Abstract
Background
Manipulation and reduction (M&R) of an injured limb is often required to reduce pain, bleeding and restore anatomical alignment, typically using procedural sedation and analgesia (PSA). Inhaled methoxyflurane (IMF) may be a suitable alternative that is simple to administer, does not require intravenous access, and has a favourable side effect profile. The objective of this systematic review and meta-analysis was to determine the effectiveness of IMF compared to PSA for M&R in acute traumatic limb injury. The primary outcome measure was procedural success rate. The secondary outcomes were to compare the length of stay, adverse events, patient satisfaction and cost-effectiveness.
Methods
This systematic review was planned, conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and is registered on PROSPERO CRD42024561087. A literature search was conducted and included studies from database inception to the search date (22 February 2024), using MEDLINE, EMBASE, Cochrane, CINAHL and PubMed platforms. All studies involving the use of IMF for M&R of acute traumatic limb injuries were included. Animal studies, case-reports, non-English language publications and opinion articles were excluded. The risk of bias was assessed using the Risk of Bias in Non-Randomised Studies of Interventions (ROBINS-I) tool. The quality of evidence was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. Meta-analysis was undertaken for the primary outcome of procedural success. Narrative synthesis was undertaken for the themes of length of stay, adverse events, patient satisfaction and cost-effectiveness.
Results
Fifty one abstracts were screened through a database search, reference review, and search of the grey literature. Following full-text review and exclusions, a total of six studies were included, with two suitable for meta-analysis. No significant differences in performance were observed between IMF and PSA (RR 0.90, 95% confidence interval 0.77–1.04, p = 0.16). Patients receiving IMF had a shorter emergency department length of stay, a low incidence of adverse events, and overall positive patient satisfaction. There may be cost-savings associated with the use of IMF.
Conclusion
Inhaled methoxyflurane was observed to perform no differently to procedural sedation for the successful manipulation and reduction of acute traumatic limb injuries in the emergency department. The overall quality of the evidence is poor, and further higher-level evidence is required.
Introduction
Manipulation and reduction (M&R) of an acute traumatic limb injury is often required to restore alignment of a fracture or dislocation in order to reduce pain, bleeding, and neurovascular injury. 1 M&R is typically performed early within the injury timeline, either pre-hospital or in the emergency department (ED), and is routinely facilitated by intravenous procedural sedation and analgesia (PSA).2–4 Standard PSA with intravenous agents carries a non-negligible airway and cardiovascular risk and requires significant monitoring and staffing levels. 5
Inhaled methoxyflurane (IMF) is a volatile anaesthetic agent with analgesic properties when used at subanaesthetic doses.6–11 IMF is simple to administer, does not require intravenous access, and has a favourable side effect profile.7,12 Consequently, IMF may be a suitable alternative to standard PSA, which may have greater utility in high-acuity and resource-limited environments.6–8,13 Current evidence comparing the effectiveness of IMF to standard PSA for M&R is limited to a few small-sample observational studies, with a paucity of higher level evidence.14–16
The objective of this systematic review and meta-analysis was to determine the effectiveness of IMF compared to PSA for M&R in acute traumatic limb injury. The primary outcome measure was procedural success rate. The secondary outcomes were to compare the length of stay (LOS), adverse events, patient satisfaction and cost-effectiveness.
Methods
This systematic literature review was planned, conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Online Supplemental Appendix 1) and is registered on PROSPERO CRD42024561087. The research question was derived using the PICO (Population, Intervention, Comparison, Outcome) framework, as recommended by The Cochrane Handbook for Systematic Reviews of Interventions. 17
The PICO framework is as follows:
Population – Patients with acute traumatic limb injury and a fracture(s) and/or dislocation(s) requiring reduction or manipulation; Intervention – IMF (which could also be part of multi-modal analgesia) to facilitate M&R; Comparison – Standard PSA (which could also be part of multi-modal analgesia) to facilitate M&R and Outcome – Procedural success rate, LOS, adverse events, patient satisfaction and cost-effectiveness.
Eligibility criteria
All studies involving the use of IMF (which could also be part of multi-modal analgesia) for M&R of acute traumatic limb injuries were included. Eligible study types included qualitative or mixed-methods studies, case series, case-control studies, cohort studies, cross-sectional studies and randomised controlled studies. Animal studies, case-reports, non-English language publications and opinion articles were excluded.
Information sources and search strategy
A literature search was conducted and included studies from database inception to the search date (22 February 2024), was conducted on the MEDLINE, EMBASE, Cochrane, CINAHL and PubMed platforms. The grey literature and the reference lists of all available studies were also reviewed to assess for eligible studies. The Medical Subject Headings (MeSH) terms included were ‘fracture’, ‘dislocation’, ‘injury’, ‘trauma’, ‘manipulation’, ‘reduction’, ‘methoxyflurane’, ‘Penthrox’, and ‘Green Whistle’. The search strategy used across databases was (Fract* OR Dislocat* OR Injur* OR Traum*) AND (Manipulat* OR Reduc*) AND (Penthrox OR Methoxyflurane OR “Green Whistle”).
Selection process
During the search strategy, duplications were manually removed, and the combined abstracts from the search strategy were reviewed independently by two authors. For abstracts meeting inclusion criteria, full texts were retrieved and reviewed by the same two authors.
