Abstract
Healthcare has entered the information age. This will deliver huge opportunities for healthcare providers to deliver more individualized treatments for patients, and as such improve outcomes. Nowhere is the prospect greater than in cancer care. Healthcare providers now need to manage the challenge of how to best capture, interpret and exploit insights from real-world clinical data. A significant aspect of cancer care is the challenge of preparing and conducting tumor boards. Currently, data are distributed across multiple systems and cannot be easily aggregated or integrated. In recognition that no suitable solution existed, the University of Missouri School of Medicine, in partnership with Roche, have co-developed and co-implemented a digital tumor board solution. This article describes the development process and the enablers and barriers for adoption from a clinician’s perspective. In addition, it reflects on some of the key factors for success and some of the future opportunities.
Medical information overload
Medical knowledge has grown exponentially and is expanding faster than our ability to assimilate and apply it effectively. By 2020, it is predicted that medical knowledge will double every 73 days, compared to 50 years in 1950. 1 In oncology, greater disease understanding has enhanced the ability of dedicated cancer treatment centers to deliver more personalized cancer care, resulting in a steady decline in cancer death rates over the last 30 years. 2 Information overload, however, is now a reality in clinical practice 3 and is increased by the multiple formats and channels for its communication. 4 In the future of oncology, this will be exacerbated by new technologies, such as next-generation sequencing, where copious amounts of data are produced, much of which has little clinical significance. 5 Well-designed digital solutions offer the opportunity to better capture, organize and present essential information and can enable advanced analytics and workflow optimizations which will be needed to meet future challenges.
Ellis Fischel Cancer Center (EFCC), at the University of Missouri (MU) Health Care, has provided comprehensive cancer care since its founding in 1940. At EFCC, physicians and other healthcare providers deliver the latest treatments and technologies to improve patient outcomes. In recognition of the growing burden of information, MU has accelerated the digitalization of its clinical practices, including implementation and innovations within the electronic medical record (EMR) system. Indeed, MU has been recognized as one of the “most wired” healthcare systems in the nation. 6 However, to truly realize the potential of digital technology, such as clinical decision support (CDS) and advanced analytics, constant innovation is required.
The legacy approach to tumor boards
A key juncture in a cancer patient’s journey is the multidisciplinary tumor board (MTB) meeting, where a team of expert clinicians discuss complex cases to determine the best possible treatment plan for a patient. MTBs are an established part of cancer care,7 –9 and a recent American Society of Clinical Oncology (ASCO) survey reported 96 percent of respondents felt such meetings were beneficial for patients and of a high educational value. 10 However, from a physician perspective, MTBs require significant preparation time from each specialty. A recent study by Stahl 11 demonstrated that nearly 47 percent of health systems reported their oncologists took as long as 2 h to prepare a complex case for review. 11 Other specialties, such as pathology and radiology, may spend even more time preparing diagnostic images, up to 6 h for a single MTB (unpublished data). Each subspecialty often has its own MTB, and up to 60–100 patients can be discussed on a weekly basis.
EFCC conducts MTBs to provide a comprehensive approach to cancer treatment planning. Subspecialty MTBs include hematopathology, breast, ENT, liver/pancreas, OB/GYN, urology, neuro-oncology, gastrointestinal pathology and musculoskeletal and thoracic (lung). Case preparation and coordination is largely undertaken using legacy processes, requiring many manual steps and information from multiple clinical disciplines, healthcare providers and electronic databases, which must be compiled for a cohesive presentation.
The manual process of MTB planning starts with physicians providing patients to create a list for review. Each specialty gathers information from diverse sources, some with poor navigability. Often this is coordinated by a specific specialty such as pathology, but there is little coordination of preparation between MTB members. A clinical leader manages the meeting, and discussion for each patient varies depending on case complexity. Residents (physician trainees) present patients, with attendings (supervising physicians) contributing additional information and querying residents and fellows as a teaching method. Radiologists present images from a picture archiving and communication system (PACS), which can be discussed at length in the surgical specialties. Pathologists provide diagnoses and include histopathology, molecular diagnostics, and biomarkers that impact diagnosis and treatment, showing digital images from tissue specimens or ancillary studies such as flow cytometry. After a consensus plan is agreed, it is documented into the EMR, which may occur sometime after the MTB. To achieve meaningful discussion and conclusions, a significant amount of information is required from a large number of discrete information systems, and after all this usually no aggregated dataset is available.
