Abstract
Results from reference laboratories are often not easily available in electronic health records. This article describes a multi-pronged, long-term approach that includes bringing send-out tests in-house, upgrading the laboratory information system, interfacing more send-out tests and more reference laboratories, utilizing the “miscellaneous assay” option offered by some reference laboratories, and scanning all remaining paper reports from reference laboratories for display in the electronic health record. This allowed all laboratory results obtained in association with a patient visit, whether performed in-house or at a reference laboratory, to be available in the integrated electronic health record. This was achieved without manual data entry of reference laboratory results, thereby avoiding the risk of transcription errors. A fully integrated electronic health record that contains all laboratory results can be achieved by maximizing the number of interfaced reference laboratory assays and making all non-interfaced results available as scanned documents.
Background and significance
Electronic health records (EHRs), defined as “a longitudinal electronic (digital format) record of patient health information generated by patient encounters in a healthcare delivery system,” 1 are replacing paper-based patient files at most hospitals and physicians’ offices.2,3 A prerequisite for the full acceptance of these systems by clinical staff is that all relevant health records are available in the EHR. 4
A significant percentage of clinical data displayed in the EHRs are generated by the clinical laboratories.1,5 Results generated in a hospital’s on-site laboratories are generally transmitted from the instruments to the laboratory information system (LIS) and from the LIS to the EHR as HL7 (Health Level Seven) messages; 6 results are then displayed in a “Results” or “Laboratory” section of the EHR together with reference ranges, interpretative comments, flags (e.g. “high,” “low,” “critical”), and other information such as the date and time of the order, of collection, and of release of results. This system generally allows error-free communication of laboratory results to clinicians within minutes of completion of analysis. Users can usually display specific data as customized tables or graphs, allowing easy determination of trends, and access results remotely from off-site locations. Due to its speed, accuracy, and remote availability, electronic communication of laboratory results has become the standard of care, expected as a matter of course by clinicians and regulators alike.1,7
Laboratory data in a hospital EHR may come from hospital laboratories, point of care testing performed at the patients’ bedside, or from outside reference laboratories. 8 It is estimated that independent/commercial (“reference”) laboratories account for 38.7 percent of the laboratory market, with annual billings of over US$12.4 billion. 9 While most hospitals are fully set up to communicate data generated by the in-house laboratories electronically to clinical staff, many institutions struggle with the accurate, timely, and complete communication of results from reference laboratories with which their LIS is not directly interfaced.7,10 As Aller et al. 1 have noted, “there is a significant risk of laboratory results being garbled as they are transmitted to and/or are displayed in the EMR.” Frequently, some of the results from such outside laboratories are manually transcribed into the LIS; this creates the risk of transcription errors and often delays the reporting of results. 11 Due to lack of sufficient staffing, lengthy reports from reference laboratories are often only partially transcribed into the LIS, and the longest and most complicated reports are sometimes summarised with comments like “results faxed/mailed to requesting clinician.” In these cases, the information in the reference laboratory report is not easily available in its entirety to all providers. This can negatively impact the quality of care and increase health care costs due to unnecessary repeat testing.
Our hospital’s goal is to use our EHR as a fully integrated health record, allowing clinicians one-stop access to all relevant patient data, including all laboratory results obtained in association with any encounter in our hospital and its clinics. This article describes the electronic reporting of all laboratory data, including reference laboratory results, in an EHR.
Methods
Site
The Columbia University Medical Center (CUMC) campus of NewYork-Presbyterian Hospital (NYPH, New York, NY, USA) is an academic medical center with over 1000 inpatient beds serving both adult and pediatric populations. The laboratories are staffed by over 470 FTEs (Full-Time Equivalents) who receive on average over 10,000 samples a day. Over 550 different assays are performed on-site in several laboratories, including Core (Hematology and Chemistry), Microbiology, Molecular Diagnosis, Immunogenetics, Cytogenetics, and several Specialty Laboratories and Satellite Laboratories. Over 15 million assays are performed annually in-house. Due to the complexity of the cases treated at our hospital, over 200,000 assays and panels are sent to over 60 different outside reference laboratories every year.
Information technology in use
Our hospital uses Cerner Millennium version 2012.1.19 (Cerner Corporation, North Kansas City, MO, USA) as the LIS. Our EHR is Eclipsys Sunrise Enterprise 5.5 (Allscripts, Chicago, IL, USA). Laboratory data generated in the hospital’s laboratories or received from reference laboratories via an interface are transmitted from the LIS to the EHR, where they are displayed in the “Results” section.