Data collection process and data items
Two authors independently undertook data extraction, with consensus agreement or adjudication by a third author for cases of disagreement. Data items were extracted into excel spreadsheet (Microsoft ® Excel for Mac, Version 16.72, 2023). The following data items were extracted; country, setting, design, sample size, patient age in years, reduction success, ED LOS in minutes, occurrence of any adverse events, patient satisfaction and any reporting of cost-effectiveness.
Risk of bias assessment
Two authors independently assessed the risk of bias and the quality of evidence, with consensus agreement or adjudication by a third author for cases of disagreement. The risk of bias was assessed using the Risk of Bias in Non-Randomised Studies of Interventions (ROBINS-I) tool. 18 The quality of evidence was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. 19
Effect measures
The primary outcome was procedural success. The end-point for this outcome was defined dichotomously as a successful or unsuccessful M&R following IMF or standard PSA.
Data are reported as numbers (percentages) and mean (+/−standard deviation) or median (interquartile range). Ratio measures (risk ratio) and mean differences were used for measures of effect. Where possible, the estimate of effect was presented along with a confidence interval and a p-value.
Narrative synthesis was undertaken for the themes of LOS, adverse events and side effects, patient satisfaction and cost-effectiveness. LOS was reported as ED LOS in minutes, adverse effects were defined as minor or major adverse events based upon standard definitions for reporting adverse events from sedation. 20
Synthesis methods