The birth of a digital tool for tumor boards
As with other forward-thinking academic medical centers, MU senior faculty and industry leaders collaborate to facilitate translation of novel technologies into frontline clinical care. A recent focus has been assimilation and effective application of complex molecular and biomarker information in cancer care, a specific and growing challenge for the MTB. 12 In recognition that no obvious “off-the-shelf” solution was available, Roche and MU in 2015 agreed to a collaborative project to co-create a digital solution to address this challenge. As MTBs are an integral part of patient cancer care, we saw a need to collaborate to develop software that would utilize informatics approaches that could translate into clinically viable improvement of tumor boards.
Co-creation is a concept popularized by Prahalad and Ramaswamy 13 in their Harvard Business Review paper “Co-Opting Customer Experience.” They defined co-creation as, “The joint creation of value by the company and the customer; allowing the customer to co-construct the service experience to suit their context” 14 (p. 8). This approach has been successfully used in several healthcare projects to date.15,16
The vision for this collaboration included digitalization of workflow and standardization of case reviews in a single dashboard accessible by all MTB members; enablement of easy access to reports, images and patient profiles; and ultimately integration with MU’s EMR. The hematopathology MTB was selected as co-development partners under the leadership of the author and served as the pilot for new prototypes and innovations in software. A team to include champions from pathology, radiology and the clinical specialties was formed to supervise the project. The author liaised with a corporate development team and enlisted the aid of numerous other clinical staff, to include residents, tumor registry staff and nursing, and created a position of a tumor board nurse navigator. This team provided input on development, usability, interface and implementation. In addition, an institutional review board (IRB)-approved clinical study was formulated to evaluate the use of software and compare it to legacy methods. These data were collected for 2 years and are currently in preparation for submission as a separate manuscript.
Value-stream mapping of the tumor board process
At the outset, Lean Six Sigma methods were used to map the activities needed to deliver a MTB. This exercise was repeated across other MTBs to gain a broader understanding of constant and variable factors related to preparation and conduction. An interesting finding was a general lack of process and/or quality metrics used to measure MTB performance and impact on patient outcome. The results of the value-mapping exercise were a high-level set of product requirements and the process and/or quality challenges they addressed (Table 1).
Digital tumor board product requirements derived from tumor board meeting process mapping of MTB process and quality challenges.
