Abstract
A Computerised Antithrombotic Risk Assessment Tool was developed for assisting the selection of antithrombotic therapy based on the risk versus benefit assessment. In view of the recent availability of the novel oral anticoagulants, this tool has been updated to CARATV2.0. To explore health professionals’ perspectives on the tool, semi-structured interviews were conducted in seven pharmacists, seven specialists, six general practitioners and six nurses, who were involved in management of antithrombotic therapy for atrial fibrillation. Three overarching themes emerged: (1) CARATV2.0 provides comprehensive structured assessment of patients and could assist with the prescription and review of antithrombotic therapy, (2) subjective issues such as health professionals’ and patients’ preferences for a particular antithrombotic therapy may affect the usefulness of CARATV2.0 and (3) CARATV2.0 requires integration into existing systems and processes. The majority of health professionals surveyed would like to use CARATV2.0 in practice, believing it would improve antithrombotic use and might reduce stroke incidence.
Introduction
The decision-making around antithrombotic therapy (e.g. anticoagulant and antiplatelet therapy) in atrial fibrillation (AF) is complex because it involves assessment of risks versus benefits. 1 For many years, warfarin was the only available OACs, but its unpredictable therapeutic effects, various food and drug interactions and the need for regular monitoring have been associated with great difficulties in its use. Such difficulties have led to a potential increase in adverse events and reluctance by clinicians to prescribe the medication.2-4 The recently marketed novel oral anticoagulants (NOACs) – dabigatran, rivaroxaban and apixaban – have substantially expanded the treatment armamentarium and are intended to overcome the limitations of warfarin. However, these new anticoagulants are not without risk because some of their so-called advantages can be regarded as potential disadvantages in specific situations. 5
To optimise the use of antithrombotic therapy in patients with AF, and specifically assist health professionals in selecting appropriate agents, an electronic decision support tool – the original Computerised Antithrombotic Risk Assessment Tool (CARAT) – was developed. 6 Its decision-making algorithm was computerised by first preparing a prototype in Microsoft Excel™ and then formatting it as a web-based interface for online access. 6 The tool generated treatment recommendations (e.g. warfarin vs aspirin therapy) for individual patients based on their risk (bleeding) versus benefit (stroke prevention) estimation, as well as the relevant medication safety considerations (e.g. drug–drug interactions, renal function and medication adherence). The original CARAT has been trialed in real-world hospital patients 7 and general practice patients 8 and evaluated by specialist clinicians for its potential clinical application in a vignette-based study. 6 CARAT has demonstrated its potential utility in practice.6,7 Although other risk assessment tools have been developed to synthesise the assessment of stroke and bleeding risks – for example, the clinical decision aid developed by LaHaye et al. 9 and the decision model developed by Casciano et al. 10 – none consider the broader medication safety issues that particularly affect the selection of therapy in the target at-risk patient population.9,10 Thus, the CARAT provides a novel, more holistic and pragmatic approach to the decision-making around antithrombotic therapy.
Our previous (i.e. original) version of CARAT was designed to address the complexity in decision-making by integrating the relevant assessments around stroke risk, bleeding risk and medication safety for individual patients; 6 at that time, the tool was able to assist in selecting among two main treatment options – warfarin and aspirin. Now that we have moved forward in time, there are additional issues to consider in the decision-making process which need to be factored into the CARAT. First, the expanded range of treatment options incorporating the NOACs (dabigatran, rivaroxaban and apixaban) has increased the number of parameters (e.g. drug–drug interactions, side effects, doses and frequency of administration) that need to be considered, further complicating decision-making. 11 Second, the range of risk assessment tools for stroke risk (e.g. CHADS2 and CHA2DS2VASc 12 ) and bleeding risk (HAS-BLED 13 and HEMORR2HAGES 14 ) has evolved. Third, the evidence-base around the effectiveness and safety of available treatment options has grown, for example, aspirin is no longer recommended for stroke prevention in AF. 15 Fourth, a broader range of health professionals is now involved in therapeutic recommendations and decision-making around antithrombotics therapy, whereby hospital specialists and general practitioners (GPs) are able to draw upon the services of nurse practitioners (NPs) 16 and consultant pharmacists (e.g. as part of medicines review process 17 ). Collectively, these issues have warranted a revision of the original CARAT, into its second version CARATV2.0, which considers the latest clinical evidence (e.g. guidelines15,18,19 and practice reviews5,20,21) and available treatments (warfarin and NOACs).
As an initial evaluation of this revised (prototype) CARATV2.0 tool, the aim of this study was to obtain feedback from a wide range of health professionals who are involved in the decision-making around antithrombotics in AF (specialist clinicians, GPs, nurses and pharmacists), to help inform the future implementation of this tool in practice. Specific topics explored were the (1) strengths and weaknesses of this tool; (2) appropriateness and relevance of the content of this tool; (3) usefulness of this tool for selecting appropriate antithrombotics, especially between warfarin and NOACs; (4) feasibility of using this tool in clinical practice; and (5) suggestions for further improvement.
