Abstract
Ownership of personal digital assistants (PDAs) and smartphones by health professionals is increasingly common. Providing the best available evidence at the point of care is important for time-poor clinical staff and may lead to benefits in the processes and outcomes of clinical care. This review was performed to investigate the usefulness of PDAs in the clinical setting. MEDLINE, Embase, CINAHL and the Cochrane Central Register of Controlled Trials were searched from 2000 to March 2010. Randomised controlled trials that evaluated the effects on the processes or outcomes of clinical care of using PDAs compared with not using a PDA were included. Two reviewers independently reviewed citations and abstracts, assessed full text articles and abstracted data from the studies. Seven trials met the review inclusion criteria, of which only three were of satisfactory quality. Studies investigated the use of PDAs either in recording patient information or in decision support for diagnoses or treatment. An increase in data collection quality was reported, and the appropriateness of diagnosis and treatment decisions was improved. PDAs appear to have potential in improving some processes and outcomes of clinical care, but the evidence is limited and reliable conclusions on whether they help, in what circumstances and how they should be used are not possible. Further research is required to assess their value and ensure full benefits from their widespread use, but the pace of technological development creates problems for the timely evaluation of these devices and their applications.
Keywords
Introduction
It is increasingly common for health professionals to have personal digital assistants (PDAs) or smartphones. 1 Access to the best available evidence at the point of care is important for time-poor clinical staff. PDAs may be able to provide health professionals with convenient rapid access to reports of research evidence to support their decision-making. The number of articles published regarding PDAs is rising. 2
PDAs are used by many clinicians for managing their diaries electronically, but they can also be used as a reference tool to obtain evidence and guidance on clinical conditions, drug calculations and other medical information, and now also to hold clinical decision support systems (CDSS). 3 PDAs can enable the transfer of CDSS from a desktop personal computer (PC) to the clinical setting. Databases can be loaded onto the devices, either for searching for information or for inputting information that can later by synchronised to an external source, so acting as a data collection tool. Many of these devices now have wireless Internet connectivity and can be used to retrieve information at the patient’s bedside or in an outpatient clinic. However, although health professionals may regard PDAs as helpful, the full benefits of their use may not be achieved without evaluation of their effectiveness.
We undertook a study to identify and assess available evidence on whether PDA use in the clinical setting improves professional practice. Specifically, we sought to identify and assess available evidence on whether PDA use in the clinical setting, compared with usual practice, improves professional practice in terms of processes and outcomes of care.
Methods
Search strategy
To identify potentially relevant journal articles, MEDLINE, Embase, Cochrane Central Register of Controlled Trials (CENTRAL) and CINAHL databases were searched from 2000 onwards. The search performed used the following search terms: palmtop*; PDA*; personal digital assistant*; pocket PC; tablet PC; mobile technolog*; computer*; hand held; decision making; computer assisted decision making; artificial intelligence; computer assisted therapy; algorithm* and computer assisted support system*. These search terms were selected from previous studies. 4 We checked articles cited by the included studies. The main searches were undertaken in March 2010.
To retrieve potentially relevant unpublished studies, 5 a search of the ProQuest Dissertation and Theses Index was undertaken in January 2012 and a search of current controlled trials was undertaken in December 2011 to look for studies that were ongoing, or completed and unpublished.
Inclusion/exclusion criteria
Studies prior to 2000 were excluded on the grounds that PDA technology was in its infancy prior to this date. Articles describing randomised controlled trials or systematic reviews of randomised controlled trials were included. We included studies (i) that used a handheld computer or smartphone; (ii) that had a control group that used no handheld computer; (iii) in which the users of the device were healthcare professionals; and (iv) in which the outcomes were either processes of care or clinical outcomes. We restricted the included studies to randomised controlled trials in order to minimise the risk of bias.
Abstracts were reviewed independently for relevance by two reviewers. Disagreements were resolved by discussion of the full text of disputed articles.