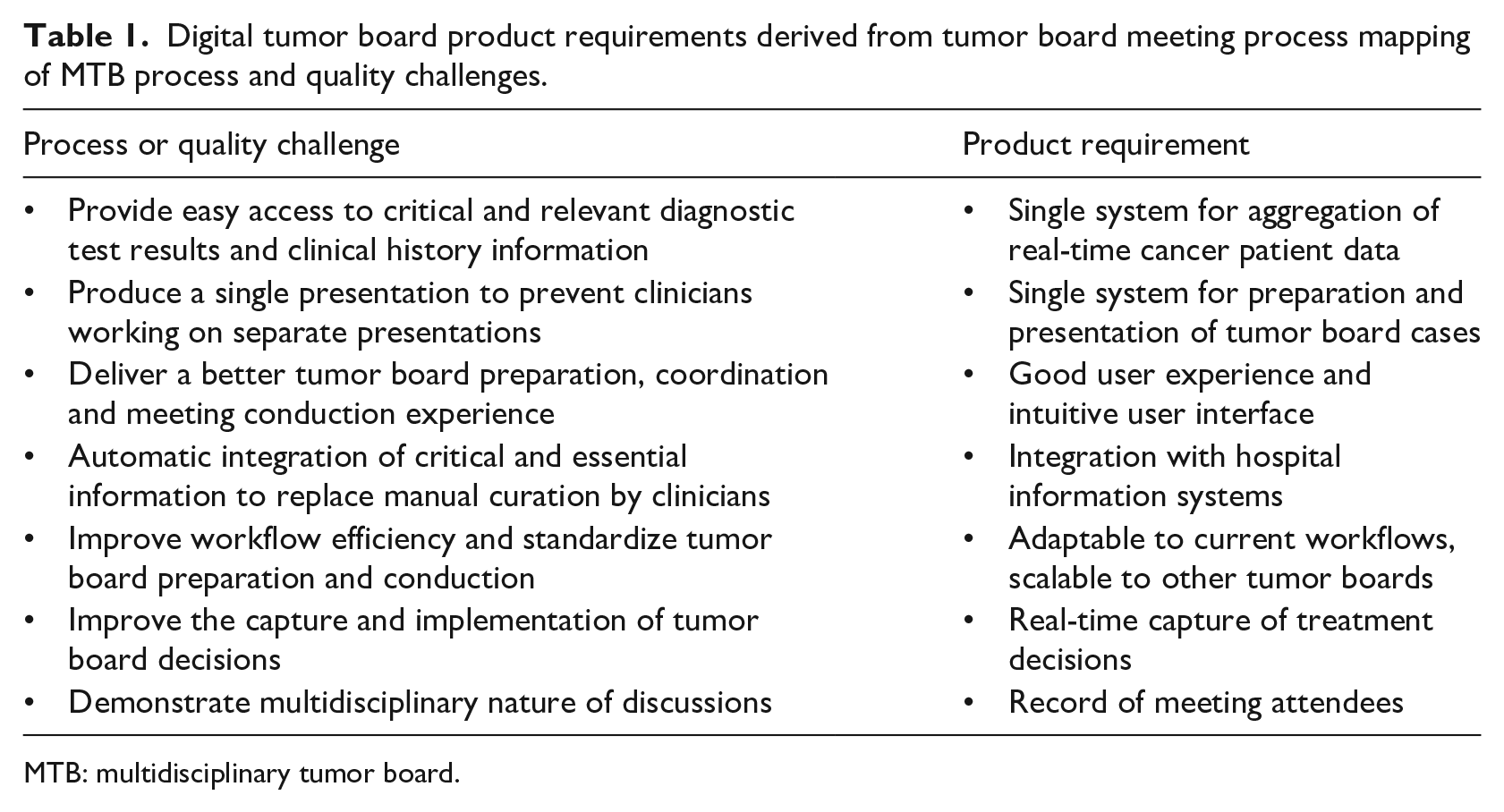
MTB: multidisciplinary tumor board.
These observations were consistent with literature, which has shown that clinical decision-making at MTBs is dependent on the quality and availability of critical information. Common factors that negatively impact meetings include unreliable teleconferencing software or connectivity; lack of pertinent test results; insufficient clinical information; non-attendance of key care team members; and poor meeting leadership.17,18 Findings that hinder the implementation of decisions include non-consideration of patient comorbidities and preferences, and/or disease progression at the time of treatment. 17 The intent was to develop a software solution that could address these negative issues and provide a cloud-based solution that placed all pertinent tests results, clinical information and disease status in a single environment that was easily accessible. The intent was this could be done in “real time” and available to clinicians prior to the meeting virtually and could provide a repository of data for access after MTB for reference.
Co-creation of a digital tumor board solution
Early concepts and the experience from another collaboration partner at Hospital del Mar in Barcelona, Spain, were presented to the team mid-2016. 19 In 2017, early cloud-based prototypes were presented for feedback. A key component for continued development was the evaluation and subsequent approval of the product concept by the compliance office and that patient data security was integral to the design principles.
The Roche team conducted scheduled feedback sessions for prototyping. Users walked through task-based interactions and described their experiences. The teams documented core function and enhancement ideas based on interactive feedback. Initially, premeditated questions were used to assess solutions for clinical content, workflow usability, interface design, feature positioning and placement. As questions were addressed, other ideas and suggestions surfaced and were considered new features for prioritization and assigned a rank score. Software evolved quickly because of the agile collaborative approach adopted by both the MU clinicians and Roche development teams.
Co-implementation of a digital tool
With use of NAVIFY® Tumor Board (a cloud-based software solution for tumor boards) clinical case information, key reports and illustrative images were available to all MTB members prior to convening. The team contributed to a single digital cloud-based platform presentation which is accessible anywhere. There is a single shared record for each patient rather than separate presentations by specialty which lack continuity and cohesion. After aggregation of all necessary information into NAVIFY, the presentation is prepared with a single click in software to generate the digitally pinned presentation. This greatly simplifies MTB preparation for subspecialties routinely involved in preparation. Figure 1.