Method
Design and setting
This qualitative study was based on face-to-face interviews. From August to October 2014, health professionals (subgroups: specialist clinicians, GPs, nurses and pharmacists) practising in the Sydney metropolitan area were involved in this study (Figure 1).
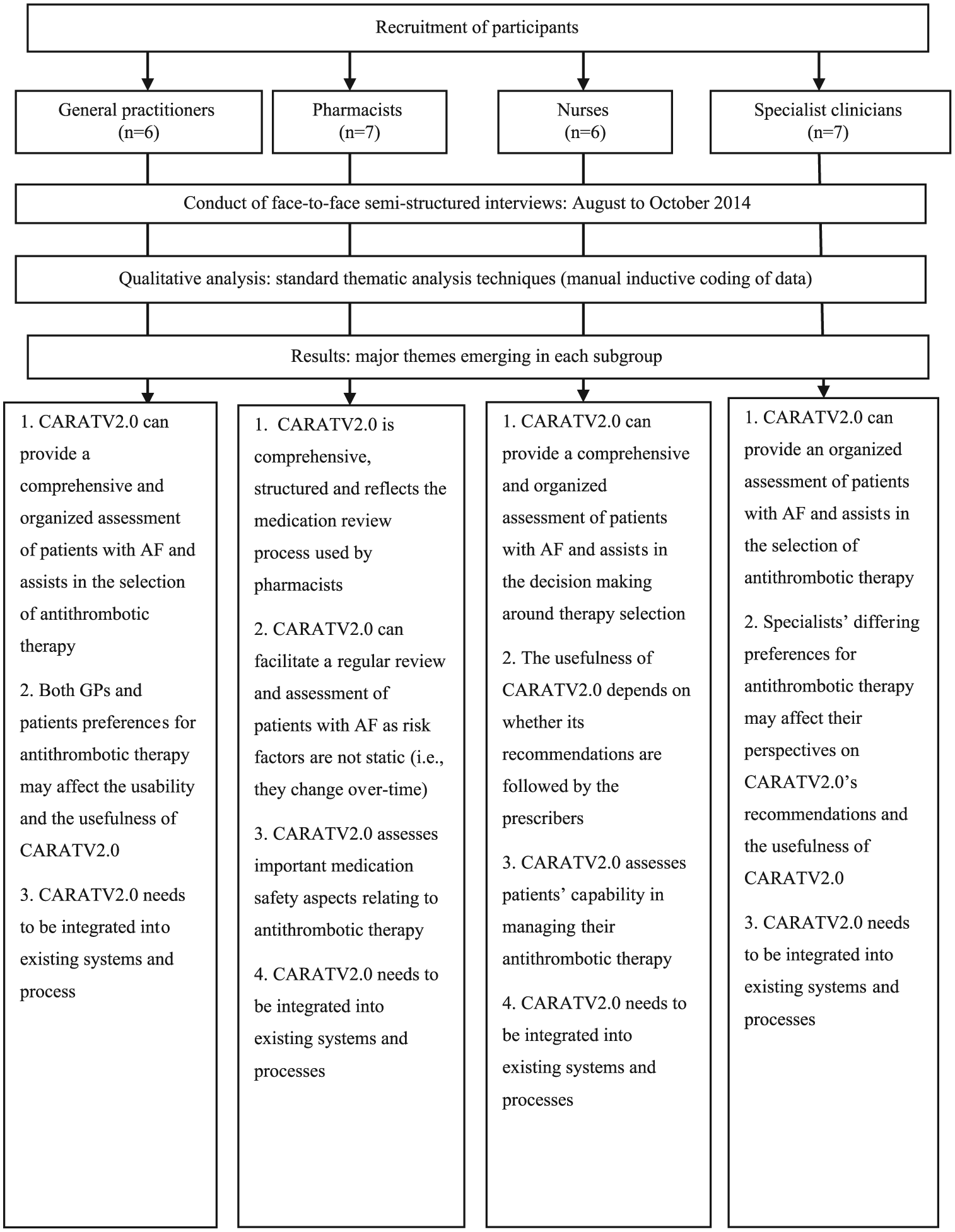
Key stages in eliciting feedback from health professionals about the decision support tool.
Participant recruitment
Purposive sampling was used to identify and recruit health professionals with experience in prescribing antithrombotics and managing antithrombotic therapy for patients with AF. 11 Specialist clinicians, hospital-based pharmacists and nurses were recruited via an invitational flyer emailed or faxed to the network of hospitals affiliated with the university. Community-based pharmacists accredited for Home Medicines Review were recruited through an emailed flyer (using contact details from the Australian Association of Consultant Pharmacy). Flyers were also emailed to community-based nurses in community health services affiliated with the university network of hospitals. By visiting family practices and medical centres in the Sydney metropolitan area, the researcher also distributed invitational flyers to GPs. Emails and faxes were also sent to GPs listed on the Internet. Eligible health professionals who agreed to participate provided written consent.
