Abstract
This study compared the cost-effectiveness of telemonitoring with standard monitoring for patients with diabetic foot ulcers. The economic evaluation was nested within a pragmatic randomised controlled trial. A total of 374 patients were randomised to either telemonitoring or standard monitoring. Telemonitoring consisted of two tele-consultations in the patient’s own home and one consultation at the outpatient clinic; standard monitoring consisted of three outpatient clinic consultations. Total healthcare costs were estimated over a 6-month period at individual patient level, from a healthcare sector perspective. The bootstrap method was used to calculate the incremental cost-effectiveness ratio, and one-way sensitivity analyses were performed. Telemonitoring costs were found to be €2039 less per patient compared to standard monitoring; however, this difference was not statistically significant. Amputation rate was similar in the two groups. In conclusion, a telemonitoring service in this form had similar costs and effects as standard monitoring.
Introduction
The prevalence of foot ulcers among individuals with diabetes mellitus in Denmark is estimated to be at least 22,000 and the annual incidence is 3000 based on cases treated in a hospital setting. 1 Approximately 15% of patients with diabetes will have at least one diabetes-related foot ulcer during their lifetime, 1 and 40%–60% of ulcers will recur within 5 years.2–4 Diabetes is associated with ischaemia, neuropathy and deformities that lead to a particularly high risk of developing foot ulcers and a low likelihood of ulcer healing. 5 These ulcers cause considerable physical discomfort and affect the patient’s mobility and quality of life. 1 The total annual direct economic cost of diagnosing and treating individuals with incident diabetic foot ulcers in Denmark has been estimated at 793 million Danish Kroner (DKK) (€106 million) per year. 5
Wound monitoring via telemedicine is currently being implemented in Denmark, also for diabetic foot ulcers, under a National Action Plan for the deployment of telemedicine and is expected to be available in all regions and municipalities by 2017.1,5,6 However, there is little evidence that telemedicine in the monitoring of diabetic foot ulcers improves ulcer healing or patient outcome in general.1,7 An optimistic so-called ‘business case’ was published in support of the National Action Plan, but no high-quality studies are available that consider the economic aspects of the use of telemedicine for ulcer monitoring.6,8
As a consequence, a large Danish pragmatic randomised controlled trial (RCT) of telemedicine consultations in the monitoring of diabetic foot ulcers was undertaken in the Region of Southern Denmark. 8 The clinical and organisational results have been published elsewhere,9,10 and the current article reports the economic evaluation. The main purpose of the trial was to generate information for the decision-making process regarding the national introduction of telemedicine consultations in the monitoring of patients with diabetic foot ulcers. The RCT was part of the RENEWING HEALTH 11 project in which nine European regions examined the effect of telemedicine in a broad, interdisciplinary perspective in patients with chronic diseases, including diabetes mellitus.11,12
Aim and objectives
The aim of this study was to perform an economic analysis, piggybacked on an RCT, to compare the costs and effects of telemonitoring (TM) with standard monitoring (SM) in individuals with diabetic foot ulcers. The following three steps were performed:
Descriptive statistics and calculation of average use of resources, average costs and average effects;
Cost-effectiveness analysis (CEA) and calculation of incremental cost-effectiveness ratio (ICER) for amputations;
Sensitivity analysis: Bias corrected bootstrap analyses on 5000 re-samples to evaluate the overall uncertainty of the estimated ICER; One-way sensitivity analyses to assess the robustness of the study results.
Methods
A recent publication by the European Wound Management Association has recommended using the Model for ASsessment of Telemedicine (MAST) when evaluating telemedicine in wound care. 13 MAST advocates the use of a multidisciplinary assessment comprising seven different domains. 14 This article uses the MAST approach and focuses on the fifth domain of economic aspects. The economic evaluation follows international guidelines for the conduct of health economic evaluation of health interventions as stated by CHEERS – Consolidated Health Economic Evaluation Reporting Standards. 15
The clinical study
The RCT was conducted in 2010–2014 at seven departments and outpatient clinics of five hospitals and included all 22 municipalities in the Region of Southern Denmark. 8 A pragmatic study design was applied, and patients diagnosed with a diabetic foot ulcer starting treatment in one of the specialised ulcer outpatient clinics were eligible for the clinical study. The aim of the clinical trial was to investigate, whether a telemedicine approach to ulcer treatment could be used as an alternative to conventional monitoring at the outpatient clinic. 16 The participants were assessed for comorbidity and the need for surgical intervention prior to randomisation. The per protocol telemedical monitoring consisted of two tele-consultations in the patient’s own home and one consultation at the outpatient clinic. SM comprised three outpatient clinic visits. The three-visit cycle continued until the study endpoint, that is, patients stayed in the study until one of the following endpoints occurred: ulcer healing, amputation, patient withdrawal of consent, patient moving to a hospital department not using telemedicine monitoring of diabetic foot ulcers, death, or 1 year (365 days) without healing (i.e. the ulcers were considered chronic).
The telemedical consultations were conducted by telephone or online written consultations between the specialised municipal nurse and doctors at the outpatient clinic. These consultations were supplemented by an uploaded screen image of the ulcer and a detailed written assessment via the online database. 17 If needed, the treatment strategy was revised, and the next consultation was carried out as required (i.e. telemedical or outpatient consultation), and the indication for further images was agreed between the nurse and physician. Patients randomised to standard care followed the usual practice and treatment provided by the outpatient clinic, where all consultations took place in the outpatient clinic. In both groups, municipal nurses provided standard daily care to the study participants in their own home under supervision of a municipality nurse specialised in ulcer care. Home care was delivered after an algorithm determined by clinical judgment, usually twice or three times a week. The healthcare professionals received training in use of the telemedicine system, 17 and diabetic foot ulcer management competencies were upgraded among the involved municipality nursing staff.
Cost estimation and prices
Healthcare sector resource use and costs were estimated using a variety of sources. The following elements were included: (1) admissions, (2) outpatient visits at hospitals, (3) emergency department visits, (4) all visits to general practitioner, (5) staff training, (6) investment and running costs of the telemedicine equipment, (7) time used by home care nurse on ulcer consultations and transport and (8) patient time used on transport to hospital for ulcer consultations. Elements 5–8 were based on questionnaires and interviews from the RCT, whereas elements 1–4 were identified for individual patients from national health administrative databases. Also, time used by staff regarding the wound-related outpatient visits in element 2 was recorded in the RCT.