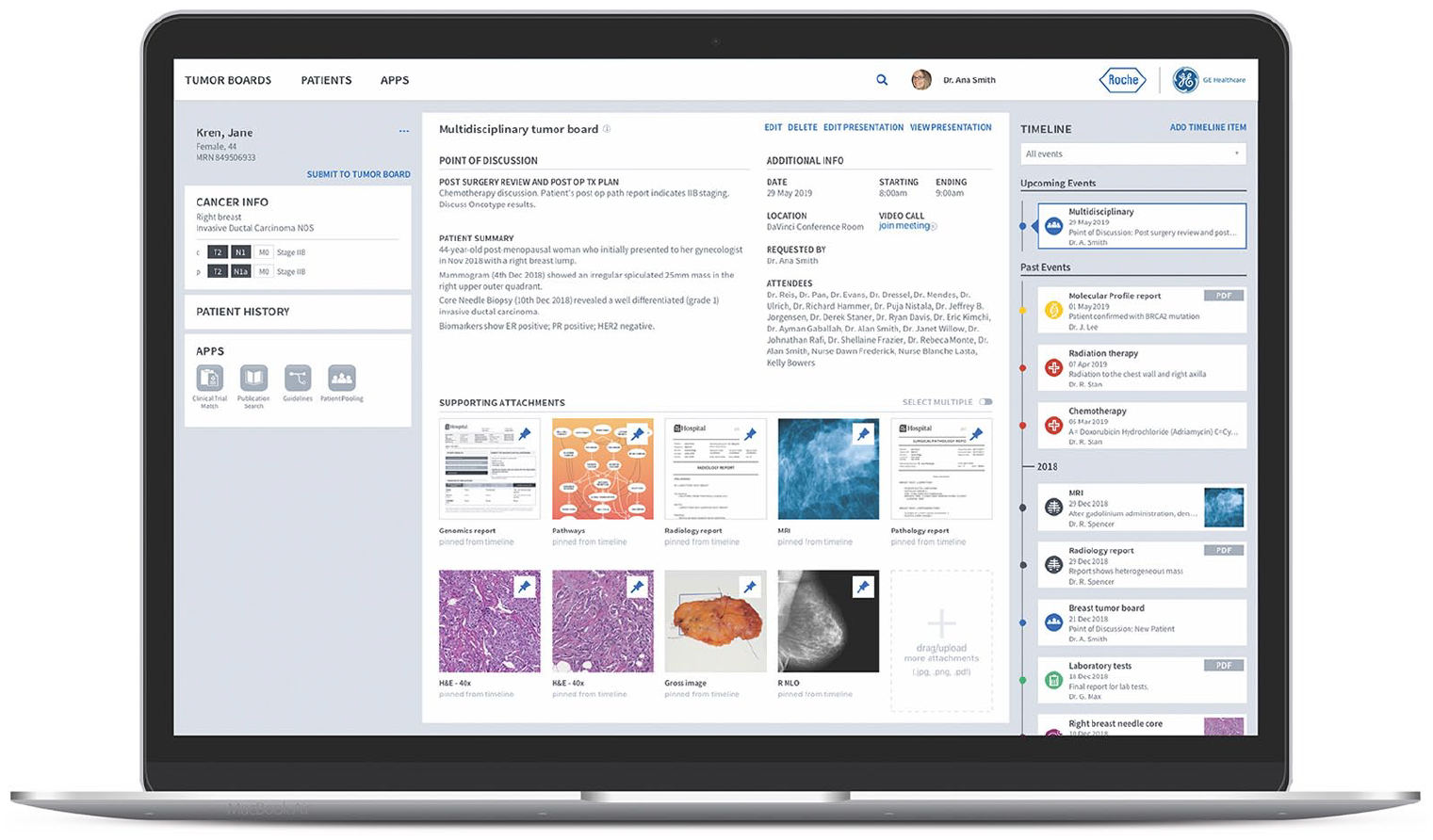
NAVIFY® Tumor Board screenshot. A comprehensive patient view for tumor boards.
The NAVIFY Tumor Board was introduced into hematopathology MTB in April 2018. To streamline the rollout, and alleviate user reluctance to the new technology, it was decided to begin with a manual upload version supported by a nurse navigator to coordinate meetings and onsite user training. As each new MTB was added, the nurse navigator repeated the support and training to the next group. By acting as a bridge between new users, experienced users and the Roche team, it quickly became apparent that the dedicated MTB nurse navigator was a key enabler for successful NAVIFY Tumor Board rollout. In addition, they were involved in tracking key metrics of clinical quality and effectiveness for the ongoing MTB clinical study (manuscript in preparation) and were extensively involved with software enhancements and EMR integration testing.