We use the RegScan document workflow solution from Streamline Health (Williamsport, PA, USA) to display paper documents in the EHR. Paper documents provided by patients or outside providers or hospitals, forms, as well as laboratory reports from reference laboratories that are not interfaced to our LIS are thereby stored and displayed in the EHR. This allows full integration of the EHR with paper-based clinical documentation; document management is seamlessly integrated with the rest of the EHR. Our clinical staff can access all clinical information remotely via a single access point, the EHR.12,13
Process of scanning paper documents for display in the EHR
Laboratory personnel enter a patient’s medical record number and encounter number in the password-protected RegScan program. The application displays the patient’s name, which the technologist verifies against the printout. S/he then chooses “Laboratory Report” as the document type and proceeds to scan the printout using a standard office scanner (Canon imageFORMULA DR-2510c document scanner; Canon USA, Melville, NY, USA). The scans are uploaded in TIFF format to a Streamline datacenter and stored there. Clinicians at our hospital can then open the “Paper Documents” tab of the EHR, select a laboratory report, and view the documents displayed by Streamline in an EHR window via a live interface.
Results
Our hospital’s goal is to provide clinicians with an integrated medical record that accurately reflects all clinical information generated during any patient encounter in our health care system. This goal had been achieved by 2008 for all laboratory data generated on-site, but not for all results from reference laboratories. At that time, three reference laboratories to which the majority of our send-out assays were sent were interfaced to our LIS. Nevertheless, there were still reference laboratory results that were communicated to our send-out department via fax or mail. These were either assays done at interfaced reference laboratories that had not been set up in our LIS or results from non-interfaced reference laboratories. Many of these reports were manually transcribed into the LIS, with the concomitant risk of transcription errors and of omission of clinically relevant information.11,14 Some of these reports were several pages long and contained long interpretative comments and tables. We did not have the personnel to transcribe all the information contained in these reports into our LIS and therefore forwarded them via fax or mail to the ordering physicians. These data were therefore not available to clinicians in the EHR, often causing unnecessary and costly repeat testing. We therefore started a long-term initiative to eliminate manual transcription for all send-out tests and to allow clinical staff to electronically retrieve from our EHR all laboratory results created in association with a visit at our hospital. The issues faced and the interventions taken are summarized in Table 1.