Table 1 presents 2012 unit costs used in the analysis. DKK were converted to EURO (€) using the exchange rate €1 = 7.5 DKK. As the magnitude of the investment costs were minor compared to the other cost components in the CEA (see Table 3), minimal efforts were put into estimating these with great precision in accordance with the recommendations. 18 Investment costs covered staff training, establishing the service infrastructure and project management. Investment costs were based on staff knowledge and details from an internal Odense University Hospital 19 document containing an overview of the budget for national implementation of telemedicine.
Unit costs (€, 2012 prices).
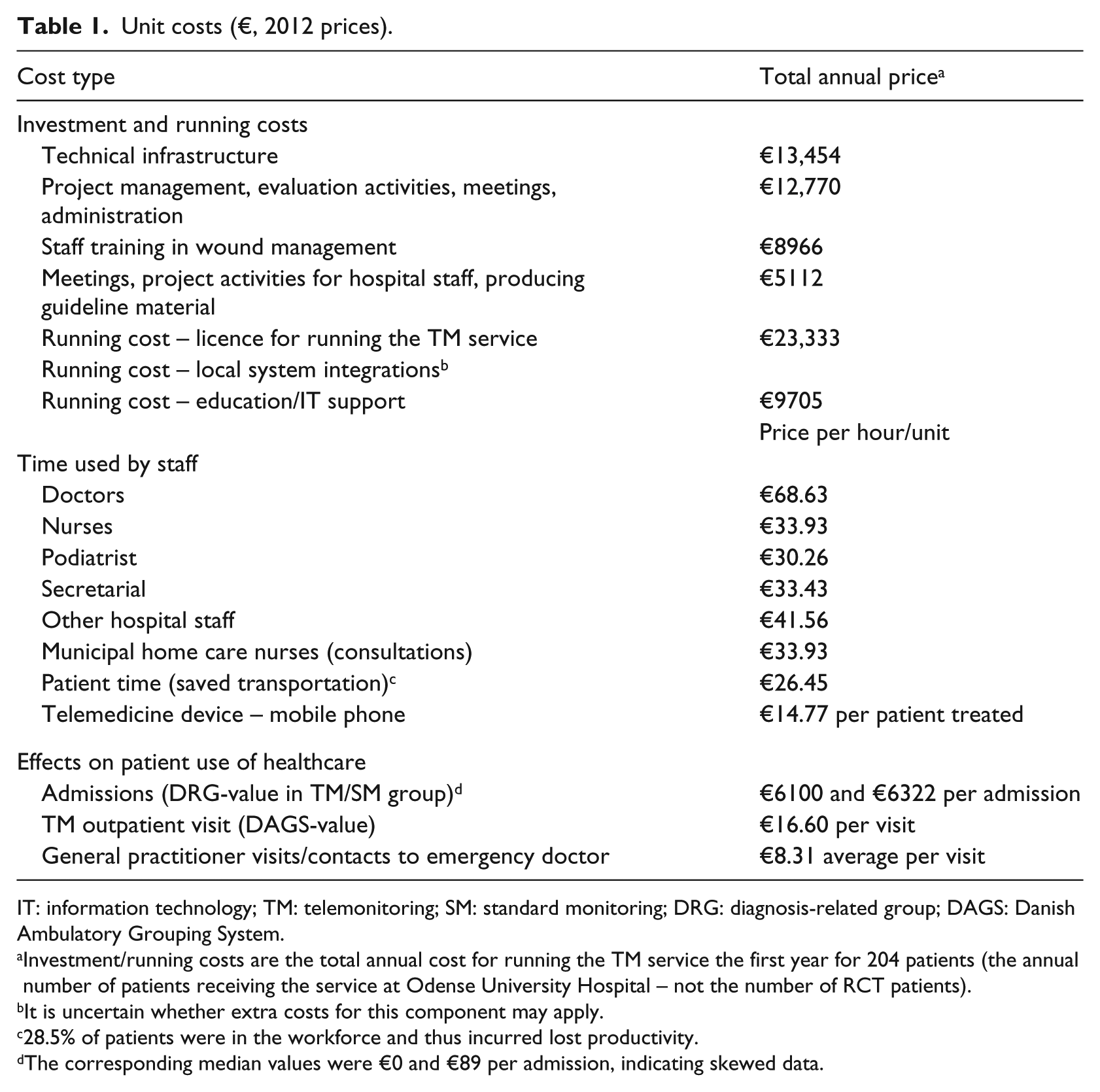
IT: information technology; TM: telemonitoring; SM: standard monitoring; DRG: diagnosis-related group; DAGS: Danish Ambulatory Grouping System.
Investment/running costs are the total annual cost for running the TM service the first year for 204 patients (the annual number of patients receiving the service at Odense University Hospital – not the number of RCT patients).
It is uncertain whether extra costs for this component may apply.
28.5% of patients were in the workforce and thus incurred lost productivity.
The corresponding median values were €0 and €89 per admission, indicating skewed data.
Hospital staff completed questionnaires about the type of consultation and the time used. Average salaries from the university hospital were used for valuing staff time, under the assumption of 1481 effective work hours per year. The number of home care consultations and the time used on consultations and transportation were recorded in questionnaires filled out by the home care nurses. The patient’s distance to hospital was recorded by hospital staff when the patient was enrolled in the study. The value of patient time used was estimated from the average national income from Statistics Denmark, under the assumption of 1481 effective work hours. Furthermore, the RCT study show that 28.5% of the patients were in the workforce, and thus had costs related to lost productivity. 20
The Equivalent Annual Cost (EAC) method 18 was used to handle hardware investments and establishment costs by converting them to an annual cost, assuming a 2-year life span for mobile phones and IT equipment and a 5-year life span for establishment costs. The above calculation assumed that 204 foot ulcers were treated at Odense University Hospital annually, based on estimates from the clinical department regarding the annual expected number of patients. Cost and effectiveness outcomes were not discounted as the analysis was limited to 6 months.
Information about patient use of healthcare services (e.g. inpatient and outpatient activity, general practitioner visits) was drawn from the Danish National Patient Register 21 that records every healthcare encounter for all Danish citizens. Each healthcare service was assigned a standardised cost (reimbursement), so that cost estimates were based on the number of encounters recorded in the Danish National Patient Register multiplied by the ascribed standardised DRG/DAGS rate 22 and fee-for-service rates for general practitioner activities reflecting average costs.