To better understand the impact of NAVIFY Tumor Board solution, a comprehensive IRB-approved clinical study was initiated at project inception to capture preparation time prior to and after the introduction of the tool. This study is scheduled to be completed in 2019, but the initial results show interesting findings. Despite a reduction in cases and increased preparation time in the immediate weeks after the software introduction, after just 4 weeks, MTBs were able to return to normal numbers. These findings support that the learning curve of a new digital process is short with appropriate training and support. Further results echo the findings of Krupinski et al., 19 who demonstrated after the introduction of NAVIFY Tumor Board in Hospital del Mar, oncologist, radiologist and surgical meeting preparation time decreased and showed less time variation. 19
A culture of continuous improvement
The co-creation/co-implementation model was not sequential but simultaneous on several levels, including clinical use, technical integration and additional software development. Iterative user testing refined software through an agile development process. Most recently, successful integration with the MU electronic record was achieved. At present, an order for MTB is entered by a provider into the EMR which automatically populates the NAVIFY platform with patient demographics and cancer tagged pathology reports. Expansion of integrated data is ongoing. As MTB preparation data are continuously collected, it will be possible to measure the impact of integration and the version changes resulting from continuous feedback.
This work has also led to a “process optimization culture” within MTB teams, whereby members actively examine elements of the value chain to seek improvements and streamline both processes and software enhancements. This is in agreement with findings from Janssen et al. 20 that real-time data and image can improve patient coordination but may not be optimally utilized by teams. For example, during pathology preparation in the legacy method, it was customary for residents to prepare photomicrographs and then store them in a dedicated departmental drive. This involved creating presentation slides and required faculty meeting with residents to review slides and sometimes retake images. By simply capturing the image at faculty sign-out, a 43-percent reduction in preparation time for the hematopathology MTB was achieved and obviated the need for additional review (Hammer, unpublished results).
Improvements with a NAVIFY Tumor Board
Already NAVIFY Tumor Board has changed and improved the preparation and conduction of MBTs at MU, examples include the following:
Clinicians can submit a patient for discussion many weeks in advance so the nurse navigator can begin preparation immediately.
When diagnostic tests take longer to receive, members can easily reschedule a patient to a later date to ensure discussion with complete information without risk of missing cases.
Attendings can easily review patient case information prior to the final agenda and proof resident presentations from remote locations.
MTB members can review patients and current therapies before conference, including relevant literature and clinical trials using the latest CDS applications, PubMed search and Clinical Trial Match, which are integrated with software. These apps provide an integrated search of literature and applicable clinical trial specific to a patient’s tumor type.
Access to relevant patient data during the meeting allows clinicians to make “real-time” presentations including tumor markers, prior cancers and recent surgeries in a timeline format. This improves participant experience and allows flexibility in accessing information.
Clinicians can edit the presentation anytime, supplementing the latest patient data or address questions based on earlier feedback from colleagues.
Residents can work asynchronously and simultaneously on a single tumor board presentation, eliminating the need to share presentation files.
All past tumor board presentations and documents reside in one place, in timeline format for easy reference, review and use in teaching. The presentation file is locked as a permanent record of the MTB.
The challenge of being the innovator
The digitalization of MTBs at MU has not been without challenges. For the MU team, the partnership was in addition to normal clinical duties, teaching responsibilities and administrative tasks. This sometimes led to significant strain on the MU team in meeting goals and deadlines. The support of the hospital and university leadership provided direction, resources and space for experimentation that was essential for success. In addition, the collaborative and transparent approach by the Roche team was an essential to maintain the trust of the clinical team and to feel their input was valued and included in product improvements.
As the industry team grew, and the project assumed higher priority for Roche, further unexpected challenges emerged. It was important to maintain continuity amid staff changes to ensure appropriate follow-up from feedback sessions. This was achieved by designating a dedicated product manager for the partnership, and the strong transparent relationship that formed with the MU team. In this way, issues were resolved quickly and momentum maintained. A true team-based collaborative relationship exists with quick access to team members as needed.