Data extraction
The articles selected underwent dual independent extraction of data. A standard data extraction form, adapted from a data extraction form used in a review of implementation studies, was used to collect data from each article. 6 The form was piloted and amendments made prior to use for the full extraction of data. The data extraction form collected information on the study participants, both professionals and patients, intervention, control, setting and relevant outcomes. The risk of bias was also assessed using seven criteria based on those used by the Effective Practice and Organisation of Care Group of the Cochrane Collaboration: 7 the reporting of baseline measurements, method and concealment of allocation, power calculation, blinding of participants and/or investigators, follow up of 80% of participants or more, and whether outcome measures were objectively measured.
Data synthesis
The data were not synthesised in a meta-analysis because of the small number and heterogeneity of the included studies. We presented the studies in tables accompanied by a narrative review. This review was reported in compliance with PRISMA guidelines. 8
Results
The search of MEDLINE found 796 results, which were reduced to 37 potentially relevant studies on assessment of the abstracts. A search of Embase found 294 results which were reduced to 17 through assessment of the abstracts. A further 13 articles were found through a search of Cochrane CENTRAL. The same search was also repeated on the CINAHL database; no new articles were retrieved. Further searches of sources of unpublished studies yielded no further suitable studies for this review.
One reviewer undertook the first assessment of abstracts; many studies were rejected at this stage as the subject heading ‘Point of care systems’ retrieved articles on mobile computed tomography scanning and other bedside technologies.
Once duplicates were removed the retained 63 articles were independently assessed for relevance by PD (all articles) and either JCS or RB. Twenty-six articles were obtained in full text and, of these, 19 were excluded based on the inclusion/exclusion criteria. All of these were reported using the English language. One of the 26 articles fell outside of the date range selected for this review, and was not a randomised controlled trial, so was excluded. 9 For excluded studies, see Table 1.
Studies that were excluded from the systematic review
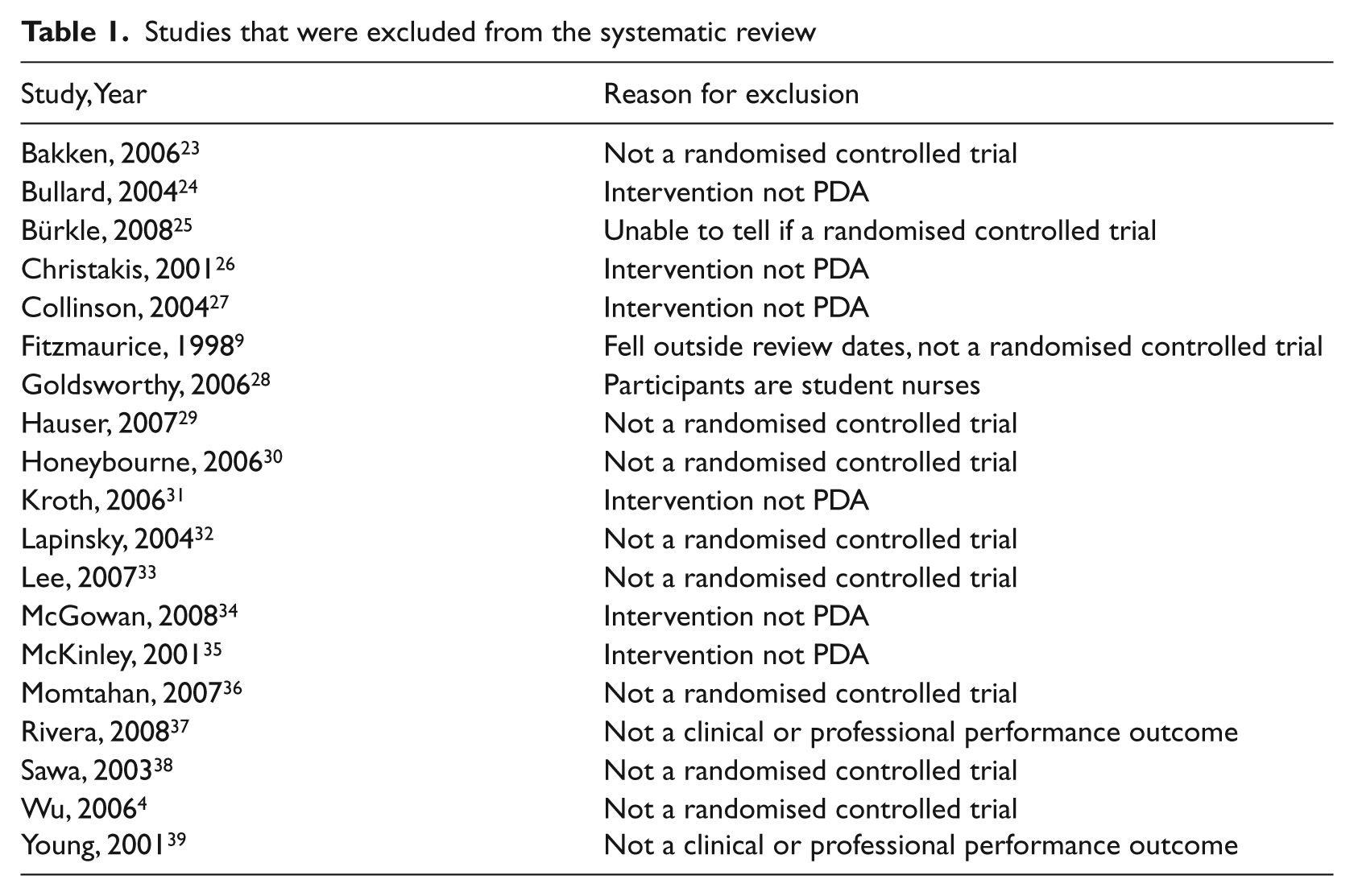
A total of seven articles were assessed as suitable for inclusion in the review (see flowchart in Figure 1). PD reviewed all studies using the data extraction screening form, and RB and JCS each reviewed half of the 26 studies. Studies were assessed on the screening form, which utilised the inclusion and exclusion criteria listed above.
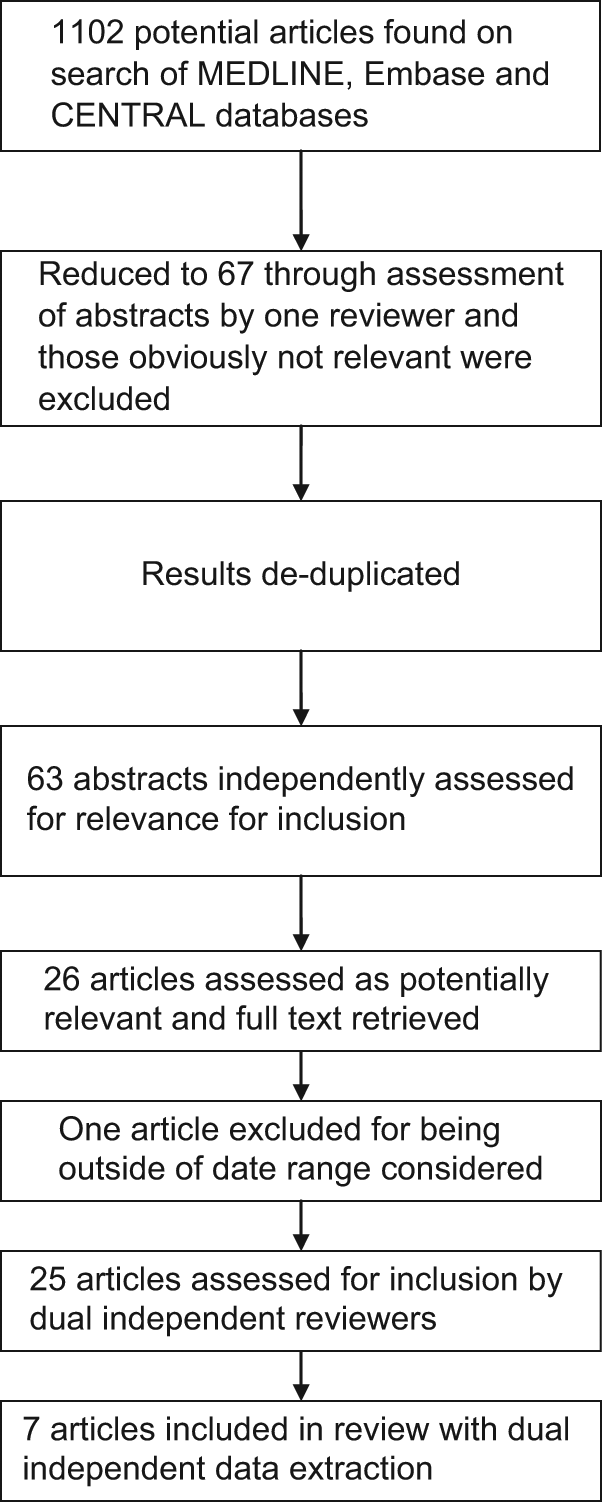
Flowchart
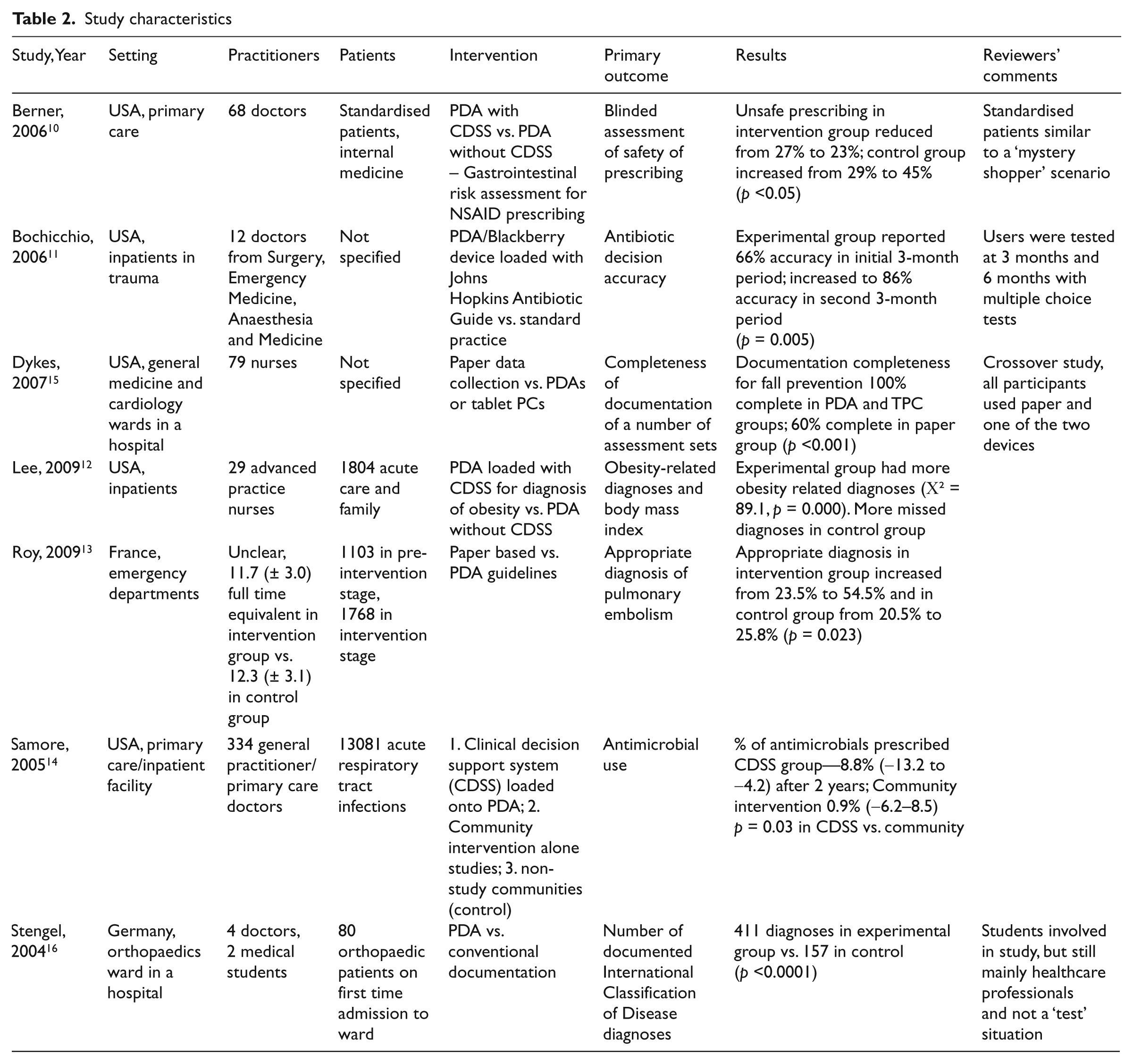