An estimated 24–40 participants (6–10 participants per group) were needed to achieve theme saturation within each subgroup of health professionals (specialist clinicians, GPs, nurses and pharmacists). 22 As this tool was developed to support a broad range of health professionals who are involved in the day-to-day management of older persons taking antithrombotics, in particular those who had previously expressed a need for assistance in decision-making,3,11 the study largely focused on canvassing feedback from GPs, nurses and pharmacists, in preference to experienced cardiologists. For this reason, relatively few cardiologists were recruited to this study.
Data collection
Semi-structured interviews (20–30 min each) were conducted by the researcher at a location convenient to each participant. At the beginning of each interview, demographic data for each participant were collected using a predesigned questionnaire. Then, the researcher presented CARATV2.0 (on the researcher’s laptop) to the participant. After this familiarisation with the tool, the participant was given the opportunity to explore CARATV2.0. Finally, the researcher used a set of open-ended questions to explore the participant’s feedback on the content of the tool and the feasibility of using CARATV2.0 in clinical practice. All questions were pretested in mock individual interviews with nonparticipants.
Prototype of CARATV2.0
The underpinning algorithm of this revised tool has been developed as a Microsoft Excel-based prototype for pretesting, with the intention of subsequently formatting the tool into an online (web-based) platform or mobile application that will enable the integration of this tool into prescribing software and/or electronic