 2.4 million deaths annually. 1 In Australia, CKD affects up to 2 million people, with 2500 commencing renal replacement therapy each year and 14 diagnosed with end-stage CKD each day.1,2 CKD is associated with increased cardiovascular disease (CVD) risk and declining kidney function is associated with reduced health-related quality of life, independent of other co-morbidities.3,4 Unlike CVD and stroke, rates of CKD are not improving.4,5 The World Health Organisation has called for targeted screening, prevention and early treatment to combat world-wide increases in the prevalence of CKD. 1
CKD is defined as kidney damage and/or reduced kidney function lasting 3 months or more. The five stages of CKD range from structural abnormalities that do not adversely impact on kidney function through to severe, end-stage disease requiring supportive palliation or renal replacement therapy (kidney transplant or dialysis). 6 Generally, symptoms do not appear until kidney function has deteriorated to stage 5, so testing for earlier stages of disease is often overlooked. In Australia, less than 10% of people with CKD know they have the condition 7 and almost 1 in 5 (18%) patients start dialysis within 3 months of discovering they have CKD. 8 CKD diagnostic and treatment delays also result in higher cardiovascular morbidity and mortality rates. 3 CKD is under-detected by general practitioners (GPs) and there is little knowledge about CKD in the community.4,7,9,10
Clinical guidelines support the early identification and treatment of CKD6,11,12. Statin medications that reduce the risk of atherosclerotic events and renin angiotensin system blockers that lower blood pressure and reduce proteinuria can delay progression of CKD, improve clinical outcomes and reduce health care costs. 13 Thus, there is significant potential to improve the trajectory of CKD through improved identification of patients at risk, or in the early stages, of the disease and through better implementation of clinical guidelines. General practice, where most Australians receive their medical care, is a key setting in which to test interventions and explore new models of care to optimise identification and management of CKD and chronic conditions more generally. 14
Models of care underpinned by technology can be drivers of successful chronic disease management
International evidence indicates that new models of care are needed to improve CKD outcomes.15,16 Successful chronic disease management has been linked to the use of ‘Chronic Care Models’ (CCMs). 17 The CCM considers the patient to be an active participant in their health care and includes planned interactions that support evidence-based care and regular review by a team of providers, each with a defined role in that patient’s care (coordinated care). 18 Primary health care encompassing at least two components of the CCM improves both health care practice and health care outcomes for patients. 19 Key elements of the CCM include decision support and clinical information systems, which are often underpinned by Health Information Technology (HIT). The challenge is to develop and implement HIT that avoids the pitfalls of the past, which include failure to adopt, scale up, spread and/or sustain use 20 and lack of use due to implementation being resource intensive 21 ).
Multifaceted quality improvement interventions and HIT
A key component of the CCM is a commitment to continuous quality improvement. Continuous quality improvement (QI) in health care is the systematic application and review of data to improve services and the delivery of health care. QI must be able to demonstrate measurable improvements in outcomes and can include processes that, over a defined time period, summarise clinical performance so that professionals can adjust behaviours and effect change. The process of audit and feedback of health data in electronic format is now a common intervention to support clinical behavioural change. 22 Studies have demonstrated that technological interventions including audit, feedback and decision support technology can facilitate improvements in CKD diagnosis and management in primary care.23,24 However, costs associated with academic detailing are high 21 and exploration of less labour intensive, and hence less expensive, interventions are required to ensure that interventions can be delivered at scale.
Our previous studies exploring implementation of a software program that interacted with existing primary care software to identify and manage patients at risk of, or diagnosed with CKD, type 2 diabetes and cardiovascular disease10,25 have provided critical learnings on many of the enablers common to successful implementation of quality improvement programs underpinned by HIT in Australian general practice:
(1) Good leadership, through identification of ‘Champions of Change’ and access to expert opinion was recognised as crucial to success.
(2) Implementation was more likely in practices with an ‘organisational culture’ supportive of QI programs.