The seven studies included 551 health care professionals, 2 medical students and a minimum of 16,068 patients (not all studies reported the number of patients involved). Study characteristics are described in Table 2. Five of the studies involved PDAs loaded with CDSS or guidelines as reference tools,10–14 and of these four were undertaken in the USA and one in France. The remaining two involved the use of PDAs as data collection tools, with one undertaken in the USA and one in Germany.15, 16
Study characteristics
The risk of bias of the included studies is shown in Table 3. The studies were mixed. Five of the studies10–12, 14, 15 did not adequately describe the method or concealment of allocation to the trial groups, although two did.13, 16 All studies reported >80% follow-up of participants. Only one study 13 met all of the risk of bias criteria (Table 3).
Risk of bias
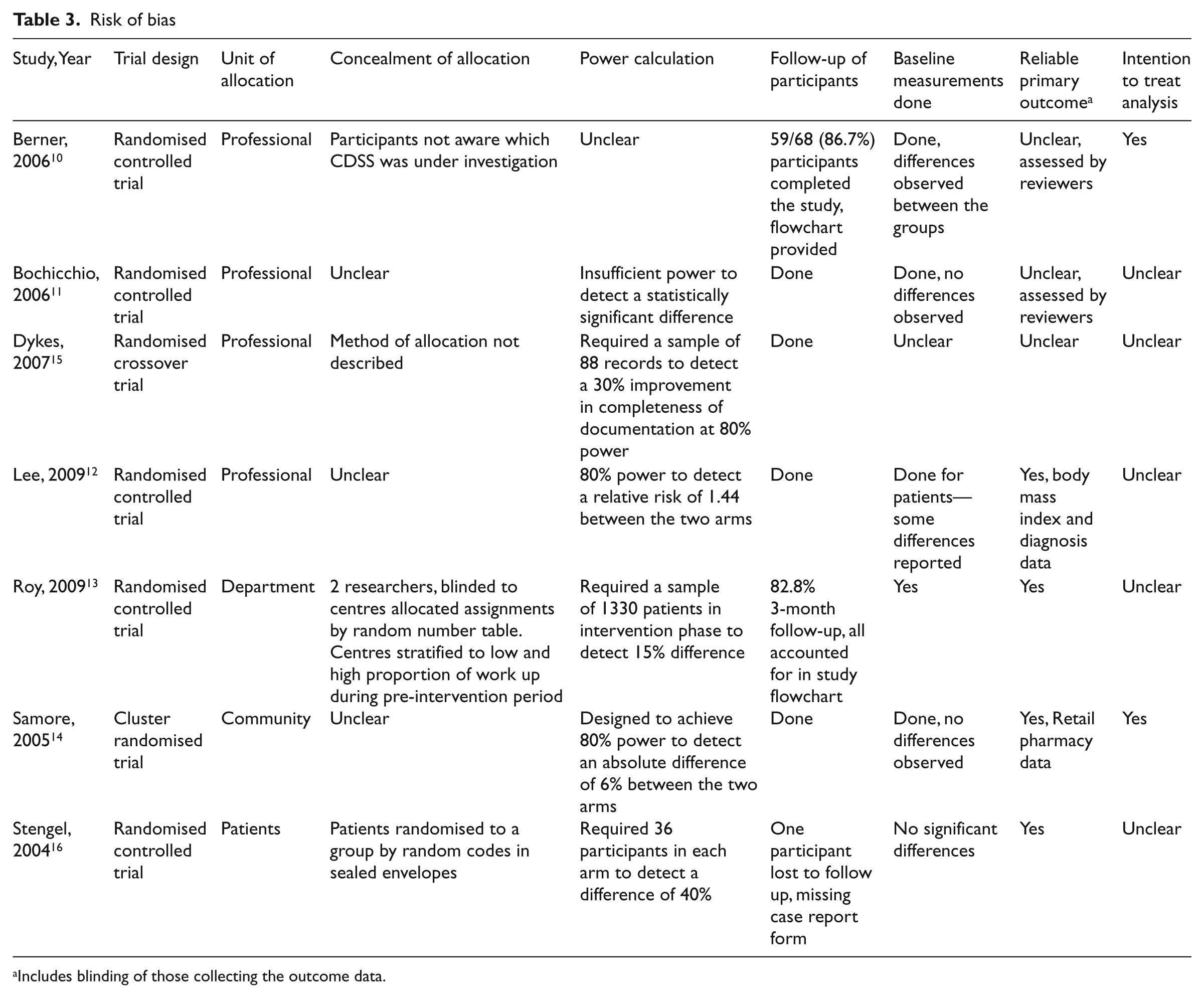
Includes blinding of those collecting the outcome data.
A statistically significant positive benefit for the outcomes of clinical care of the use of PDAs as clinical decision support tools was shown in three studies.10, 13, 14 The results from the fourth study on PDAs for decision support were unclear with regard to statistical significance. 11 Both studies that investigated the use of PDAs for data collection found statistically significant improvement in the intervention arm.15, 16
Study themes