Data were synthesised as per the PRISMA guidelines. Studies were assessed clinically (PICO) and methodologically (study design, comparability, outcome ascertainment and risk of bias). A narrative synthesis and summary of effect measures were conducted when heterogeneity was deemed too substantial for meaningful meta-analysis. All analysis and plotting were executed with Stata (Stata, version 18MP, 2024, Stata Corp, College Station TX, USA; www.stata.com).
The standard risk ratio (RR) was calculated as a measure of the relative risk of achieving the primary end-point (procedural success). The null value for the RR (i.e. the value at which IMF and standard PSA performed statistically equally) was 1.0. Confidence intervals (CIs) were calculated at the 95% level. Failure of a 95% CI to include the null value was interpreted as an indication of statistical significance.
The meta-analysis plan for the primary outcome of procedural success with IMF compared to PSA was reported as a forest plot. All studies compatible with the primary outcome comparing IMF to PSA were included in the meta-analysis.14,15 Meta-analysis of this outcome called for the utilisation of a random-effects (RE) model approach. After the search identified a very small study number (n = 2), the Sidik-Jonkman RE approach was selected as a preferred approach to minimise bias in small-study meta-analysis as this outperforms other methods.21,22 Analysis was undertaken using the available data, and no missing data was reported in the two studies included in the meta-analysis.14,15 Freeman-Tukey variance-stabilizing transformation of the effect size was used to minimise risk of inappropriate down-weighting of studies with proportions close to 0 or 1. With the understanding that heterogeneity assessment was limited by low study N, we used Cochrane I2 cutoffs: 30–60% ‘may represent moderate heterogeneity’.21,23 Low study N also precluded techniques that had been planned for use to explore small-study (publication) and similar bias.
Results
Study selection