Analysis
Handling of missing data
In the main CEA analysis, missing values were handled by mean imputation conditional on the assigned group, that is, replacing a missing value with the mean value of the available cases in either the TM or the SM group. 23
The economic evaluation
The primary outcome for the CEA was incremental cost per 1% change in amputation rate. Follow-up for amputations was approximately 6 months and for costs 6 months after study inclusion. In the data analysis, differences in the average use of resources, costs and effects per patient in the intervention and control groups were calculated. Differences in costs and number of amputations were compared in the two groups and combined in a CEA. The ICER, which is the additional cost per additional health outcome, that is, amputation, was then calculated according to the standard formula: ICER = (C1−C2)/(E1−E2), where C1 and E1 were the cost and effect in the intervention group, and C2 and E2 were the cost and effect in the control group.
Data handling, sensitivity analysis, statistical methods and assumptions
It was intended to keep all patients in the analysis in order to analyse data by the intention-to-treat principle (ITT). According to ITT, all randomised patients in a study should be analysed regardless of whether they have completed the study or not or received the treatment. The full ITT principle is only applicable when complete measurements on the primary outcomes for all randomised patients exist. 24 In this case, no measurements on the primary outcomes exist for the dropout patients, and it is further considered unethical to use data from patients who have withdrawn their consent, and hence ITT is not performed.
Data were entered into EpiData version 3.1 25 before transfer to the statistical package Stata version 12 for analysis. The two patient groups were compared using appropriate statistical tests for testing differences in costs and effects, that is, t-test for normally distributed continuous data, χ2-test for categorical data and the non-parametric Mann–Whitney test for non-normally distributed data. Statistical significance was set at p < 0.05. The analyses were performed on means for all variables included in the economic evaluation. The overall uncertainty of the estimated ICER was evaluated through probabilistic sensitivity analysis in terms of a 5000 bootstrap re-sample with a 95% confidence interval. A cost-effectiveness plane was used to illustrate the 5000 bootstrap re-sample of ICER.
Results
The data and descriptive statistics
Data for 24 patients were not included in the economic (and clinical) evaluation; 2 patients withdrew consent after randomisation, 8 had participated before, 3 did not have an ulcer on the foot and 11 had insufficient data and were excluded for a variety of reasons. 10 Hence, the CEA was carried out on individual data from a total of 374 patients (181 in the SM group and 193 in the TM group). Most variables were 100% complete, and imputed values were used for three elements in the CEA (home care visits were 63% complete while total outpatient visits and distance from patient’s home to hospital were 95% complete).
The patients in the two groups had no significant differences at baseline on demographic and clinical characteristics, that is, age, gender, height, weight, body mass index, smoking, comorbidities and years with diabetes. A statistically significant difference in mortality rate in the two groups was observed; see the clinical study for a discussion of this result. 10 Average costs per patient 6 months before inclusion in the study were calculated based on admissions, outpatient visits, emergency department visits and general practitioner visits and showed no statistically significant differences in the two groups. Hence, no adjustments for baseline values were made in the following analyses.
Average resource use and average costs
Table 2 shows a statistically significant difference in total staff time used on outpatient consultations (156 min for the TM group compared with 266 min for the SM group). This is expected, given the study design. Furthermore, the total time used by staff on telemedicine consultations was 68 min for the TM group. The between-group differences in staff time used for telephone consultations and other outpatient visits were not statistically significant, but the number of telephone contacts was significantly higher in the TM group (0.3 contacts vs. 0.2 contacts). Again, this is expected given the study design. Total staff time (time spent on the four consultation types combined) was statistically significantly higher in the SM group (306 min) than the TM group (255 min), due to fewer outpatient visits in the TM group and less time used by nurses and podiatrists.
Average use of resources per patient in the TM and SM group.
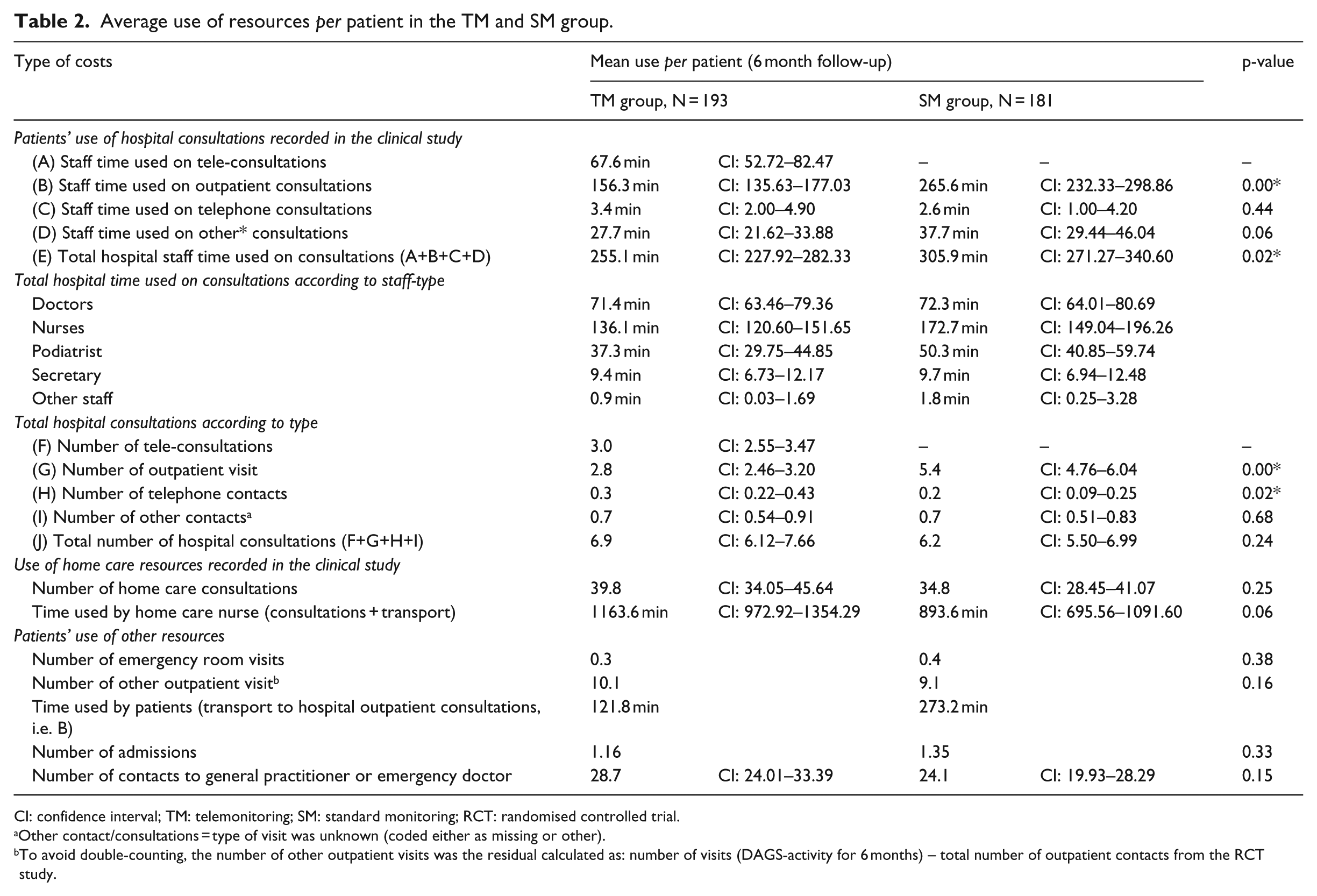
CI: confidence interval; TM: telemonitoring; SM: standard monitoring; RCT: randomised controlled trial.