(3) Audit and benchmarking reports were important motivators for QI.
(4) Health outcomes were more likely to be documented where clinical systems were automated or streamlined.
In common with the findings from other QI studies, competing clinician’ priorities and the resources required for academic detailing were major barriers to scaling-up the project. 26 Additional learnings suggested that HIT-chronic disease management systems could be improved by streamlining QI and integrating point of care decision aids and access to guidelines.10,25
An Integrated Knowledge Translation approach may be a facilitator of HIT implementation
The Knowledge to Action (KTA) cycle is a widely recognised guiding framework of knowledge translation based on more than 30 planned-action theories. 27 It consists of the knowledge funnel, through which information becomes more refined and valid and the action cycle, the process of knowledge implementation. 27 This can be used to inform the development and implementation of HIT. Despite decades of research that have applied KTA principles, significant knowledge-action gaps remain. A research strategy that is inclusive of its stakeholders is considered more likely to be accepted, more likely to be adopted into policy and practice and more likely to be disseminated to audiences amenable to change.28,29 ‘Co-design’ takes this approach so that end-users (health professionals and/or patients who use research results to inform their decisions 28 ) are empowered to become active participants, rather than passive recipients, in the design process. Where co-design includes knowledge production and integration, researchers, stakeholders and end-users collaborate through the entire research process and the knowledge-action gap is bridged most effectively.28,29 This is known as Integrated Knowledge Transfer (IKT). Levels of engagement by end-users may be proportional to the strength of outcomes that can be achieved 30 but the lack of reporting of theoretical underpinnings in IKT studies 31 means that the mechanisms for success, or failure, are not clearly understood. To advance the strength of evidence around IKT, Gagliardi et al. 32 recommend that future studies provide more detailed reporting of: the preparatory phase of partnership formation before embarking on the study; the proposed processes of interaction between researchers and end-users (meetings and their mode); the roles and realms of engagement of research users in the research process; and the theoretical underpinnings that will support evaluation.
This paper reports the protocol for the development of a technological platform called Future Health Today (FHT). FHT is a software program that aims to optimise the early detection and management of chronic disease at a practice population (via the FHT dashboard) and individual patient level (via point of care tool), reduce variation in practice by facilitating access to clinical guidelines and underpin quality improvement activities in general practice. Our initial focus will be on CKD, but the platform will be expanded to include other chronic diseases in the future. This paper describes the protocol for the co-design and initial pilot work of FHT. This is the first stage in a planned program of work which will subsequently include a larger feasibility study and evaluation in a randomised controlled trial.
Methods/design
Aims and research questions
The aim of this study is to co-design and explore the implementation of FHT in two general practices to optimise and tailor FHT to the needs of ‘real world’ practice (Aim 1). Our secondary aim is to examine the impact of FHT on screening people at risk of CKD and identifying, documenting and managing people with CKD (Aim 2).
The key research questions that will be addressed are:
What are the key components which FHT should include in order to implement quality improvement activities for chronic disease in general practice?
What are the barriers and facilitators to the implementation of the Future Health Today technology platform in general practice?
What is the impact of the Future Health Today technology platform on (a) screening for CKD in people with risk factors; (b) diagnosis of CKD; and (c) pharmacological management of diagnosed CKD?
Study design
This study will employ a mixed methods approach, conducted over 12 months, in two phases (see Figure 1). The first will be the development of the FHT platform using co-design and informed by previous quality improvement studies (Aim 1). The second phase is a pilot study in two general practices that will be invited to test the FHT prototype (Aim 2; Figure 1).
