Abstract
Background:
Incisional hernia formation has been reported as high as 20% within 1 year following midline laparotomy. Since hyperthermic intraperitoneal chemotherapy is likely to impair wound healing, we sought to investigate the incidence of incisional hernia formation and abdominal wall rupture following cytoreductive surgery and hyperthermic intraperitoneal chemotherapy.
Methods:
Consecutive patients with radiographic evidence of peritoneal metastases were scheduled for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy at the Comprehensive Cancer Center, University Hospital Tuebingen, Germany. Clinical data were retrospectively analyzed.
Results:
Between May 2005 and May 2014, 271 patients underwent cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Within follow-up, 19 (7%) incisional hernias and 11 (4%) abdominal wall ruptures were observed. Age ⩾70 years, cardio-pulmonary comorbidity, the presence of pseudomyxoma peritonei or mesothelioma, and postoperative abdominal wall rupture were detected as risk factors for hernia formation. However, Cox multivariate analysis only confirmed the presence of pseudomyxoma peritonei or mesothelioma and postoperative abdominal wall rupture as independent risk factors.
Conclusion:
Our data do not suggest that cytoreductive surgery and hyperthermic intraperitoneal chemotherapy is necessarily associated with a higher incidence of incisional hernia formation. However, patients suffering from pseudomyxoma peritonei or mesothelioma and patients with postoperative abdominal wall rupture seem to be at risk for developing incisional herniation.
Keywords
Introduction
Up to the early 1990s, peritoneal surface malignancy (PM) was considered as final stage of disease with a poor prognosis and was treated only in palliative intent. Paul H. Sugarbaker noticed that gastrointestinal tumors which have metastasized to the peritoneum rarely present with simultaneous distant metastases. In order to cure those patients, he proposed the concept of complete cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC) (1). CRS and HIPEC has been shown to be capable of improving overall survival in selected patients (2–4). For therapy of colorectal cancer patients, CRS and HIPEC has been included in the 2014 German S3-Evidence-Based Guideline for colorectal cancer (5).
CRS and HIPEC is often associated with extensive surgery in order to achieve complete cytoreduction. Laparotomy starting above the xiphi-sternal junction to pubis is mandatory for an appropriate assessment of the abdominal cavity evaluating tumor load and estimate the probability of complete resection (6).
Abdominal wall hernias are reported with a frequency of 9%–20% within 1 year following median laparotomy (7). Höer et al. highlighted in a retrospective study of 2.983 laparotomy patients that almost 90% of incisional hernias occur within 5 years after surgery. Accepted risk factors are male gender, smoking, previous surgery, and wound complications (8).
Since HIPEC has previously been shown to be associated with increased postoperative infectious complications (9), we sought to investigate the incidence of incisional hernia formation and postoperative abdominal wall rupture following CRS and HIPEC.
Materials and Methods
Patients
Between June 2005 and May 2014, consecutive patients were enrolled in the Peritoneal Surface Malignancy Program at the Comprehensive Cancer Center, University of Tuebingen, Germany. Preoperative diagnostics consisted of a clinical examination, blood test, computed tomography (CT) scan and laparoscopy in some cases to rule out distant metastases and local irresectability, which was defined as infiltration of the mesenteric axis, retroperitoneum, or diffuse extensive tumor growth on the entire small bowel surface. All patients that did not show signs of irresectability were scheduled for CRS and HIPEC during a weekly interdisciplinary oncologic team meeting, presenting patient demographics and imaging results. Pre- and postoperative data were prospectively collected in an institutional database and retrospectively analyzed. The presence of chronic obstructive pulmonary disease or coronary heart disease was summarized as cardio-pulmonary comorbidity. Surgical site infection (SSI) was defined as either wound redness, wound dehiscence with secretion either of putrid or caliginous smelly fluid, or requiring antibiotic treatment or surgical intervention. After surgery, patients were followed up daily during hospital stay. After discharge, patients underwent physical examination every 3 months to assess for recurrent disease including physical examination and CT. This investigation was performed in accordance with the local ethical guidelines.