Other contact/consultations = type of visit was unknown (coded either as missing or other).
To avoid double-counting, the number of other outpatient visits was the residual calculated as: number of visits (DAGS-activity for 6 months) – total number of outpatient contacts from the RCT study.
The number of total hospital consultations was fairly similar in the TM and SM groups (6.9 vs. 6.2), but the TM group had more municipal home care consultations (40 contacts vs. 35 contacts), and hence higher staff time. On average, a TM patient used less time than an SM patient on transport for outpatient visits (122 vs. 273 min). The TM group had fewer hospital admissions. As Table 2 shows, only four resource elements showed a statistically significant difference.
Unit costs were multiplied by the quantities to calculate total costs. Table 3 shows that from a broad healthcare sector perspective, the total costs of TM were €2039 lower per patient treated than in the SM group. This was mainly due to fewer admissions and lower outpatient costs in the 6-month follow-up period. This difference was not statistically significant, however.
Average treatment costs per patient in the telemedicine (TM) and standard monitoring (SM) groups (€, 2012 prices).
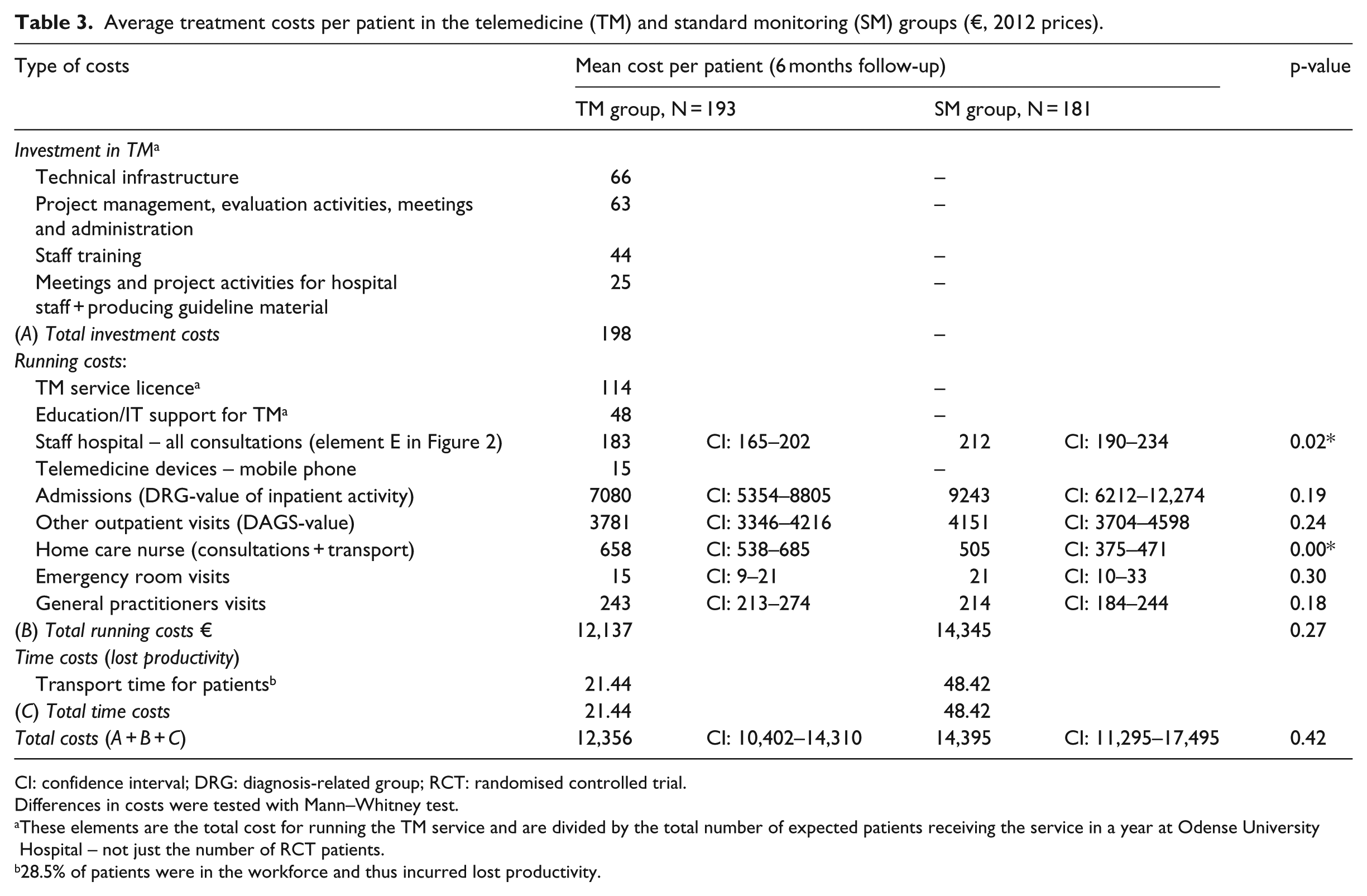
CI: confidence interval; DRG: diagnosis-related group; RCT: randomised controlled trial.