Fifty one abstracts were screened through a database search, reference review, and search of the grey literature. Following full-text review and exclusions, a total of six studies were included (Figure 1).14–16,24–26 There were no disagreements between authors at any stage.
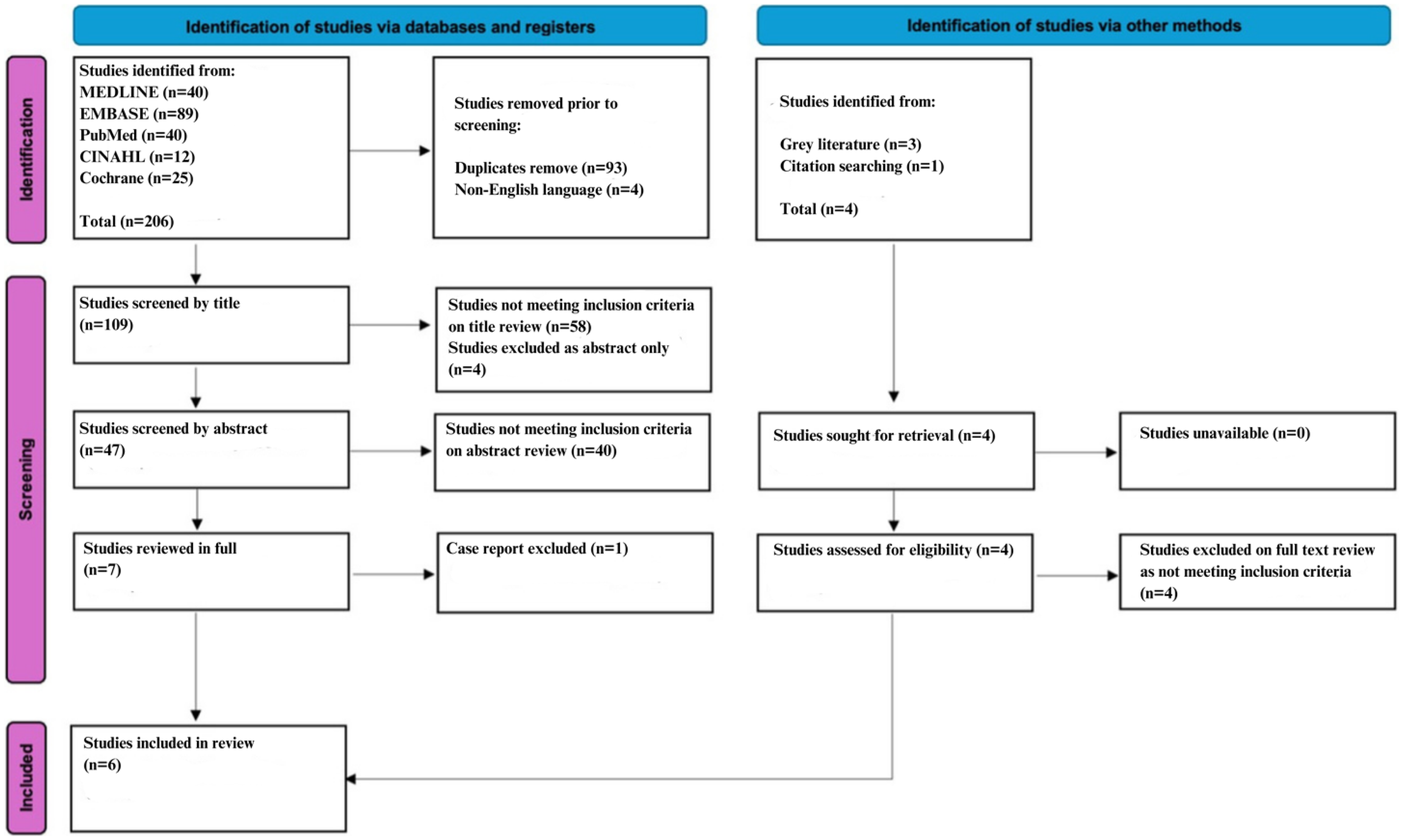
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram on the use of inhaled methoxyflurane for the manipulation and reduction of acute traumatic limb injuries.
Study characteristics
The six studies included were methodologically heterogenous and most of these were undertaken in the ED; one study was a small prospective case series, 24 one study was a prospective case-control undertaken as a service evaluation, 16 one a mixed-methods questionnaire and interview, 25 and three were retrospective observational studies with two of these directly comparing IMF and standard PSA for M&R (Table 1).14,15,26
Study characteristics for studies on the use of inhaled methoxyflurane for the manipulation and reduction of acute traumatic limb injuries.
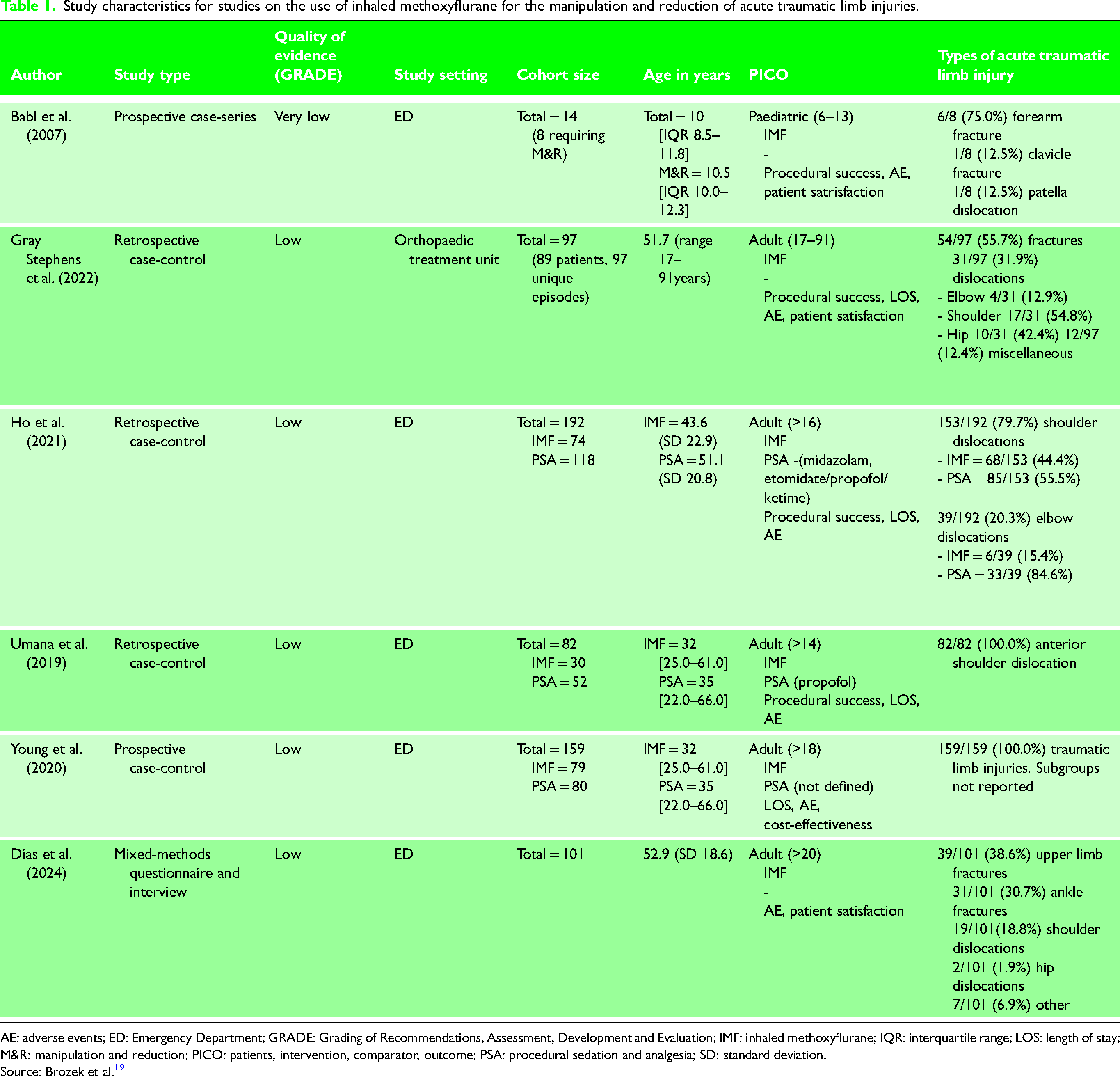
AE: adverse events; ED: Emergency Department; GRADE: Grading of Recommendations, Assessment, Development and Evaluation; IMF: inhaled methoxyflurane; IQR: interquartile range; LOS: length of stay; M&R: manipulation and reduction; PICO: patients, intervention, comparator, outcome; PSA: procedural sedation and analgesia; SD: standard deviation.
Source: Brozek et al. 19
Primary outcome: Procedural success
Two studies were appropriate for meta-analysis of the primary outcome of procedural success (Table 2). The output for the RR calculations for the studies by Ho et al. and Umana et al. are shown in the Online Supplementary Appendix 2.14,15
Outcomes of individual studies on the use of inhaled methoxyflurane for the manipulation and reduction of acute traumatic limb injuries.
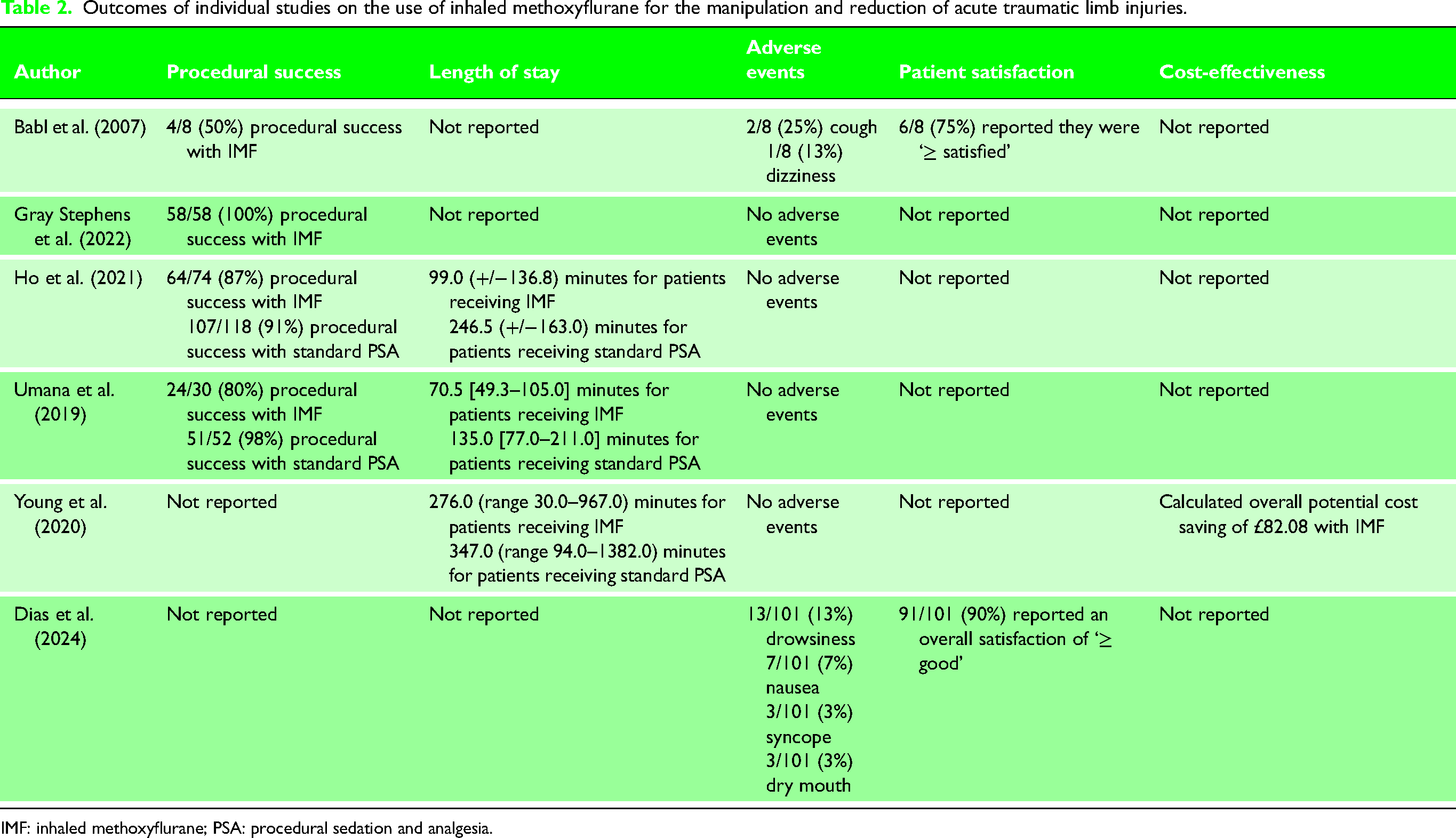
IMF: inhaled methoxyflurane; PSA: procedural sedation and analgesia.