There were three distinct themes to the articles selected for inclusion in this review: those investigating diagnosis using a CDSS as a primary outcome measure;12, 13 those investigating the appropriateness of the treatment using CDSS;10, 11, 14 and those investigating PDAs for the accuracy of record keeping.15, 16
Both studies measuring diagnostic accuracy using CDSS on PDAs versus other means found that the PDA group identified more diagnoses than the control.12, 13 Roy et al. found that the intervention increased diagnostic accuracy, which was statistically significant. Lee et al. also found statistically significant results in favour of the experimental group. However, Roy et al. investigated PDAs versus paper guidelines and Lee et al. investigated PDAs with a CDSS versus PDAs without.
All three studies investigating the appropriateness of treatment decisions using a PDA for decision support found the use of a CDSS loaded onto a PDA improved treatment decisions. Bochicchio et al. used examinations to test participants’ understanding of prescribing accuracy and found the use of the PDA improved knowledge and understanding at a statistically significant level (p = 0.005) [11]. Berner et al investigated gastrointestinal risk assessment for non-steroidal anti-inflammatory drug (NSAID) prescribing and found that unsafe prescribing was significantly (p = 0.001) reduced in the PDA experimental group compared with unsafe practice in the control group. 10 Samore et al. found a significant difference in favour of the use of a PDA with a CDSS versus a community intervention for antimicrobial prescribing levels (p = 0.03).
The studies that investigated the use of PDAs for keeping and amending patient records and documentation found that more patient information was documented in the PDA use groups than in the control groups. Participants reported that they found the devices more easy to use than paper records.15, 16 Dykes et al. found a statistically significant difference in completion of patient documentation in the PDA groups versus the paper documentation groups, while Stengel et al. found a statistically significant difference (p = 0.0001) in the number of documented diagnoses using a PDA rather than paper records.
Discussion
The findings from the small number of studies included in this review suggest that PDAs can be helpful in improving the processes and outcomes of care, and can improve the quality of data collection and record-keeping. However, the number of studies and their methodological quality preclude definitive judgement on the effectiveness of use of PDAs in decision support or data collection. Nevertheless, there is growing interest in the potential of PDAs. A search of MEDLINE for articles on PDAs by year of publication, using the same strategy as for this systematic review, showed that in 2000, 879 articles that mentioned PDAs or handheld computers had been published; in 2009 the number had almost doubled to 1644.