Differences in costs were tested with Mann–Whitney test.
These elements are the total cost for running the TM service and are divided by the total number of expected patients receiving the service in a year at Odense University Hospital – not just the number of RCT patients.
28.5% of patients were in the workforce and thus incurred lost productivity.
The distribution of total cost per patient in the two groups 6 months after study inclusion is shown in Figure 1. As noted by Drummond, 18 cost data are often non-normally distributed with a skewed and heavy right-hand tail of individuals who make frequent use of healthcare services. This was also the case in our data. The mean total cost in the TM and SM groups was €12,356 and €14,395, respectively; the corresponding median numbers were €6503 and €7677. One TM patient and one SM patient had a total cost above €100,000; this difference is explored in more detail in the sensitivity analysis.
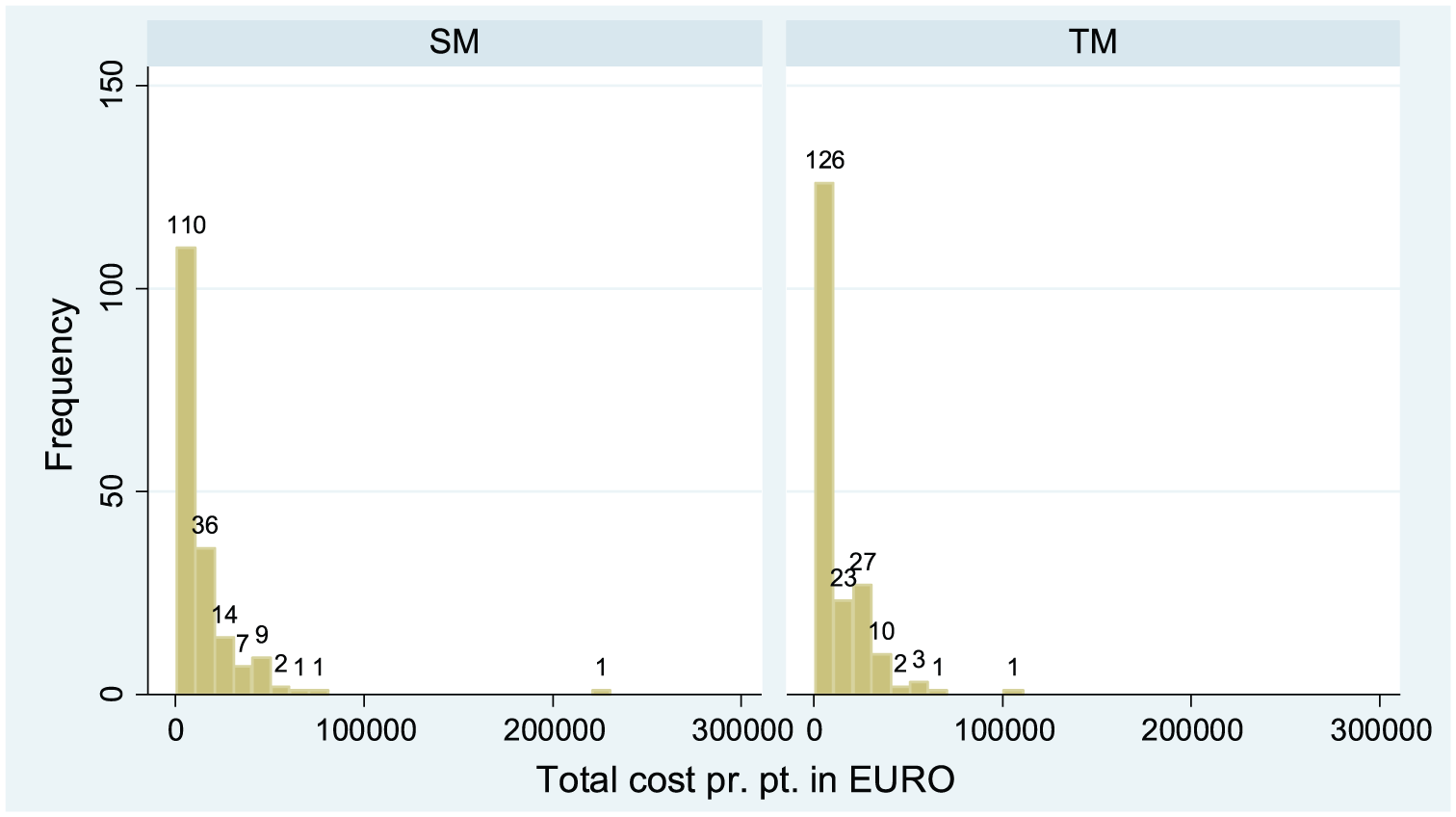
Histogram of total costs per patient in the telemonitoring group and the standard monitoring group.
Incremental cost-effectiveness ratio
No statistically significant difference could be detected between the TM and SM groups in the primary outcome, that is, amputations, but there was an absolute difference in the primary outcome. The following results of the CEA should thus be interpreted with great caution.
Based on data from 374 patients, we calculated the ICER as the cost per avoided amputation. Table 4 indicates that on average, a patient in the TM group cost €2039 less than an SM patient. Furthermore, 11% and 14% of patients had amputations in the TM and SM groups, respectively. Combining the above information, the result was a mean saving of approximately €67,973 to avoid one amputation.
Incremental cost-effectiveness ratio (ICER) for amputations in the telemedicine (TM) and standard monitoring (SM) groups.