health data management systems. The prototype comprises four distinct sections: (1) stroke risk assessment, that is, CHADS2 23 and CHA2DS2VASc; 12 (2) bleeding risk assessment, that is, HAS-BLED 13 and HEMORR2HAGES; 14 (3) medication safety issues, for example, mini–mental state examination 24 for cognition, estimated glomerular filtration rate (eGFR) (MDRD and Cockcroft and Gault equation 25 ) for renal function and Child-Pugh score 26 for liver function; and (4) therapy recommendations and advice. The application of the tool requires the user to input relevant data into the cells, which auto-populates the formulae underpinning the decision-making algorithm, and which, in turn, generates a treatment recommendation. A patient is considered to be eligible for OACs whenever the risk of stroke is equal to or more than the risk of bleeding, otherwise the patient is deemed to be unsuitable for OACs. CARATV2.0 also provides initial advice around any identified medication safety issues which need to be addressed by the clinician. This study explores feedback on the data inputs and usability of CARATV2.0.
Data analysis
The interviews were digitally recorded (audio) and transcribed verbatim by the researcher. The accuracy of the transcripts was confirmed by listening to the digital records and reviewing the transcripts. The transcripts were analysed for themes, using standard thematic analysis techniques (manual inductive coding). 27 The two authors independently reviewed the transcripts and identified themes before reaching a consensus through discussion. The accuracy and reliability of the analysis was confirmed by inter-researcher validation (with three other independent researchers).
Results