During the early stages of development, there was fear of failure, in particular, of implementing software that might be perceived by clinical colleagues as inferior to the standard approach, and therefore rejected. As development continued and software surpassed expectations, the challenge became one of change management, as with any new technology. An important validation for the project was the institution’s leadership. Most importantly, however, was the commitment and support of the hematopathology tumor board team members, who acted as additional champions for the work. Indeed, one senior oncologist pushed the product capabilities even further by mandating his residents use the NAVIFY Tumor Board as a “patient cockpit” to coordinate patient care pathways beyond therapy decisions.
Co-creation between healthcare providers and industry partners should become a standard for digital tool development. Despite being a challenging experience and significant additional commitment, the chance to transform the process of clinical decision-making as a digital innovator/early adopter is extremely rewarding. It is a roller coaster of highs and lows and markedly different from routine clinical practice. The collaborative process was a learning process for both the medical institution and industry partner. Insights into workflow and implementation strategies were the reward for the struggles faced. It is exciting to see how the current product will develop further to realize other significant opportunities. We currently have incorporated software into six tumor boards, created a new tumor board using NAVIFY and are in the process of EMR integration to automate the data flow into software in a Health Insurance Portability and Accountability Act (HIPAA) compliant environment. Current integration includes patient demographics, pathology reports, radiology reports and radiologic imaging. Additional integration features to include digital images from pathology and molecular data are planned on the future roadmap.
The opportunity of a cloud-based tumor board solution
A well-designed digital tool for the preparation and conduction of MTB meetings presents opportunities for care providers to enhance the quality of care delivered to patients and to optimize care coordination by and between hospitals. These tools are at the forefront of current healthcare development. Cloud-based solutions, however, can take these benefits much further. By complementing hub-and-spoke care delivery models, information exchange between sites is simplified and could ultimately facilitate virtual asynchronous MTBs. MU intends to experiment with this functionality in 2020 with remote partner hospitals in a virtual environment. The issues faced by us in this endeavor have been discussed by Munro and Swartzman. 8
Another benefit for cloud solutions is the ability to connect with and provide greater support to patients and their caregivers. This could include validating a patient’s premorbid status and care preferences prior to MTBs, symptom monitoring during treatment and/or providing educational support throughout their cancer journey. Moreover, a future opportunity could be to bring the patient and their caregivers into a virtual MTB so they are more engaged in the decision-making process, an exciting but daunting proposition for many clinicians. An important function may be to provide the patient access to their files as a tool to manage their own care and ease availability for second opinions or other treatment sites.
A growing concern for healthcare payors and providers is the spiraling cost of care. Data collection and greater connection to patients will facilitate the growing movement toward value-based reimbursement. As Porter 21 stated, “the only way to truly contain costs in healthcare is to improve outcomes: in a value-based system, achieving and maintaining good health is inherently less costly than dealing with poor health.” 21 Notwithstanding, it is very difficult to mandate such models unless data collection can truly capture patient outcomes. Well-designed digital tools allow capture of both clinical and metadata that can provide analysis of quality metrics to optimize care and monitor performance.
User-centered design methodology 22 fosters development of tools that mesh with clinical work rather than overburden already busy care teams. The ability to integrate CDS applications into the workflow solution will allow teams to implement tools that enhance clinical judgment. Indeed, in September 2018, tools for automatic clinical trial matching and smart literature searching were introduced into the NAVIFY workflow. In the future, descriptive, diagnostic, predictive and ultimately prescriptive analytic tools will become available to clinicians.
To meet the challenges of increasing volume and complexity of medical knowledge, care providers must embrace digital transformation and advanced analytics. Instead of accepting current digital tools with poor user experience and limited functionality, we must demand that industry builds solutions that place users at the center of the design process and seek to capture the opportunities of the latest technologies and medical science. This effort toward collaboration was highlighted in the 2014 ACMI debate where it was resolved that the lack of interaction between health IT vendors and academic units is stifling innovation. 23 The current collaboration between Roche and MU highlights the productive nature of such co-development that can result in impactful technology in healthcare.
Footnotes
Acknowledgements
The example digital tool used to frame this article has been developed as part of an ongoing collaboration between the University of Missouri Medical School and Roche.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Hammer is a member of the Roche DSI Clinical Advisory Board and has received reimbursement for travel expenses and honorariums. Dr Prime is employed by Roche.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
IRB approval
This project was approved by the University of Missouri IRB under all appropriate guidelines.