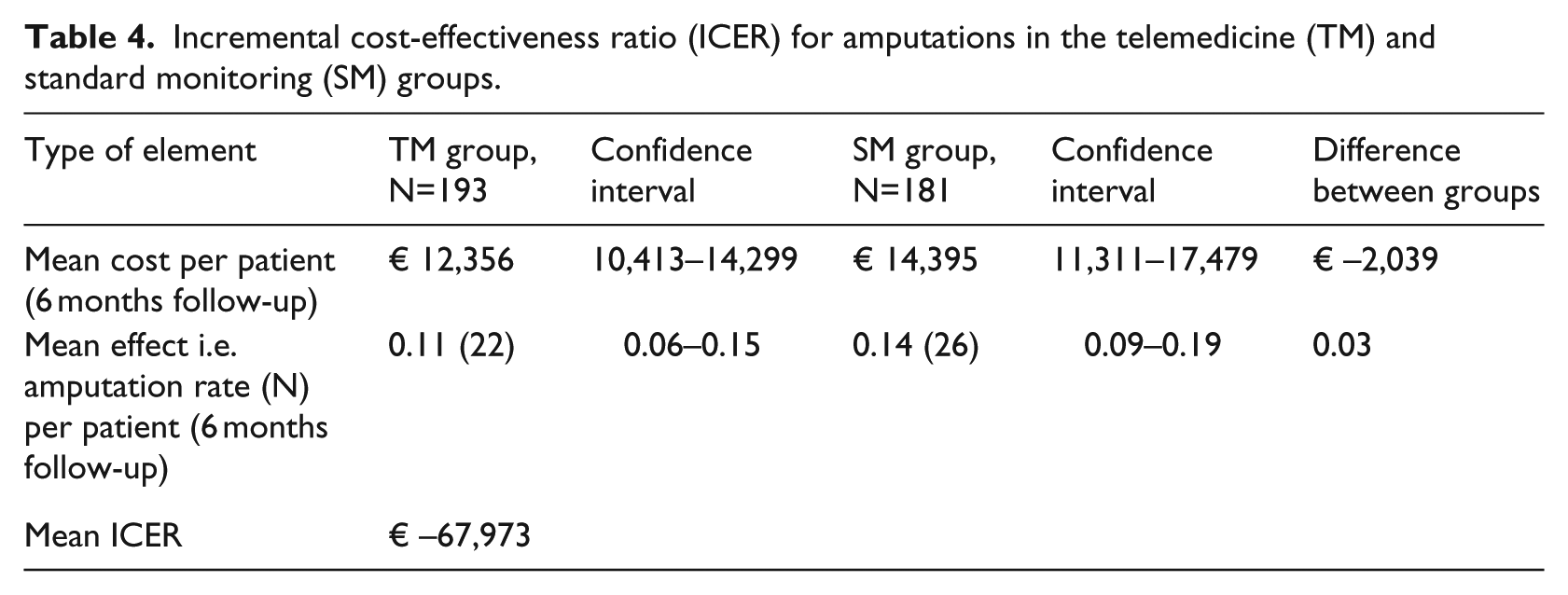
Figure 2 shows that most of the 5000 replications were in the fourth quadrant of the figure, indicating that TM was less expensive and associated with fewer amputations than SM, that is, TM was dominant. It should be noted that this observed result in Table 4 and Figure 2 may be due to chance alone (due to no statistically significant differences in cost or amputation rate).
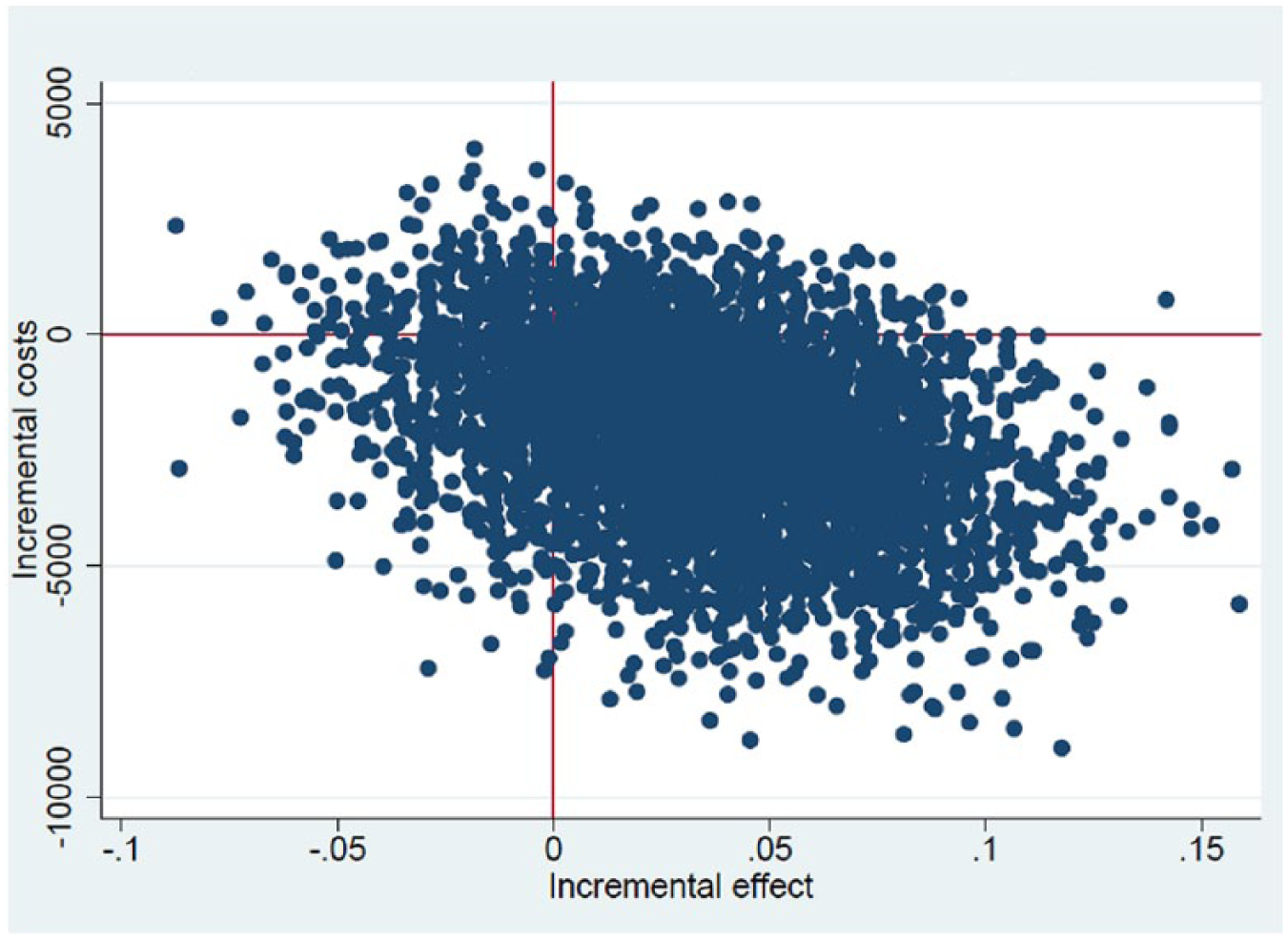
Cost-effectiveness plane.