Overall, 26 participants comprising 7 specialist clinicians, 6 GPs, 7 pharmacists and 6 nurses were interviewed (Table 1). Similar themes were identified among the four subgroups, with three overarching themes emerging (Tables 2 to 4 and Figure 1).
Participant characteristics.
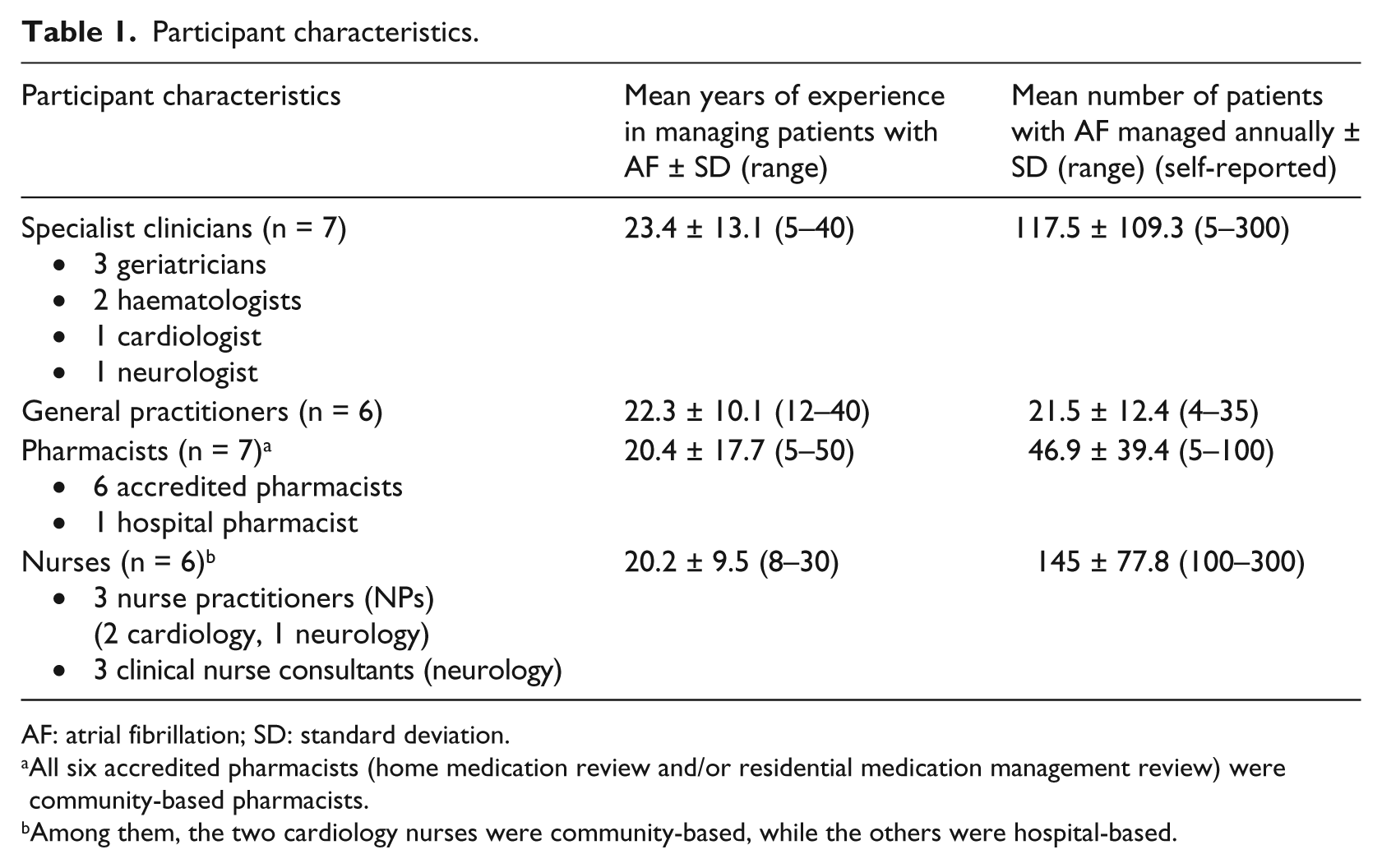
AF: atrial fibrillation; SD: standard deviation.
All six accredited pharmacists (home medication review and/or residential medication management review) were community-based pharmacists.
Among them, the two cardiology nurses were community-based, while the others were hospital-based.
Theme 1: need for comprehensive structured assessment of patients to assist with the prescription and review of antithrombotic therapy.
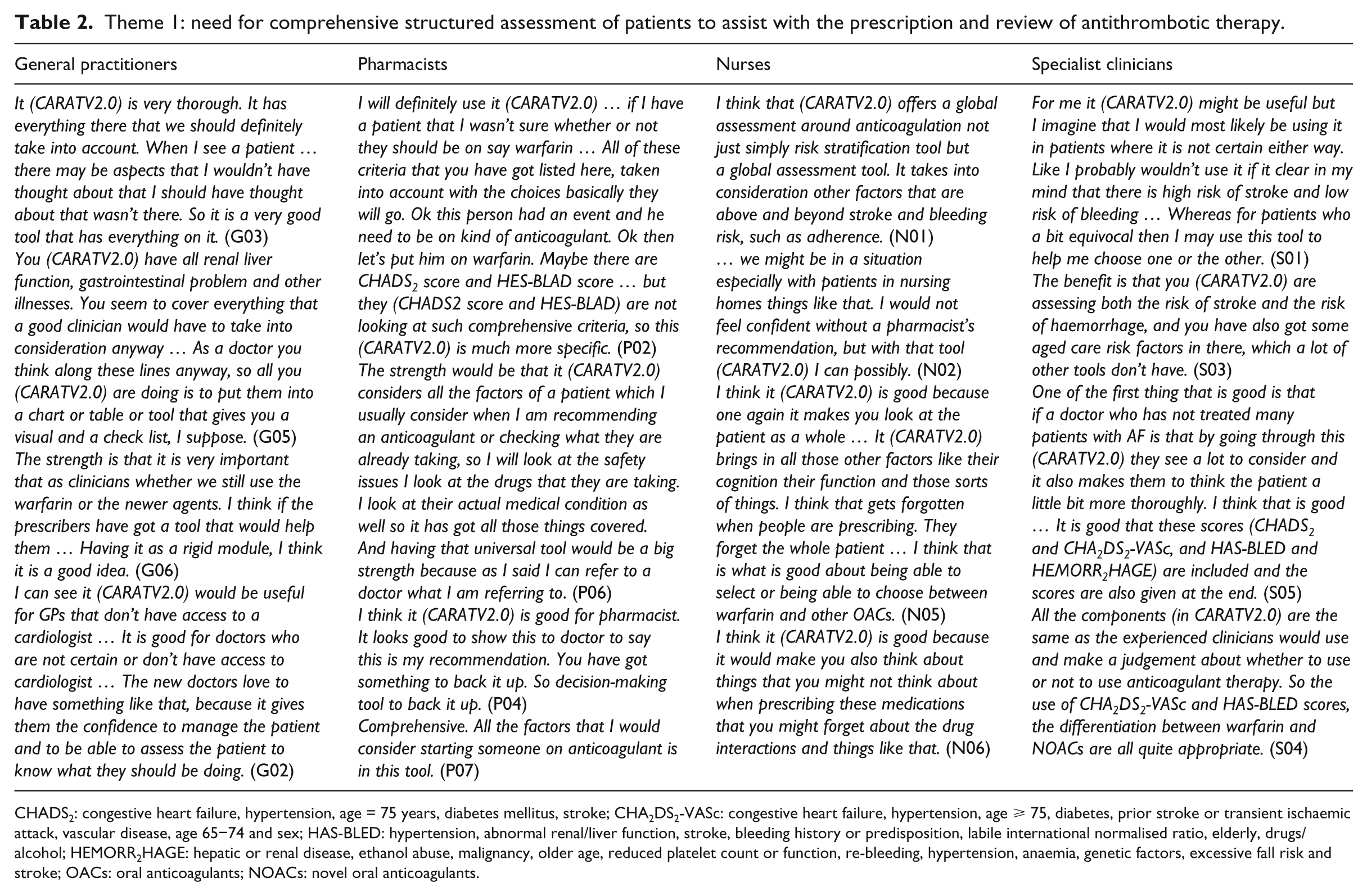
CHADS2: congestive heart failure, hypertension, age = 75 years, diabetes mellitus, stroke; CHA2DS2-VASc: congestive heart failure, hypertension, age ⩾ 75, diabetes, prior stroke or transient ischaemic attack, vascular disease, age 65−74 and sex; HAS-BLED: hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalised ratio, elderly, drugs/alcohol; HEMORR2HAGE: hepatic or renal disease, ethanol abuse, malignancy, older age, reduced platelet count or function, re-bleeding, hypertension, anaemia, genetic factors, excessive fall risk and stroke; OACs: oral anticoagulants; NOACs: novel oral anticoagulants.
Theme 2: health professionals’ and patients’ preferences for a particular antithrombotic therapy.
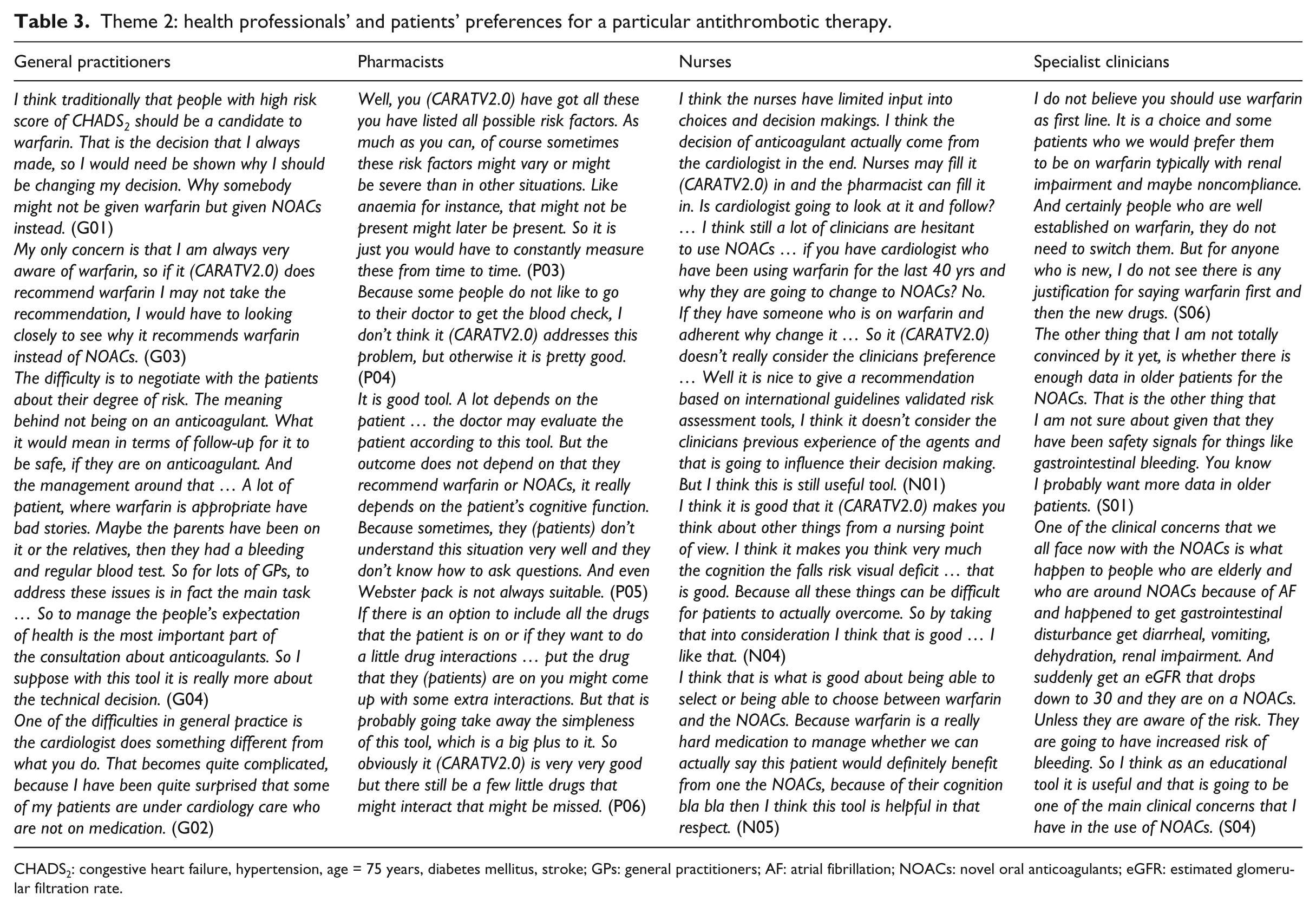
CHADS2: congestive heart failure, hypertension, age = 75 years, diabetes mellitus, stroke; GPs: general practitioners; AF: atrial fibrillation; NOACs: novel oral anticoagulants; eGFR: estimated glomerular filtration rate.