Surgical Procedure
After laparotomy through a midline incision, complete adhesiolysis, and exploration of the entire abdominal cavity, the peritoneal carcinomatosis index (PCI) was determined following the criteria described by Jacquet and Sugarbaker (10). If peritoneal metastases were found to be resectable, CRS was started aiming for complete cytoreduction (CC-0, CC-1) (CC-0 meaning no visible disease; CC-1 meaning nodules smaller than 0.25 cm.)
After complete cytoreduction and fashioning of intestinal anastomoses, if necessary, HIPEC with Cisplatin 50 mg/m2 for 90 min at 42°C (gastric cancer, recurrent ovarian cancer, and mesothelioma) or Mitomycin 35 mg/m2 (colorectal cancer, pseudomyxoma peritonei) was administered using the open coliseum technique. Since February 2012, we performed the open-closed technique using Oxaliplatin 300 mg/m2 intraperitoneally (i.p.) combined with 5-FU (400 mg/m2) intravenously (i.v.) for 30 min in colorectal cancer and pseudomyxoma peritonei patients. Cisplatin (75 mg/m2 i.p.) combined with Doxorubicin (15 mg/m2 i.p.) was used in gastric cancer, recurrent ovarian cancer, and mesothelioma. Abdominal wall closure was performed either by PDS II 1 (Ethicon, Norderstedt, Germany) running suture or 1 Vicryl (Ethicon, Norderstedt, Germany) interrupted single stitches.
Definition of Incisional Hernia and Abdominal Wall Rupture
Incisional hernia was defined as either (1) a fascial gap or protrusion with a relaxed and tensed abdominal wall in supine and standing position after completed superficial wound healing or (2) a fascial gap or separation in axial images on CT. Abdominal wall rupture was defined as missing continuity of abdominal fascia in combination with wound dehiscence with consecutive relapse operation.
Statistics
Data are presented as median (min.–max.) or n (%) unless otherwise stated. Qualitative differences were compared using Chi-square test, quantitative differences using Mann–Whitney U test. Risk factors for hernia formation were assessed by univariate and multivariate analysis (Cox regression analysis). A p-value less than 0.05 was considered significant. SPSS version 24.0 software (SPSS, Chicago, IL, USA) was used for all statistical analysis.
Results
Operative Outcomes
In total, 271 consecutive patients with a median age of 55 (17–76) years were analyzed. The median follow-up was 38 (2–110) months. Patients were retrospectively categorized with respect to incisional hernia formation (hernia vs non-hernia group). Both groups were comparable for demographic and intraoperative characteristics. In the hernia group, however, patients were older, showed more often cardio-pulmonary comorbidities, developed more frequently abdominal wall rupture, and suffered more often from pseudomyxoma peritonei or mesothelioma. However, there was no difference for SSI, technique of abdominal wall closure, or HIPEC-related mild leucopenia (Table 1).
Demographic and intraoperative data.