Summary of the Integration Project.
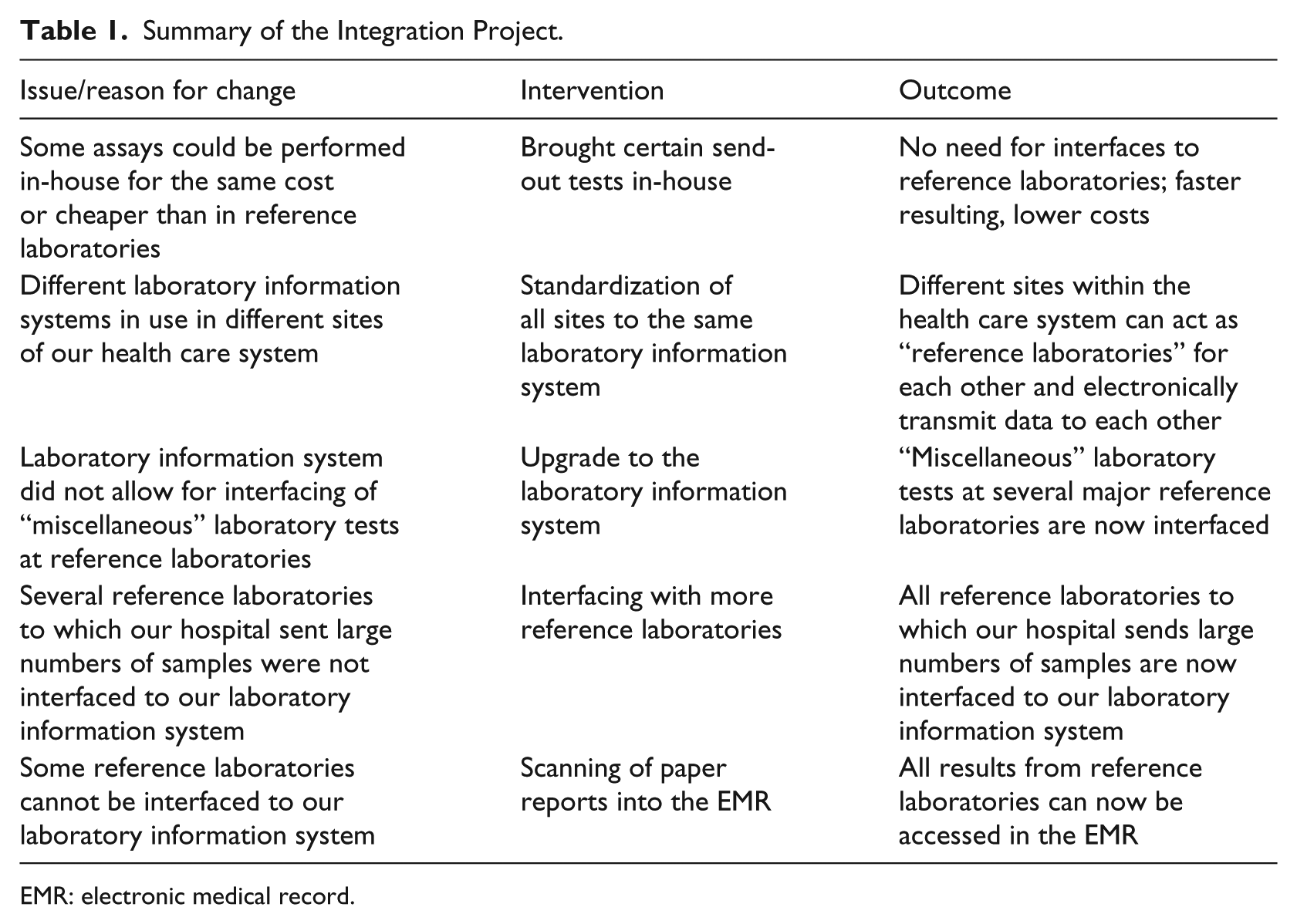
EMR: electronic medical record.
Bringing send-out tests in-house
Careful analysis of our send-out statistics showed that a number of high-volume tests were sent to reference laboratories which could be done at lower cost and with a shorter turnaround time in-house. Annually, we sent approximately 2700 samples for Vitamin D testing, 5400 samples for Epstein–Barr virus (EBV) testing, and 2700 samples for cytomegalovirus (CMV) testing to reference laboratories. Performing these assays in-house led to savings of over US$800,000. Performing these assays in the on-site laboratories allowed us to provide better service at lower cost; it also enabled us to result these assays directly from our instruments into our LIS and from the LIS into the laboratory section of the EHR, making the results available faster and avoiding transcription errors associated with manual result entry.
LIS upgrade and standardization
For historical reasons, different LISs were in use at the different geographical sites of our organization. Some of these systems did not offer all features expected in a modern LIS. In a multi-year effort, all our sites implemented the same state-of-the-art LIS with a shared database. Our laboratories can now easily exchange samples with each other. Receiving sites can download orders from other sites to their instruments, read the specimens’ barcodes, and result the samples directly into the shared LIS. This allows all sites to act as “reference laboratories” for each other or to provide emergency backup testing during instrument downtimes. This reduces the number of samples that need to be sent to outside laboratories.
Interfacing more reference laboratories and send-out assays to the LIS
When designing and building our new LIS, we increased the number of interfaced assays at the three reference laboratories with which we already had an interface. We also interfaced three additional reference laboratories to the new LIS. This significantly increased the number of send-out tests that were individually interfaced and that could be reported via HL7 in the EHR like any in-house test.
Miscellaneous assay
Three of the reference laboratories that are interfaced to our LIS (ARUP Laboratories, Salt Lake City, UT, USA; Mayo Medical Laboratories, Rochester, MN, USA; and Quest Diagnostics, Madison, NJ, USA) offer a “miscellaneous” or “generic” order. The use of these orders allows the electronic reporting of all assays performed by the reference laboratory, not only of assays specifically built in the LIS of the ordering institution. This facilitates the electronic reporting of orders that are not requested with sufficient frequency to justify setting them up as individually named orderables in the LIS. By maximizing the usage of this option, we were able to further increase the number of send-out tests that were reported electronically in our EHR and to decrease the number of non-interfaced laboratory results.