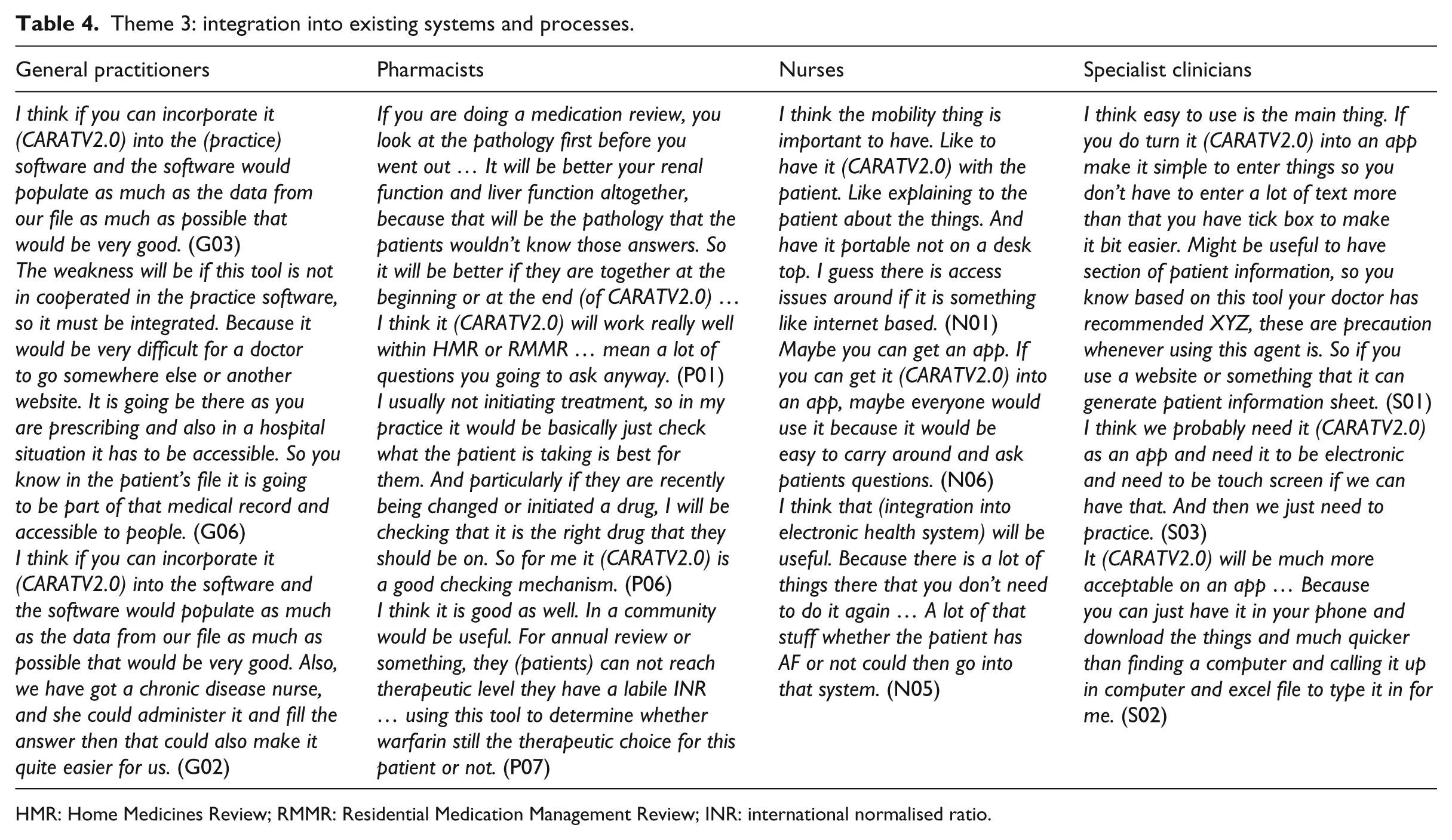
Theme 3: integration into existing systems and processes.