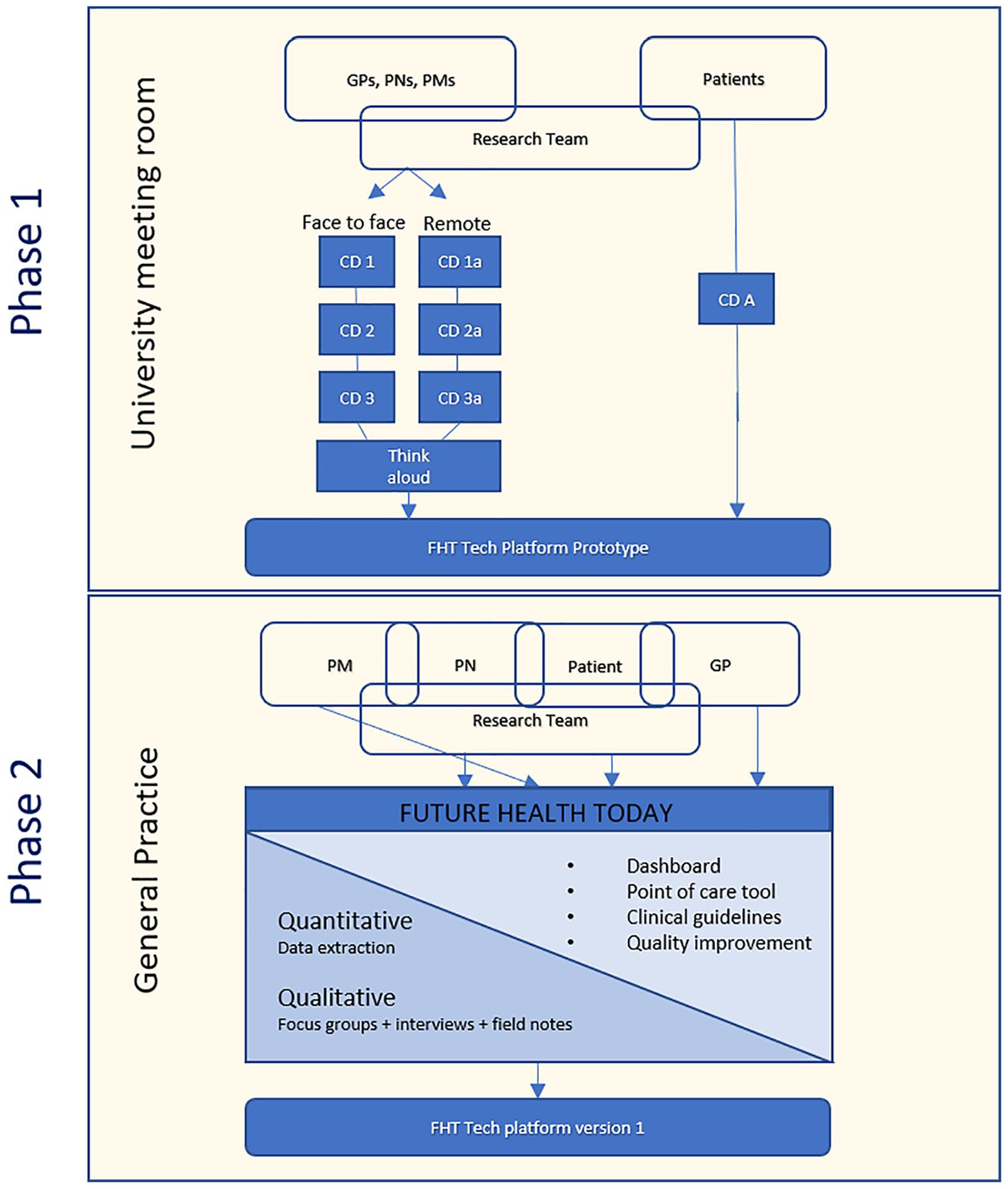
Overview of study design.
PHASE 1: Development of FHT
Preparatory phase/partnership and research team
The research team for this project includes: renal and other disease specialists, academic GPs (including one who works at the site selected for Phase 2 of this study), an on-site GP with an interest in HIT, health informaticians, academic lawyers, a project officer, a statistician, recruitment experts and HIT experts including a business analyst, analyst programmers and a technical architect. Clinicians and clinician researchers on the team have collaborated previously on QI programs utilising HIT in general practice.