Ho et al. 14 observed successful M&R (defined as the successful reduction of joint dislocation at first attempt) to be achieved in 64/74 (87%) receiving IMF and 107/118 (91%) receiving standard PSA, p = 0.365. Umana et al. 15 observed successful M&R (defined as successful reduction of joint dislocation without the need for PSA) in 24/30 (80%) of the IMF group and 51/52 (98%) of the standard PSA group. Figure 2 depicts a forest plot resulting from the random-effects meta-analysis. Heterogeneity was a potential source of concern but Cochrane's Q was non-significant (p = 0.15). The meta-analysis did not identify a statistically significant difference between the pooled effect estimate and the null value for RR (of 1), p = 0.16.
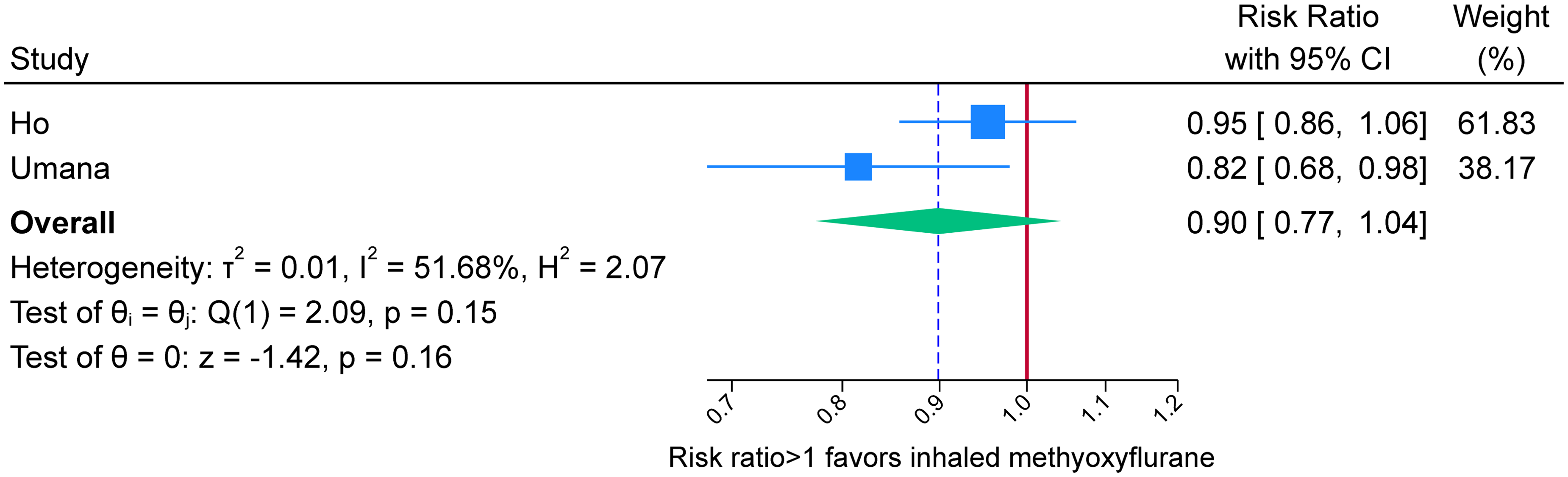
Forest plot for the end-point of procedural success using methoxyflurane compared to standard procedural sedation and analgesia.