HMR: Home Medicines Review; RMMR: Residential Medication Management Review; INR: international normalised ratio.
Theme 1: need for comprehensive structured assessment of patients to assist with the prescription and review of antithrombotic therapy
The most highly appreciated feature of CARATV2.0 was that it provides comprehensive assessment of a patient’s risk versus benefit of using antithrombotics. Perhaps more importantly, the ability of CARATV2.0 to provide guidance and assistance in selecting among OACs, especially between warfarin and the NOACs, was highlighted by health professionals. Overall, health professionals considered this tool helpful in the decision-making for antithrombotic therapy and hoped that it could help reduce the incidence of strokes.
Specifically, many GPs and specialists felt that CARATV2.0 validated or organised their own decision-making process. Interested in using this tool for the prescription of antithrombotics, GPs and specialists tended to see the tool as most useful in those cases in which there are clinical dilemmas (i.e. where the risk versus benefit of using OACs is not clear-cut). With regard to selection among OACs, both GPs and specialists appreciated that the tool offered a specific recommendation among the OACs (especially either warfarin or an NOAC). The doctors considered this useful because they perceived that the differences in the benefits and risks of individual anticoagulant agents were not clear to many doctors. One GP mentioned that this tool could be useful for the initiation of therapy.
Similarly, the senior accredited and hospital-based pharmacists (with ⩾40 years of experience) also stated that CARATV2.0 validated or organised their own decision-making process. In contrast, nurses and the junior accredited pharmacists felt that they could use this tool as a reference for their medication reviews and patient assessments, especially when choosing among OACs. Pharmacists and nurses also emphasised that patients’ risk factors associated with antithrombotic therapy were not static; therefore, they tended to see this tool as most useful for regular reviews of patients. This aspect of tool use seemed to be overlooked by the GPs and specialists.
Theme 2: health professionals’ and patients’ preferences for a particular antithrombotic therapy
Health professionals’ opinions on CARATV2.0’s recommendations were underpinned by whether they perceived the tool as preferring any particular antithrombotic therapy and whether this therapy was the one they preferred to use. While warfarin was preferred by the majority of health professionals, one neurologist, two haematologists, one GP and one nurse stated that they preferred using NOACs. Some pro-NOAC health professionals perceived that CARATV2.0 was biased towards warfarin and thus distrusted CARATV2.0’s recommendation when it did not recommend their preferred therapy. Similarly, several pro-warfarin health professionals questioned and disliked CARATV2.0’s recommendations because it did not allow negotiation with their preference. Because patients were routinely referred to either GPs (in remote and regional areas) or specialists (in metropolitan areas) for the prescription of antithrombotic therapy, many health professionals believed that the usefulness of CARATV2.0 in improving antithrombotic selection would depend on whether the tool’s recommendations are followed by the GPs and hospital doctors.
Some GPs believed that CARATV2.0 might be able to assist in negotiations with patients by providing evidence (e.g. stroke risk score) for explanations. However, one GP argued that CARATV2.0 could not help in persuading patients to take certain OACs because the negotiation to persuade or convince patients to take antithrombotic therapy involves managing individualised health expectations rather than only presenting scientific evidence about this form of therapy.
Pharmacists’ and nurses’ perspectives on the usefulness of CARATV2.0 was largely determined by whether they thought this tool considered important issues in medication management when selecting antithrombotic therapy for individual patients. While pharmacists focused more on the medication safety issues (e.g. drug–drug interactions, adherence and international normalised ratio (INR)) when using this tool, nurses paid more attention to the tool’s assessment of patients’ capability to manage their medications (e.g. mobility, cognitive function and lifestyle). Although pharmacists and nurses believed that CARATV2.0 comprehensively assessed the major medication management issues, they also pointed out that CARATV2.0 did not consider every issue, for example, use of fish oil supplements or binge drinking. But they also admitted that the selective inclusion of the most important medication management issues ensured the simplicity and ease of use of this tool.
Theme 3: integration into existing systems and processes
Hospital-based health professionals (including specialists, nurses and pharmacists) and GPs suggested integration of CARATV2.0 into existing systems and processes due to the limited time available for making clinical decisions. Most hospital-based health professionals and GPs recommended that the tool’s usefulness could be improved if it was integrated into or linked with electronic medical records or the electronic prescribing software used in hospitals and general practices. They recommended it be accessible through mobile phones, computers and tablets in order to self-populate the medical records and databases and to make the tool easily accessible and portable. However, both GPs and hospital-based health professionals were worried that some of the practice computer software might not interact well with CARATV2.0. They were also concerned that some of the information required by CARATV2.0 might not be available in the electronic health system, which would mean that such information would require manual entering. To solve this problem, some suggested that pharmacists, junior medical residents, medical students or practice staff (e.g. nurses) could populate CARATV2.0 manually, allowing senior clinicians more time to review CARATV2.0’s recommendations.