Sensitivity analysis
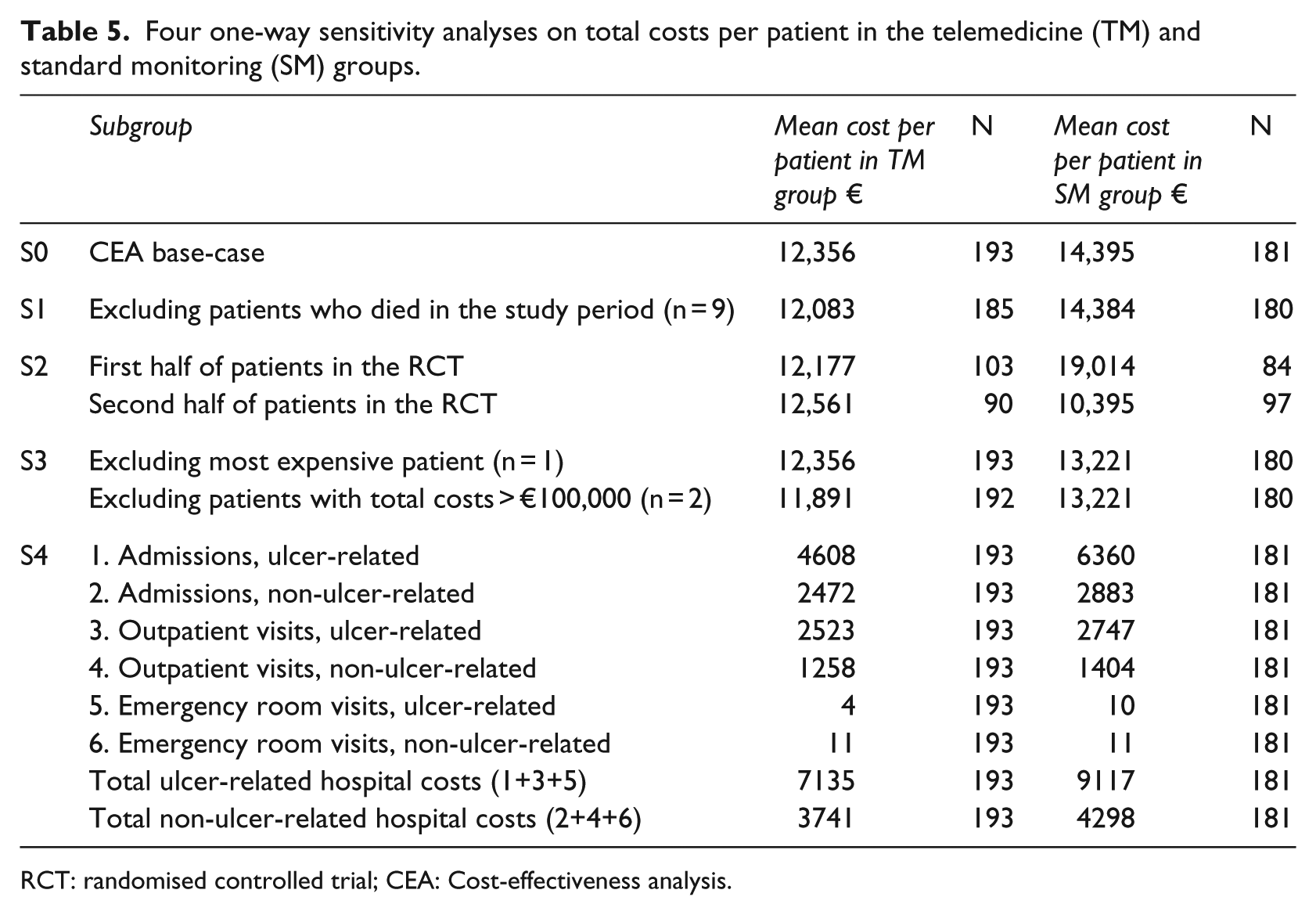
Table 5 presents four different sensitivity analyses, S1–S4 (the first row, S0, is the results from the main analysis reported above, for comparison).
Four one-way sensitivity analyses on total costs per patient in the telemedicine (TM) and standard monitoring (SM) groups.
RCT: randomised controlled trial; CEA: Cost-effectiveness analysis.
As the clinical analysis found a significant difference in mortality between the TM and SM groups, 10 we tested whether the difference in total cost between the two groups was due to this difference in mortality rate. In S1, we excluded from the economic analysis all patients who died during the study period, but this did not change the overall results. S2 investigated whether the average total costs per patient changed over time in the two groups. The results indicated that costs in the TM group appeared robust over time, with little change from the first half of patients included in the RCT to the second half. A different pattern was observed in the SM group, where the second half of the patients included in the RCT were less costly to treat, with a mean cost of €10,395 compared to the first half patients with a mean cost of €19,014.
In S3, we first excluded one patient with the highest total cost and subsequently patients with a total cost of more than €100,000 (one in each group). The result was highly sensitive to this change. Excluding one patient lowered the average total cost by approximately €1100 in the SM group, while excluding two patients resulted in a lower total cost of approximately €500 per patient in the TM group and €1200 in the SM group.
Finally, the difference in hospital resource use was explored in more detail. As can be seen from the average total costs in the main analysis in Table 3, the difference in total costs was primarily due to differences in number of admissions and outpatient visits in the two groups. S5 investigated this difference further by subdividing hospital costs (admissions, outpatient and emergency) into ulcer-related or non-ulcer-related costs. This categorisation was done by two doctors with extensive wound care experience using the departmental codes for hospital visits. The results of S5 show that SM patients had higher ulcer-related and non-ulcer-related costs, suggesting that the overall cost difference between a TM and an SM patient was robust. On the other hand, because the same pattern was seen for both ulcer and non-ulcer elements, it is unlikely that the difference was due to the TM consultations for treatment of diabetic foot ulcers.
Discussion
This CEA comparing TM and SM for individuals with diabetic foot ulcers found that TM cost is €2039 less per patient treated than SM; however, this difference was not statistically significant. Cost savings were due to differences in the number of admissions and outpatient visits, and TM remained less expensive in all sensitivity analyses. The amputation rates were similar in the two groups, and the incremental cost-effectiveness ratio showed a mean saving of approximately €67,973 to avoid one amputation. On the basis of these results, we conclude that a telemonitoring service in this form has similar costs and effects as standard outpatient monitoring. Cost savings by providing TM care for individuals with diabetic foot ulcers are thus uncertain.