In the study by Ho et al., 14 analgesia prior to IMF or PSA was not reported. Umana et al. 15 reported analgesia received prior to IMF or PSA. At triage 11/30 (37%) in the IMF group received ibuprofen or paracetamol, whilst 4/30 (13%) received either difene, tapentadol, solpadol, or tramadol. 15 In the PSA group 38/58 (66%) received ibuprofen or paracetamol whilst 6/52 (12%) received difene or solpadol. 15 The timing of prior medication administration was not reported. 15
Two additional included studies explored M&R success with IMF.24,26 In a case series that included eight children receiving IMF for M&R, Babl et al. observed 4/8 (50%) were successful. 24 These results are from a small heterogeneous cohort of children. Success definitions were not defined, and environmental confounders such as parental presence or the utilisation of play specialists were not controlled for. 24 Two patients had received ibuprofen or paracetamol and codeine 1 hour before IMF and one patient had received parenteral morphine 2.5 hours before IMF. 24 Gray Stephens et al. 26 described the expedited care of 89 trauma patients with acute traumatic limb injury who, in total, had 97 unique episodes of M&R utilising IMF during the COVID-19 pandemic. All cases of fracture manipulation (n = 54) and reduction of elbow dislocations (n = 4) were successful. 26 Other reductions of joint dislocations were treated with variable success, with lower success rates reported for proximal joint reductions. 26 This included 11/17 (65%) successful anterior shoulder reductions and 3/10 (30%) successful hip reductions. 26 Gray Stephens et al. 26 did not report analgesia received prior to IMF. 26
Length of stay
ED LOS for patients undergoing M&R with IMF and/or PSA was reported in three studies.14–16 Ho et al. 14 observed ED LOS (defined as the time from triage to discharge or admission) to be significantly shorter in the IMF group compared to the standard PSA group: 99.0 (+/−136.8) minutes and 246.5 (+/−163.0) minutes, respectively, p≤ 0.001. Umana et al. also observed the ED LOS (undefined) to be shorter in the IMF group, 70.5 [49.3–105.0] minutes compared to the standard PSA group of 135.0 [77.0–211.0] minutes, p = < 0.001. 15 Similarly, Young et al. 16 observed that ED LOS (defined as overall time spent in the ED) for patients requiring M&R was reduced by 71.0 minutes compared to standard PSA; IMF 276.0 (range 30.0–967.0) minutes compared to standard PSA 347.0 (range 94.0–1382.0) minutes, p = 0.038.
The duration of the procedure and the time taken to recover from sedation are all important determinants of ED LOS. Ho et al. 14 reported the duration of the procedure, and observed this to be shorter amongst the IMF group (16.0 (+/−17.0) minutes) compared to the standard PSA group (32.0 (+/−40.3) minutes), p = < 0.001. Umana et al. 15 reported the time to recovery with IMF and observed patients to have a recovery time of 30.0 [19.3–44.0] minutes compared to 47.0 [32.0–68.0] minutes in the standard PSA group, p = < 0.005.
Adverse events
All six studies described the frequency of major and minor adverse events. Four studies did not observe any adverse events.14–16,26 The majority of these studies were retrospective and relied on clinician recall or contemporaneous recording of adverse events within the notes.14,15,26
Babl et al. 24 prospectively recorded adverse events and assessed for a number of airway, cardiovascular and neurological complications during IMF administration. There were no major adverse events and 3/8 (38%) minor self-resolving adverse events, with 2/8 (25%) having a cough and 1/8 (13%) dizziness. 24 Moreover, no adverse events were reported at the 1-week telephone follow up. 24
Dias et al. 25 explored patient-reported outcomes in a questionnaire. When asked whether they had experienced any adverse events, the majority of respondents did not report any 75/101 (74%). 25 Of patients who did report minor adverse events, drowsiness was the most reported 13/101 (13%), followed by nausea (7/101 (7%)). 25
Patient satisfaction
Two studies explored the theme of patient/parent satisfaction.24,25 Babl et al. 24 prospectively recorded patient satisfaction using a five-point Likert scale. Satisfaction was recorded following IMF recovery and 1 week after discharge with no patients lost to follow up. 24 Overall, 6/8 (75%) reported they were ‘≥ satisfied’. 24 In 2/8 (25%) reported they were ‘≤ dissatisfied’. 24
Dias et al. 25 specifically explored patient satisfaction in a mixed-methods quantitative and qualitative study informed by a patient focus group. Quantitative data were gathered using the five-point Likert scale, whilst qualitative data were gathered using the Trickett short interview method. 25 The efficacy of IMF for pain reduction was rated as ‘good’ by 90/101 (89%) and ‘excellent’ by 53/101 (52%). 25 In terms of overall satisfaction, 91/101 (90%) reported an overall satisfaction of ‘≥ good’. Most patients (95/101 (94%)) reported they would use IMF again. 25 Potential areas of improvement were identified by 15/101 (15%) of patients. 25 Suggested improvements included earlier administration (10/101 (10%)), alternative analgesia (2/101 (1%)), an improved explanation of side effects (1/101 (1%)), no side effects (1/101 (1%)), and delayed manipulation (1/101 (1%)). 25 The qualitative analysis confirmed an overall positive experience. 25
Cost-effectiveness
One study explored the potential cost implication of using IMF for M&R in the ED. 16 In the ED of a London major trauma centre, Young et al. 16 estimated the cost of IMF to be £73.39 based on the staff and medication costs. Compared to the price of £155.47 for standard PSA, estimated from the NHS supply chain, Young et al. 16 calculated potential savings of £82.08 per M&R.
Risk of bias and quality of evidence
All included studies had a critical risk of bias. This was largely related to confounding and the selection of participants (Table 3). Overall, the quality of evidence was very low to low. This is based upon the risk of bias, inconsistency, indirectness and imprecision. The study graded as very low quality was a result of the small sample size (n = 8) and risk of bias from confounders (Table 1).
Risk of Bias in Non-Randomised Studies of Interventions (ROBINS-I) assessment for studies on the use of inhaled methoxyflurane for the manipulation and reduction of acute traumatic limb injuries.