Scanning of paper documents for display in the EHR
Our Information Technology (IT) system allows us to scan documents for display in the “Paper Documents” section of the EHR. This function was initially mainly used for forms or clinically relevant information that patients presented on admission to the hospital or during outpatient clinic visits, for example, insurance cards, referral letters, and electrocardiogram, radiology, or laboratory results from other institutions. Since 2011, we expanded the use of this functionality to all reports from non-interfaced reference laboratories that we receive as hardcopies. After the reference laboratory results have been scanned in, the original order is resulted in the LIS with a comment indicating where in the EHR the scanned document can be found and which reference laboratory performed the testing. The reports are then accessible in their original format, with all reference ranges, interpretative comments, and references to the literature, to any clinician with access to our EHR. This function is especially useful for genetic testing, where an extensive explanation of the limitations and clinical significance of the findings can only be conveyed by the full original report, and where manual transcription into the LIS is practically impossible.
Outcome for reference laboratory results
The result of our multi-pronged approach to the resulting of reference laboratory results is that all laboratory data associated with any visit at our health care system are now available in the EHR (Figure 1, Table 2). Of the over 50,000 samples sent annually to reference laboratories that would not have been interfaced before the Integration Project, over 40,000 are now sent to three newly interfaced reference laboratories. Over 4000 samples are resulted via the “miscellaneous” order option by previously interfaced reference laboratories, and over 6000 reports are scanned into the EHR. No results are manually transcribed. Our clinicians have access to a one-stop source for all laboratory information for their patients, as long as their patients’ samples are sent to our hospitals’ laboratories for testing or shipping to a reference laboratory. Since all laboratory results are electronically transmitted either as an HL7 message or as a TIFF file, the possibility of transcription errors has been eliminated. Khoury et al. 15 described a median transcription error rate of 2 percent. Assuming that our transcription error rate is similar to the one described by Khoury et al., we are annually avoiding over 1000 transcription errors with potentially significant impact on patient care.
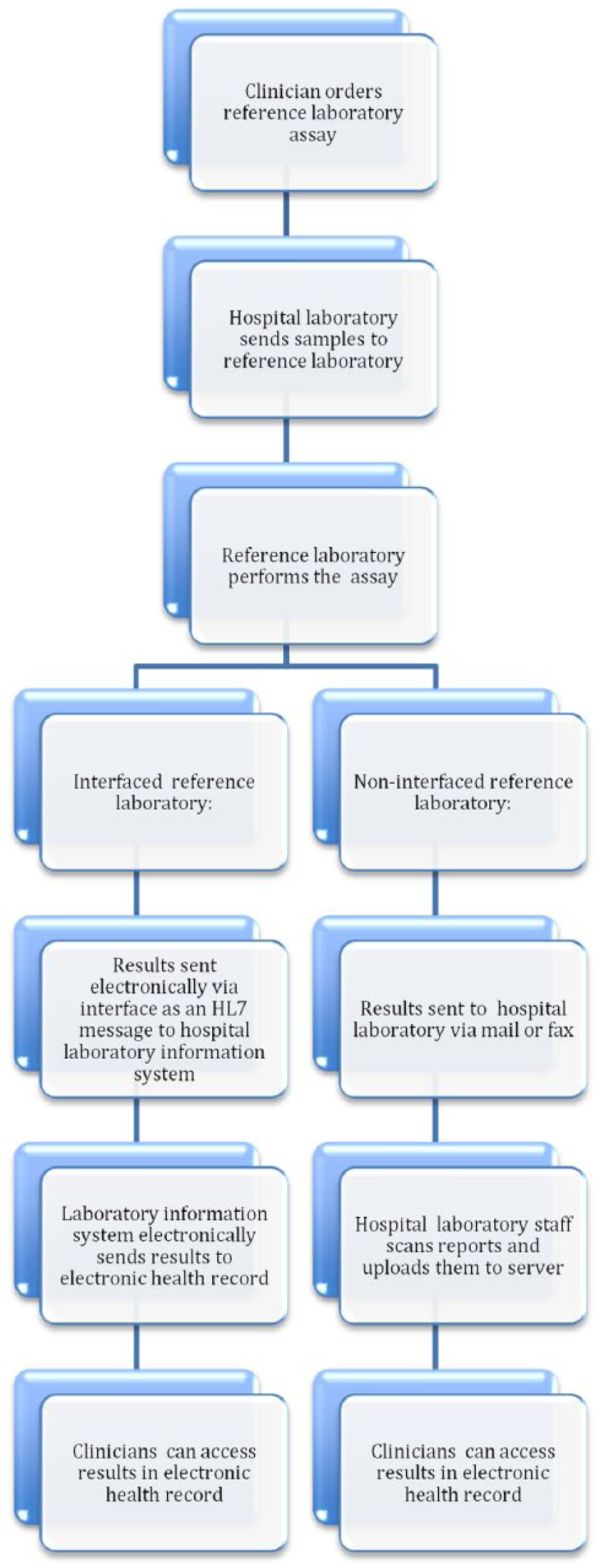
Reporting of reference laboratory results in a fully integrated EHR.