This study is the first RCT-based economic evaluation of TM in individuals with diabetic foot ulcers. A few other studies have addressed the costs of telemedicine in wound care, but none addressed cost-effectiveness in accordance with international guidelines on economic evaluation alongside a trial.10,26 A Swedish study by Tennvall et al. concluded that inpatient costs for foot complications cannot be accurately estimated from the Inpatient Registry when based exclusively on the primary diagnosis. However, they concluded that fairly good estimates can be made with a combination of foot-related diagnoses together with codes for diabetes. 27 We have taken this observation by Tennvall et al. into account and have used clinical expert opinion in performing sensitivity analysis on costs to investigate whether admission, outpatient and emergency visits are ulcer-related.
The conclusion that the costs and effects of TM are comparable to those of SM may seem counter-intuitive given the non-significant major difference in total costs in absolute terms. However, our findings may reflect that the study was powered after the clinical study to detect a change in number of emergency department visits, and not differences in the costs of outpatient or inpatient visits. Hence, the chosen study design in the clinical study poses a key limitation to the economic study because emergency department visits are only a very small element in the total costs. The data variability is likely to be much higher for total costs than for the number of emergency department visits reducing the possibility of identifying statistically significant changes. Thus, we cannot expect to find a statistically significant difference in the overall costs – even if such a difference was present.
Conversely, some evidence challenges that the absolute difference in costs was due to use of TM. Sensitivity analysis was used to investigate the difference between the two groups in admissions and outpatient visits. When hospital costs (admissions, outpatient and emergency) were subdivided into ulcer- or non-ulcer-related costs, the same reduction in costs was observed in both groups (ulcer- or non-ulcer-related costs). Excluding the most expensive patient from the analysis and the difference in total costs decreased substantially. These findings suggest that the absolute difference in costs may not be ulcer-related (and thus TM-related) and that the potential for future cost savings with TM may be limited.
Originally, a cost-utility analysis was planned as the main analytical framework, but results from this analysis are omitted due to incomplete data. First, we had a very low completion rate on the SF-36 questionnaires, and quality-adjusted life years (QALYs) could be calculated for only 65 patients. Second, the representativeness of these 65 patients was low compared to that of the 274 patients in the cost-effectiveness analyses. Overall, the use of randomised design in economic evaluation of telemedicine is a matter of dispute. On one hand, a small-scale test of telemedicine within one or few hospitals having a positive attitude towards telemedicine and an un-blinded staff and patients may result in positive bias. On the other hand, optimal use of telemedicine may require major organisational changes that a hospital testing a new intervention in a small-scale study is not ready to make. In addition, implementation of telemedicine may require large investment in training and IT equipment, and this may result in higher costs compared to a large-scale implementation. A possible solution to this could be observational studies estimating the costs per patient before and after a hospital or country implemented telemedicine on a large scale. However, one should not forget the value of pragmatic randomised trials which are quite distinct from traditional RCT and better suited for the changing environment of telemedicine interventions and the complex nature of the intervention.28,29
Regarding transferability of results, it can be argued that some of the included costs may be overestimated and avoidable if the service was implemented in a different setting or in a day-to-day running of the telemedicine service, that is, not protocol-driven. For example, the cross-sectorial collaboration between hospital and municipal health services is essential and an important aspect of the TM. 16 Consequently, some of the investment costs in the economic analysis cover more than a narrow implementation of telemedicine ulcer treatment (e.g. staff training in general wound care, project management, establishing an infrastructure for the service and evaluation activities), and there is an argument for leaving some of these components out of the CEA. Costs related to project management and evaluation activities may be avoidable in a day-to-day running of a telemedicine service, but since they constitute less than 1% of the total costs per patient not influencing the overall results, we decided to keep them in the analysis. When assessing total cost differences between the two groups in the long term, it is more fair to omit investment costs altogether.
Furthermore, the results reported here are based on experiences from a research trial. In routine clinical use, it may be possible to optimise the telemedicine consultations to make them faster and thus less costly. Also, the time used for patient transportation is rather low reflecting that on an average, a TM patient has 18 km to the hospital and an SM patient has 21 km. This is valid in an urbanised area like Denmark, but in a typical rural environment, distances are likely to be much longer making TM more cost-effective. The cost for home care nurses may also be higher in a more rural setting due to longer distances. Thus, the average treatment cost per patient in the TM group may be even lower.
Despite the above-mentioned weaknesses, the study brings new evidence to an area with little previous research and is the first with a methodologically strong economic evaluation of TM for individuals with diabetic foot ulcers. 18
Footnotes
Acknowledgements
We thank all collaborators from Odense University Hospital for their contribution to this manuscript, and especially data specialist Ditlev F. G. Jensen for always delivering speedy and competent help with data access and handling of register data. HTA consultant Mette Bøg Horup reviewed a draft of the manuscript and provided valuable comments. Medical student Mads Rye Bjerregaard helped with data management in the clinical study and data handling issues. Innovation consultant Lisbeth Irene Jørgensen was always very helpful when we had difficulties in understanding or finding economic information about the project. Furthermore, we thank Johnny Frøkjær for allowing us access to data from the clinical study. From Aalborg University, we thank Professor Lars H. Ehlers for competent feedback, theoretical advice and guidance in the role of supervisor on Marie Gerstrøm’s master’s thesis. Marie Gerstrøm’s thesis constitutes an earlier and preliminary health economic analysis of this study. The study was performed according to the Declaration of Helsinki II. Approval from the Danish regional ethic committees was not required for this kind of study (committee consulted). Danish Data Protection Agency 2008-58-0035. The trial is registered at the US National Institutes of Health (ClinicalTrials.gov) # NCT01608425
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Regional implementation of telemedicine in wound management was funded by: (1) ABT Fund (Applied Citizen Technology) Ministry of Finance, Denmark, (2) ‘ABT funds’ from the Region of Southern Denmark, (3) European Union (EU) project RENEWING HEALTH. Furthermore, the trial was partly funded by the Faculty of Health Sciences at the University of Southern Denmark through a PhD stipend. Odense University Hospital supported the telemedicine service and the clinical services. The funders had no part in study design, data collection, analysis, interpretation of data, and the writing of the article. Also, they had no influence on the decision to submit for publication. Writing assistance was used and was paid for by the University of Southern Denmark.