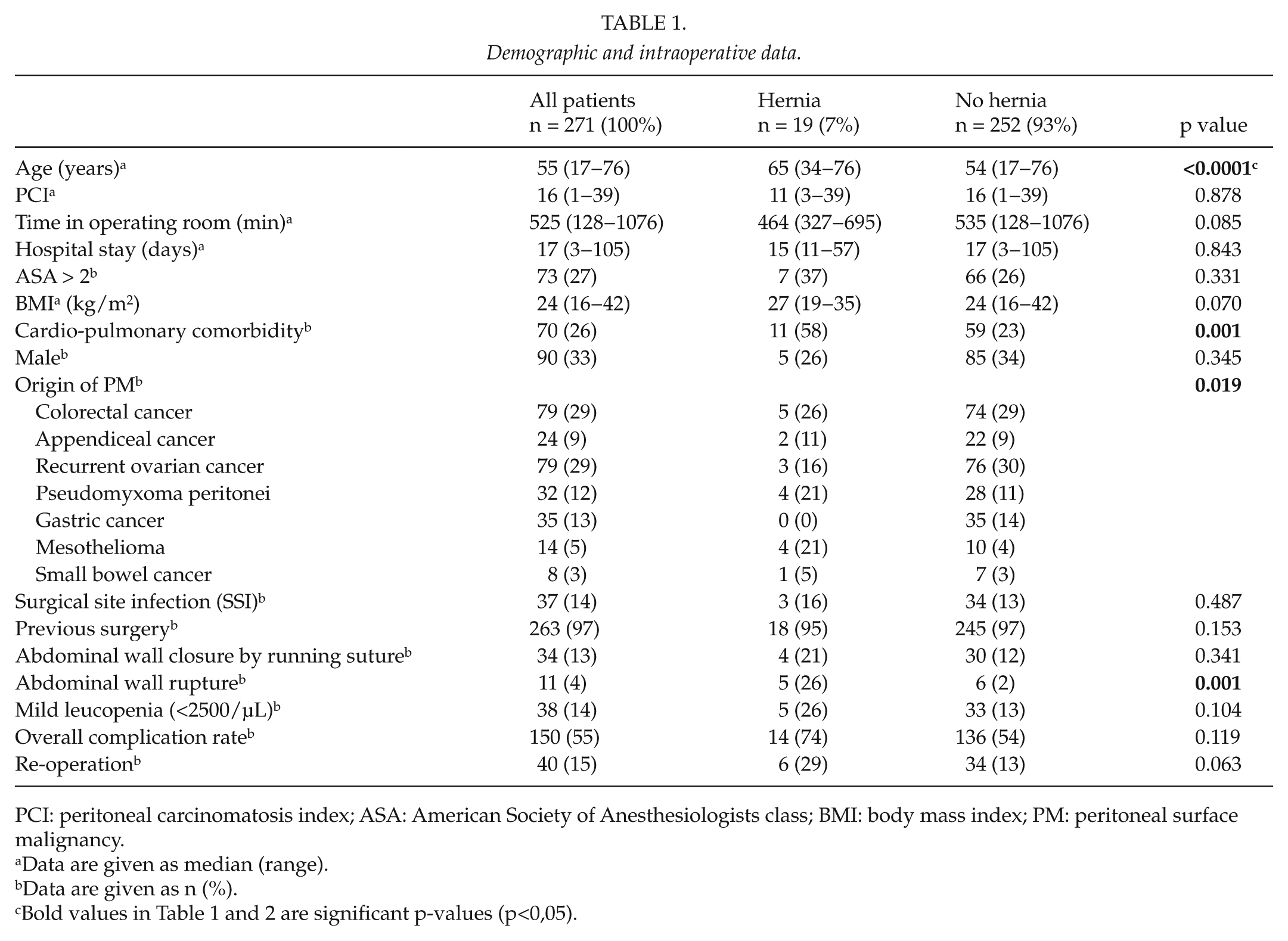
PCI: peritoneal carcinomatosis index; ASA: American Society of Anesthesiologists class; BMI: body mass index; PM: peritoneal surface malignancy.
Data are given as median (range).
Data are given as n (%).
Bold values in Table 1 and 2 are significant p-values (p<0,05).
In all, 19 patients (7%) developed incisional herniation within follow-up. Hernia formation occurred in median within 3.7 (0.3−42) months after surgery. Even though no incarceration was observed, hernia repair had to be performed in 12 patients (63%).
In all, 11 patients (4%) experienced abdominal wall rupture within follow-up. Among those 11 patients, 5 (26%) developed an incisional hernia (Table 1).
Risk Factors for Hernia-Formation and Abdominal Wall Rupture
Univariate analysis demonstrated age ⩾70 years, cardio-pulmonary comorbidity, the presence of pseudomyxoma peritonei or mesothelioma, and abdominal wall rupture as risk factor for hernia formation.
However, only the presence of pseudomyxoma peritonei or mesothelioma and abdominal wall rupture remained significant risk factors in multivariate analysis (Table 2).
Risk factors for incisional hernia formation.

Cox regression analysis was used for multivariate analysis with a p < 0.05 considered significant.