Theoretical underpinnings
Clinical Performance Feedback Intervention Theory (CP-FIT), a theory of clinical performance improvement that underpins the action phases of the Knowledge to Action Cycle, has been selected to explore the effectiveness of the implementation of the FHT program. 33 CP-FIT enhances IKT by providing a detailed, clinically focused lens on factors that impact and enhance IKT to create best possible clinical performance outcomes. CP-FIT, designed from a qualitative meta-synthesis of 30 existing behaviour change theories, states that feedback on clinical performance works within a ‘cycle of sequential processes’ and that failure of a single process within the feedback cycle risks diminishing or destabilising the entire process. The feedback cycle is influenced by ‘Feedback’, ‘Recipient’ (usually health professional) and ‘Context’ variables, which contribute a total of 42 hypotheses, operating by seven ‘Mechanisms’, to provide a comprehensive explanation of how feedback in health care works. CP-FIT will inform the development of interview schedules and will be utilised in a framework analysis of the focus group and semi-structured interviews in Phase 2 (Figure 1).
Processes of interaction, roles and realms of engagement
Research strategy for co-design and implementation of Future Health Today
Phase 1 focusses on the co-design of the FHT software (Months 1–6). We used ‘Service design’ as our co-design research strategy. 34 Service design thinking places the end-user (in this case, GPs, practice nurses, practice managers and patients) ‘front and centre’, although all stakeholders are included in the design process (specialists, reference groups, software developers). Service design thinking encompasses co-design and takes a holistic approach that considers the whole environment (e.g. including the patient’s home environment). Tools, such as patient journey maps, storyboards and service prototypes are used to visualise inter-related sequences of events and evidence is gathered along the way to make the service tangible. The process is iterative, tests and refines prototypes, gathering end-user, stakeholder and expert feedback through multiple cycles 35 and provides a natural fit with the ‘Action-Cycle’ of the KTA framework. Software developers (staff on this project) and researchers will participate in the co-design meetings so that, in addition to active engagement, they are able to communicate whether the design elements suggested by participants are able to be incorporated into the technology platform.
Recruitment, participants and data collection
Approximately twenty health professional participants (GPs, practice nurses and practice managers) for Phase 1 will be recruited via e-newsletter and email from general practices in an established primary care research network representative of the breadth of Victorian general practice in terms of location (metropolitan, regional and rural), structure (small private and corporate general practices, community health centres) and billing structures (Medicare ‘bulk billing’, privately co-funded or mixed billing. Three meetings will be conducted, with the offer of either face-to-face (held on a university site) or ‘remotely’, via a university organised videoconference link, according to participant’s location and ability to attend. Patient-consumers (patients with a chronic disease, at risk of chronic disease or caring for someone with a chronic disease) will also be invited to a face-to-face meeting at a university site.
Meetings are anticipated to last 1.5–2 h and will be digitally recorded. to capture participants’ interaction with patient journey maps, storyboards and service prototypes during the sessions. Four health professionals that participated in the co-design sessions will be invited to participate in a ‘Think Aloud’ 36 session in a simulation environment where they will talk their thoughts aloud whilst using the first version of FHT-prototype and make suggestions for improvement.
Audio recordings will be transcribed and de-identified before analysis. A multilevel analysis will be conducted in this phase of the project. The first stage of analysis will involve thematic and descriptive analyses, initially identifying the technological requirements and preferences of participants for FHT. Second stage analysis will examine themes arising from the data relating to contextual variables, including technological capability, current recall and audit practices and quality improvement activities. These contextual variables will be further analysed using the CP-FIT framework. 33 The analysis will also examine potential mechanisms facilitating or impeding implementation. A codebook will be developed from themes arising from the data and from the CP-FIT framework. Coding will be undertaken by two researchers, with cross-checking and co-validation occurring at code development and initial data analysis.