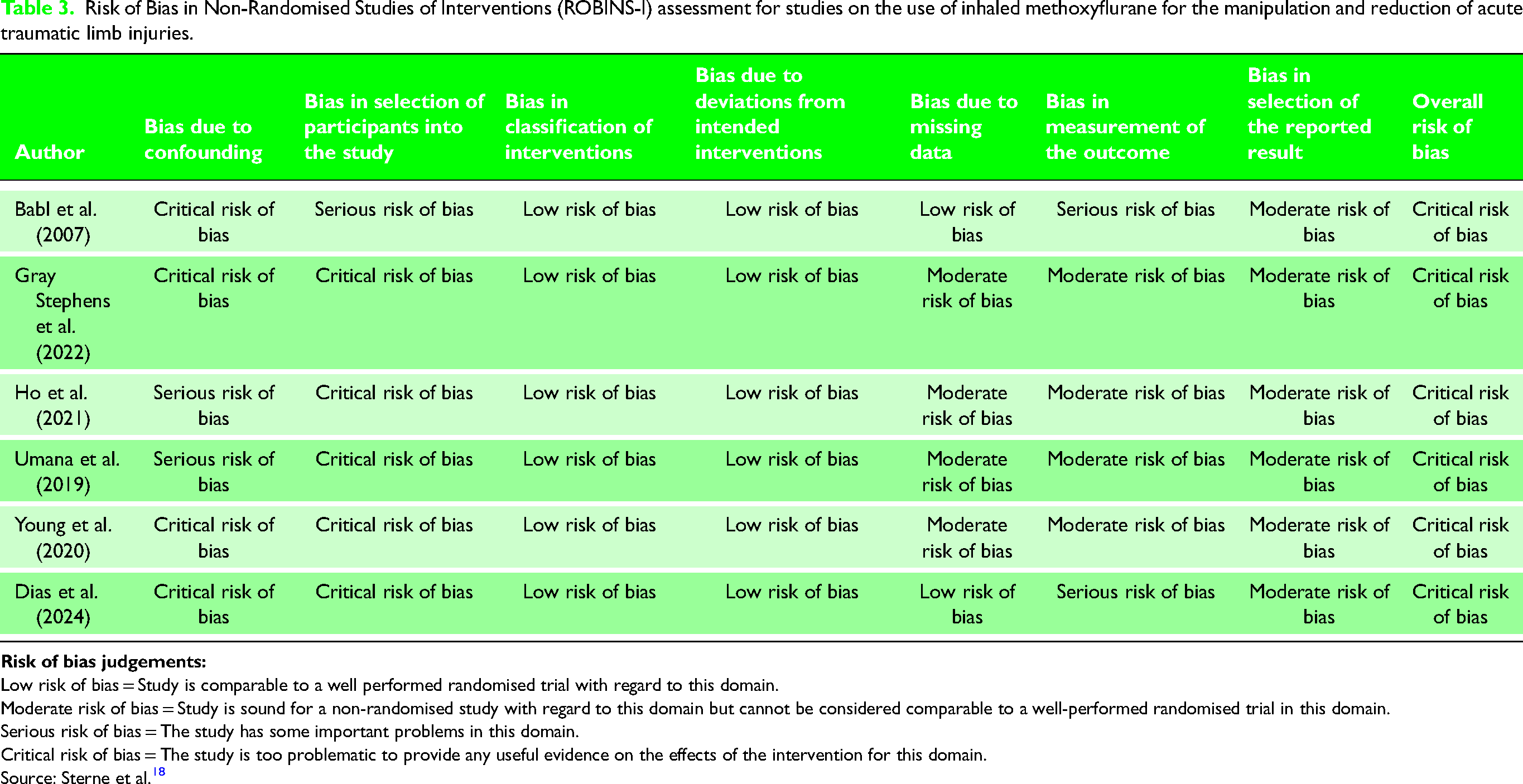
Low risk of bias = Study is comparable to a well performed randomised trial with regard to this domain.
Moderate risk of bias = Study is sound for a non-randomised study with regard to this domain but cannot be considered comparable to a well-performed randomised trial in this domain.
Serious risk of bias = The study has some important problems in this domain.
Critical risk of bias = The study is too problematic to provide any useful evidence on the effects of the intervention for this domain.
Source: Sterne et al. 18
Discussion
IMF was observed to perform no differently than standard PSA for successful M&R of acute traumatic limb injuries. The review also observed a shorter ED LOS, a low rate of adverse events, and overall positive patient satisfaction in patients receiving IMF. There may also be cost-savings associated with the use of IMF.