In contrast to hospital-based health professionals, the time needed for populating CARATV2.0 was not raised as a major issue by community-based pharmacists and community-based nurses. Some of these practitioners actually thought that CARATV2.0 would save them time because it considers all the antithrombotics indicated for AF and integrates many relevant risk assessment tools into one tool. They paid more attention to how the tool’s usability could be improved by incorporation into their medication review and patient assessment processes within an electronic format (e.g. ‘apps’, websites or software).
Discussion
The results from this study show that CARATV2.0 is generally welcomed by health professionals and that they consider it can potentially improve prescription of OACs and clinical outcomes of patients. This is consistent with the evidence that computerised decision support tools can significantly improve prescription among clinicians and can improve the quality and safety of care provided.28,29 Although decision support tools that focus on the assessment of stroke and bleeding risk are widely available, 30 so far CARATV2.0 is the only tool that integrates stroke risk assessment, bleeding risk assessment and medication safety assessment and that considers both the traditional antithrombotic agents, warfarin and the NOACs.
Since decision-making is an emotive process, comprehensive risk versus benefit assessment, systematic documentation and communication of decisions can assist in the selection of optimal therapy for individual patients. 3 However, due to limited experience with the use of newly available NOACs, especially NOAC use in elderly patients, the risk versus benefit assessment of using OACs in these, and other, patients is a complex task for many health professionals. CARATV2.0’s comprehensive risk versus benefit assessment of individual patients provides guidance and a reference for, and confidence in, not only the decision on whether a patient should be treated with antithrombotics but also choosing the appropriate therapy among various OACs. Furthermore, because CARATV2.0 is based on the latest clinical guidelines, the tool can also reinforce the use of clinical guidelines by health professionals.
The study found that subjective issues, including clinician and patient preferences for particular antithrombotic therapies, can have a substantial impact on the clinical decision-making process. 31 Studies have shown that clinicians tend to override recommendations made by a decision support tool if they have a strong preference for a particular medication. 32 Also, patients’ preference of therapy has been reported to substantially affect the clinical decision-making for therapy. 33 Although it is widely recognised that computerised decision support tools have the potential to improve the behaviour of clinicians in terms of prescription and consistency of decision-making, evidence supporting the long-term impact of decision support tools on clinicians’ prescribing behaviour is lacking. 34 Given these subjective issues, the impact of CARATV2.0 on decision-making for antithrombotic therapy in AF needs to be further explored.
The suggestion of integrating CARATV2.0 into existing systems and processes shows that the health professionals valued the tool as an effective support for clinical decision-making. 32 According to a systematic review by Kawamoto et al., 35 a successful decision support tool needs to be computer-based, to have automatic provision of decision support as part of clinician workflow, to provide recommendations rather than just assessments and to provide decision support at the time and location of decision-making. However, this suggestion also reflects that clinicians are reluctant to prioritise and allocate time for the initial decision-making around antithrombotics, which contrasts with the time spent in managing the adverse outcomes of poor or suboptimal prescription. Also, ‘pharmacotherapy’ as an intervention follows a less structured decision-making process than other interventions such as surgery.
Some limitations of the study need to be acknowledged. The participating specialist clinicians, GPs, pharmacists and nurses in this study were volunteers who showed interest in the study. This could have biased their feedback on CARATV2.0. Also, the sampling strategy affects to some extent the generalisability of the study findings beyond these participants. Furthermore, CARATV2.0 inputs are based on available evidence from guidelines and reviews, which may not be relevant to all patient populations and may change over time as new evidence emerges. Since the findings from this research are restricted to the content and feasibility of this tool, the potential clinical and economic impact of the tool and the feasibility of using the tool in real-world clinical practice require further evaluation.
Overall, the feedback from health professionals identifies that the only drawback of this tool is the time needed to complete the assessment (i.e. input the relevant data). To address this issue for future application in practice, CARATV2.0 may be integrated into other systems (e.g. electronic medical records) to enable the auto-population of patient data into the tool. Furthermore, the tool may be used by other health professionals (e.g. NPs and consultant pharmacists) where comprehensive patient assessment and medication review are part of their targeted services.17,36
Conclusion
CARATV2.0 was regarded by a variety of health professionals as a potentially useful tool that provided a systematic assessment around the decision-making for antithrombotic therapy in patients with AF. The tool also shows potential for rationalising the use of antithrombotics and for improving the clinical outcomes of patients with AF. Future research should evaluate the impact of this tool on the prescription of antithrombotics in clinical practice. The main drawback of this prototype tool is that it requires the manual input of data, which may not be time-efficient for busy health professionals. Therefore, processes for the auto-population of the tool with relevant patient data need to be explored, for example, the integration of CARATV2.0 into electronic databases or prescribing software or/and re-formatting it into a mobile online application.
Footnotes
Acknowledgements
The authors would like to thank Ekta Pandya, Shamsher Singh, Riana Rahmawati and Leigh Findlay.
Authors’ contribution
All authors have made substantial contributions to all of the following: (1) the conception and design of the study, acquisition of data, analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval was given by University of Technology Sydney (UTS HREC REF NO. 2013000338).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