PHASE 2: A pilot study in general practice invited to test the FHT prototype (Aim 2)
Recruitment and participants
Phase 2 (Months 7–11) is a pilot and feasibility implementation of FHT, which will be conducted in a single, large, metropolitan general practice (Practice 1). The practice is situated in an area with higher than average levels of socio-economic disadvantage. 37 The majority of consultations (more than 80%) are directly billed to the public insurer –Medicare Australia 38 – and do not incur out of pocket fees for patients (described as ‘mixed billing’). This will be supplemented by technical testing and user feedback (focus group) in a second general practice (Practice 2) utilising a different general practice electronic health record (EHR) system.
GPs working at Practice 1 (including two members of the research team) have an existing relationship with the hospital research partner, an interest in the use of technology in general practice and a commitment to research. The study aims to recruit key staff (three GPs, one practice nurse, a health assistant involved in chronic disease recalls and the practice manager) to provide feedback about FHT.
Implementation includes installation of FHT onto relevant staff computers (those recruited to the study) and education regarding utility and QI processes.
Data collection and analysis
Qualitative data
It is anticipated that semi-structured interviews will be conducted fortnightly for the first 6 weeks post-implementation, with a focus group to be conducted 3–4 months post-implementation. Focus groups and interviews will explore health professionals’ level of interest and engagement utilising FHT, opinions regarding the barriers and facilitators of implementation, improvements in practice that they attribute to the program, any adverse consequences experienced, perceived and potential medicolegal risks and recommendations to improve the program (e.g. software modifications, education requirements, optimisation of feedback reports).
As part of routine clinical care, patients identified as at risk of CKD, who have pathology consistent with CKD or who have CKD that may benefit from optimisation of management at this practice will be invited to make an appointment for review. After obtaining informed consent, a sub-set of patients will be recruited into a focus group (or interview where this is not possible) by an academic GP-researcher who works in the practice. This aims to explore the experience of patients that have been recalled to attend the practice as a result of utilisation of the FHT tool. Patients that regularly consult with this GP will be excluded from the study to avoid any perceived coercion to participate.
All participants will be asked to provide written (verbal consent where interview is over the phone), informed consent. Focus groups and interviews will be recorded and transcribed by a professional transcription service. Transcripts will be de-identified prior to analysis and stored as password protected digital text files on a secure server at the University to protect participants’ privacy. Field notes from participant observations, practice meetings and transcripts will be analysed in NVivo 39 using an inductive and deductive coding structure informed by CP-FIT.
The analysis strategy for this phase of the project will utilise the coding framework identified in Phase 1, and build it further using a codebook developed using the CP-FIT data dictionary 33 and codes arising from the Phase 2 data. This consolidated codebook will be reviewed by multiple researchers to verify the veracity of the coding structure. Data collected in this phase will be analysed by two researchers, with cross-checking and co-validation occurring at code development and initial data analysis. It is anticipated that analyses will identify barrier and enabling mechanisms influencing the implementation of the feedback/audit tool and the use of the tool, within given contexts.
Quantitative data
Patient data will be extracted from the practice EHR using GRHANITE® a privacy protecting extraction tool that de-identifies data at the source of its extraction. 40 The deidentified patient data is stored in Patron, a general practice data repository, created and operated by the Department of General Practice, University of Melbourne, 41 on a secure server at the University of Melbourne. 42 Analysis will only include active patients, defined as those that have attended the GP practice at least three times within the past two years. 43 Active patients will be included if 18 years of age or older and excluded if they are already receiving dialysis treatment or have a kidney transplant. Baseline is defined as the day that the intervention is first introduced into the practice. Baseline demographics and clinical factors from all patients meeting these criteria on the day of intervention commencement will be summarised using descriptive statistics. Demographic and clinical characteristics will include age in years, sex, as well as known risk factors for CKD including Aboriginal and Torres Strait Islander identification, body mass index, smoking status, family history of CKD, hypertension, diabetes, cardiovascular disease and acute kidney injury.