None of the included studies reported cost-effectiveness data. Buying the PDA devices, loading them with the appropriate applications, the cost of replacing devices that are lost, training staff in their use, and the cost of software and hardware support, if appropriate, are all factors to be taken into consideration. However, PDA use may also save time and cost, and this should be fully explored in future research in comparison with other means of improving clinical decision-making before PDAs can be recommended for widespread use for decision support and data collection.
Strengths and limitations of this review
There are several limitations to this review. The results are limited by the quality of studies included and meta-analysis was not possible owing to the heterogeneity of the included studies. Only randomised controlled trials were included in the review, and it may be that other study designs, such as controlled before and after studies, and interrupted time series studies, would have contributed useful evidence. We sought to limit the review to studies with the strongest methodological design to reduce the risk of bias as much as possible. It is virtually impossible to blind study subjects to the fact that they are using PDAs, and other study designs may be less successful at limiting this source of bias.
Hand-searching key journals, potentially useful in identifying other articles, was not carried out owing to time constraints. The reference lists of articles found in the searches were checked and this did identify relevant background studies to the review, although no other randomised controlled trials were identified by this means. Consequently, we did not include potentially relevant unpublished studies, such as Joseph 17 on the usability of handheld computing. However, our review was specifically focused on the process and outcomes of care rather than user acceptability.
There is a risk of publication bias 18 in this review, as studies with negative findings may not have been submitted for publication. Furthermore, the studies in the review took place in a limited range of settings; research in more diverse clinical settings is required. By limiting the studies returned in the database searches to those published in the English language only, some research in other clinical settings may have been missed.
User acceptability was not reported in any of the included trials in this review. This does need to be a consideration for the future design of CDSS for use in clinical care. The Technology Acceptance Model (TAM) 19 has been demonstrated to be applicable in studies looking at the user acceptance of PDAs for complex documentation. 20 Acceptance of PDA technologies in healthcare may lead to more rapid knowledge creation, synthesis and transfer, 21 thus improving the cycle of evidence-based practice.
Implications for policy
As technology develops and more clinicians and hospitals adopt the use of handheld computing and resources, more research is likely to be carried out. However, as handheld computer technology advances quickly, there is a risk that evaluation studies will not be completed before the technology concerned become obsolete. Timely evaluation studies are needed before new technologies are adopted, if technologies are to be used as effectively as possible and resources are not diverted to invest in these technologies rather than potentially more effective alternatives.
As clinicians become more accustomed to using handheld devices as a part of their working and personal lives, there will be implications for hospital policies. The use of wireless networks will become more important, and those hospitals without wireless networks will find they need to install the correct infrastructure to allow access to PDA and smartphone devices. Correct infrastructure and system security are important issues to consider in organisational uptake of devices. 22 Our review shows that PDAs do have clinical applications and to optimise performance for those using them, the right infrastructure and software needs to be available. This may mean in-house-designed clinical decision support systems or integration with electronic patient care records, which will need to be carefully planned and rolled out for each patient care setting. The findings of this review justify encouraging hardware and software developers to take the needs of health environments into account when designing new products.
Conclusion
Further research is necessary to ensure that using handheld PDAs or computers has a positive impact on clinical outcomes. Wu and Strauss 4 state that innovations in informatics and technology are being implemented in the clinical setting without rigorous evaluation. However, PDAs under evaluation in studies may have been superseded by new devices by the time the studies are complete. To help in reducing the problem of rapid technology obsolescence, researchers should design studies to evaluate the basic attributes of handheld devices rather than the features of individual devices.
Footnotes
Funding
JC-S and RB are funded by the National Institute for Health Research (NIHR) Collaboration for Leadership in Applied Health Research and Care (CLAHRC) for Leicestershire, Northamptonshire and Rutland (LNR). The views expressed in this article are not necessarily those of the NIHR or the Department of Health.
Conflicts of interest
The authors have no conflicts of interest to declare.