For analgesia in trauma, IMF was compared to placebo in the STOP! Study. 27 This randomised double-blinded controlled trial involving 300 patients with moderate trauma injuries demonstrated that patients treated with IMF had significantly greater and faster reduction in pain scores. 27 This study also reported few adverse effects and that both patients and professionals were satisfied with IMF use. 27 However, although useful in demonstrating safety and utility, the study lacked an active comparator arm. 27 The inMEDIATE study, MEDITA study, and PenASAP are all randomised controlled trials undertaken at multiple centres which compare IMF to other active standard treatments for pain.12,28,29 All these studies demonstrated IMF to be significantly greater to standard treatments for the acute management of moderate to severe trauma pain and that there were few adverse effects.12,28,29 However, these studies were focused on moderate to severe trauma pain, and a gap exists for patients with acute traumatic limb injuries requiring M&R, with these patients typically requiring PSA.12,28,29
This systematic review and meta-analysis is the first to consider the effectiveness of IMF compared to standard PSA to facilitate M&R of acute traumatic limb injuries. Despite the small number of studies for meta-analysis, the results support the use of IMF as an alternative to standard PSA; however, the results are based on poor-quality evidence. This may be presentation-specific with particular applicability for patients with fractures, shoulder dislocations and elbow dislocations, whilst a deeper level of sedation and potentially muscle relaxation may be required to reduce more proximal large-joint injuries such as hip dislocation.14–16,26 However, IMF may still have utility in the context of proximal large-joint injuries with the option to escalate to intravenous PSA in the event of unsuccessful M&R under IMF.14–16,26
Implications for practice, policy and future research
In the United Kingdom, IMF has been incorporated into analgesia and sedation guidelines by the Joint Royal Colleges Ambulance Liaison Committee (JRCALC) and the Royal College of Emergency Medicine (RCEM).30,31 Internationally, IMF is used extensively in the prehospital arena for the management of acute pain in trauma, endorsed by both the European Society for Emergency Medicine and licensed by the Therapeutic Goods Administration in Australia.8–10,32,33 IMF offers numerous practical advantages for this environment. The IMF administration device is small, lightweight, and stable at a wide range of temperatures.6,7,10 The quick onset allows rapid interventions to occur and is not associated with the delays or monitoring requirements that are associated with standard PSA.7,8,10 Recent major incidents globally have highlighted a need for additional prehospital analgesic agents, that may facilitate the splinting and traction of injured limbs without the need for monitoring. 34 IMF may pose an attractive solution to this problem and this review supports the development of expanding the role of IMF in prehospital clinical practice for this indication. Moreover, the overall paucity of evidence suggests that expansion of the literature base with larger observational studies and noninferiority randomised controlled trials may further demonstrate the utility of IMF for M&R of acute traumatic limb injuries, compared to traditional PSA.
All three studies that evaluated ED LOS demonstrated significantly shorter LOS for patients who received IMF compared to PSA.14–16 Whilst formal cost-analysis was only performed in one centre, these costs were estimated based on the United Kingdom national average and did not include a complete cost-effectiveness analysis; therefore, the true cost implications are likely to differ between hospitals. 16 In addition to the non-tangible time and personnel costs for patients undergoing procedural sedation, the reduction in the use of consumables such as cannulation, intravenous fluid, oxygen and anaesthetic equipment is likely to be significant. It is expected that these would lead to substantial cost-savings for healthcare providers; however, with the overall lack of formal cost-effectiveness analysis, this should form a key component of future work.
Limitations
This review highlights the paucity of evidence despite a detailed systematic search of the literature. All included studies were single-centre, observational studies with no randomisation, and were at critical risk of confounding and selection bias.14–16,24–26 There was a further risk of confounding and bias; for example, the retrospective design risks recall bias and the Hawthorne effect, which may be particularly relevant when interpreting the self-reporting of minor adverse events. Moreover, for patient satisfaction reduction success may confound satisfaction levels, whilst other confounders such as analgesia dosage prior to IMF and M&R were not controlled, which may have influenced the results.
The small number of studies for meta-analysis is a limitation, however, the methodology for this is robust, using the Sidik-Jonkman RE to account for the small-study meta-analysis and this provides the biggest sample size to date (n = 274). Moreover, heterogeneity in the limbs requiring M&R reflects the utility of IMF. No formal economic analysis was performed and therefore further work is required to explore the cost-effectiveness of IMF in this cohort. 16 Moreover, all studies were performed in an acute hospital setting, predominantly in the ED,14–16,24–26 and studies not in the English language were excluded (n = 4). This may reduce the generalisability of the results to other settings such as prehospital, which given the acuity, inherent environmental challenges, small staffing numbers and difficulties with monitoring, is arguably the arena in which IMF use may have the greatest potential to benefit injured patients.6,7,13
Conclusion
Inhaled methoxyflurane was observed to perform no differently to procedural sedation for the successful manipulation and reduction of acute traumatic limb injuries in the emergency department. Patients receiving methoxyflurane had a shorter emergency departmental LOS, and although formal cost-analysis is needed, this may be associated with cost-savings. The overall quality of the evidence is poor, and further higher-level evidence is required.
Supplemental Material
sj-docx-1-tra-10.1177_14604086241309051 - Supplemental material for The effectiveness of inhaled methoxyflurane compared to procedural sedation for the reduction of acute traumatic limb injuries – a systematic review and meta-analysis
Supplemental material, sj-docx-1-tra-10.1177_14604086241309051 for The effectiveness of inhaled methoxyflurane compared to procedural sedation for the reduction of acute traumatic limb injuries – a systematic review and meta-analysis by Joe Dowsing, James Price, Owen Hibberd, Stephen H. Thomas and Edward B.G. Barnard in Trauma
Footnotes
Contributorship
All authors contributed to the study conception and design. Material preparation and data collection were undertaken by JD, JP and OH. Statistical analysis was performed by ST. The first draft of the manuscript was written by JD, JP and OH, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors have no competing interests to declare that are relevant to the content of this article.
Funding
The authors did not receive support from any organisation for the submitted work.
Informed consent
No informed consent was required.
Supplemental material
Supplemental material for this article is available online.
Trial registration
PROSPERO CRD42024561087.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