Reporting of reference laboratory results after EMR integration.

EMR: electronic medical record.
Clinician reaction to the display of all reference laboratory results in the EHR
The clinician reaction to the increased number of assays done in-house or interfaced directly to the LIS has been overwhelmingly favorable. A survey of 30 clinicians indicated that 8 (26.6%) “highly approved” of the system, 16 (53.3%) “approved,” 4 (13.3%) disapproved, and 2 (6.6%) had noticed no change. Comments included “very, very, very much approve” and “ability to access all laboratory data via the same portal works for me.” Two physicians criticized that the scanned reports were in a different section of the EHR than the interfaced laboratory results (“too many clicks”) and that the scanned reports can only be accessed from the hospital’s official EHR and not from other EHRs used in outpatient settings in our physicians’ offices (“would want scanned reports to also be available in other electronic health records”).
Discussion
By using a multi-pronged, long-term approach, our hospital has been able to achieve a fully integrated health record for all laboratory results obtained in association with an encounter in our health care system. This provides our clinical staff a one-stop source for all laboratory results, with the confidence that the risk of transcription errors due to manual data entry has been eliminated. Unnecessary repeats of often expensive tests have been minimized, since all licensed providers have electronic access to all previous reports.
The risks of manual data entry are well known. Many regulatory agencies therefore require ongoing control mechanisms such as double-keying or supervisory reviews of all manual data entries. 16 In our institution, any manual data entry must be reviewed by a second technologist before the information is released into the EMR. Our elimination of manual data entry for all send-out tests eliminates manual data entry as a source of error for reference laboratory results and dispenses with the need for time-intensive and costly review required by regulations.
Limitations of our approach include that some reference laboratory results are transmitted as TIFF files, not as HL7 messages. These results are therefore not displayed in the “Results” section of the EHR, but in the “Paper Documents” section, and cannot be displayed in the form of tables or graphs. Our clinical staff were less satisfied with the display of the scanned-in documents than with the display of interfaced results; a similar lack of satisfaction with scanned reports has been reported by Lærum et al. 4 in a study of Norwegian physicians. Nevertheless, the clinical advantages of displaying every detail of these often lengthy and complicated reports may outweigh the disadvantages of the alternative approach, namely, to manually transcribe a small part of the report into the LIS or to send hardcopies of the reports to the ordering providers.
Our EHR is only fully integrated for laboratory results generated by our laboratories or our send-out services. We cannot integrate laboratory findings generated when physicians or patients choose to send samples to outside laboratories on their own, bypassing our laboratories and send-out department.
The ability to access all relevant clinical information easily in one place should enhance the adoption of EHRs and provide a competitive advantage to health care systems that take a leadership role in achieving integration. Every institution will need to assess the obstacles it faces to being able to report all reference laboratory data in its EHR. Constant communication with clinical staff to assess needs and close cooperation with all stakeholders during implementation are crucial for the success of any effort that involves significant changes to the way laboratories communicate results to their customers.
Conclusion
The universal adoption of EHRs will depend on the timely and accurate availability of all relevant health information associated with a patient encounter in the EHR. This work describes a multi-pronged approach that includes bringing send-out tests in-house, interfacing of multiple reference laboratories to the LIS, the use of the “miscellaneous assay” option offered by some commercial laboratories, and scanning of reference laboratory results into the EHR. This allows the electronic reporting of all laboratory results, including results obtained at non-interfaced reference laboratories, in the EHR, without the need for manual data entry. Other hospitals and health care systems may find this approach useful for making all laboratory results available in the EHR, thereby creating a truly integrated health record, and for the elimination of transcription errors associated with manual data entry.
Footnotes
Acknowledgements
The author thanks Ms Elenor Pikus, Mr Jeffrey Raheb, and Ms Irina Lutinger for helpful discussions and advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