Baseline numbers and percentages of patients in the following CKD-related categories will also be reported. These categories have been informed by precursor studies10,25 and will be finalised in the co-design process which has been described above.
Categories include:
Patients at risk of CKD without up-to-date diagnostic testing for CKD
Patients with a possible diagnosis of CKD (based on a single abnormal pathology test which has not had a subsequent test to confirm CKD, performed at least 3 months apart) with no coded CKD diagnosis
Patients with pathology tests consistent with CKD (based on at least two pathology tests at least 3 months apart) with no coded CKD diagnosis
Patients with a coded diagnosis of CKD with no prescribed ACE inhibitor or angiotensin 2 receptor antagonist in the presence of hypertension, albuminuria or high absolute cardiovascular risk
Patients with a coded diagnosis of CKD with no prescribed statin, at high absolute cardiovascular risk and aged less than 75 years old, or if aged 75 years or older with coded diagnosis of cardiovascular disease
The practice will select one of the CKD-related categories above as the focus of a quality improvement audit activity. Eligible patients at baseline will also be assessed at 4–5 months post start of intervention. The number and percentage of patients at target with respect to the quality improvement activity will be the outcome. This outcome will be summarised in a 2 × 2 contingency table pre and post intervention. Differences in the paired proportions will be tested using McNemar’s chi-square statistic with p-value reported. Odds ratios will be calculated using the ratio of the discordant pairs and be reported with their respective 95% confidence intervals.
We also plan to do an interrupted time series analysis on the same cohort of patients incorporating expanded time frames before and after initiation of the intervention. Interrupted time series studies can be used effectively to study non-controlled interventions, offering the ability to assess whether factors other than the intervention itself may have contributed to any effect.44,45 The interrupted time series analysis using segmented regression modelling will be conducted using fortnightly outcome measurements across three time periods (segments): (i) Pre-intervention segment-10 months prior to commencement of the intervention; (ii) Transitional segment-5 month duration starting from commencement of the intervention; (iii) Post-effect segment-5 month duration commencing at the end of the transitional period. Proportions of patients meeting the criteria for each of the CKD-related categories listed above will be assessed. Level and trend changes between these time periods will be reported (with 95% confidence intervals and p-values) in a forthcoming publication. All statistical analysis will be conducted using Stata MP®. 46
This proof of concept study will be used to inform subsequent iterations of this intervention that will be tested on a much larger scale.
Discussion
FHT is an ambitious attempt to realise the opportunity that HIT offers towards improving the management of chronic diseases. Our approach involves the careful and systematic use of co-design with service users, clinicians and digital technologists to increase the likelihood of adoption of the new technology. 47 We anticipate benefits of co-design for FHT to include generation of new ideas, optimised cooperation between stakeholders and users, ensuring that FHT meets the needs of endusers, optimised project management and high satisfaction of users. 48 Previous attempts at implementation of digital technologies to improve chronic disease management have failed to appreciate the complex requirements and the dynamics that can arise between researchers and service users at the level of service models.49,50 Whilst our initial focus is on CKD, we aim to expand the platform to include other chronic diseases in the future.
This study protocol provides a clear description of an IKT process consistent with Gagliardi et al.’s 32 proposal including a clearly defined theoretical framework. It is, therefore, likely to make a valuable contribution towards elucidating the components that work in IKT research and further promote the structured steps involved in the early development of new HIT that is anticipated to lead to optimised functionality and use prior to larger trials and implementation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Paul Ramsay Foundation; and the Australian Government’s Medical Research Future Fund (MRFF) Rapid Applied Research Translation Program in conjunction with the Melbourne Academic Centre for Health.