Bold values in Table 1 and 2 are significant p-values (p<0,05).
Discussion
Incisional hernia formation is a frequent long-term complication following median laparotomy. It occurs in up to 20% of patients following abdominal surgery (11). Suitable diagnostic tools for the detection of incisional hernias are clinical examination, ultrasound, and CT (12). Accepted risk factors include obesity, age, male gender, SSI, suture techniques, and tension (8, 13).
The influence of HIPEC on abdominal wall wound strength has not been investigated in humans so far. Valle et al. (9) showed that CRS and HIPEC is associated with an increased risk of postoperative infections which might be due to the length of operative time, intraoperative contamination in case of digestive tract resections, and HIPEC-related immunosuppression.
Aarts et al. (14) demonstrated a decrease in abdominal wall wound strength following CRS and HIPEC in an animal model which might—at least in part—explain the higher incidence of abdominal wall rupture following CRS and HIPEC. Two multicenter trials (INSECT (15) and ISSAAC trial (16)) showed a rate of burst abdomen after elective midline laparotomy of 2.8% and 2.0%, respectively, as opposed to 4% in our patient cohort. The combination of chemotherapy and hyperthermia is further known to increase cellular death and induce apoptosis (17). Partial resection of the abdominal wall which might be necessary for complete tumor resection additionally compromises abdominal wall closure and strength.
In our collective of patients, SSIs were not associated with a significantly increased risk of abdominal wall herniation. Similarly, as opposed to the literature, we observed only a statistical trend for an elevated body mass index as well as re-operation increasing the risk of incisional hernia formation. However, these findings might be explained by the low number of patients included. Postoperative mild leucopenia (<2500/µL) which might correlate with systemic and local immunosuppression and consecutive impairment of wound healing did not show a significantly increased risk of incisional hernia formation in our collective of patients.
The length of operative time which might correlate with an increased risk of postoperative infections as proposed by Valle et al. did not show any influence on the risk of abdominal wall herniation.
In univariate analysis, age ⩾years, cardio-pulmonary comorbidity, the presence of pseudomyxoma peritonei or mesothelioma, and postoperative abdominal wall rupture were detected as risk factors for incisional hernia formation. In multivariate analysis, only the presence of pseudomyxoma peritonei or mesothelioma and abdominal wall rupture remained significant risk factors.
In case of postoperative abdominal wall rupture, reconstruction of the abdominal wall is challenging, resulting many times in non-tension-free repair. Since high suture tension is likely to result in collagen disbalance, favoring collagen type III with less tensile strength compared to type I, hernia formation might be the long-term consequence (18). The finding that patients suffering from pseudomyxoma peritonei or mesothelioma are at higher risk of hernia formation is interesting. However, involvement of the parietal peritoneal layer is more likely in these entities which might also result in removing parts of the abdominal wall in order to achieve complete cytoreduction. This could also explain that the intra-abdominal distribution of the tumor rather than a high PCI is associated with an increased risk of abdominal wall morbidity. Again, abdominal wall closure might be more difficult in these cases with higher suture tension resulting in lower wound strength and impaired long-term healing.
The overall low incidence of incisional herniation of 7% in our patient cohort might be due to different causes. On the one hand, survival of some patients is limited, so they will not survive long enough to suffer incisional herniation. On the other hand, the number of patients suffering a hernia might increase as follow-up proceeds and follow-up in our collective is inhomogeneous (2–110 months). As already mentioned in the introduction, 90% of all hernias are seen within 5 years of follow-up (8). Therefore, the incidence of abdominal wall morbidity is likely to further increase when follow-up progresses.
Even though the number of hernias in our cohort was low (7%) with no observed incarceration, hernia repair had to be performed in 12 patients (63%) because of clinical symptoms.
Conclusion
In summary, our study suggests that the overall risk of suffering from incisional hernia formation after CRS and HIPEC might not necessarily be increased. Patients suffering from pseudomyxoma peritonei or mesothelioma and those with postoperative abdominal wall rupture seem to be at higher risk of incisional hernia formation within follow-up.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
